Abstract
Introduction
The decision whether to operate on patients with intracranial tumors is complex and influenced by patient-specific factors, including the preoperative functional status. This work assesses the risks for mortality and complications, and post-operative recovery in functionally dependent patients undergoing microsurgical resection of intracranial tumors.
Methods
Observational two-center study, analyzing institutional registry data. Dependency was defined as admission Karnofsky Performance Scale (KPS) of ≤ 50. The primary endpoint was in-hospital mortality. Secondary endpoints were rate and type [Clavien-Dindo grade (CDG)] of complications, as well as postoperative change in KPS until the 3-month follow-up (M3).
Results
Of n = 1951 patients, n = 98 (5.0%) were dependent. Mortality rates were 2.0% for dependent and 0.4% for independent patients (p = 0.018). In univariable analysis, dependent patients were more likely than independent patients to die in hospital (OR 5.49, 95% CI 1.12–26.8, p = 0.035). In a multivariable model, the effect was slightly attenuated (OR 4.75, 95% CI 0.91–24.7, p = 0.064). Dependent patients tended to experience more postoperative complications. They were more likely to suffer from a severe complication (CDG 4 and 5; OR 3.55, 95% CI 1.49–8.46, p = 0.004). In 40.8 and 52.4% of cases, dependent patients regained functional independence at discharge and M3, respectively.
Conclusions
In operated patients with intracranial tumors presenting functionally dependent at admission, the risk for in-hospital mortality and complications is elevated. However, if conducted successfully, surgery may lead to regain of independence in every second patient within 3 months.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
There are many factors that influence the decision-making process for the operative treatment of patients with intracranial tumors. To estimate its benefits and risks, patient- (e.g., age, comorbidity, functional status) and disease-specific factors (e.g., tumor dignity and location) need to be evaluated thoroughly. Owing to development of modern operative techniques and improvements in the perioperative management, microsurgical resection of intracranial tumors has become possible even in most difficult locations and patients with advanced age or comorbidity. The feasibility issue of neurosurgical treatment has become less relevant nowadays. In clinical practice, however, physicians are often confronted with the question whether or not patients admitted in a functionally dependent status will also benefit from surgical treatment.
This uncertainty may result from the lack of experience and data: for decades, dependent patients with intracranial tumors have a priori been excluded from microsurgical resection. They have traditionally received diagnostic biopsies (plus adjuvant treatment) and were not included in randomized surgical trials [1]. For these reasons, there is limited literature for neuro-oncology. Recently, one prospective study on a limited patient sample showed significant improvement of short-term Quality of Life (QoL) in a notable fraction of functionally dependent patients with glioblastoma, but also an increased risk for morbidity and mortality [2]. Correspondingly, previous studies in other surgical disciplines showed that functional dependency was associated with an increased risk for mortality, postoperative complications and undesired outcome [3,4,5].
For the lack of data on this important question, the present study assesses the risks for mortality and complications, and reports post-operative recovery in functionally dependent patients undergoing microsurgical resection of intracranial tumors.
Methods
We retrospectively analyzed prospectively acquired patient data from institutional registries established independently at two Neurosurgical Departments. The methodology of data collection and validation for both registries has been described previously [6, 7].
Patient identification
All patients with full datasets undergoing microsurgical procedures aiming at maximum safe resection of intracranial tumors were considered. Patients undergoing diagnostic biopsies were excluded. We included surgical procedures performed between 01/2013 and 01/2017 at the Department of Neurosurgery, University Hospital of Zurich, Switzerland (USZ), and between 01/2012 and 12/2014 at the Second Division of Neurosurgery, Fondazione IRCCS Istituto Neurologico Carlo Besta of Milan, Italy (FINCB).
Surgical technique and patient management
Operations were generally performed under general anesthesia. Intraoperative neuronavigation was used in most cases and both intraoperative ultrasound (ioUS) and ioMRI were regularly applied to maximize extend of resection (EOR) [8]. For tumors located in eloquent areas, intraoperative neurophysiological monitoring alongside with preoperative functional MRI was frequently used to minimalize the risk for postoperative neurologic deficits. Awake surgery was performed if language monitoring was required during tumor resection.
