Abstract
Background
Recently exposure to ionizing radiation driven by artificial radiation sources such as Medical X-rays and Nuclear medicine has increased hastily. Ionizing radiation-induced the DNA damage and activate the DNA damage response signaling pathways. The aim of this study was to evaluate the role of miR-21 and miR-625 in response to low-dose ionizing radiation.
Materials and methods
In this study, the blood sample of 38 volunteer patients who underwent Cardiac scans before and after 99mTc-MIBI injection were used. The WBC of patients was used for RNA extraction and after cDNA synthesis by the poly-A method the expression level of miR-21 and miR-625 was evaluated by real-time PCR method.
Results
The results of this study indicated that miR-21 and miR- 625 were significantly upregulated under exposure to low-dose ionizing radiation. The expression level of these miRNAs was not significantly correlated with the age and BMI of patients. More ever the bioinformatics analysis indicated that SP1 was a common target of both miRNAs and had the highest degree between hub genes.
Conclusion
In summary miR-21 and miR-625 can contribute to the response to acute low dose ionizing radiation by targeting the SP1. However further studies should be carried out on the molecular mechanism of effects of miR-21 and miR-625 in response to low dose ionizing radiation by targeting the SP1.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Human exposure to ionizing radiation is inevitable. The radiation source included natural sources such as drinks, foods, soil, internal radioactive sources, and cosmic rays, and artificial radiation sources such as Medical X-rays and Nuclear medicine [1]. Exposure to ionizing radiation-induced the DNA damage directly or indirectly through the reactive oxygen species (ROS) generation [2]. With the increasing use of artificial radiation sources, especially medical sources, concerns about the effects of ionizing radiation on human health have increased [3]. In recent years using the CT-scan and nuclear medicine diagnostic methods as high-dose procedures has grown hastily [4].
Exposure to ionizing radiation activates important signaling pathways including ATM, ATR, PI3k-Akt, NF-κB; activation of these pathways leads to induce the DNA repair and cell cycle arrest and inhibit apoptosis [5]. Low-dose ionizing radiation leads to stimulation of stem cell proliferation and increasing the synthesis of RNA, protein, and DNA [6]. Pearce et al. in their study indicated that low dose ionizing radiation exposure in pediatric imaging increases brain cancer and leukemia [7].
Myocardial perfusion imaging as a non-invasive diagnostic molecular medicine method is widely used for evaluating regional blood flow perfuse the myocardium. Generally, Technetium-99 m methoxy isobutyl isonitrile (99mTc-MIBI) is the most favorable agent for Myocardial perfusion imaging [8, 9]. 99mTc with a physical half-time of 6 h emits gamma rays with an energy of about 140 keV [10]. So like the other diagnostic molecular medicine methods myocardial perfusion imaging considers as a low-dose ionizing radiation exposure method [1].
Recent studies indicated that the expression level of genes and especially microRNAs (miRNAs) in a biological sample such as blood was dysregulated due to the ionizing radiation so they can be used as a potential biomarker of irradiation [11, 12]. miRNAs as a collection of small non-coding RNAs play a significant role in the pathogenesis of various diseases. Numerous biological processes such as proliferation, invasion, migration, apoptosis, and differentiation have been regulated through the miRNA [13, 14]. miRNAs are about 18–24 nucleotides long and regulate their target gene expression. miRNAs exert their effect by destroying the target mRNA or inhibiting its translation into a protein by binding to their complement region in the target mRNA [15, 16]. Recent studies indicated that miRNAs have an essential role in response to irradiation of living cells. Ionizing radiation leads to transient miRNAs dysregulation dose-dependently [17]. DNA damage due to ionizing radiation regulates miRNA expression level through transcription activation or miRNA procession enhancement. Upon activation of ATM as a DNA damage sensor due to the DNA damage, the expression level of miRNAs was changed through the KH-type splicing regulatory protein (KSRP) or P53 activation [18]. Recent studies evaluate the dysregulation of miRNAs as a result of low-dose ionizing radiation. According to these results, the expression level of miR-21 in human skin fibroblasts cells was up-regulated significantly under low dose ionizing radiation 10 cGy. more ever exposing the human fibroblasts cells low-dose γ-radiation 100 cGy leads to upregulation of miR-625. Considering the results of these in-vitro studies which indicate a change in the expression of miR-21 and miR-625 due to the low-dose ionizing radiation, we hypothesize that these miRNAs having a role in reply to low dose gamma-radiation. This study intended to evaluate the effects of low-dose gamma radiation on the expression level of miR-21 and miR-625 in human peripheral blood.
