Abstract
Photodynamic therapy (PDT) is a minimally invasive approach, in which a photosensitizer compound is activated by exposure to light. The activation of the sensitizer drug results in several chemical reactions, such as the production of reactive oxygen species and other reactive molecules, which presence in the biological site leads to the damage of target cells. Although PDT has been primarily developed to combat cancerous lesions, this therapy can be employed for the treatment of several conditions, including infectious diseases. A wide range of microorganisms, including Gram-positive and Gram-negative bacteria, viruses, protozoa, and fungi, have demonstrated susceptibility to antimicrobial PDT. This treatment might consist in an alternative for the management of fungal infections. Antifungal photodynamic therapy has been successfully employed against Candida species, dermatophytes, and Aspergillus niger. Chromoblastomycosis is an infection that involves skin and subcutaneous tissues caused by the traumatic inoculation of dematiaceous fungi species, being that the most prevalent are Fonsecaea pedrosoi and Claphialophora carrionii. In the present work, the clinical applications of PDT for the treatment of chromoblastomycosis are evaluated. We have employed methylene blue as photosensitizer and a LED (Light Emitting Diode) device as light source. The results of this treatment are positive, denoting the efficacy of PDT against chromoblastomycosis. Considering that great part of the published works are focused on in vitro trials, these clinical tests can be considered a relevant source of information about antifungal PDT, since its results have demonstrated to be promising. The perspectives of this kind of treatment are analyzed in agreement with the recent literature involving antifungal PDT.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Photodynamic therapy (PDT) is a treatment that employs a photosensitizer compound, which is activated by exposure to light of a wavelength that is excitatory to this compound. The activation of the sensitizer drug results in several chemical reactions, such as the production of reactive oxygen species (type II reaction) and other reactive molecules (free radicals, type I reaction). The presence of these molecules in the site to be treated leads to the damage of target cells [1]. PDT was firstly developed for the treatment of cancer, but recently it has been employed against several microorganisms, including bacteria, virus, and fungi [2–13]. Antifungal photodynamic therapy has been successfully employed against Candida species, dermatophytes, and A. niger [14–20].
The great interest in alternative therapies for the treatment of fungal infections comes from the fact that the number of antifungal agents available for chemotherapy is very restrict when compared with the number of antibacterial drugs. Furthermore, the cases of recurrent infections are a major issue for certain kinds of disease, such as candidiasis, dermatophytosis, and chromoblastomycosis. Antifungal photodynamic therapy is a developing area of research [21]. Qiao et al. [22] developed an interesting review focusing on in vivo PDT for the treatment of superficial mycoses; however, the majority of the literature in this area concerns to in vitro experiments. Considering the potential of this technique in the treatment of fungal infections and the importance of developing new antifungal strategies, PDT can be considered an alternative of great interest to future researches.
Chromoblastomycosis is an infection that involves skin and subcutaneous tissues caused by the traumatic inoculation of a specific group of dematiaceous fungi. Several species have been implicated as etiological agents; however, the most prevalent in endemic areas are F. pedrosoi and C. carrionii. F. compacta, P. verrucosa, R. aquaspersa, or E. dermatitidis may also be causative agents of the disease [23]. These saprophyte fungi habit the soil and decayed materials, such as woods and leaves, which can explain the greater incidence of the disease in farm laborers.
The treatment of chromoblastomycosis can be performed by surgical removal of the lesions, cryosurgery, employment of antifungal agents, or a combination of these treatments. However, it is important to register that the complete healing is difficult and recurrences are common [23]. The administration of antifungal agents for long-term treatments can cause several undesirable side effects and toxicity to the patient, and the surgical treatment can be mutilating and painful. Considering these facts, the application of PDT for the treatment of chromoblastomycosis is an issue of interest, especially in recurrent fungal infections, even if employed as adjutant to conventional antifungal therapy. The procedure is safe and does not cause additional damage to the patients. In the present work, 10 patients with chromoblastomycosis were treated with PDT, employing methylene blue as photosensitizer and a LED (Light Emitting Diode) device as light source.
Materials and Methods
Patients
Ten patients infected with chromoblastomycosis were selected from the Ambulatory of Chromoblastomycosis in the Infectious/Parasitic Diseases Service of the Federal University of Maranhão (UFMA), São Luís, Maranhão, Brazil. The patients selected did not receive antifungal treatment in the past 6 months before the treatment and did not presented any kind of additional disease or predisposing condition for other infections. The chromoblastomycosis infection was diagnosed by a dermatologist through physical examination, direct observation of fungal structures in the microscope, cultivation, and direct observation of the fungus in the lesion. No particular stage of the disease was used as selection criterion. The protocol of the study was approved by the Ethics Committee of the institution, and a consent formulary was signed by each patient.
