Abstract
Cranial chordomas are rare tumors that have been difficult to study given their low prevalence. Individual case series with decades of data collection provide some insight into the pathobiology of this tumor and its responses to treatment. This meta-analysis is an attempt to aggregate the sum experiences and present a comprehensive review of their findings. We performed a comprehensive review of studies published in English language literature and found a total of over 2,000 patients treated for cranial chordoma. Patient information was then extracted from each paper and aggregated into a comprehensive database. The tumor recurrences in these patients were then stratified according to age (<21 vs. >21 years), histological findings (chondroid vs. typical) and treatment (surgery and radiation vs. surgery only). Data was analyzed via Pearson chi-square and t-test. A total of 464 non-duplicated patients from 121 articles treated for cranial chordoma met the inclusion criteria. The recurrence rate among all patients was 68% (314 patients) with an average disease-free interval of 45 months (median, 23 months). The mean follow-up time was 39 months (median, 27 months). The patients in younger group, patients with chordoma with chondroid histologic type, and patients who received surgery and adjuvant radiotherapy had significantly lower recurrence rate than their respective counterparts. The results of our systematic analysis provide useful data for practitioners in objectively summarizing the tumor recurrence in patients with cranial chordomas. Our data suggests that younger patients with chondroid type cranial chordoma treated with both surgery and radiation may have improved rates of tumor recurrence in the treatment of these tumors.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Chordoma is an extra-axial tumor that is assumed to develop from the remnants of the notochord [1]. This midline neoplasm has been observed intracranially (35%), in the mobile segments of the spine (15%), but has most commonly been reported in the sacrum (50%) [2–5]. Skull based chordomas are almost always associated with the clivus [6–8]. Chordoma typically presents with lower cranial nerve palsies due to direct compression of the surrounding neural structures. Local invasion of the mass results in high morbidity and mortality as a result of direct brainstem compression. Given their anatomical location and local aggressiveness, cranial chordomas remain problematic for neurosurgeons. Currently, the mainstay of therapy is surgical resection combined with high dose radiation therapy. Despite combined therapy, the recurrence rate remains quite high even with gross total resection. A large volume of literature has been published describing factors influencing the prognosis of patients with cranial chordomas. Much of the data regarding outcomes following treatment has been reported by small case series and case reports which lack statistical power to derive significant conclusions about appropriate management of these tumors. Several factors are thought to be important in the recurrence of chordomas, including extent of resection, adjuvant radiation therapy as well as histologic subtype. In this analysis we compiled a comprehensive systematic analysis of the English language literature to evaluate the factors associated with recurrence in cranial chordomas.
Methodology
Article selection
Articles were identified via a Pub Med search using the key words “cranial chordoma,” “clival chordoma,” “skull base chordoma,” alone and in combination with “recurrence” as Boolean searches. We then searched all the references in these papers.
Inclusion criteria were: (1) All articles had to specifically follow patients for recurrence (2) Articles had to have enough information of presentations, treatment and follow-up for each patient in order to be disaggregated.
Pathological assessment
Chordoma and its chondroid subtype are analyzed in this analysis. In reports that present patient data gathered before the advent of chondroid subtype, all references to the term “chordoma” are assumed to be considered the standard subtype designation. In more recent studies where the chondroid designation was used, pathological confirmation was evaluated but was not always observed. In these cases we assumed that the use of the “chondroid” terminology was based upon pathological confirmation. Data regarding the “dedifferentiated” subtype of chordoma were too scarce and sporadically mentioned to extract this data subset reliably.
Data extraction
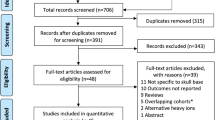
Our search resulted in over 2,000 patients treated for cranial chordoma. Of this compiled data set, 760 patients had sufficient date to be disaggregated. Of these 760 patients, 464 of them were followed up for recurrence. All these patients were disaggregated and all references for these articles further scrutinized to ensure the non-duplication of patients and the completion of a thorough and comprehensive review of the literature. In those cases where patients had more than one treatment for recurrence only the initial treatment was considered. In cases where histology was confirmed without any specifications we assume the histology was characteristic of typical chordoma. The age recorded for patients with recurrence, was age of first presentation. All cases that did not have any follow-up data (and no mortality noted) were all excluded. Article data sets that were thought to be incomplete, or that were unable to be disaggregated due to averaged data sets were eliminated from our data sets. Data were analyzed as a whole and stratified into 3 subgroups. The first subgroup divided the data according to patient age with a cut-off age of 21 years. A second stratification divided data based on histological features, group one included those chordomas with typical or conventional histology and group two were those patients with chondroid chordomas (chordomas with prominent chondroid features). The final subgroup analysis stratified the data according to treatment: one group comprised of those patients who received surgery alone as their initial treatment and the second group included those patients who received surgery with adjuvant radiation.
Statistical analysis
Pearson Chi-squared test was used for statistical evaluation of the data. A P value of less than 0.05 (5%) was considered to be significant.
