Abstract
Case reports and animal models suggest that chemotherapy, corticosteroids and radiotherapy (RT) may increase the risk of herpes simplex encephalitis (HSE). We retrospectively examined cases of HSE at an academic hospital devoted to cancer care. Patients were identified by positive herpes simplex virus (HSV) polymerase chain reaction (PCR) in cerebrospinal fluid (CSF) or by brain pathology. There were seven patients with HSE over a 12 year period, four of whom had received cranial RT. During this time, a total of 997 patients were treated with cranial RT, suggesting a greater incidence than the expected risk of two to four cases per million people per year in the general population. Five patients had recently received chemotherapy and three were on dexamethasone. MRI findings were typical; four patients had bilateral anterior temporal lesions and three had unilateral-temporal lesions. Four patients had a normal CSF white blood cell count, three of whom had prior RT and dexamethasone. Four patients were positive for HSV-1, and two for HSV-2. One patient had a negative CSF PCR for HSV, but autopsy confirmed active HSE. Though still rare, the risk of HSE may be increased in patients with cancer, especially in those receiving cranial RT. MRI findings were typical, but CSF white blood cell count was normal in four patients and one had negative CSF testing, suggesting that CSF results may be misleading in this population.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Herpes simplex encephalitis (HSE) is the most common identifiable cause of viral encephalitis, occurring in two to four patients per million people annually in the general population as consistently reported from multiple studies [1–3]. It is unclear whether immunosuppression is a risk factor for HSE, and immunosuppressive chemotherapies and corticosteroids are used in many cancer patients. HSE has been reported previously in individual patients with cancer, with a majority of reported episodes occurring shortly after radiotherapy (RT) to the brain [4–8]. One autopsy study of 78 bone marrow transplant recipients identified two with HSE [7]. However, estimates of the incidence of HSE in people with cancer compared to the general population are not available. Thus, we performed a review of patients diagnosed with HSE at Memorial Sloan-Kettering Cancer Center (MSKCC).
Methods
We conducted an Internal Review Board approved retrospective study using an institutional database of all CSF test results for herpes simplex virus (HSV) by polymerase chain reaction (PCR) from the period between January 1, 1998 and October 31, 2009. The medical records of patients with CSF results positive for HSV were reviewed and data collected for clinical details. One additional case found at autopsy during this time period was also included.
Results
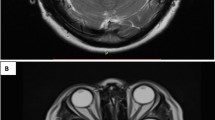
We identified seven patients with HSE, five of whom were women (Table 1). Their ages ranged from 45 to 76 (median 55) years old. Two patients had breast cancer and one patient each had primary CNS lymphoma (PCNSL), non-small-cell lung cancer, melanoma, renal and thyroid cancer. Five patients had received chemotherapy within 3 months of their HSE. Three patients were taking dexamethasone at the time of their HSE. Four patients had brain metastases and received whole brain RT within 3 months of their HSE; one patient had oral ulcers possibly due to mucosal HSV at the end of RT 3 weeks prior to the development of HSE. The patient with PCNSL had not received cranial RT. In all patients, MRI findings typical of temporal lobe encephalitis prompted CSF studies and acyclovir therapy. On MRI FLAIR sequences, four patients had bilateral anterior temporal lesions and three had unilateral temporal lesions. No lesion was enhancing, and these abnormalities could be distinguished clearly from intracranial tumor. All seven patients underwent lumbar puncture; CSF WBC ranged from 1 to 349 Cells/mcL (median 6), and four patients had a normal CSF white blood cell count, three of whom had prior RT and dexamethasone. Systemic white blood cell counts (median 6,400 Cells/mcL, range 4,500–10,900 Cells/mcL) and absolute neutrophil counts (median 5,700 Cells/mcL, range 3,300–9,900 Cells/mcL) were normal on admission. Absolute lymphocyte counts were in the normal range for the majority of patients, even those on chemotherapy (median 500 Cells/mcL, range 400–1,500 Cells/mcL, normal range is 500–5,300 Cells/mcL). Six of seven had an elevated CSF protein concentration. Four patients had CSF positive for HSV-1, two had CSF positive for HSV-2. One patient had negative CSF PCR for HSV, but typical MRI findings and autopsy confirmed active HSE. In the autopsy material there was a paucity of inflammation but viral inclusions staining for HSV were found, in accordance with previous reported autopsy findings in immunocompromised patients with HSE [4].