Materials and methods
Sample collection
This study was performed in the nuclear medicine section of the Farshchian medical center of Hamadan University of Medical Sciences. The sample consisted of 38 patients (32 females and 6 males) who underwent Cardiac scans and accepted Informed consent and participation in this study. The average patients’ age and BMI were 53.92 ± 9.74 years and 28.76 ± 4.25 respectively. This study was approved by the Ethics Committee of the Hamadan University of Medical Sciences (IR.UMSHA.REC.1398.418).
Assessment of absorbed doses
In this study Micro-silica beads (TrueInvivo Ltd., UK), types of TLD tablets, were used to assess the participants’ absorption radiation dose. Therefore, TLDs were prepared and calibrated by a theratron-60 source, and both individual and group calibration factors were calculated for them. A TLD 7103 reader (TLD 7103 RSD Co., Iran) was used to read the TLDs.
A blood sample was taken from the patients before drug injection, then three TLDs tablets were placed on the patients’ necks and 25–30 mCi of 99mTc-MIBI injected to the patients to perform the stress test of the heart. Patients were asked to have the TLDs on their necks for up to 24 h when they would come back to the hospital to perform their heart rest test. Therefore, the superficial dose of skin in the neck site is measured using these TLD tablets placed on the patient's body for 24 h. Then another blood sample was taken from the patients to compare the results with the first sample. The inlet surface dose is obtained by multiplying the number obtained from the readings of each tablet in the individual and group calibration factors of each dosimeter. It has been estimated that the absorbed dosage in the body is equivalent to the inlet dose of the surface which dosimeters measure [19].
RNA extraction and real-time PCR
In order to evaluate the expression level of miRNAs in white blood cells (WBC) before extraction of total RNA the whole blood of patients were treated with red blood cell lysis buffer. The resulting WBC Pellets were used for RNA extraction. Total RNA was extracted using the RiboEx Total RNA Solution (GeneAll, South Korea) according to the recommended protocol. The quality, quantity, and purity of extracted RNA were assessed using 1% agarose gel electrophoresis and NanoDrop (Thermo Fisher Scientific, USA).
cDNA was synthesized using the one-step miRNA cDNA synthesis kit (ExirGene, Iran) based on the manufacturer’s protocol. In this method, miRNAs were polyadenylated at 3′ end and reverse transcribed to converted into cDNA. Finally, the expression level of has-miR-21-5p (Accession number: MIMAT0000076) and has-miR-625-5p (Accession number: MIMAT0003294) was evaluated using miRNA specific primer pairs (ExirGene, Iran) and RealQ Plus Master Mix Green (Amplicon, Denmark) according to the manufacturer’s protocol with Roche LightCycler® 96 system (Roche, Germany). In this study U6, snRNA was used as an internal control for the normalization of miRNA expression level. Relative miRNAs expression levels were calculated using the \({2}^{-\Delta \Delta CT}\) equation [20].
Bioinformatics analysis of miRNA target genes
In order to predict the low-dose gamma radiation effects on target genes, the mirwalk software was used to determine the valid target genes of evaluated miRNAs (http://zmf.umm.uni-heidelberg.de/apps/zmf/mirwalk2/). The string software was used to depict the protein–protein interaction (PPI) network of target genes of miR-21 and miR-625. Finally the Cytoscape software (V. 3.6.0) was used to visualize and analyze the depicted network.