Results
Results of the systematic analysis
A total of 121 articles with 464 non-duplicated chordoma patients met the inclusion criteria for this systematic analysis [9–54, 55–94, 95–129]. The recurrence rate among these patients was 68% (314 patients) with an average disease-free interval of 45 months (median, 23 months).
Clinical variables on recurrence
A total of 53 patients were younger than 21 years of age at the initial time of presentation, while 348 patients were older than 21 years. Surprisingly, the recurrence rate was higher for patients who were older than 21 years old of age, when compared to the younger patients [74% vs. 60%, P = 0.04 (Fig. 1)]. Hence younger patients indicated an association with a lower rate of recurrence. A total of 429 patients in our analysis had chordomas with typical histology, while 32 patients had a histological confirmation of chondroid chordoma. The recurrence rate was higher for those patients with chordomas of typical histology [77% vs. 38%, P < 0.0001 (Fig. 2)]. Patients with the rarer chondroid chordoma histopathology were associated with an improved rate of recurrence.
The effect treatment on recurrence
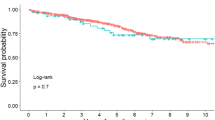
A total of 226 patients had surgery alone, while 128 patients had adjuvant radiation in addition to surgery. The recurrence rate was higher in the group of patients that had surgery alone [79% vs. 60%, P < 0.0001 (Fig. 3)]. This statistically significant difference confirms the importance in utilizing a radiation modality in the treatment of cranial chordoma. In our data analysis, more patients were treated with surgery alone despite this lower rate of recurrence associated with adjuvant radiation.
Discussion
Local recurrence is considered by many to be the most significant predictor of mortality in patients suffering from cranial chordomas. In this study we performed a systematic analysis of recurrence in a large population of patients who have undergone treatment for skull base chordomas.
The over-all recurrence rate in our meta-analysis was 68%. This was a little higher than those quoted in the literature by larger studies. For example, one chordoma study of 60 patients with a median follow-up time of 1.9–30 years had a recurrence ranging from 12 to 60% [130]. The average survival time in our study for treated chordoma patients was 3.3 years. This was similar to the survival times reported in the literature after surgery or radiation therapy, or both that ranges from 3.6 to 6.6 years [5, 101, 131]. The disease-free interval in our study, on average was 45 months, with several cases recurring as long as 18 years after the initial treatment [116]. The review of literature reported very similar figures with a 2–3 years time period to recurrence after initial treatment and a few cases of recurrence appearing greater than 10 years after the primary treatment [12].
Chondroid chordomas
The chondroid chordoma subtype diagnosis is a recent addition to the accepted pathological classification system. Given its even more rare incidence, little is known about its pathology in comparison to the standard subtype. One interesting finding in our analysis is that chondroid chordoma was much less likely to recur than chordoma of typical histology. Heffelfinger et al. [132] proposed in 1973, a more benign clinical course for the variant chondroid of the chordoma that he himself had defined. Nonetheless, in 1993 Mitchell et al. [133] refuted the fact that there was a difference in the clinical course between the chondroid and the typical histological variants and attributed this apparent difference to the age of the patient at the time of presentation. Authors stated that it was the high prevalence of chondroid chordoma in younger patients that accounted for the more favorable prognosis and not merely the histology itself. However, our study does not support Mitchell and agrees with the initial observations reported by Heffelfinger. This is demonstrated in the fact that the age range of patients in our analysis with chondroid chordoma ranges from 4 to 72 years and yet still the over-all recurrence rate in the chondroid patients were lower as a group when compared to the group of patients that presented with the conventional chordoma histology. Our data suggests that the biological behavior for these histological variants is different and the chondroid variant has a more benign clinicopathological course.
Out of a total of 461 patients in our analysis, 429 patients were identified as having typical histology, whereas 32 patients (7.4%) had histological confirmation of the chondroid chordoma subtype. Obviously data gathered from patients prior to the 1980s would not reflect this histopathologic subtype. This prevalence rate observed in this multi-decade meta-analysis is similar to our most recent 10 year institutional experience (unpublished data). At our institution from the years of 1989 to 2008, there were approximately 100 histological confirmed cases of skull base chordomas. In the most recent 10 years (1999–2008) in which the diagnosis of chondroid chordoma was an accepted available diagnosis, there were 5 confirmed cases of chondroid chordomas out of a total of 51 newly diagnosed chordoma tumors (9.8%). In a tertiary institution in which neurosurgical tumors are commonly identified and treated, 1 chondroid chordoma is diagnosed on average every 2 years.
Given the recent acceptance of the chondroid subtype as a distinct pathological variant, it is impossible to cleanly define a prevalence and recurrence rate for this tumor. Data gathered in this meta-analysis covers several decades of case reports and institutional series. The chondroid variant has been an arguably accepted identifiable subtype in the past 1–2 decades. Consequently, it is likely that typical chordoma numbers are overestimated in the early years of reporting the occurrence rates. Recent data may be a more accurate assessment of this tumors prevalence and prognostic significance. Only a multi-institutional prospective analysis will be able to address these questions.