Assessment of functional status and study groups
The Karnofsky performance status (KPS) [9] was assessed in all patients before surgery, as well as at time of discharge. Additional data on the 3-month outcome, including KPS, was available for the USZ patient cohort. Based on the KPS before surgery, we dichotomized the combined patient cohort into functionally dependent (KPS ≤ 50) and functionally independent (KPS ≥ 60) patients.
Recorded variables
Besides KPS, we recorded age, sex, type of surgery, and histopathologic diagnosis in each patient. Preoperative MRI images were reviewed and maximum tumor size, eloquent (defined as: motor, sensory, language or visual areas, hypothalamus, thalamus, internal capsule, brainstem, and pineal region) and posterior fossa location was determined, according to Ferroli et al. [6].
Any deviation from the normal postoperative course was considered a complication, including adverse events that are sometimes expected (e.g. transient motor deficit after resection of a glioma in the supplementary motor area), despite the fact that they may be regarded as acceptable or unavoidable. The severity of complication was rated using the Clavien–Dindo grading system (CDG; Supplementary Table 1) [10]. CDG 4 and 5 were considered as severe complications. In cases with multiple documented complications, the most severe one was chosen for statistical analysis.
The etiology of complications was categorized according to Ferroli et al. [6].
Endpoints and statistical considerations
The primary endpoint was in-hospital mortality. Secondary endpoints were the postoperative change in KPS, the rate, type (CDG) and etiology of postoperative complications.
Demographic baseline data were described using frequencies and percentage for categorical variables. Ordinal variables were described as medians and interquartile range (IQR). Interval variables were described as group means and standard deviations (SD). Imbalances between the dependent and independent groups were tested using Pearson χ2 tests, Wilcoxon rank sum tests or student’s t tests, as appropriate. Logistic regression analysis was performed to estimate the effect size of the relationship between the dependent and independent variables of interest. First, a univariable model was built to analyze the direct relationship. Then, a multivariable model was adjusted for baseline group differences. Odds ratios (OR) and 95% confidence intervals (CI) were analyzed for changes and sensitivity analyses were made. P-values < 0.05 were regarded as statistically significant. Statistical analyses were performed with Stata version 14.2 for Mac (College Station, TX: StataCorp LP) and Matlab version R2016b.
Ethical considerations
The scientific workup of registry data was approved by the institutional review boards of both institutions and the patient’s informed consent was waived. At USZ, the local ethics committee (Kantonale Ethikkommission KEK-ZH 2012–0244) approved the protocol that was registered at clinicaltrials.gov (NCT01628406) and follows the STROBE recommendation for observational studies. The authors report no relevant conflicts of interest.
Results
A total of n = 1951 consecutive patients were available, from which 98 (5.0%) were functionally dependent at the time of surgery. The median KPS at presentation in the dependent group was 50 (IQR 40–50) and 90 (IQR 80–90) in the independent group.
Patient- and disease-specific information is presented in Table 1. There were some significant differences with respect to age, histopathologic diagnosis and tumor size between both groups. The study groups were balanced for all other baseline variables, including sex, type of surgery, and surgery in eloquent or posterior fossa locations.
Analysis of the primary endpoint
In-hospital mortality was higher in the dependent (2.0%) than in the independent group (0.4%; p = 0.018). Accordingly, dependent patients had a 5.49 times greater odds of dying during hospitalization (OR 5.49; 95% CI 1.12–26.8; p = 0.035). In a multivariable model, adjusted for baseline differences, the effect was slightly attenuated and with loss of statistical significance (OR 4.75, 95% CI 0.91–24.7, p = 0.064; Table 2). Sensitivity analyses indicated robustness of the model.
Analysis of the secondary endpoint: postoperative change in KPS
At discharge, functionally dependent patients showed a median improvement of 10 points on the KPS (IQR 0–30; range − 40 to 60; Fig. 1), whereas the KPS remained unchanged in independent patients (median: 0, IQR 0–0; range − 90 to 30). Of the 98 dependent patients, 10 (10.2%) reached a KPS of 60, 10 (10.2%) a KPS of 70, 12 (12.2%) a KPS of 80 and 8 (8.2%) a KPS of 90 points.