Results
Absorbed doses measurement
These compounds of TLD make it almost equivalent to air or tissue and have an effective atomic number which is very near to the tissue effective atomic number. TLDs that have minimum fading, high sensitivity, and high thermoluminescence are used exclusively for external dosimetry of the patient. More ever, due to their small size and proper structure, they do not cause any harm to the patient [21, 22]. Assessment of The TLD tablets placed on the patient’s body indicated that the absorbed radiation dose of patients is 7.72 ± 4.73 mSv.
Effects of ionizing radiation on the miRNA expression level
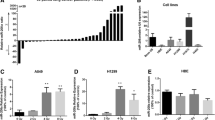
As seen in Fig. 1 Evaluation of miRNAs expression level by real-time PCR by pair sample t-test indicated that low dose ionizing radiation exposure leads to a significant increase of miR-21 expression level (P-Value = 0.05). more ever miR-625 was upregulated significantly after exposure to low-dose gamma radiation (P-Value = 0.001).
More ever the expression level of miR-21 and miR-625 was not significantly different between males and females (P-Value = 0.30 and 0.20 respectively). the expression level of miR-21 and miR-625 was not significantly different between two age groups less and more than 54 (P-value 0.09, and 0.29 respectively).based on the BMI the expression level of miR-21 and miR-625 was not significant between two BMI groups below and above 28.76 (P-value 0.30 and 0.55 respectively.
Effects of ionizing radiation on miRNA target genes
Using the mirwalk software the 1039 valid targets of miR-21 and miR-625 were determined. miR-21 and miR 625 had 832 and 207 target genes respectively. As seen in Fig. 2 the network of miR-21 and miR-625 and their valid target was visualized using Cytoscape software. Network analysis indicated that Formin-binding protein (FNBP1), Peptidylprolyl Isomerase F (PPIF), Transducin (β)-like 1 X-linked receptor 1 (TBL1XR1), The insulin-like growth factor 1 receptor (IGF1R), Argonaute 2 (AGO2), Sprouty RTK Signaling Antagonist 4 (SPRY4), N-Alpha-Acetyltransferase 50(NAA50), specificity protein 1 (SP1), and SRY (sex-determining region Y)-box 2 (SOX2) were the common target of both miRNAs. Using the string software, the PPI network of target genes was created considering the confidence score threshold of 0.7. The constructed PPI network of target genes was evaluated with the CytoHuuba plugin under Cytoscape software. Based on the degree top 10 hub genes including STAT3, MYC, EGFR, PIK3R1, PTEN, BRCA1, VEGFA, SKP2, UBE2V1, and SP1 were selected. The SP1 as a hub gene was the common valid target of miR-21 and miR-625 (Fig. 3).
Discussion
Recently, there are many concerns about the dangers of low-dose ionizing radiation from natural and artificial sources. Some studies indicated that exposure to low-dose Ionizing radiation affects the immune system and associates with cancer risk [23, 24]. More ever, the miRNAs expression level was dysregulated under ionizing radiation [25]. Considering the role of miRNAs in response to ionizing radiation, in this study we evaluate the effects of low-dose gamma rays of 99mTc on the expression level of miR-21 and miR-625 in Patients examined with myocardial perfusion imaging.
The Results of the present study indicated that miR-21 and miR-625 were upregulated under low dose gamma radiation in WBC of patients. Upregulation of miR-21 as a potential oncomiR was reported in several cancers. This miRNA has role in response to treatment and associates reciprocally with cancer patients’ prognosis [26]. Peng et al. in their study indicated that the radioresistance of esophageal squamous cell carcinoma by targeting the PTEN [27]. In liver tissues and hepatocytes, expression level of miR-21 under short-term and long term exposure to Ionizing radiation was increased through the AP-1 And EGFR-Stat3 pathways respectively. More ever low and high LET radiation leads to continuous increase in miR-21 expression level in brain tissue due to the positive feedback between EGFR and miR-21 [28, 29]. Mir-625 which having role in tumorigenesis, invasion and metastasis,was dysregulated in several tumors [30, 31]. some studies indicated that this miRNA was associated with chemoradioresistance of tumor cells. on the other hand expression level of miR-625 was dysregulated in gastric tumors under radiotherapy [32, 33]. The results of the present study, in line with the previous in-vitro study, showed an increase in the expression of miR-21 and miR-625 in reply to low-dose gamma radiation [25, 34].