The duration of patient symptoms prior to acyclovir therapy ranged from 1 to 3 days (mean 2.4 days). One patient (case 6) with HSV-2 encephalitis also had multiple subcortical infarcts and beading of cerebral blood vessels on imaging, suggesting cerebral vasculitis from HSV in addition to HSE. Case 6 also had simultaneous left C6/7 dermatomal vesicular skin lesions that tested positive for HSV. Five patients died without significant improvement a median of 59 days from presentation (range 7–74). One patient recovered with mild residual deficits, but died of her breast cancer 430 days later. One patient was sent to hospice due to declining condition 59 days later, and no further information was available.
During the same 12 year period, 997 patients received RT to the brain at MSKCC, four of whom developed HSE, giving a risk of approximately 1 in 250 patients treated with cranial irradiation. This apparent risk is significantly greater than the expected annual incidence of two to four patients with HSE per million people annually or the 1.3–3.1 cases of encephalitis of any cause per 1,00,000 people annually between 2000 and 2008 in New York City [1, 9]. A firm denominator for our overall cancer population in this time period is not available, since many patients are seen for single consultations or receive only partial but not ongoing care at our institution.
Discussion
This retrospective study suggests an increased incidence of HSE in patients with cancer, particularly those receiving RT to the brain. Functional immunosuppression from chemotherapy or corticosteroids may also be contributing factors, but steroids could have affected only three of our seven patients. The 0.4% incidence we observed among patients receiving cranial irradiation far exceeds the expected incidence reported from large, population-based studies [1]. We found 34 cases of HSE in people with cancer reported in the literature. Information on brain radiation was provided in 23 of these patients and 15 had received cranial RT within 3 months of their HSE (summarized in Table 2), lending support to our observation that RT may increase susceptibility to HSE. The immunosuppressant effects of dexamethasone or other therapies used together with or in proximity to brain RT or RT itself may induce local effects on immune control of latent HSV in the CNS or impaired defense against reinfection. However, CNS RT alone does not explain all the increased vulnerability to HSE in our population as three of our patients did not receive cranial irradiation; however, all three had received chemotherapy or dexamethasone within 3 months of HSE, either of which may be sufficient to disrupt host immunity.
MRI abnormalities were typical of HSE in our patients; imaging provided the key to diagnosis, as some other features of their HSE were atypical. However, the MRI findings triggered the search for HSE, so patients with atypical neuroimaging features and HSE may not have been identified. CSF pleocytosis was absent in more than half our patients as has been observed previously in this population, and did not correlate with current dexamethasone use [5]. A number of features of HSE were atypical in our cohort as well as cases reported in the literature, including absence of CSF pleocytosis and a possibly higher incidence of HSV-2 as the causative agent. Six of twelve cancer patients with HSE and CSF results reported in the literature also had a normal CSF cell count (Table 2). CSF PCR for HSV was negative in one of our patients and inflammatory infiltrates were absent at autopsy in this patient. Furthermore, HSV-2 normally causes only 6% of HSE, but was seen in two of our seven patients and three of the twelve cases reported in the literature that described HSV type [1].
Systemic administration of cyclophosphamide and dexamethasone to rabbits latently infected with HSV produced active CNS HSV infection in the temporal lobes, suggesting that immunosuppression without RT may increase risk of HSE [10]. In a prospective study of infectious encephalitis due to any cause, co-existing cancer was a significant predictor of fatal outcome [2]. Oral HSV lesions can develop during RT to the head and neck, and may have occurred in one of our patients, although the viral etiology of that patient’s oral ulcers was not confirmed [11]. HSE has also been reported rarely following craniotomy; however, it occurred within days of neurosurgery in all reported cases and peri-operative dexamethasone use may have contributed [12]. Although five of our patients had intracranial neoplasms (four brain metastases), none had a recent craniotomy. Toll-like receptor activation of the innate immune system is important in the CNS immune response to HSV as proven by increased susceptibility to HSE in people with genetic defects in toll-like receptor-3 pathways, and is potently impaired by corticosteroids [13–15]. Animal models suggest that radiation does not significantly alter toll-like receptor signaling and may in fact activate the innate immune system [16, 17]. Three of our patients were known to be taking glucocorticoids at the time of HSE. There are conflicting data from animal models suggesting that dexamethasone and corticosterone may be beneficial or harmful in HSE depending on the timing of administration, and a large trial using dexamethasone together with acyclovir for acute HSE is currently underway [10, 13, 18].
The retrospective nature of our study and small number of patients are major limitations. However, the similarity between our observations and individual cases reported in the literature supports our hypothesis. Our study may be an underestimate of HSE, as it only included patients diagnosed with HSE at our institution. Patients treated at our institution, but who may have been hospitalized elsewhere for HSE, would not have been included in our report. We would have also missed patients who did not undergo CSF testing or pathologic examination for HSV.
