Abstract
Effects of mycorrhizal association on improving tolerance of host plant under stress environments have received attentions in recent years. In this paper, six isolates of AMF (arbuscular mycorrhizal fungus) were inoculated to Casuarina equisetifolia seedlings under glasshouse conditions to investigate the effects of AMF on growth and drought tolerance of host plants. All the six isolates which belong to Glomus showed high mycorrhizal colonization (88.5–96.0%) with C. equisetifolia seedlings. Seedlings were subjected to drought stress without watering for 7 days and survival of the seedlings inoculated with Glomus caledonium Gc90068, G. versiforme Gv9004 and G. caledonium Gc90036 increased by 36.6, 23.3 and 16.6%, respectively compared with uninoculated seedlings. Limited influence of AMF on seedling height growth was found, but the effects of AMF on total biomass increment were very significant; the increment ranged from 25.7 to 118.9% compared with uninoculated treatment, and it was noted that AMF exerted more influences on root biomass than shoot biomass. Based on the changes in physiological and biochemical parameters among different treatments caused by drought stress, it was concluded that AM (arbuscular mycorrhizal) associations improve the drought tolerance of C. equisetifolia seedlings by means of some physiological and biochemical responses, such as lowering permeability of plasma membrane and MDA (malondialdehyde) contents, enhancing concentrations of P nutrition, soluble sugar, soluble protein and activities of POX (Peroxidase) of C. equisetifolia seedlings.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Casuarina equisetifolia L. Johnson is a nitrogen-fixing tree of considerable social, economic and environmental importance in coastal area of southern China. Plantations of C. equisetifolia play very important roles as windbreaks, in stabilization of moving sands, provision of fuel wood and reclamation of coastal ecosystems (Pinyopusarerk et al. 2004). The coastal sand dunes in tropical and subtropical areas of southern China are characterized by drought, infertility and poor water holding capacity that often result in low survival and poor growth of casuarina plantations. High priority has been given on reforestation of coastal dunes in southern China and there is a need to enhance the survival of casuarina seedlings by improving their drought tolerance.
Members of Casuarinaceae are infected either by ectomycorrhizal fungi or arbuscular mycorrhizal fungi (AMF) in the fields (Rose 1980; Gardner et al. 1986; Reddell et al. 1986; Raman and Elumalai 1991; Khan 1993; Sambandan et al. 1994), but the susceptibility of Allocasuarina and Casuarina species to mycorrhizal infection is variable depending on the type of mycorrhizal fungi. It has been reported that Allocasuarina species formed ectomycorrhizas more commonly than Casuarina species, inversely a higher percentage of arbuscular mycorrhizas (AM) was observed within Casuarina (Reddell et al. 1986). The genus of the AM Glomus was the dominant native species found in field surveys in southern China (Zhong et al. 2002).
In recent years, there has been a growing appreciation of the importance of mycorrhizal associations, especially of AMF on improving drought tolerance of host plants (Mukerji and Kapoor 1986; Cuenca et al. 1997; Caravaca et al. 2003). Based on the research of Nelsen and Safir (1982) on onion, improved phosphorus nutrition is a crucial factor for increased drought tolerance of mycorrhizal plants. Exposure of plants to drought conditions led to increases in free praline, soluble sugar, peroxidate (POX) activities and malondialdehyde (MDA) concentration, and inhibitions of protein synthesis have been proved in many literatures (Dhindsa 1991; Zhang and Kirkham 1994; Arji and Arzani 2008), so comparison of the changes in these parameters between non-mycorrhizal and mycorrhizal plants under drought stress can reveal the effects of mycorrhizal association on improving drought tolerance. AMF have been used in vegetation establishment in stressful and disturbed ecosystems (Estaún et al. 1997; Requena et al. 2001). However, little work has been done on improving drought tolerance of casuarinas using AM associations.