The results of bioinformatics analysis indicated that FNBP1, PPIF, TBL1XR1, IGF1R, AGO2, SPRY4, NAA50, SOX2, and SP1 are the common targets of miR-21 and miR-625. On the other hand among the 10 hub genes, the SP1 was a hub gene which was the common valid target of miR-21 and miR-625. FNBP1 is composed of four domains including SH3, FBH, FCH, and, HR1. the FNBP protein through the actin cytoskeleton regulation may have a role in the control of the nuclear division and cell polarity [35]. PPIF affects apoptosis and necrosis by regulating mitochondrial permeability. On the other hand, the protein encoded by this gene has a role in the progression and tumor cell migration of endometrial cancer [36]. PPIF has antagonism effects on p53 and p21 which regulate cell proliferation [37]. TBL1XR1 as an essential compartment of the NCOR/SMRT complex regulates several signaling pathways such as nuclear factor-κB, Wnt/β-catenin, and Notch pathways. the protein encoded by this gene has a role in the control of inflammation, apoptosis, and proliferation [38, 39]. IGF1R as a cell membrane receptor regulates cell proliferation and cell apoptosis through the control of several downstream signaling pathways. More ever IGF1R can control the response to ionizing radiation by regulating the DNA damage response, proliferation,and apoptosis [14, 40]. Among human Argonaute proteins, AGO2 is of great importance due to its endonuclease properties. In addition to the important role of AGO2 in silencing target genes of RNAi, this protein can regulate several cellular processes such as transcription, splicing, polyadenylation, DNA methylation, and DNA repair. This protein can promote DNA repair through the recruitment of RAD51 at ionizing radiation-induced DSB [41, 42]. SPRY4 as a tumor suppressor gene can reduce the proliferation of tumor cells [43]. NAA50 is one Element of the N-alpha-acetyltransferase complex which acetylated the protein with methionine at their N-terminal. This protein has a pivotal role in chromosome condensation [44]. SOX2 as a Stemness marker plays an essential role in the control of apoptosis, DNA damage response, and telomerase function. Some studies indicated that this protein is associated with response to radiation [45, 46]. Transcription factor SP1 through binding to promoter leads to transcription activation of numerous genes. SP1 has an essential role in diverse cellular processes such as proliferation, apoptosis, and DNA damage response. This protein regulates the response to ionizing radiation through accumulation of some critical proteins needed for DSBs repair [47]. Deng et al. in their study indicated that in cervical cancer, SP1 playing role in response to ionizing radiation by targeting the CDK1 as the important determining factor of G2/M checkpoint [48].
In summary, assessment of micro RNAs expression level under low dose ionizing radiation indicated that miR-21 and miR-625 upregulated after exposure to low dose gamma radiation. on the other hand bioinformatics analysis for evaluation of miR-21 and miR-625 target genes indicated that SP1 as the common valid target of miR-21 and miR-625 has the highest degree between hub genes. It seems that miR-21 and miR-625 contribute to the response to acute low-dose gamma radiation by targeting the SP1. However, it is recommended that further study should be carried out on the molecular mechanism of effects of miR-21 and miR-625 in response to low-dose ionizing radiation by targeting the SP1.
Data availability
Data available on request due to privacy/ethical restrictions.