It has been suggested that age is a significant prognostic factor in patients suffering from skull base chordomas [20, 134, 135]. Many reporters describe a more aggressive behavior for chordomas in children highlighting the hypercellularity, pleomorphism and high levels of mitotic activity seen in this age group [134]. Moreover, Borda et al. [20] reported that the prognosis is worse in these younger patients because of the extremely diverse and malignant pathological appearance of these tumors observed in children and adolescents. Our analysis does not support these observations and reveals that the recurrence rate was lower in the younger patients younger than 21 years of age. The reason for this observation is unclear but a few authors have previously postulated a worse prognosis for the older population when compared to patients younger than 40 years of age [6]. In our study, both major histological variants (typical and chondroid) as well as the two major treatment modalities (surgery, surgery and radiation) were similarly represented in both groups of patients.
Patients who had surgery and adjuvant radiation treatment had lower recurrence rates than those patients who had surgery alone as several observers have reported increasingly high recurrence rates in surgical patients treated for cranial chordoma, even after radical resection [136]. Although our 60% recurrence rate for patients treated with the combination modality of both surgery and radiation was actually higher than those quoted in the literature of 15–32% [32, 137–142] we strongly believe that cautious resection followed by post-operative proton-beam radiotherapy epitomizes the best therapeutic approach. Proton-beam therapy is thought to be a more effective means of radiation therapy as it gives a more concentrated dose to the tumor bed without much spread to surrounding tissues. The narrow beam of proton-beam therapy allows significant doses to be given to areas of the skull base without toxic doses spreading to nearby brainstem structures. Furthermore, although adjuvant radiation is associated with a lower rate of recurrence for cranial chordoma, most patients in this analytical series were treated with surgery only instead of surgery with radiation. Our data suggests that radiation therapy should be seriously considered as an adjuvant treatment for cranial chordomas.
Conclusion
In conclusion, we report our results from a large disaggregated systematic analysis of the English language literature regarding recurrence among cranial chordoma patients. We hope that by using such a large data set, we are able to summate the aggregated data set and identify recurring characteristics in the pathobiology of this tumor and its response to treatment. Our data confirms that adjuvant radiation is an important factor in a lower rate of recurrence and also suggests that younger patients with a chondroid subtype may have a lower rate of recurrence.
References
Colli BO, Al-Mefty O (2001) Chordomas of the skull base: follow-up review and prognostic factors. Neurosurg Focus 10:E1
Bergh P, Kindblom LG, Gunterberg B, Remotti F, Ryd W, Meis-Kindblom JM (2000) Prognostic factors in chordoma of the sacrum and mobile spine: a study of 39 patients. Cancer 88:2122–2134
Kaiser TE, Pritchard DJ, Unni KK (1984) Clinicopathologic study of sacrococcygeal chordoma. Cancer 53:2574–2578
Miles WK, Chang DW, Kroll SS, Miller MJ, Langstein HN, Reece GP, Evans GR, Robb GL (2000) Reconstruction of large sacral defects following total sacrectomy. Plast Reconstr Surg 105:2387–2394
Rich TA, Schiller A, Suit HD, Mankin HJ (1985) Clinical and pathologic review of 48 cases of chordoma. Cancer 56:182–187
Forsyth PA, Cascino TL, Shaw EG, Scheithauer BW, O’Fallon JR, Dozier JC, Piepgras DG (1993) Intracranial chordomas: a clinicopathological and prognostic study of 51 cases. J Neurosurg 78:741–747
Laws EJ Cranial chordomas. In: Wilkins RHRS (ed) Neurosurgery. McGraw-Hill, New York, pp 927–929
Laws EJ Clivus chordomas. In: Sekhar LNJI (ed) Surgery of cranial base tumors. Raven Press, New York, pp 679–685
Allen KL, Kerr WA (1968) Chordomas of the clivus and the cervical spine. S Afr Med J 42:1165–1174