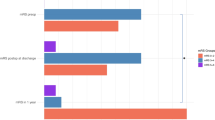
Change in the KPS between admission and discharge (grey) or 3-month follow-up (white) in functionally dependent patients undergoing microsurgical resection of an intracranial tumor. Note that even though the majority of patients is discharged without significant change in the KPS, considerable improvement is possible in the early period already. At the 3-month follow-up the spread of the data is larger
At the 3 month follow-up, functionally dependent patients showed a median improvement of 20 points on the KPS as compared to preoperative (IQR − 10 to 40; range − 50 to 80; Fig. 1), and independent patients remained unchanged on the KPS (median: 0, IQR 0–10; range − 90 to 40). Of the 82 dependent patients with 3 month follow-up, 7 (8.5%) reached a KPS of 60, 5 (6.1%) a KPS of 70, 10 (12.2%) a KPS of 80, 18 (22.0%) a KPS of 90 and 3 (3.7%) a KPS of 100 points. The relative frequency and magnitude of the change in the postoperative KPS is illustrated in detail in Fig. 1.
In 40.8% of cases, dependent patients were discharged in functionally independent condition (Fig. 2). At the 3 month follow-up, 52.4% of originally dependent patients had regained their functional independence (Fig. 2).
Graph illustrating the distribution of independent patients at discharge or 3-month follow-up after maximal safe resection of a brain tumor, in relation to functional dependence at admission (grey). Note that 40.7% of dependent patients before surgery were discharged in an independent condition and the proportion of independence increased to 52.4% at 3 months postoperative
Detailed postoperative changes of KPS, stratified by histopathologic diagnosis, are presented in Supplementary Table 2. At discharge, the highest median improvement on the KPS was observed in patients with low-grade gliomas (LGG; + 30 points), followed by anaplastic astrocytoma (AA) and adenoma (both + 15 points). Previously dependent patients with AA showed the highest rate of independence (67%), followed by adenomas (60%) and LGGs (57%). At M3, the highest median improvement on the KPS was observed in patients with adenomas, AA and LGG (+ 40 points), followed by other tumor types (+ 35 points). Previously dependent patients with adenomas and other tumor types showed the highest rate of independence (75%), followed by LGG (71%) and both AA and meningioma (60%). Previously dependent patients with metastases showed some improvement at discharge (+ 10 points; 40% independent), but performed relatively poor at the M3 follow-up (− 20 points; 32% independent).
Analysis of the secondary endpoint: postoperative complications
Postoperative complication data is presented in Supplementary Table 3. Any type of complication until discharge was recorded in 56.1% of patients of the dependent, compared to 34.9% in the independent group (p = 0.069). The majority of complications (531/689 = 77% CI [74–80%]) were treated without invasive treatment (CDG 1 and CDG 2).
The most common type of complication in the dependent group was CDG 2 (requiring pharmacological treatment only; 34.5%), followed by CDG 1 (not requiring any treatment; 20.0%), whereas in the independent group this order was reversed. Severe complications occurred more often in the dependent group. In a multivariable model, adjusted for baseline differences, dependent patients had a 3.55 times greater odds of suffering from a severe complication (CDG 4 and 5; OR 3.55, 95% CI 1.49–8.46, p = 0.004; Supplementary Table 4).
The etiologies of complications are presented in Supplementary Table 5. The most common cause of complication in the dependent group was internal medicine-related (37.2%; e.g. urinary tract infection, pneumonia, pulmonary embolism, renal insufficiency), followed by direct surgical trauma (25.6%) and sepsis (11.6%). Conversely, the most frequent etiology in the independent group was direct surgical trauma (42.9%), followed by internal medicine-related (13.0%) causes and hemorrhagic events (9.1%).
Complications, length of stay and KPS at discharge
The grade of the complications registered at discharge correlated with the length of stay and the functional performance (Fig. 3). The median postoperative length of hospitalization was significantly higher in cases with a complication (9 vs. 6 days, p < 0.001). The length of stay and the CDG grade were correlated with Spearman’s rho = 0.44 (p < 0.001). The linear fit had a slope of 2.3 days per increment of CDG (Fig. 3b). This prolongation in stay was to be expected for a therapy-oriented grading scale like CDG. When considering functional performance, the difference between KPS at admission and KPS at discharge (ΔKPS) decreased with the CDG-grade of the complication. The ΔKPS and the CDG grade were correlated with Spearman’s rho = − 0.45 (p < 0.001). This correlation remained unchanged if the deceased patients (CDG 5) were excluded. The linear fit had a slope of − 4.8 KPS points per increment of CDG (Fig. 3c).