References
Bahreyni-Toossi MT, Vosoughi H, Azimian H, Rezaei AR, Momennezhad M (2018) In vivo exposure effects of (99m)Tc-methoxyisobutylisonitrile on the FDXR and XPA genes expression in human peripheral blood lymphocytes. Asia Oceania J Nucl Med Biol 6(1):32–40
Borrego-Soto G, Ortiz-López R, Rojas-Martínez A (2015) Ionizing radiation-induced DNA injury and damage detection in patients with breast cancer. Genet Mol Biol 38(4):420–432
Morgan WF, Bair WJ (2013) Issues in low dose radiation biology: the controversy continues. A perspective. Radiat Res 179(5):501–510
Mettler F, Huda W, Yoshizumi T, Mahesh M (2008) Effective doses in radiology and diagnostic nuclear medicine: a catalog 1. Radiology 248:254–263
Ouellette MM, Yan Y (2019) Radiation-activated prosurvival signaling pathways in cancer cells. Prec Radiat Oncol 3(3):111–120
Liang X, So YH, Cui J, Ma K, Xu X, Zhao Y et al (2011) The low-dose ionizing radiation stimulates cell proliferation via activation of the MAPK/ERK pathway in rat cultured mesenchymal stem cells. J Radiat Res 52(3):380–386
Pearce MS, Salotti JA, Little MP, McHugh K, Lee C, Kim KP et al (2012) Radiation exposure from CT scans in childhood and subsequent risk of leukaemia and brain tumours: a retrospective cohort study. Lancet (London, England) 380(9840):499–505
Lee JC, West MJ, Khafagi FA (2013) Myocardial perfusion scans. Aust Fam Physician 42(8):564–567
Sun Y, Ma P, Bax JJ, Blom N, Yu Y, Wang Y et al (2003) 99mTc-MIBI myocardial perfusion imaging in myocarditis. Nucl Med Commun 24(7):779–783
Gottschalk A (1969) Technetium-99m in clinical nuclear medicine. Annu Rev Med 20:131–140
Song M, Xie D, Gao S, Bai CJ, Zhu MX, Guan H et al (2020) A biomarker panel of radiation-upregulated miRNA as signature for ionizing radiation exposure. Life (Basel, Switzerland) 10(12):361
O’Brien G, Cruz-Garcia L, Majewski M, Grepl J, Abend M, Port M et al (2018) FDXR is a biomarker of radiation exposure in vivo. Sci Rep 8(1):684
Afshar S, Afshar S, Warden E, Manochehri H, Saidijam M (2019) Application of artificial neural network in miRNA biomarker selection and precise diagnosis of colorectal cancer. IBJ 23(3):175–183
Afshar S, SedighiPashaki A, Najafi R, Nikzad S, Amini R, Shabab N et al (2020) Cross-resistance of acquired radioresistant colorectal cancer cell line to gefitinib and regorafenib. Iran J Med Sci 45(1):50–58
Jovanovic M, Hengartner MO (2006) miRNAs and apoptosis: RNAs to die for. Oncogene 25(46):6176–6187
Bahreini F, Saidijam M, Mousivand Z, Najafi R, Afshar S (2021) Assessment of lncRNA DANCR, miR-145-5p and NRAS axis as biomarkers for the diagnosis of colorectal cancer. Mol Biol Rep 48(4):3541–3547
Moertl S, Mutschelknaus L, Heider T, Atkinson MJ (2016) MicroRNAs as novel elements in personalized radiotherapy. Transl Cancer Res 5:S1262–S1269
Wang Y, Taniguchi T (2013) MicroRNAs and DNA damage response: implications for cancer therapy. Cell Cycle 12(1):32–42
Eckerman K, Harrison J, Menzel HG, Clement CH (2013) ICRP publication 119: compendium of dose coefficients based on ICRP publication 60. Ann ICRP 42(4):e1–e130
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods (San Diego, Calif) 25(4):402–408
Jafari SM, Jordan TJ, Distefano G, Bradley DA, Spyrou NM, Nisbet A et al (2015) Feasibility of using glass-bead thermoluminescent dosimeters for radiotherapy treatment plan verification. Br J Radiol 88(1055):20140804
Nikzad S, Pourkaveh M, Vesal N, Gharekhanloo F (2018) Cumulative radiation dose and cancer risk estimation in common diagnostic radiology procedures. Iran J Radiol. In Press.
