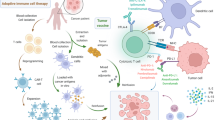
Abstract
Genomic profiling of tumors has become the mainstay for diagnosis, treatment monitoring and a guide to precision medicine. However, in clinical practice, the detection of driver mutations in tumors has several procedural limitations owing to progressive disease and tumor heterogeneity. The current era of liquid biopsy promises a better solution. This diagnostic utility of liquid biopsy has been demonstrated by numerous studies for the detection of cell-free DNA (cfDNA) in plasma for disease diagnosis, prognosis, and prediction. However, cfDNAs are limited in blood circulation and still hurdles to achieve promising precision medicine. Malignant pleural effusion (MPE) is usually detected in advanced lung malignancy, which is rich in tumor cells. Extracellular vesicles and cfDNAs are the two major targets currently explored using MPE. Therefore, MPE can be used as a source of biomarkers in liquid biopsy for investigating tumor mutations. This review focuses on the liquid biopsy approaches for pleural effusion which may be explored as an alternative source for liquid biopsy in lung cancer patients to diagnose early disease progression.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Lung cancer has been estimated to be the most common cause of cancer deaths across the world [1]. The healthcare costs and burden attributed to lung cancer were significant as per the global burden of disease study conducted in 2016 [2]. The estimated 5-year survival rate of 17.8% was lower than that of all the other cancers [3]. More than 50% of the lung malignancies are diagnosed at the advanced stage, which causes a high mortality rate with a five-year survival rate of 4% [4]. Unfortunately, early stages of lung cancers can be asymptomatic, which makes early diagnosis misinterpreted and dismissed immediately [5]. Various invasive and non-invasive diagnostic procedures are used for lung cancer. Non- invasive methods such as computer tomography (CT) scans, low-dose CT scans, chest X-rays and positron emission tomography (PET scans) are used to diagnose non-small-cell lung carcinoma (NSCLC) [6]. Constant monitoring of patients exposed to radiation can, however, lead to radiation-induced cancer [7]. The statistics concerning the prevalence and survival rate suggest the novel screening methods to detect lung cancer in earlier stages in the general population and thereby improve lung cancer survival rates.
Although new technologies are being developed, tissue biopsy and characterization of histology have always remained the gold standard for the detection of NSCLC [7]. Currently, the tissue biopsy method is used to obtain molecular information from the tumors of NSCLC patients [8]. However, being invasive and an obstacle to frequent sampling, the efficacy of the tissue biopsy method is limited. In addition, local tissue sampling can be biased due to tumor heterogeneity and difficult to detect distant metastasis. Advances in genomics technologies are slowly shifting the future of diagnosis, prognosis, and selection of drug regimen towards liquid biopsy [9].
Liquid biopsy method is minimally invasive in comparison to tissue biopsy and can deliver real-time dynamics of lung cancer through utilizing biomarkers in the circulation. It includes cell-free DNA (cfDNA), circulating tumor DNA (ctDNA), extracellular vesicles (EVs), mRNA (messenger RNA), miRNA (microRNA), circulating tumor cells (CTCs) and exosomes [9,10,11].
Even though blood-based liquid biopsies have the potential to produce tumor molecular profiles, challenges remain due to the limited amount of plasma ctDNA [12]. Hence, other body fluids that contain ctDNA including malignant pleural effusion (MPE), ascites, and cerebrospinal fluid is being used as potential alternatives for liquid biopsy samples [13]. MPE is rich in tumor cells and its collection is minimally invasive than a tissue biopsy. Therefore, MPE has been explored as a suitable liquid biopsy specimen [14].
The first-line treatment for metastatic NSCLC often involves platinum-based combination therapy such as carboplatin/paclitaxel with or without immunotherapy. However, in many cases, it is observed that patients with advanced NSCLC developed mutations in the tyrosine kinase domain of the epidermal growth factor receptor (EGFR) which could confer sensitivity to the available targeted therapy for EGFR tyrosine kinase inhibitors (TKI) such as gefitinib and erlotinib [15]. It has also been reported that the EGFR T790M mutation can be detected in ctDNA before clinical progression, thus indicating an early intervention [16]. Hence, testing for mutations in EGFR proves to be an essential step in the treatment decision pathway [15]. The first published case report in 2005 used MPE for targeting EGFR mutation as a liquid biopsy specimen [17, 18]. Cobas® EGFR Mutation Test v2 (Roche Molecular Systems, Pleasanton, CA, USA) was the first liquid biopsy test approved by the U. S. Food and Drug Administration in 2016 [19].
Recent innovations such as next-generation sequencing (NGS), droplet digital polymerase chain reaction (ddPCR), and microfluidic chip-based technologies have developed promising clinical test results, which recommend that liquid biopsy-based technologies will be a mainstay for precision medicine in the future.