Complications and KPS at discharge. a In 689/1951 cases (35%), a complication of any grade was noted at discharge but the majority of those (77%) was treated without invasive treatment (CDG 1 and CDG 2). b The length of stay and the CDG grade were correlated with Spearman’s rho = 0.44 (p < 0.001). The linear fit had a slope of 2.3 days per increment of CDG. c The difference between KPS at admission and discharge (ΔKPS) decreased with the CDG-grade of the complication. The ΔKPS and the CDG grade were correlated with Spearman’s rho = − 0.45 (p < 0.001). The linear fit had a slope of − 4.8 KPS points per increment of CDG. AE adverse event
Discussion
This study set out to explore the relationship between functional dependence before maximum safe microsurgical resection of intracranial tumors and postoperative mortality, complications and recovery in a large two-center dataset. As the data concerning microsurgical therapy of patients that preoperatively depend on care is limited, the present report provides robust estimates of the dangers, but also benefit that can be expected from surgical treatment. The most striking findings were that first, in dependent patients the risk for in-hospital mortality was fourfold higher than in independent patients. Second, dependency could be identified as strong, independent predictor of severe complications leading to ICU management or death. Third—and importantly—if successfully conducted, surgical treatment led to regain of functional independence in a considerable fraction of patients.
Study groups were built by dichotomizing the KPS, using a pragmatic cut-off. Patients rated KPS 50 were counted as dependent for this study, as those require “considerable assistance and frequent medical care”. Even though a previous study chose the cutoff for dependency at KPS 60 [2], we considered patients “requiring occasional assistance, but able to care for most personal needs” as (mostly) independent. The credibility of our observations is substantiated by internal consistencies both within and between the two independent datasets, as well as by validity between actual observed and theoretically expected outcomes conferred through the tested variable. Internal consistency is evident by a strong relationship between the severity of postoperative complications and both the change in patient condition at discharge and length of hospitalization (Fig. 3). The outcome measures demonstrated improved results in the independent group compared with the dependent group, commencing at discharge and persisting to M3. Validity (expected compared with observed) can be appreciated at two levels. Surgery leads to marked improvement on the KPS, independent of the study groups. This improvement was graduated, measurable at discharge and more substantial and with larger spread of data at the M3 follow-up (Fig. 1). It corresponds to the typical postoperative course after microsurgical resection of intracranial tumors [11], lending credibility to the results. The second level of validity pertains to patient performance by group. Based on the previous literature, it could be expected that if a group difference was observed, this difference would be in favor of the independent patients and measurable on the applied outcome metrics (Fig. 2).
The functional level of cancer patients as determined by the KPS is strongly linked to the disease burden, and can be used as prognostic predictor in general oncology [12, 13]. In patients with intracranial tumors, however, poor functional level may be linked to the location of the lesion than to the general dissemination of the disease [2]. This concept has been substantiated by demonstrating that the prognostic impact of functional status was shown to be independent from tumor volume [14, 15]. In addition Chambless et al. could demonstrate that compared to the preoperative KPS, the postoperative KPS had superior predictive capacity for overall survival, also pointing out the influence of intracranial mass lesions on the KPS in non-operated patients [11]. It is conceivable that negative treatment selection explains to some extend the particularly poor prognosis of functionally dependent patients with intracranial tumors. They are often exempt from clinical trials and likely to undergo diagnostic biopsies only, leading to a self-fulfilling prophecy [2, 16]. In dependent patients, tumor location tends to be more often eloquent, which can be appreciated from our data as well (Table 1). If the underlying pathology is suspected to be malignant and/or disseminated, palliative microsurgical resection remains controversial in light of the modest expected improvement and increased surgical risk. Here, diagnostic biopsy is usually favored to achieve histopathologic diagnosis and enable adjuvant treatment while preserving short-term function and quality of life [2, 17]. Our patient cohort is likely to be a selected one: we commonly propose microsurgical resection in dependent patients that fulfill the following criteria: (1) the general health status allows for a neurosurgical intervention, (2) the tumor dissemination is focal, or diffuse but non-eloquent, (3) the tumorous lesion is the major limiting factor concerning life expectancy, (4) the reduction of mass effect and/or edema following microsurgical treatment allows for significant recovery.