The objective of this study was to investigate the effects of AM associations on enhancement of drought tolerance of C. equisetifolia seedlings under glasshouse conditions, and to screen superior isolates for further field tests.
Materials and methods
AMF inoculum preparation
Six isolates of AMF were included in inoculation experiment (Table 1). To acquire AMF inoculum, the spores of various AMF isolates were inoculated to Trifolium repens for 6 months. Inoculum consisted of spores, mycelium, infected root fragments and potting media (1.5:1:2 vermiculite:peat:river sands on a volume:volume basis) which was sterilized in pressure steam sterilizer before being used for AMF propagation.
Inoculation and transplanting
Casuarina equisetifolia seedlings were used as host plant for AM association. Seeds which were collected from a plantation of C. equisetifolia were surface-sterilized in 0.1% HgCl2 for 10 min, rinsed several times in sterile deionized water, and sown in germination tray containing the potting media mentioned above. One month after seed sowing, uniform seedlings about 6 cm in height were transplanted into 250 ml plastic cups, one seedling per cup, and 10 g of the inoculum was added near the roots of each seedling. The seedlings were watered once daily with tap water. In addition, 20 ml solution of potassium ammonium phosphate compound fertilizer (N:P:K 15:15:15) in 1% concentration was applied to each seedling every 2 weeks. The experiment was conducted in a glasshouse, day/night temperature about 36/25°C and relative humidity 65–95%.
Experimental design, measurement and data analysis
A randomized complete block design with seven replicates was employed for seven treatments (six AMF isolates and one control, uninoculated) as shown in Table 1. Each treatment plot consisted of ten seedlings per replicate. In total, 490 seedlings were involved in this experiment. Four of the seven replicates were used for seedling height measurements and low osmotic potential solution treatments. The other three replicates were used for seedling root sampling and survival test after watering was stopped for 7 days.
After transplanting for 120 days, four seedlings from each treatment in the four-replicate group were randomly selected and treated with low osmotic potential solution for 4 days to simulate drought stress conditions. The low osmotic potential solution consisted of PEG-6000 (polyethylene glycol 6000) and water. Two osmotic potential solutions (0 MPa, water only) and −0.5 MPa (PEG-6000 150 g/L) were prepared for drought stress treatments.
In order to examine the effects of AMF on improving drought tolerance, after transplanting for 140 days watering of the seedlings in the three-replicate group was stopped for 7 days, and resumed thereafter. Survival of the seedlings was assessed after growing for another 7 days.
Height of seedlings in the four-replicate group was measured for the first time 15 days after transplanting. Nine more seedling height measurements were carried out thereafter at 15 days interval. After the last height measurement (150 days after transplanting), all seedlings were harvested for shoot and root fresh weight. Dry weight of shoots and roots was obtained after oven drying at 80°C for 48 h.
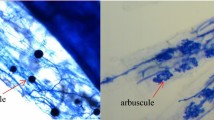
The roots of the seedlings in the three-replicate group were sampled to determine mycorrhizal colonization (MC) after growing for 120 days. Mycorrhizal colonization was estimated using the root fragment method of Brundrett et al. (1996). Root samples were cut into 1-cm long segments, soaked in 10% (w/v) KOH at 90°C for 1 h in a water bath, rinsed three times in tap water, and acidified in 2% (v/v) HCl. They were rinsed again three more times, then stained with Trypan blue at 90°C for 15 min, and washed out excess Trypan blue. Thirty root segments from each subsample were examined under the microscope for MC test.
Relative permeability, content of MDA, content of soluble sugar, content of soluble protein and POX of branchlets were measured after seedlings were subjected to low osmotic potential solution. The relative permeability of seedling branchlets was measured as follows. Fresh branchlets were cut into 2-cm long segments and 2 g of branchlet segments were placed in a glass tube containing 25 ml of deionized water. The electrolytic conductivity of the bathing solution was measured with a conductivity meter (DDS-307 model) after soaking branchlet segments in the water for 15 h. The branchlet segments were then heated to boil. When the boiling solution had cooled to room temperature, electrolytic conductivity was measured again. The relative permeability of branchlet plasma membrane was calculated using the following formula (Zwiazek and Blake 1991):
Relative permeability(%) = electrolytic conductivity of solution at 15 h before heating/electrolytic conductivity of solution after heating × 100.