Eisenberg MJ, Afilalo J, Lawler PR, Abrahamowicz M, Richard H, Pilote L (2011) Cancer risk related to low-dose ionizing radiation from cardiac imaging in patients after acute myocardial infarction. CMAJ 183(4):430–436
Lumniczky K, Impens N, Armengol G, Candéias S, Georgakilas AG, Hornhardt S et al (2021) Low dose ionizing radiation effects on the immune system. Environ Int 149:106212
Chaudhry MA, Omaruddin RA, Kreger B, de Toledo SM, Azzam EI (2012) Micro RNA responses to chronic or acute exposures to low dose ionizing radiation. Mol Biol Rep 39(7):7549–7558
Puccetti MV, Adams CM, Dan TD, Palagani A, Simone BA, DeAngelis T et al (2019) MicroRNA-21 is required for hematopoietic cell viability after radiation exposure. Int J Radiat Oncol Biol Phys 104(5):1165–1174
Peng J, Lv Y, Wu C (2020) Radiation-resistance increased by overexpression of microRNA-21 and inhibition of its target PTEN in esophageal squamous cell carcinoma. J Int Med Res 48(4):300060519882543
Zhu Y, Yu X, Fu H, Wang H, Wang P, Zheng X et al (2010) MicroRNA-21 is involved in ionizing radiation-promoted liver carcinogenesis. Int J Clin Exp Med 3(3):211–222
Shi Y, Zhang X, Tang X, Wang P, Wang H, Wang Y (2012) MiR-21 is continually elevated long-term in the brain after exposure to ionizing radiation. Radiat Res 177(1):124–128
Gong X, Xu B, Zi L, Chen X (2019) miR-625 reverses multidrug resistance in gastric cancer cells by directly targeting ALDH1A1. Cancer Manage Res 11:6615–6624
Fang W, Fan Y, Fa Z, Xu J, Yu H, Li P et al (2017) microRNA-625 inhibits tumorigenicity by suppressing proliferation, migration and invasion in malignant melanoma. Oncotarget 8(8):13253–13263
He J, Hua J, Ding N, Xu S, Sun R, Zhou G et al (2014) Modulation of microRNAs by ionizing radiation in human gastric cancer. Oncol Rep 32(2):787–793
Salendo J, Spitzner M, Kramer F, Zhang X, Jo P, Wolff HA et al (2013) Identification of a microRNA expression signature for chemoradiosensitivity of colorectal cancer cells, involving miRNAs-320a, -224, -132 and let7g. Radiother Oncol 108(3):451–457
Bae S, Kim K, Cha HJ, Choi Y, Shin SH, An IS et al (2015) Low-dose γ-irradiation induces dual radio-adaptive responses depending on the post-irradiation time by altering microRNA expression profiles in normal human dermal fibroblasts. Int J Mol Med 35(1):227–237
Katoh M, Katoh M (2004) Identification and characterization of human FNBP1L gene in silico. Int J Mol Med 13(1):157–162
Yang L, Cui Y, Sun X, Wang Y (2021) Overexpression of TICRR and PPIF confer poor prognosis in endometrial cancer identified by gene co-expression network analysis. Aging 13(3):4564–4589
Bigi A, Beltrami E, Trinei M, Stendardo M, Pelicci PG, Giorgio M (2016) Cyclophilin D counteracts P53-mediated growth arrest and promotes Ras tumorigenesis. Oncogene 35(39):5132–5143
Zhao Y, Lin H, Jiang J, Ge M, Liang X (2019) TBL1XR1 as a potential therapeutic target that promotes epithelial-mesenchymal transition in lung squamous cell carcinoma. Exp Ther Med 17(1):91–98
Gu J-F, Fu W, Qian H-X, Gu W-X, Zong Y, Chen Q et al (2020) TBL1XR1 induces cell proliferation and inhibit cell apoptosis by the PI3K/AKT pathway in pancreatic ductal adenocarcinoma. World J Gastroenterol 26(25):3586–3602
Valenciano A, Henríquez-Hernández LA, Moreno M, Lloret M, Lara PC (2012) Role of IGF-1 receptor in radiation response. Transl Oncol 5(1):1–9
Gao M, Wei W, Li M-M, Wu Y-S, Ba Z, Jin K-X et al (2014) Ago2 facilitates Rad51 recruitment and DNA double-strand break repair by homologous recombination. Cell Res 24(5):532–541
Li X, Wang X, Cheng Z, Zhu Q (2020) AGO2 and its partners: a silencing complex, a chromatin modulator, and new features. Crit Rev Biochem Mol Biol 55(1):33–53