Analyzing the present data we observed tendencies for certain subgroups of dependent patients to benefit more from surgical treatment.
Tumor type
We noticed a tendency towards better functional recovery in those patients with benign or less malign histopathological diagnoses (LGGs, AAs and adenomas, besides meningiomas; Supplementary Table 2). Given the nature of the underlying disease, those patients are also likely to have a more sustained mid- and long-term benefit from surgical treatment. Our data suggest that even carefully selected dependent patients with glioblastomas can benefit from microsurgical treatment, and the current data help to estimate its short- to mid-term effect.
In contrast, dependent patients with brain metastases carried a rather poor prognosis in general and concerning benefit from surgery (Supplementary Table 2). They tend to present in a poor health state linked to the intracranial dissemination, in conjunction with the primary tumor’s disease burden, and the effects of (neo)adjuvant therapies (chemotherapy, radiation) besides other factors (fatigue, paraneoplastic syndromes, etc.) [18, 19]. For those patients, the therapeutic options comprise stereotactic radiosurgery, whole brain radiation therapy and chemotherapy, and the indication for surgery should be critically evaluated, given the present findings [20, 21]. More research is needed to select those patients with brain metastases that will likely show a positive and sustained response from microsurgical treatment, and to distinguish them from patients that should preferably be managed non-operatively.
Tumor size
Tumor size has been reported to be a relevant factor for the preoperative functional level, since the mass effect on the brain is higher with larger tumors [22]. Also in our cohort, functional dependent patients had larger tumors (Table 1). While there was no higher likelihood for mortality in patients with larger tumors (Table 2), multivariable regression analysis did show a higher likelihood for severe complications for each increase in tumor size category (OR 2.22, 95% CI 1.24–3.97, p = 0.007; see Supplementary Table 4). The effect size of this finding is moderate, probably as small but eloquent tumor or those difficult to dissect (e.g. skull-base) can lead to severe disability, whereas removing large tumors in non-eloquent areas is usually less challenging.
Patient age
Despite the current era of advanced micro-neurosurgery and sophisticated perioperative management, surgery for elderly patients with brain tumors still is associated with increased surgical morbidity and mortality [23, 24]. The effect is believed to be conveyed by the increasing prevalence of comorbidities with higher age [25, 26]. Our present results indicate no higher likelihood for mortality or severe complications across the age categories, paralleling recent observations of our group for various neurosurgical pathologies [27]. Other factors (such as dependency) appear more relevant, but detailed analysis of the exact association between age and both, mortality and complications is warranted to rule out a type-II error.
Strengths and limitations
The database combined two large populations, which reduces cultural bias on perception of functional dependency and produces results of high external validity. The variable definitions were unified in both centers. Simple but robust statistical methods using a solid primary endpoint were applied to test a pre-defined hypothesis, and the number of statistical tests was kept at a minimum in order to prevent type-I errors.
There are limitations of this study. First, as this was not a randomized trial, the answer whether or not dependent patients should be operated cannot be answered, whereas the study provides estimates for the postoperative outcome that can be expected in a contemporary series. Second, the studied cohort was heterogeneous, especially concerning the histopathological diagnostic. Third, as postoperative functional recovery may take weeks/months but was available for the short-term only, questions concerning the mid- or long-term course remain unanswered. Also, we are unable to estimate the effects of surgery independently from steroid application, in-patient physio- or occupational therapy that are known to influence the outcome [28]. Regarding the KPS, median improvement was higher in the dependent compared to the independent group. As for this secondary outcome, both the dependent and independent variable was the KPS, interpretation needs to be made carefully.
Conclusion
In operated patients with intracranial tumors presenting functionally dependent at admission, the risk for in-hospital mortality and complications is higher. However, if conducted successfully, surgery may lead to regain of independence in every second patient within 3 months postoperative.