MDA contents were measured using the method of Zhao et al. (1994). Proline contents were measured using the method of Troll and Lindsley (1955). Soluble sugar contents were measured using the anthrone method described by Fales (1951). POX and Soluble protein contents were evaluated as described by Hao et al. (2004). P concentrations in dried shoots were determined using an autoanalyzer ICP-DES (McLeod 1982).
Data were statistically analyzed using SAS software package for analysis of variances (ANOVA). Duncan’s multiple range test at 5% level was used to compare the significant difference between treatment means. Arcsin transformations were performed for percentage data for ANOVA, but the percentage data presented in the result tables are non-transformed values.
Results
The ANOVA results are summarized in Table 2. There were significant differences (P < 0.05–0.001) between the AMF treatments in most of the physiological and biochemical parameters assessed, indicating that AMF can produce physiological and biochemical changes in casuarina seedlings regardless whether they were under drought stress or not.
Effect of AMF on MC and Survival of seedlings
Roots of C. equisetifolia seedlings inoculated with AMF had significantly higher mycorrhizal colonization than those of the uninoculated seedlings (Table 3). The MC recorded for the uninoculated seedlings (T7) was 11.1% only compared to 88.5–96.0% of the inoculated seedlings (T1–T6). There were significant differences between the six treatments inoculated with different AMF, with T3 and T4 recording the highest MC implying that there was superior compatibility between C. equisetifolia and some AMF involved in the experiment. It was noteworthy that only root external hyphae, vesicles and spores were found, but no arbuscule structures were detected.
Survival differed considerably between treatments and not all seedlings treated with AMF survived significantly better than the untreated seedlings (Table 3). T3 and T4 isolates had the highest survival (93.3 and 80%, respectively), which were significantly higher than other treatments. Interestingly it was the T3 treatment which had the lowest survival at 46.7% although it was not significantly different from the control at 56.7%. The results indicate that certain AMF isolates such as T3 and T4 can markedly improve drought tolerance of C. equisetifolia seedlings.
Effects of AMF on height growth of seedlings
As shown in Fig. 1, there were no significant differences in seedling height among treatments after inoculating for 15 days, but marked differences were found from the 30th day on ward. The results implied that it would take some time to establish mycorrhizal association between host plant and AMF. In the subsequent 120 days of observations, only the seedlings of T1 and T3 were taller than those of T7.
Effect of AMF on biomass and P nutrition of seedlings
Various AMF exerted different influences on biomass accumulation of C. equisetifolia seedlings as shown in Table 4. AMF had more effect on root biomass than on shoot biomass in C. equisetifolia seedlings. For example, root fresh weight in T4 increased by 142.86% compared to T7, and root dry weight increased by 131.17%, but shoot fresh weight and shoot dry weight only increased by 91.63 and 109.17%, respectively, In terms of total biomass, T4 achieved best mycorrhizal effectiveness, which gained biomass increment by 127.45%. The remaining treatments were ranked as T5 (79.6%), T6 (70.9%), T2 (28.2%), T1 (25.7%) and T3 (24.8%). In contrast to the biomass, the P concentrations of seedlings in different treatments showed a similar trend to total biomass, suggesting that favorable AMF were very helpful in improving the P nutrition of seedlings, consequently promoting the growth of host plants.