Antifungal Photodynamic Therapy
The photosensitizer delivery was carried out as described by Calzavara-Pinton et al. [18] with modifications. A 20% methylene blue preparation in Eucerin cream (Beiersdorf, Hamburg, Germany) was applied to the skin lesions for 4 h under a gauze occlusive dressing, which was removed prior to the irradiation. After this period, the lesions were irradiated with red LED (GaAlAs, wavelength absorption 660 nm, energy dose of 28 J/cm2) for 15 min. If the lesion extension exceeded the LED reaching area, the irradiation was repeated in the same patient until all the compromised area to be treated. The light intensity was uniform in all the areas treated, and the treated areas varied between the patients. Any undesirable side effect was registered during the treatment.
The lesions were clinically and microscopically observed after 7 days. The treatment was repeated for five additional weeks. The lesions were recorded for comparison before and after the treatment. Patients returned after 1 week for follow-up and for treatment with PDT. The total follow-up period is 2 years after the complete remission of the disease and negative mycological tests.
Results and Discussion
All of the ten patients treated presented reduction in the compromised area after six PDT applications, considering clinical and microscopical aspects. The reduction was measured as about 80–90% of improvement in the aspect of the lesion, i.e., reduction in the lesion volume and tissue cicatrization in 80–90% when compared with the previous condition of the lesion.
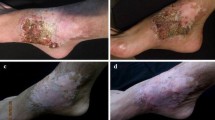
Figure 1 corresponds to the evolution of the healing in one of the patients treated. The efficacy of this alternative treatment was verified by expertise doctors and confirmed by biopsy. The treatment was well tolerated by all patients evaluated, and no side effects were registered. Patients reported that the pain sensation diminished, as well as the disappearance of the odor and rapid reduction in the lesions. They also felt hopeful and excited about the treatment. Patients with severe lesions reported that they were less worried about the aspect of their lesions. However, the complete healing was not achieved in any patient, and the mycological tests were still positive, except for one patient. These positive tests showed the necessity of adjutant treatment with itraconazole after the period of treatment with PDT. Figure 2 shows histopathological images of the lesions. The use of PDT as a coadjutant treatment does not make this therapy irrelevant, since it can reduce the size of the compromised areas and the degree of infection. This can lead to an improvement in the efficacy of the antifungal chemotherapy, as well as in reduction in the time of antifungal administration, less toxicity to the patient, and eventually in diminishing the relapses cases. Furthermore, the quality of life of the patients is significantly improved during the therapy, decreasing the number of patients that abandon the treatment.
Clinical aspect of chromoblastomycosis lesion in one patient, before and after antifungal photodynamic therapy. a Initial aspect of the lesion before treatment. b Aspect of the lesion after 1 week of treatment. Methylene Blue was applied in the skin. c Aspect of the lesion after 4 weeks of treatment. d Aspect of the lesion after 1 additional week of follow-up
The results obtained in this clinical trial do not allow the elucidation of the mechanism involved in the healing process. However, we could infer that PDT may act by paralyzing the growth, killing the dematiaceous fungi, or even by interfering in the immunologic mechanism. In any case, additional studies are necessary to clarify the particular action of PDT against chromoblastomycosis infections.
Despite this preliminary results obtained with methylene blue do not achieve the complete healing, it indicates promising applications for PDT in chromoblastomycosis. Other photosensitizer agents as well as other therapeutic schemes might be tested to obtain better results.
References
Moreira LM, Santos FV, Lyon JP, Maftoum-Costa M, Pacheco-Soares C, Silva NS. Photodynamic therapy: porphyrins and phthalocyanines as photosensitizers. Aus J Chem. 2008;61:741–54.
Calzavara-Pinton PG, Venturini M, Sala R. A comprehensive overview of photodynamic therapy in the treatment of superficial fungal infections of the skin. J Photochem Photobiol B: Biol. 2005;78:1–6.
Zeina B, Greenman J, Purcell WM, Das B. Killing of cutaneous microbial species by photodynamic therapy. Br J Dermatol. 2001;144:274–8.
Zeina B, Greenman J, Corry D, Purcell WM. Cytotoxic effects of antimicrobial photodynamic therapy on keratinocytes in vitro. Br J Dermatol. 2002;146:568–73.