References
Metcalfe SE, Grant R (2001) Biopsy versus resection for malignant glioma. Cochrane Database Syst Rev. https://doi.org/10.1002/14651858.CD002034
Jakola AS, Sagberg LM, Gulati S, Solheim O (2015) Perioperative quality of life in functionally dependent glioblastoma patients: a prospective study. Br J Neurosurg 29(6):843–849. https://doi.org/10.3109/02688697.2015.1054355
Kilic A, Beaty CA, Merlo CA, Conte JV, Shah AS (2013) Functional status is highly predictive of outcomes after redo lung transplantation: an analysis of 390 cases in the modern era. Ann Thorac Surg 96(5):1804–1811. https://doi.org/10.1016/j.athoracsur.2013.05.080 (discussion 1811)
Crawford RS, Cambria RP, Abularrage CJ, Conrad MF, Lancaster RT, Watkins MT, LaMuraglia GM (2010) Preoperative functional status predicts perioperative outcomes after infrainguinal bypass surgery. J Vasc Surg 51(2):351–358. https://doi.org/10.1016/j.jvs.2009.08.065 (discussion 358–359)
de la Fuente SG, Bennett KM, Scarborough JE (2013) Functional status determines postoperative outcomes in elderly patients undergoing hepatic resections. J Surg Oncol 107(8):865–870. https://doi.org/10.1002/jso.23335
Ferroli P, Broggi M, Schiavolin S, Acerbi F, Bettamio V, Caldiroli D, Cusin A, La Corte E, Leonardi M, Raggi A, Schiariti M, Visintini S, Franzini A, Broggi G (2015) Predicting functional impairment in brain tumor surgery: the Big Five and the Milan Complexity Scale. Neurosurg Focus 39(6):E14. https://doi.org/10.3171/2015.9.FOCUS15339
Sarnthein J, Stieglitz L, Clavien PA, Regli L (2016) A patient registry to improve patient safety: recording general neurosurgery complications. PLoS ONE 11(9):e0163154. https://doi.org/10.1371/journal.pone.0163154
Neidert MC, Hostettler IC, Burkhardt JK, Mohme M, Held U, Kofmehl R, Eisele G, Woernle CM, Regli L, Bozinov O (2016) The influence of intraoperative resection control modalities on survival following gross total resection of glioblastoma. Neurosurg Rev 39(3):401–409. https://doi.org/10.1007/s10143-015-0698-z
Karnofsky D, Burchenal J (1949) The clinical evaluation of chemotherapeutic agents in cancer. Columbia University Press, New York
Dindo D, Demartines N, Clavien PA (2004) Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240(2):205–213
Chambless LB, Kistka HM, Parker SL, Hassam-Malani L, McGirt MJ, Thompson RC (2015) The relative value of postoperative versus preoperative Karnofsky Performance Scale scores as a predictor of survival after surgical resection of glioblastoma multiforme. J Neuro-oncol 121(2):359–364. https://doi.org/10.1007/s11060-014-1640-x
Angelo K, Dalhaug A, Pawinski A, Haukland E, Nieder C (2014) Survival prediction score: a simple but age-dependent method predicting prognosis in patients undergoing palliative radiotherapy. ISRN Oncol 2014:912865. https://doi.org/10.1155/2014/912865
Paesmans M, Sculier JP, Lecomte J, Thiriaux J, Libert P, Sergysels R, Bureau G, Dabouis G, Van Cutsem O, Mommen P, Ninane V, Klastersky J (2000) Prognostic factors for patients with small cell lung carcinoma: analysis of a series of 763 patients included in 4 consecutive prospective trials with a minimum follow-up of 5 years. Cancer 89(3):523–533
Kreth FW, Thon N, Simon M, Westphal M, Schackert G, Nikkhah G, Hentschel B, Reifenberger G, Pietsch T, Weller M, Tonn JC, German Glioma N (2013) Gross total but not incomplete resection of glioblastoma prolongs survival in the era of radiochemotherapy. Ann Oncol 24(12):3117–3123. https://doi.org/10.1093/annonc/mdt388
Marko NF, Weil RJ, Schroeder JL, Lang FF, Suki D, Sawaya RE (2014) Extent of resection of glioblastoma revisited: personalized survival modeling facilitates more accurate survival prediction and supports a maximum-safe-resection approach to surgery. J Clin Oncol 32(8):774–782. https://doi.org/10.1200/JCO.2013.51.8886