Alonso WA, Black P, Connor GH, Uematsu S (1971) Transoral transpalatal approach for resection of clival chordoma. Laryngoscope 81:1626–1631
Alvarado R, Gomez J, Morale SG, Davis CP (2004) Neck pain: common complaint, uncommon diagnosis—symptomatic clival chordoma. South Med J 97:83–86
Amendola BE, Amendola MA, Oliver E, McClatchey KD (1986) Chordoma: role of radiation therapy. Radiology 158:839–843
Arnautovic KI, Al-Mefty O (2001) Surgical seeding of chordomas. Neurosurg Focus 10:E7
Arnold H, Herrmann HD (1986) Skull base chordoma with cavernous sinus involvement. Partial or radical tumour-removal? Acta Neurochir (Wien) 83:31–37
Asano S, Kawahara N, Kirino T (2003) Intradural spinal seeding of a clival chordoma. Acta Neurochir (Wien) 145:599–603 (discussion 603)
Auger M, Raney B, Callender D, Eifel P, Ordonez NG (1994) Metastatic intracranial chordoma in a child with massive pulmonary tumor emboli. Pediatr Pathol 14:763–770
Bartal AD, Heilbronn YD (1970) Transcervical removal of a clivus chordoma in a 2-year-old child. Reversal of quadriplegia and bulbar paralysis. Acta Neurochir (Wien) 23:127–133
Berryhill BH, Armstrong BW (1984) Extracranial presentation of craniocervical chordoma. Laryngoscope 94:1063–1065
Blevins NH, Jackler RK, Kaplan MJ, Gutin PH (1995) Combined transpetrosal-subtemporal craniotomy for clival tumors with extension into the posterior fossa. Laryngoscope 105:975–982
Borba LA, Al-Mefty O, Mrak RE, Suen J (1996) Cranial chordomas in children and adolescents. J Neurosurg 84:584–591
Bowyer J, Natha S, Marsh I, Foy P (2003) Visual complications of proton beam therapy for clival chordoma. Eye 17:318–323
Brooks LJ, Afshani E, Hidalgo C, Fisher J (1981) Clivus chordoma with pulmonary metastases appearing as failure to thrive. Am J Dis Child 135:713–715
Canalis RF, Black K, Martin N, Becker D (1991) Extended retrolabyrinthine transtentorial approach to petroclival lesions. Laryngoscope 101:6–13
Chang SD, Martin DP, Lee E, Adler JR Jr (2001) Stereotactic radiosurgery and hypofractionated stereotactic radiotherapy for residual or recurrent cranial base and cervical chordomas. Neurosurg Focus 10:E5
Cloward RB, Passarelli P (1979) Removal of giant clival chordoma by an anterior cervical approach. Surg Neurol 11:129–134
Cocke EW Jr, Robertson JH, Robertson JT, Crook JP Jr (1990) The extended maxillotomy and subtotal maxillectomy for excision of skull base tumors. Arch Otolaryngol Head Neck Surg 116:92–104
Couldwell WT, Stillerman CB, Rice D, Maceri D, Sherman R, Fukushima T, Hinton DR (1996) Malignant clival chordoma with postoperative cutaneous metastases. Skull Base Surg 6:61–66
Crockard A, Macaulay E, Plowman PN (1999) Stereotactic radiosurgery. VI. Posterior displacement of the brainstem facilitates safer high dose radiosurgery for clival chordoma. Br J Neurosurg 13:65–70
Cummings BJ, Hodson DI, Bush RS (1983) Chordoma: the results of megavoltage radiation therapy. Int J Radiat Oncol Biol Phys 9:633–642
Danziger J, Allen KL, Bloch S (1974) Intracranial chordomas. Clin Radiol 25:309–316
De Grood MP, Moffie D (1964) Cranial chordoma. Acta Neurochir (Wien) 11:653–662
Debus J, Schulz-Ertner D, Schad L, Essig M, Rhein B, Thillmann CO, Wannenmacher M (2000) Stereotactic fractionated radiotherapy for chordomas and chondrosarcomas of the skull base. Int J Radiat Oncol Biol Phys 47:591–596
Delgado TE, Garrido E, Harwick RD (1981) Labiomandibular, transoral approach to chordomas in the clivus and upper cervical spine. Neurosurgery 8:675–679
DeMonte F, Diaz E Jr, Callender D, Suk I (2001) Transmandibular, circumglossal, retropharyngeal approach for chordomas of the clivus and upper cervical spine. Technical note. Neurosurg Focus 10:E10
Diaz-Gonzalez FJ, Padron A, Foncea AM, Garcia de Sola R, Naval L, Rubio P (1999) A new transfacial approach for lesions of the clivus and parapharyngeal space: the partial segmented Le Fort I osteotomy. Plast Reconstr Surg 103:955–959
Doglietto F, Sabatino G, Pallini R (2005) Clivus chordoma associated with a pontine arachnoid cyst. Lancet Oncol 6:536
Eisemann ML (1980) Sphenooccipital chordoma presenting as a nasopharyngeal mass. A case report. Ann Otol Rhinol Laryngol 89:271–275
Erbengi A, Tekkok IH, Acikgoz B (1991) Posterior fossa chordomas—with special reference to transoral surgery. Neurosurg Rev 14:23–28