The atypical clinical features we observed suggest that patients with cancer who develop altered mental status or seizures with or without fever and have new temporal lobe abnormalities on FLAIR MRI should undergo lumbar puncture for HSV PCR and be started on acyclovir until testing is definitive, even if CSF pleocytosis is absent. In patients with typical clinical and radiographic features, treatment may be continued even if the CSF PCR result is negative as a false negative result was observed in one of our patients. While most of our patients died within 2 months of their HSE despite treatment, one patient (who had no intracranial tumor, never received cranial RT and was not taking glucocorticoids) survived more than a year with modest residual neurologic dysfunction, succumbing finally to her systemic cancer, suggesting that treatment of HSE may still be effective in this population. Acyclovir-resistant HSV may also be a consideration in these patients as acyclovir resistance is more common in immunosuppressed and bone marrow transplant patients (14% in one study) [19]. Vigilance for this diagnosis and early institution of therapy may help improve outcome.
References
Hjalmarsson A, Blomqvist P, Skoldenberg B (2007) Herpes simplex encephalitis in Sweden, 1990–2001: incidence, morbidity and mortality. Clin Infect Dis 45:875–880
Mailles A, Stahl JP (2009) Infectious encephalitis in France in 2007: a national prospective study. Clin Infect Dis 49:1838–1847
Puchhammer-Stockl E, Presterl E, Croy C et al (2001) Screening for possible failure of herpes simplex PCR in cerebrospinal fluid for the diagnosis of herpes simplex encephalitis. J Med Virol 64:531–536
Schiff D, Rosenblum MK (1998) Herpes simplex encephalitis (HSE) and the immunocompromised. Hum Pathol 29:215–222
Jacobs DH (1999) Herpes simplex virus encephalitis following corticosteroids and cranial irradiation. Neurology 52:1108–1109
Spacca B, Mallucci C, Riordan A et al (2007) HSV encephalitis in a child with brain stem glioma: a rare complication of therapy: case report and review of the neurosurgical literature. Childs Nerv Syst 23:1347–1350
Patchell RA, White CL, Clark AW et al (1985) Neurologic complications of bone marrow transplantation. Neurology 35:300–306
Kocher M, Kunze S, Eich HT et al (2005) Efficacy and toxicity of postoperative temozolomide radiochemotherapy in malignant glioma. Strahlenther Onkol 181:157–163
https://a816healthpsi.nyc.gov/epiquery/EpiQuery/CDSS/index.html. Accessed 14 Feb 2011
Stroop WG, Schaefer DC (1986) Production of encephalitis restricted to the temporal lobes by experimental reactivation of herpes simplex virus. J Inf Dis 153:721–731
Oakley C, Epstein JB, Sherlock CH (1997) Reactivation of oral herpes simplex virus: implications for clinical management of herpes simplex virus recurrence during radiotherapy. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 84:272–278
Aldea S, Joly LM, Roujeau T et al (2003) Postoperative herpes simplex virus encephalitis after neurosurgery: case report and review of the literature. Clin Infect Dis 36:e96–e99
Sergerie Y, Boivin G, Gosselin D et al (2007) Delayed but not early glucocorticoid treatment protects the host during experimental herpes simplex virus encephalitis in mice. J Infect Dis 195:817–825
Sancho-Shimizu V, Zhang SY, Abel L, Tardieu M, Rozenberg F, Jouanguy E, Casanova JL (2008) Genetic susceptibility to herpes simplex virus 1 encephalitis in mice and humans. Curr Opin Allergy Clin Immunol 7:495–505
McCoy CE, Carpenter S, Palsson-McDermott EM, Gearing LJ, O’Neill LAJ (2008) Glucocorticoids inhibit IRF3 phosphorylation in response to toll-like receptor-3 and -4 by targeting TBK1 activation. J Biol Chem 283:14277–14285
Turrin NP, Plante M-M, Lessard M, Rivest S (2007) Irradiation does not compromise or exacerbate the innate immune response in the brains of mice that were transplanted with bone marrow stem cells. Stem Cells 25:3165–3172
Roses RE, Xu M, Koski GK, Czerniecki BJ (2008) Radiation therapy and toll-like receptor signaling: implications for the treatment of cancer. Oncogene 27:200–207
Martinez-Torres F, Menon S, Pristch M et al (2008) Protocol for German trial of acyclovir and corticosteroids in Herpes-Simplex-Encephalitis (GACHE): a multicenter, multinational, randomized, double-blind, placebo-controlled German, Austrian and Dutch trial. BMC Neurol 8:40