Effects of AMF on physiological and biochemical changes of seedlings
No significant differences in relative electrolytic conductivities between inoculated treatments and T7 was detected under non-drought condition, but relative electrolytic conductivities in some treatments inoculated with AMF under drought stress declined distinctly compared with T7 (Fig. 2). For instance, T4, T5 and T6 reduced by 48.0, 20.2 and 15.1%, respectively compared with T7. No significant differences were found between T1, T2, T3 and T7. The results suggested that favorable AMF can alleviate the detrimental effects of drought stress on plasma membrane of host plants by reducing permeability of branchlets.
There were similar MDA contents among various treatments under non-drought stress condition, but MDA contents of all treatments increased inconsistently under drought stress (Fig. 3). MDA contents of treatments inoculated with drought-tolerant AMF increased less than that of treatments inoculated with inferior AMF or without AMF.
Treatments inoculated with AMF under non-drought stress condition uniformly achieved higher soluble sugar contents in branchlets than that of T7. T2, T4, T5 and T6 increased much more than other treatments, viz. increased by 30.3, 52.1, 72.8 and 53.9%, respectively compared with T7 (Fig. 4). However, under drought stress, soluble sugar contents of all treatments decreased compared with that under non-drought condition, especially T2, T5 and T6 decreased more significantly.
Under non-drought condition, branchlets soluble protein contents of treatments inoculated with AMF enhanced evidently than that of T7. In contrast, under drought stress, soluble protein of all treatments was lower than that under non-drought condition, but T4 and T5 still were detected the higher soluble protein, which increased by 48.1 and 29.9%, respectively than that of T7 (see Fig. 5).
As shown in Fig. 6, only POX activities of T4, T5 and T6 were higher than that of T7 under non-drought condition. However, under drought stress, the POX activities of all treatments improved obviously than that of the treatments under non-drought condition, but only T4, T5 and T6 had higher POX activities than T7.
Discussion
Examination of various root samples after inoculating with 6 AMF for 120 days showed that all the treatments had high mycorrhizal colonization, implying that it is easy to establish mycorrhizal association between C. equisetifolia and AMF of the Glomus genus. The results are consistent with previous research by Chung and Liu (1986) and Sidhu et al. (1990). Compared with other tree species, the mycorrhizal effectiveness of six AMF on improvement of height growth and biomass in different treatments was not as good as expected. It is probable that high mycorrhizal colonization does not necessarily mean superior mycorrhizal effectiveness. It is thus essential to select specific isolates of AMF for host plants in terms of different tasks or targets.
One of the most severe factors limiting the growth of plants in southern China is P deficiency of soil. Plant roots infected by Arbuscular mycorrhizae are known to be more efficient in taking up soil P than uninfected roots (Pacovsky et al. 1985). Raju et al. (1990) found that most of the observed changes in the growth of the sorghum resulting from mycorrhizal infection are attributable to increased uptake of P and other nutrients. The increment of total biomass and P concentration in C. equisetifolia seedlings infected by AMF indicated that AMF play an important role in improving P absorption of host plants.
It is accepted that plasma membrane plays an important role in regulating micro-environment and maintaining well-balanced metabolism in cell, and electrolytic conductivity of plasma membrane under adverse conditions reflects the stability of plasma membrane. Some researcher considered that cell-membrane stability can be treated as the main evaluation factor of cereal crop when selecting drought-resistance variety (Tripathy et al. 2000). In this study, our results demonstrated that specific isolates of AMF can improve plasma membrane stability of C. equisetifolia seedlings under drought stress.