Fichardt T, de Villiers PC (1974) Chordoma. S Afr Med J 48:383–391
Firooznia H, Pinto RS, Lin JP, Baruch HH, Zausner J (1976) Chordoma: radiologic evaluation of 20 cases. AJR Am J Roentgenol 127:797–805
Fisch U (1978) Infratemporal fossa approach to tumours of the temporal bone and base of the skull. J Laryngol Otol 92:949–967
Fischbein NJ, Kaplan MJ, Holliday RA, Dillon WP (2000) Recurrence of clival chordoma along the surgical pathway. AJNR Am J Neuroradiol 21:578–583
Fournier H, Mercier P (2000) A limited anterior petrosectomy with preoperative embolization of the inferior petrosal sinus for ventral brainstem tumor removal. Surg Neurol 54:10–17 (discussion 17–18)
Frank G, Sciarretta V, Calbucci F, Farneti G, Mazzatenta D, Pasquini E (2006) The endoscopic transnasal transsphenoidal approach for the treatment of cranial base chordomas and chondrosarcomas. Neurosurgery 59:ONS50–57 (discussion ONS50–57)
Freeman J (1950) Cranial chordoma. Arch Otolaryngol 51:237–244 (illust)
Frempong-Boadu AK, Faunce WA, Fessler RG (2002) Endoscopically assisted transoral-transpharyngeal approach to the craniovertebral junction. Neurosurgery 51:S60–S66
Gates GA, Sertl GO, Grubb RL, Wippold FJ II (1994) Closure of clival cerebrospinal fluid fistula with biocompatible osteoconductive polymer. Arch Otolaryngol Head Neck Surg 120:459–461
Gherardi G, Marveggio C, Cola C, Redaelli G (1994) Decisive role of immunocytochemistry in aspiration cytology of chordoma of the clivus: a case report with review of the literature. J Laryngol Otol 108:426–430
Goel A (1995) Chordoma and chondrosarcoma: relationship to the internal carotid artery. Acta Neurochir (Wien) 133:30–35
Goel A (1995) Middle fossa sub-Gasserian ganglion approach to clivus chordomas. Acta Neurochir (Wien) 136:212–216
Goel A, Nadkarni TD, Kirtane MV (1996) Clival chordoma in an infant. J Postgrad Med 42:51–53
Gonzalez-Martinez JA, Guthikonda M, Vellutini E, Zamorano L, Li Q, Kupski W, Diaz FG (2002) Intradural invasion of chordoma: two case reports. Skull Base 12:155–161
Guiot G, Rougerie J, Bouche J (1968) The rhinoseptal route for the removal of clivus chordomas. Johns Hopkins Med J 122:329–335
Handa J, Suzuki F, Nioka H, Koyama T (1987) Clivus chordoma in childhood. Surg Neurol 28:58–62
Hara T, Kawahara N, Tsuboi K, Shibahara J, Ushiku T, Kirino T (2006) Sarcomatous transformation of clival chordoma after charged-particle radiotherapy. Report of two cases. J Neurosurg 105:136–141
Harbour JW, Lawton MT, Criscuolo GR, Holliday MJ, Mattox DE, Long DM (1991) Clivus chordoma: a report of 12 recent cases and review of the literature. Skull Base Surg 1:200–206
Haridas A, Ansari S, Afshar F (2003) Chordoma presenting as pseudoprolactinoma. Br J Neurosurg 17:260–262
Harsh GRt, Sekhar LN (1992) The subtemporal, transcavernous, anterior transpetrosal approach to the upper brain stem and clivus. J Neurosurg 77:709–717
Harwick RD, Miller AS (1979) Craniocervical chordomas. Am J Surg 138:512–516
Jho HD, Carrau RL, McLaughlin MR, Somaza SC (1997) Endoscopic transsphenoidal resection of a large chordoma in the posterior fossa. Acta Neurochir (Wien) 139:343–347 (discussion 347–348)
Kaneko Y, Sato Y, Iwaki T, Shin RW, Tateishi J, Fukui M (1991) Chordoma in early childhood: a clinicopathological study. Neurosurgery 29:442–446
Keohane SG, Pye IF (1994) Intracranial chordoma with a novel symptom. J Neurol Neurosurg Psychiatry 57:503–504
Kinoshita T, Okudera T, Shimosegawa E, Yoshida Y, Yasui N, Ogawa T, Hatazawa J (2001) Chordoma with postoperative subcutaneous implantation and meningeal dissemination: MRI. Neuroradiology 43:763–766
Kitai R, Yoshida K, Kubota T, Sato K, Handa Y, Kasahara K, Nakajima H (2005) Clival chordoma manifesting as nasal bleeding. A case report. Neuroradiology 47:368–371
Kline LB, Glaser JS (1981) Bilateral abducens nerve palsies from clivus chordoma. Ann Ophthalmol 13:705–707
Kombogiorgas D, St George EJ, Chapman S, English M, Solanki GA (2006) Infantile clivus chordoma without clivus involvement: case report and review of the literature. Childs Nerv Syst 22:1369–1374
Kondziolka D, Lunsford LD, Flickinger JC (1991) The role of radiosurgery in the management of chordoma and chondrosarcoma of the cranial base. Neurosurgery 29:38–45 (discussion 45–36)