Stranska R, Schuurman R, Nienhuis E et al (2005) Survey of acyclovir-resistant herpes simplex virus in the Netherlands: prevalence and characterization. J Clin Virol 32:7–18
Silvano G, Lzazzari G, Resta F et al (2007) A herpes simplex virus-1 fatal encephalitis following chemo-radiotherapy, steroids and prophylactic cranial irradiation in a small cell lung cancer patient. Lung Cancer 57:243–246
Peng T, Blakely J, Cingolani E et al (2007) Herpes simplex encephalitis in a patient with recurrent pituitary adenoma receiving radiation therapy. Am J Clin Oncol 30:664–665
Manz HJ, Phillips TM, McCullough DC (1979) Herpes simplex type 2 encephalitis concurrent with known cerebral metastases. Acta Neuropathol 47:237–240
Suzuki HI, Hangaishi A, Hosoya N et al (2008) Herpes simplex encephalitis and subsequent cytomegalovirus encephalitis after chemoradiotherapy for central nervous system lymphoma. Int J Hematol 87:538–541
Tohyama Y, Sako K, Daita G et al (1997) Dissociation of 99mTc-ECD and 99mTc-HMPAO distributions in herpes simplex encephalitis. Childs Nerv Syst 13:352–355
Molloy S, Allcutt D, Brennan P, Farrell MA, Perryman R, Brett FM (2000) Herpes simplex encephalitis occurring after chemotherapy, surgery, and stereotactic radiotherapy for medulloblastoma. Arch Pathol Lab Med 124:1809–1812
Riel-Romero RM, Baumann RJ (2003) Herpes simplex encephalitis and radiotherapy. Pediatr Neurol 29:69–71
Koudriavtseva T, Onesti E, Tonachella R et al (2010) Fatal herpetic encephalitis during brain radiotherapy in a cerebral metastasized breast cancer patient. J Neurooncol 100:137–140
Dragoje S, Tolnay M, Dalquen P et al (1995) Brain irradiation and antioedematous dexamethasone treatment-risk factors for herpes simplex encephalitis. Schweiz Arch Neurol Psychiatr 146:277–280
Saran F, Baumert BG, Creak AL et al (2008) Hypofractionated stereotactic radiotherapy in the management of recurrent or residual medulloblastoma/PNET. Pediatr Blood Cancer 50:554–560
Cathomas E, Pelosi E, Smart J (2005) Herpes simplex encephalitis as a complication of adjuvant chemotherapy treatment for breast cancer. Clin Oncol (R Coll Radiol) 17:292–293
Nolan RC, Van Gessel H, Byrne M (2004) An unusual complication of chemotherapy: herpes simplex meningoencephalitis and bilateral acute retinal necrosis. Clin Oncol 16:81–82
Graber JJ, Rus H, Zivin-Tutela T et al (2009) Herpes encephalitis in monoclonal gammopathy. Infect Med 26(3):78–81
Rommee R, Brunstein CG, Wiesdorf DJ et al (2010) Herpes simplex virus encephalitis after allogeneic transplantation: an instructive case. Bone Marrow Transpl 45:776–778
McDermott SS, McDermott PF, Skare J et al (2000) Positive CSF HSV PCR in patients with GBM: a note of caution. Neurology 54:746–749
Calderoni A, Aebi S (2002) Combination chemotherapy with high-dose methotrexate and cytarabine with or without brain irradiation for primary central nervous system lymphomas. J Neurooncol 59:227–230
Schütt P, Ebeling P, Buttkereit U et al (2005) Thalidomide in combination with dexamethasone for pretreated patients with multiple myeloma: serum level of soluble interleukin-2 receptor as a predictive factor for response rate and for survival. Ann Hematol 84:594–600
Klingelhöfer J, Prange HW, Schipper HI (1984) Herpes simplex encephalitis and plasmacytoma: chance coincidence or causal relation (in German). Nervenarzt 55:86–89
Chim CS, Ho PL, Yuen KY et al (1999) Herpes simplex encephalitis in multiple myeloma. Aust N Z J Med 29:381–382
Schmidt-Hieber M, Schwender J, Heinz WJ et al (2011) Viral encephalitis after allogeneic stem cell transplantation: a rare complication with distinct characteristics of causative agents. Haematalogica 96:142–149
Acknowledgments
The authors would like to thank Galina Yusim for her able assistance with data collection for this project, Dr. Peter Mead for his helpful advice and Judith Lampron for her tireless editorial expertise.
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Graber, J.J., Rosenblum, M.K. & DeAngelis, L.M. Herpes simplex encephalitis in patients with cancer. J Neurooncol 105, 415–421 (2011). https://doi.org/10.1007/s11060-011-0609-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-011-0609-2