In order to tolerate drought stress, plants will accumulate high concentration of low-molecular-mass organic solutes such as proline, betaine, soluble sugars or amino acids to regulate the osmotic potential of cells aiming at improving absorption of water under drought stress. Moreover, plant will enhance the activity of antioxidant enzymes to clean the free radicals produced in plants under adverse condition. In this study, concentrations of soluble sugar and soluble protein in C. equisetifolia seedlings inoculated with superior AMF were improved significantly compared with that of untreated seedlings. It was regarded as a consequence of high photosynthesis efficiency of host plants from mycorrhizal association (Wright et al. 1998), resulting in lowering osmotic potential of host plants so as to improve the drought-tolerant ability. POX activities of C. equisetifolia seedlings inoculated with superior AMF increased significantly compared with that of uninoculated seedlings whether the host plants were under drought condition or not. This suggests that AMF can activate the enzymatic activity of antioxidant system not always under adverse conditions. In contrast to POX activities, MDA contents did not vary obviously among all treatments under non-drought condition, but under drought stress, MDA contents of C. equisetifolia seedlings inoculated with superior AMF decreased significantly compared with that of uninoculated seedlings. This demonstrated that AMF can only reduce abnormally high MDA contents caused by drought stress, and had no effect on normal MDA contents.
In conclusion, selecting suitable mycorrhizal fungi for plant revegetation on the drought-proned coastal dune is a high priority. It has been found that AMF of the Glomus genus is the dominant species in coastal plantations of C. equisetifolia in southern China (Zhong et al. unpublished data), but the differences of mycorrhizal effectiveness between different species or isolates was noteworthy, so further collection and selection of favorable isolates are warranted.
References
Arji I, Arzani K (2008) Effect of water stress on some biochemical changes in leaf of five olive (Olea europaea L.) cultivars. Acta Hortic 791(2):523–526
Brundrett M, Bougher N, Dell B, Grove T, Malajczuk N (1996) Working with mycorrhizas in forestry and agriculture. ACIAR Monogr 32:373
Caravaca F, Barea JM, Palenzuela J, Figueroa D, Alguacil MM, Roldan A (2003) Establishment of shrub species in a degraded semiarid site after inoculation with native or allochthonous arbuscular mycorrhizal fungi. Appl Soil Ecol 22(2):103–111
Chung HH, Liu SC (1986) Frankia and endomycorrhizae association in coastal windbreaks plantation of casuarina, proceedings of 18th IUFRO world congress, Div 2, Vol II. Forest Plant and Forest Protection 455–468 pp
Cuenca G, Andrade ZD, Escalante G (1997) Arbuscular mycorrhizae in the rehabilitation of fragile degraded tropical lands. Biol Fertil Soils 26(2):107–111. doi:10.1007/s003740050351
Dhindsa RS (1991) Drought stress, enzymes of glutathione metabolism, oxidation injury, and protein synthesis in Tortula ruralis. Plant Physiol 95(2):648–651
Estaún V, Savé R, Biel C (1997) AM inoculation as a biological tool to improve plant revegetation of a disturbed soil with Rosmarinus officinalis under semi-arid conditions. Appl Soil Ecol 6:223–229
Fales FW (1951) The assimilation and degradation of carbohydrates by yeast cells. J Biol Chem 193(1):113–124
Gardner WK, Parbery DG, Barber DA (1986) The acquisition of phosphorus by lupinus albus L. I. Some characteristics of the soil/root interface. Plant Soil 68:19–32
Hao ZB, Chang Z, Xu Z (2004) Experiment Guide of Plant Physiology (in Chinese) Haerbin Industrial University Press, Haerbin, p 115
Khan AG (1993) Occurrence and importance of mycorrhizae in aquatic trees of New South Wales, Australia. Mycorrhiza 3:31–38. doi:10.1007/BF00213465