Zhang E, Han L, Yin D, He X, Hong L, Si X et al (2017) H3K27 acetylation activated-long non-coding RNA CCAT1 affects cell proliferation and migration by regulating SPRY4 and HOXB13 expression in esophageal squamous cell carcinoma. Nucleic Acids Res 45(6):3086–3101
Reddi R, Saddanapu V, Chinthapalli DK, Sankoju P, Sripadi P, Addlagatta A (2016) Human Naa50 protein displays broad substrate specificity for amino-terminal acetylation: detailed structural and biochemical analysis using tetrapeptide library. J Biol Chem 291(39):20530–20538
Rachmadi L, Siregar NC, Kanoko M, Andrijono A, Bardosono S, Suryandari DA et al (2019) Role of cancer stem cell, apoptotic factor, DNA repair, and telomerase toward radiation therapy response in stage IIIB cervical cancer. Oman Med J 34(3):224–230
Shen L, Huang X, Xie X, Su J, Yuan J, Chen X (2014) High expression of SOX2 and OCT4 indicates radiation resistance and an independent negative prognosis in cervical squamous cell carcinoma. J Histochem Cytochem 62(7):499–509
Beishline K, Kelly CM, Olofsson BA, Koduri S, Emrich J, Greenberg RA et al (2012) Sp1 facilitates DNA double-strand break repair through a nontranscriptional mechanism. Mol Cell Biol 32(18):3790–3799
Deng Y-R, Chen X-J, Chen W, Wu L-F, Jiang H-P, Lin D et al (2019) Sp1 contributes to radioresistance of cervical cancer through targeting G2/M cell cycle checkpoint CDK1. Cancer Manage Res 11:5835–5844
Acknowledgements
The study was funded by Vice-chancellor for Research and Technology, Hamadan University of Medical Sciences (No. 9807024907).
Funding
The study was funded by Vice-chancellor for Research and Technology, Hamadan University of Medical Sciences (No. 9807024907).
Author information
Authors and Affiliations
Contributions
SA, SN, MS, and MA conceived and designed the analysis. RM and SA collected the data. SA, RM, and SN contributed to analysis tools. SA, RM, LT, and SN preformed the analysis. SA, MS, LT, MA and SN contributed to the interpretation of the results. SA wrote the manuscript in consultation with all authors.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
Ethical issues (Including plagiarism, informed consent, misconduct, data fabrication and/or falsifcation, double publication and/or submission, redundancy, etc.) have been completely observed by the authors. The ethical protocol of this study was approved by the Ethics Committee of Hamadan University of Medical Sciences. (Ethical code: IR.UMSHA.REC.1398.418) and written informed consent was obtained from all patients to participate in the study.
Consent to participate
The ethical protocol of this study was approved by the Ethics Committee of Hamadan University of Medical Sciences (Ethical code: IR.UMSHA.REC.1398.418) and written informed consent was obtained from all patients to participate in the study.
Consent to publish
Ethical issues (Including plagiarism, informed consent, misconduct, data fabrication and/or falsifcation, double publication and/or submission, redundancy, etc.) have been completely observed by the authors. All authors agreed with the content and that all gave explicit consent to submit and that they obtained consent from the responsible authorities at the institute/organization where the work has been carried out, before the work is submitted.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mahmoudi, R., Saidijam, M., Nikzad, S. et al. Human exposure to low dose ionizing radiation affects miR-21 and miR-625 expression levels. Mol Biol Rep 49, 1321–1327 (2022). https://doi.org/10.1007/s11033-021-06960-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-021-06960-3