Zeina B, Greenman J, Corry D, Purcell WM. Antimicrobial photodynamic therapy: assessment of genotoxic effects on keratinocytes in vitro. Br J Dermatol. 2003;148:229–32.
Marotti J, Aranha AC, Eduardo Cde P, Ribeiro MS. Photodynamic therapy can be effective as a treatment for herpes simplex labialis. Photomed Laser Surg. 2009;27:357–63.
Rossi R, Bruscino N, Ricceri F, Grazzini M, Dindelli M, Lotti T. Photodynamic treatment for viral infections of the skin. G Ital Dermatol Venereol. 2009;144:79–83.
Kacerovska D, Pizinger K, Kumpova M, Cetkovska P. Genital warts treated by photodynamic therapy. Skinmed. 2007;6:295–7.
Akilov OE, Kosaka S, O’Riordan K, Hasan T. Parasiticidal effect of delta-aminolevulinic acid-based photodynamic therapy for cutaneous leishmaniasis is indirect and mediated through the killing of the host cells. Exp Dermatol. 2007;16:651–60.
Asilian A, Davami M. Comparison between the efficacy of photodynamic therapy and topical paromomycin in the treatment of Old World cutaneous leishmaniasis: a placebo-controlled, randomized clinical trial. Clin Exp Dermatol. 2006;31:634–7.
Bristow C, Hudson R, Paget TA, Boyle RW. Potential of cationic porphyrins for photodynamic treatment of cutaneous leishmaniasis. Photodiag Photodyn Ther. 2006;3:162–7.
Akilov OE, Kosaka S, O’Riordan K, Hasan T. Photodynamic therapy for cutaneous leishmaniasis: the effectiveness of topical phenothiaziniums in parasite eradication and Th1 immune response stimulation. Photochem Photobiol Sci. 2007;6:1067–75.
Silva NS, de Ribeiro CM, Machado AH, Pacheco-Soares C. Ultrastructural changes in T. foetus after treatments with AlPcS4 and photodynamic therapy. Vet Parasitol. 2007;146:175–81.
Bliss JM, Bigelow CE, Foster TH, Haidaris CG. Susceptibility of Candida species to photodynamic effects of photofrin. Antimicrob Agents Chemother. 2004;48:2000–6.
Munin E, Giroldo LM, Alves LP, Costa MS. Study of germ tube formation by C. albicans after photodynamic antimicrobial chemotherapy (PACT). J Photochem Photobiol B: Biol. 2007;88:16–20.
Giroldo LM, Felipe MP, Oliveira MA, Munin E, Alves LP, Costa MS. Photodynamic antimicrobial chemotherapy (PACT) with methylene blue increases membrane permeability in C. albicans. Lasers Med Sci. 2009;24:109–12.
Dovigo LN, Pavarina AC, Oliveira M, Garcia E, Giampaolo ET, Vergani CE, Bagnato VS. Fungicidal effect of photodynamic therapy against fluconazole-resistant C. albicans and C. glabrata. Mycoses. 2009;54:123–30.
Calzavara-Pinton PG, Venturini M, Capezzera R, Sala R, Zane C. Photodynamic therapy of interdigital mycoses of the feet with topical application of 5-aminolevulinic acid. Photodermatol Photoimmunol Photomed. 2004;20:144–7.
Smijs TGM, Schuitmaker HJ. Photodynamic Inactivation of the dermatomycete T. rubrum. Photochem Photobiol. 2003;77:556–60.
Friedberg JS, Skema C, Baum ED, Burdick J, Vinogradov SA, Wilson DF, Horan AD, Nachamkin I. In vitro effects of photodynamic therapy on A. fumigatus. J Antimicrob Chemother. 2001;48:105–7.
Donnelly RF, McCarron PA, Tunney MM. Antifungal photodynamic therapy. Microbiol Res. 2008;163:1–12.
Qiao J, Li R, Ding Y, Fang H. Photodynamic therapy in the treatment of superficial mycoses: an evidence-based evaluation. Mycopathologia. 2010;170:339–43.
Queiroz-Telles F, Esterre P, Perez-Blanco M, Vitale RG, Salgado CG, Bonifaz A. Chromoblastomycosis: an overview of clinical manifestations, diagnosis and treatment. Med Mycol. 2009;47:3–15.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lyon, J.P., de Maria Pedroso e Silva Azevedo, C., Moreira, L.M. et al. Photodynamic Antifungal Therapy Against Chromoblastomycosis. Mycopathologia 172, 293–297 (2011). https://doi.org/10.1007/s11046-011-9434-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11046-011-9434-6