McLeod S (1982) Routine analytical method in notes on soil techniques No. 4 CSIRO division of soils, Canberra, Australia
Mukerji KG, Kapoor A (1986) Occurrence and importance of vesicular-arbuscular mycorrhizal fungi in semiarid regions of India. For Ecol Manage 16:117–126
Nelsen CE, Safir GnR (1982) Increased drought tolerance of mycorrhizal onion plants caused by improved phosphorus nutrition. Planta 154(5):407–413. doi:10.1007/BF01267807
Pacovsky RS, Fuller G, Paul EA (1985) Influence of soil on the interactions between endo-mycorrhizae and Azospirillum on sorghum. Soil Biol Biochem 17:521–531
Pinyopusarerk K, Kalinganire A, Williams ER, Aken KM (2004) Evaluation of international provenance trials of Casuarina equisetifolia. ACIAR technical reports canberra, No. 58 p 9
Raju PS, Clark RB, Ellis JR (1990) Effects of species of VA-mycorrhizal fungi on growth and mineral uptake of sorghum at different temperatures. Plant Soil 121:165–170. doi:10.1007/BF00012308
Raman N, Elumalai S (1991) Studies of mycorrhizal and actinorhizal association in Casuarina equisetifolia in Coramandel coastal region. J Trop For 7:138–150
Reddell P, Bowen GD, Robson AD (1986) Nodulation of Casurinaceae in relation to host species and soil properties. Aust J Bot 34(4):435–444. doi:10.1071/BT9860435
Requena N, Estefania PS, Concepcion AA et al (2001) Management of indigenous plant-microbe symbioses aids restoration of desertified ecosystems. Appl Environ Microbiol 67(2):495–498
Rose SL (1980) Mycorrhizal associations of some actinomycete nodulated nitrogen-fixing plants. Can J Bot 58(13):1449–1454. doi:10.1139/b80-176
Sambandan K, Kannan K, Raman N (1994) Vesicular-arbuscular mycorrhizae of casuarina equisetifolia Forst. in four different soil types in Tamil Nadu. Indian Forester 120(6):510–514
Sidhu OP, Behl HM, Gupta ML, Janardhanan K (1990) Occurrence of VAM in Casuarina equisetifolia L. Curr Sci 59(8):422–423
Tripathy JN, Zhang J, Robin S, Nguyen TT, Nguyen HT (2000) QTLs for cell-membrane stability mapped in rice (Oryza sativa L.) under drought stress. Theor Appl Genet 100:1197–1202. doi:10.1007/s001220051424
Troll W, Lindsley J (1955) A photometric method for the determination of praline. J Biol Chem 215:655–660
Wright DP, Read DJ, Scholes JD (1998) Mycorrhizal sink strength influences whole plant carbon balance of Trifolium repens L. Plant Cell Environ 21:881–891
Zhang J, Kirkham MB (1994) Drought-stress-induced changes in activities of superoxide dismutase, catalase, and peroxidase in wheat species. Plant Cell Physiol 35(5):785–791
Zhao SJ, Xu ZC, Zhou C (1994) Improved method of MDA determination in plant tissue. Bulletin of Plant Physiol 30:207–210
Zhong CL, Gong MQ, Lin SQ, Chen Y, Wang FZ (2002) Investigation of AM fungi under casuarina plantations and inoculating experiment for casuarina junghuhnian seedlings. For Res 15(4):427–431
Zwiazek JJ, Blake TJ (1991) Early detection of membrane injury in black spruce (Picea mariana). Can J For Res 21(3):401–404
Acknowledgments
This study was financially supported by the International Foundation for Science (IFS grant No. D/3691-1), Youth Foundation of research Institute of Tropical Forestry (2007-24), the 11th National Five-Year Plan for Forestry Projects (2006BAD01A1605), the 863 Program (2002AA241091) and Ministry of Science and Technology’s Extension Program (2007GB24320424). AMF isolates of G. mosseae, G. etunicatum and G. intraradices were kindly provided by Prof. Wang Youshan, Plant Nutrition and Resource Institute, Agriculture and Forestry Academy of Beijing, China. Mr Aljoy Abarquez of CSIRO Plant Industry, Australia was thanked for his valuable comments on early draft of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, Y., Zhong, C.L., Chen, Y. et al. Improving drought tolerance of Casuarina equisetifolia seedlings by arbuscular mycorrhizas under glasshouse conditions. New Forests 40, 261–271 (2010). https://doi.org/10.1007/s11056-010-9198-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11056-010-9198-8