Kontozoglou T, Qizilbash AH, Sianos J, Stead R (1986) Chordoma: cytologic and immunocytochemical study of four cases. Diagn Cytopathol 2:55–61
Koshiyama H, Sakamoto M, Fujiwara K, Kim YC, Teraura T, Koh T (1992) Chondroid chordoma presenting with hypopituitarism. Intern Med 31:1366–1369
Krishnan S, Foote RL, Brown PD, Pollock BE, Link MJ, Garces YI (2005) Radiosurgery for cranial base chordomas and chondrosarcomas. Neurosurgery 56:777–784 (discussion 777–784)
Kumar PP, Good RR, Skultety FM, Leibrock LG (1988) Local control of recurrent clival and sacral chordoma after interstitial irradiation with iodine-125: new techniques for treatment of recurrent or unresectable chordomas. Neurosurgery 22:479–483
Kyoshima K, Oikawa S, Kanaji M, Zenisaka H, Takizawa T, Goto T, Takasawa H, Watanabe A, Tokushige K, Sakai K (2003) Repeat operations in the management of clival chordomas: palliative surgery. J Clin Neurosci 10:571–578
Lalwani AK, Kaplan MJ, Gutin PH (1992) The transsphenoethmoid approach to the sphenoid sinus and clivus. Neurosurgery 31:1008–1014 (discussion 1014)
Larson TC III, Houser OW, Laws ER Jr (1987) Imaging of cranial chordomas. Mayo Clin Proc 62:886–893
Leproux F, de Toffol B, Aesch B, Cotty P (1993) MRI of cranial chordomas: the value of gadolinium. Neuroradiology 35:543–545
Levi AD, Kucharczyk W, Lang AP, Schutz H (1991) Clival chordoma presenting with acute brain stem hemorrhage. Can J Neurol Sci 18:515–518
Lountzis NI, Hogarty MD, Kim HJ, Junkins-Hopkins JM (2006) Cutaneous metastatic chordoma with concomitant tuberous sclerosis. J Am Acad Dermatol 55:S6–S10
Lovely TJ, Buchheit WA (1991) Syringomyelia as a postoperative sequela of the resection of a chordoma of the clivus: case report. Neurosurgery 28:431–433
Lowery RD (1999) A case report: maxillotomy for removal of a clival chordoma. J Neurosci Nurs 31:303–308
Maira G, Pallini R, Anile C, Fernandez E, Salvinelli F, La Rocca LM, Rossi GF (1996) Surgical treatment of clival chordomas: the transsphenoidal approach revisited. J Neurosurg 85:784–792
Masui K, Kawai S, Yonezawa T, Fujimoto K, Nishi N (2006) Intradural retroclival chordoma without bone involvement—case report. Neurol Med Chir (Tokyo) 46:552–555
Matsuoka S, Yokota A, Yasukouchi H, Harada A, Kadoya C, Wada S, Ishikawa T, Okuda S (1993) Clival chordoma associated with pathological laughter. Case report. J Neurosurg 79:428–433
Mekari-Sabbagh ON, DaCunha RP (2001) Crossed eyes in a six-year-old girl. Surv Ophthalmol 45:331–334
Moriki T, Takahashi T, Wada M, Ueda S, Ichien M, Miyazaki E (1999) Chondroid chordoma: fine-needle aspiration cytology with histopathological, immunohistochemical, and ultrastructural study of two cases. Diagn Cytopathol 21:335–339
Morrill KW, Foster J, Haid RW (1993) The Le Fort I osteotomy as an approach to the midcranial base for tumor resection: case report. J Oral Maxillofac Surg 51:82–84
Nagib MG, Wisiol ES, Simonton SC, Levinson RM (1990) Transoral labiomandibular approach to basiocciput chordomas in childhood. Childs Nerv Syst 6:126–130
Nakau R, Kamiyama H, Kazumata K, Andou M (2003) Subarachnoid hemorrhage associated with clival chordoma—case report. Neurol Med Chir (Tokyo) 43:605–607
Niida H, Tanaka R, Tamura T, Takeuchi S (1994) Clival chordoma in early childhood without bone involvement. Childs Nerv Syst 10:533–535
Nishigaya K, Kaneko M, Ohashi Y, Nukui H (1998) Intradural retroclival chordoma without bone involvement: no tumor regrowth 5 years after operation. Case report. J Neurosurg 88:764–768
Nolte K (1979) Malignant intracranial chordoma and sarcoma of the clivus in infancy. Pediatr Radiol 8:1–6
Oexle K, Dammann O, Bechmann B, Vortmeyer AO, Probst EN (1992) Intracranial chordoma in a neonate. Eur J Pediatr 151:336–338
Ogi H, Kiryu H, Hori Y, Fukui M (1995) Cutaneous metastasis of CNS chordoma. Am J Dermatopathol 17:599–602
Oikawa S, Kyoshima K, Goto T, Iwashita T, Takizawa T, Kobayashi S, Ito M (2001) Histological study on local invasiveness of clival chordoma. Case report of autopsy. Acta Neurochir (Wien) 143:1065–1069
Oppenheim JS, Rosenblum BR, Sacher M, Wolfe D, Wallace S, Rothman AS (1991) Intracranial chordoma in a preadolescent. Case report. Surg Neurol 35:230–233