Stummer W, Pichlmeier U, Meinel T, Wiestler OD, Zanella F, Reulen HJ, Group AL-GS (2006) Fluorescence-guided surgery with 5-aminolevulinic acid for resection of malignant glioma: a randomised controlled multicentre phase III trial. Lancet Oncol 7(5):392–401. https://doi.org/10.1016/S1470-2045(06)70665-9
Wick W, Platten M, Meisner C, Felsberg J, Tabatabai G, Simon M, Nikkhah G, Papsdorf K, Steinbach JP, Sabel M, Combs SE, Vesper J, Braun C, Meixensberger J, Ketter R, Mayer-Steinacker R, Reifenberger G, Weller M, Society NOASGoN-oWGoGC (2012) Temozolomide chemotherapy alone versus radiotherapy alone for malignant astrocytoma in the elderly: the NOA-08 randomised, phase 3 trial. Lancet Oncol 13(7):707–715. https://doi.org/10.1016/S1470-2045(12)70164-X
Goldberg ID, Bloomer WD, Dawson DM (1982) Nervous system toxic effects of cancer therapy. JAMA 247(10):1437–1441
Warfel BS, Lachmann E, Nagler W (1993) Physiatric evaluation of the cancer patient. J Back Musculoskelet Rehabil 3(2):60–68. https://doi.org/10.3233/BMR-1993-3210
Kalkanis SN, Linskey ME (2010) Evidence-based clinical practice parameter guidelines for the treatment of patients with metastatic brain tumors: introduction. J Neuro-oncol 96(1):7–10. https://doi.org/10.1007/s11060-009-0065-4
Norden AD, Wen PY, Kesari S (2005) Brain metastases. Curr Opin Neurol 18(6):654–661
Prados MD, Berger MS, Wilson CB (1998) Primary central nervous system tumors: advances in knowledge and treatment. CA: Cancer J Clin 48(6):331–360, 321
Grossman R, Mukherjee D, Chang DC, Purtell M, Lim M, Brem H, Quinones-Hinojosa A (2011) Predictors of inpatient death and complications among postoperative elderly patients with metastatic brain tumors. Ann Surg Oncol 18(2):521–528. https://doi.org/10.1245/s10434-010-1299-2
Cornu P, Chatellier G, Dagreou F, Clemenceau S, Foncin JF, Rivierez M, Philippon J (1990) Intracranial meningiomas in elderly patients. Postoperative morbidity and mortality. Factors predictive of outcome. Acta Neurochir 102(3–4):98–102
Piccirillo JF, Vlahiotis A, Barrett LB, Flood KL, Spitznagel EL, Steyerberg EW (2008) The changing prevalence of comorbidity across the age spectrum. Critical Rev Oncol/Hematol 67(2):124–132. https://doi.org/10.1016/j.critrevonc.2008.01.013
Grossman R, Mukherjee D, Chang DC, Bennett R, Brem H, Olivi A, Quinones-Hinojosa A (2011) Preoperative charlson comorbidity score predicts postoperative outcomes among older intracranial meningioma patients. World Neurosurg 75(2):279–285. https://doi.org/10.1016/j.wneu.2010.09.003
Maldaner N, Sarnthein J, Bozinov O, Regli L, Neidert MC (2017) Neurosurgery in octogenarians: a prospective study of perioperative morbidity, mortality, and complications in elderly patients. World Neurosurg. https://doi.org/10.1016/j.wneu.2017.10.154
Marciniak CM, Sliwa JA, Spill G, Heinemann AW, Semik PE (1996) Functional outcome following rehabilitation of the cancer patient. Arch Phys Med Rehabil 77(1):54–57
Acknowledgements
The authors thank all resident and faculty neurosurgeons who entered and validated the patient data in the institutional patient registries that were basis of this research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Stienen, M.N., Zhang, D.Y., Broggi, M. et al. The influence of preoperative dependency on mortality, functional recovery and complications after microsurgical resection of intracranial tumors. J Neurooncol 139, 441–448 (2018). https://doi.org/10.1007/s11060-018-2882-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-018-2882-9