Perzin KH, Pushparaj N (1986) Nonepithelial tumors of the nasal cavity, paranasal sinuses, and nasopharynx. A clinicopathologic study. XIV: Chordomas. Cancer 57:784–796
Plaut HF, Blatt ES (1967) Chordoma of the clivus. A report of four cases. Am J Roentgenol Radium Ther Nucl Med 100:639–649
Plese JP, Borges JM, Nudelman M, Lefevre AB, Sallum J (1978) Unusual subarachnoid metastasis of an intracranial chordoma in infancy. Childs Brain 4:251–256
Plotkin M, Amthauer H, Eisenacher J, Wurm R, Michel R, Wust P, Stockhammer F, Rottgen R, Gutberlet M, Ruf J, Felix R (2005) Value of 123I-IMT SPECT for diagnosis of recurrent non-astrocytic intracranial tumours. Neuroradiology 47:18–26
Poskitt KJ, Sargent MA, Norman MG, Fink AM, Stringer DA (1995) Pediatric case of the day. Chordoma of the clivus. Radiographics 15:1507–1509
Rabadan A, Conesa H (1992) Transmaxillary-transnasal approach to the anterior clivus: a microsurgical anatomical model. Neurosurgery 30:473–481 (discussion 482)
Raffel C, Wright DC, Gutin PH, Wilson CB (1985) Cranial chordomas: clinical presentation and results of operative and radiation therapy in twenty-six patients. Neurosurgery 17:703–710
Sassin JF, Chutorian AM (1967) Intracranial chordoma in children. Arch Neurol 17:89–93
Schonegger K, Gelpi E, Prayer D, Dieckmann K, Matula C, Hassler M, Hainfellner JA, Marosi C (2005) Recurrent and metastatic clivus chordoma: systemic palliative therapy retards disease progression. Anticancer Drugs 16:1139–1143
Seifert V, Laszig R (1991) Transoral transpalatal removal of a giant premesencephalic clivus chordoma. Acta Neurochir (Wien) 112:141–146
Sekhar LN, Nanda A, Sen CN, Snyderman CN, Janecka IP (1992) The extended frontal approach to tumors of the anterior, middle, and posterior skull base. J Neurosurg 76:198–206
Sekhar LN, Pranatartiharan R, Chanda A, Wright DC (2001) Chordomas and chondrosarcomas of the skull base: results and complications of surgical management. Neurosurg Focus 10:E2
Sen C, Triana A (2001) Cranial chordomas: results of radical excision. Neurosurg Focus 10:E3
Sen CN, Sekhar LN, Schramm VL, Janecka IP (1989) Chordoma and chondrosarcoma of the cranial base: an 8-year experience. Neurosurgery 25:931–940 (discussion 931–940)
Smink KW, Hekster RE, Bots GT (1977) Clivus chordoma with distinct vascularity demonstrated by angiography. Neuroradiology 13:273–277
Soo MY (2001) Chordoma: review of clinicoradiological features and factors affecting survival. Australas Radiol 45:427–434
Stevenson GC, Stoney RJ, Perkins RK, Adams JE (1966) A transcervical transclival approach to the ventral surface of the brain stem for removal of a clivus chordoma. J Neurosurg 24:544–551
Stuer C, Schramm J, Schaller C (2006) Skull base chordomas: management and results. Neurol Med Chir (Tokyo) 46:118–124 (discussion 115–124)
Swearingen B, Joseph M, Cheney M, Ojemann RG (1995) A modified transfacial approach to the clivus. Neurosurgery 36:101–104 (discussion 104–105)
Takahashi T, Asai T, Isayama Y, Tamaki N, Matsumoto S (1983) Chordoma. J Clin Neuroophthalmol 3:251–257
Takami T, Ohata K, Goto T, Tsuyuguchi N, Nishio A, Hara M (2006) Surgical management of petroclival chordomas: report of eight cases. Skull Base 16:85–94
Tamaki N, Nagashima T, Ehara K, Motooka Y, Barua KK (2001) Surgical approaches and strategies for skull base chordomas. Neurosurg Focus 10:E9
Tashiro T, Fukuda T, Inoue Y, Nemoto Y, Shakudo M, Katsuyama J, Hakuba A, Onoyama Y (1994) Intradural chordoma: case report and review of the literature. Neuroradiology 36:313–315
Tewfik HH, McGinnis WL, Nordstrom DG, Latourette HB (1977) Chordoma: evaluation of clinical behavior and treatment modalities. Int J Radiat Oncol Biol Phys 2:959–962
Tomlinson FH, Scheithauer BW, Forsythe PA, Unni KK, Meyer FB (1992) Sarcomatous transformation in cranial chordoma. Neurosurgery 31:13–18
Tzortzidis F, Elahi F, Wright D, Natarajan SK, Sekhar LN (2006) Patient outcome at long-term follow-up after aggressive microsurgical resection of cranial base chordomas. Neurosurgery 59:230–237 (discussion 230–237)
Uggowitzer MM, Kugler C, Groell R, Lindbichler F, Radner H, Sutter B, Ranner G (1999) Drop metastases in a patient with a chondroid chordoma of the clivus. Neuroradiology 41:504–507
Volpe NJ, Liebsch NJ, Munzenrider JE, Lessell S (1993) Neuro-ophthalmologic findings in chordoma and chondrosarcoma of the skull base. Am J Ophthalmol 115:97–104
Warakaulle DR, Anslow P (2002) A unique presentation of retroclival chordoma. J Postgrad Med 48:285–287
Wenig BL, Sciubba JJ, Goldstein MN, Abramson AL (1985) Chondroid chordoma of the skull base: a better prognosis based on histologic criteria. Otolaryngol Head Neck Surg 93:559–563
Williams WG, Lo LJ, Chen YR (1998) The Le Fort I-palatal split approach for skull base tumors: efficacy, complications, and outcome. Plast Reconstr Surg 102:2310–2319
Wold LE, Laws ER Jr (1983) Cranial chordomas in children and young adults. J Neurosurg 59:1043–1047
Yadav YR, Kak VK, Khosla VK, Khandelwal N, Radotra BD (1992) Cranial chordoma in the first decade. Clin Neurol Neurosurg 94:241–246
Yoshii Y, Tsunoda T, Hyodo A, Nose T, Tsujii H, Tsuji H, Inada T, Maruhashi A, Hayakawa Y (1993) Proton radiation therapy for clivus chordoma—case report. Neurol Med Chir (Tokyo) 33:173–176
Zorlu F, Gurkaynak M, Yildiz F, Oge K, Atahan IL (2000) Conventional external radiotherapy in the management of clivus chordomas with overt residual disease. Neurol Sci 21:203–207
Gay E, Sekhar LN, Rubinstein E, Wright DC, Sen C, Janecka IP, Snyderman CH (1995) Chordomas and chondrosarcomas of the cranial base: results and follow-up of 60 patients. Neurosurgery 36:887–896 (discussion 887–896)
Kamrin RP, Potanos JN, Pool JL (1964) An evaluation of the diagnosis and treatment of chordoma. J Neurol Neurosurg Psychiatry 27:157–165
Heffelfinger MJ, Dahlin DC, MacCarty CS, Beabout JW (1973) Chordomas and cartilaginous tumors at the skull base. Cancer 32:410–420
Mitchell A, Scheithauer BW, Unni KK, Forsyth PJ, Wold LE, McGivney DJ (1993) Chordoma and chondroid neoplasms of the spheno-occiput. An immunohistochemical study of 41 cases with prognostic and nosologic implications. Cancer 72:2943–2949
Coffin CM, Swanson PE, Wick MR, Dehner LP (1993) Chordoma in childhood and adolescence. A clinicopathologic analysis of 12 cases. Arch Pathol Lab Med 117:927–933
Menezes AH, Gantz BJ, Traynelis VC, McCulloch TM (1997) Cranial base chordomas. Clin Neurosurg 44:491–509
Menezes A Tumors of the craniocervical junction. In: Youmans JR (ed) Neurological surgery. WB Saunders, Philadelphia, pp 3041–3072
Austin JP, Urie MM, Cardenosa G, Munzenrider JE (1993) Probable causes of recurrence in patients with chordoma and chondrosarcoma of the base of skull and cervical spine. Int J Radiat Oncol Biol Phys 25:439–444
Austin-Seymour M, Munzenrider J, Goitein M, Verhey L, Urie M, Gentry R, Birnbaum S, Ruotolo D, McManus P, Skates S et al (1989) Fractionated proton radiation therapy of chordoma and low-grade chondrosarcoma of the base of the skull. J Neurosurg 70:13–17
Berson AM, Castro JR, Petti P, Phillips TL, Gauger GE, Gutin P, Collier JM, Henderson SD, Baken K (1988) Charged particle irradiation of chordoma and chondrosarcoma of the base of skull and cervical spine: the Lawrence Berkeley Laboratory experience. Int J Radiat Oncol Biol Phys 15:559–565
Castro JR, Linstadt DE, Bahary JP, Petti PL, Daftari I, Collier JM, Gutin PH, Gauger G, Phillips TL (1994) Experience in charged particle irradiation of tumors of the skull base: 1977–1992. Int J Radiat Oncol Biol Phys 29:647–655
Hug EB, Loredo LN, Slater JD, DeVries A, Grove RI, Schaefer RA, Rosenberg AE, Slater JM (1999) Proton radiation therapy for chordomas and chondrosarcomas of the skull base. J Neurosurg 91:432–439
Rhomberg W, Bohler FK, Novak H, Dertinger S, Breitfellner G (2003) A small prospective study of chordomas treated with radiotherapy and razoxane. Strahlenther Onkol 179:249–253
Acknowledgments
Funding/source of support: Howard Hughes Medical Institute Research Training Fellowship—SJH; National Research Service Award and UCSF Clinical and Translational Scientist Training Research Award–IY; Reza and Georgianna Khatib Endowed Chair in Skull Base Tumor Surgery—ATP.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jian, B.J., Bloch, O.G., Yang, I. et al. Adjuvant radiation therapy and chondroid chordoma subtype are associated with a lower tumor recurrence rate of cranial chordoma. J Neurooncol 98, 101–108 (2010). https://doi.org/10.1007/s11060-009-0068-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-009-0068-1