This review focuses on the effectiveness of using MPE as a specimen for liquid biopsy for treatment monitoring of lung cancer. The study was framed after an exhaustive literature search from 84 articles collected through PubMed and Google search using the keywords ‘liquid biopsy’, ‘pleural effusion’, ‘malignant pleural effusion’, ‘lung cancer’, ‘cfDNA’, ‘ctDNA’, ‘extracellular vesicles’ ‘miRNA’ in combination with the Boolean operators “AND/OR.”
Malignant pleural effusion
Pleural effusion or “water on the lungs” is the build-up of extra fluid in the space between the lungs and chest wall, which is referred to as pleural space. Pleural effusion is a convenient clinical sample with high clinical diagnostic significance [18]. MPE is a common in metastatic lung cancer and is defined as the presence of malignant cells in the pleural space. The incidence of MPE in the USA is estimated as more than 150,000 cases annually [20]. The common causes of MPE are lung cancer (37.5%), breast cancer (16.8%), and lymphoma (11.5%) [21]. It is assessed that 8 to 15% of lung cancer patients have MPE, in which lung adenocarcinoma is more associated because it grows in the lung periphery and easily invades the pleural cavity which shows the advanced disease stage or progression [22]. In contrast to other invasive techniques like tissue biopsy, MPE is very easy to collect. Moreover, the mutation rate in patients with MPE related to lung adenocarcinoma were much higher compared to mutation rates in surgically resected specimen [21]. It may be an alternative source supplying useful information about the mutation status of the EGFR gene. If EGFR gene mutation determination can be achieved with more attainable pleural effusion samples, then targeted drug therapy will be possible for advanced NSCLC patients, which will contribute to vital clinical and practical value [23].
Diagnosis and collection of malignant pleural effusion
Malignancies in the lung may be asymptomatic or patients may experience cough, dyspnoea and decreased exercise tolerance during early stages. In these cases, MPE could be detected on imaging studies. Standard chest radiographs can detect as little as 50 mL of pleural fluid (PF) on a lateral view, which is used for the diagnosis of MPE [24]. Pleural effusions are evaluated using chest ultrasonography because it detects small volumes (5 mL) of pleural fluid which can identify MPE following thoracentesis and chest-catheter insertion [25]. Patients with suspected MPE can be detected using CT scans and better imaging of soft tissues can be detected with magnetic resonance imaging (MRI) [26]. MRI with triple-echo pulse sequences shows highly sensitive for small effusions thus can differentiate from exudative and transudative effusions [27]. Thoracentesis is a routinely performed minimally invasive method used in patients with PE to relieve their clinical symptoms. It also used for diagnosis and possible further ancillary tests, like molecular studies [28]. Tissue biopsy, which was considered to be the gold standard for the molecular detection and decision making regarding the treatment of NSCLC patients before 2016 might fail sometimes because of multiple reasons like unavailability of the tumor tissue due to its invasiveness or failure in getting enough of tumor tissues for further gene detection [29]. For the patients resistant to targeted therapy, re-biopsy is very difficult because of the suboptimal clinical conditions. On the other hand, liquid biopsy has proven to be comparatively non-invasive, easily accessible and repeatable for conducting molecular profile testing and monitoring drug resistance. The combination of imaging techniques like CT or MRI along with the molecular techniques involving the liquid biopsy can increase the sensitivity, cost-effectiveness and early detection of lung cancers [30].
Studies conducted by using malignant cells (cell blocks or cell pellets) from MPE using real-time PCR concluded with high concordance in comparison with tumor samples in terms of sensitivity and specificity [31]. In contrast, only a few studies have investigated genetic mutations in MPE samples using amplification refractory mutation system (ARMS) and/or PCR [32,33,34] or by next-generation sequencing (NGS) method [35,36,37,38,39].
Circulating cancer biomarkers in malignant pleural effusion
Cell-free DNA
Generally, normal and cancer cells release small fragmented DNA into the circulation, during the course of events such as necrosis and apoptosis [12, 40,41,42]. These fragments of DNA are known as cfDNA irrespective of the cell origin. cfDNA released by cancerous cells is referred to as ctDNA. ctDNA is a good source to investigate tumor mutations, changes in methylation, CNVs, or viral sequences associated with the tumor [43,44,45,46,47]. ctDNA can be isolated in serum and plasma. However, they are reported to be circulating in other body fluids like breast milk, urine, stool, sputum, cerebrospinal fluid, peritoneal fluid, lymphatic fluid, bone marrow, ascites and even in gastric and biliary juice [10].
A study by Kimura et al. indicated that cfDNA in pleural effusion fluid can act as a predictor of EGFR mutations in patients and correlate to the EGFR tyrosine kinase inhibitor responsiveness in patients [39]. Kawahara et al. reported that the cytology cfDNA approach has high sensitivity and specificity (88% and 100%, respectively) when compared to the analysis DNA isolated from tumor tissue, which suggests that this can act as an accurate method for detecting the activating EGFR mutations [48]. Numerous studies have shown the efficacy of using cfDNA from PE and MPE as a source of cfDNA for investigating tumor mutations in lung cancer and advanced lung cancer patients respectively. We have summarized these studies in Table 1.
Extracellular vesicles
EVs are migratory packages of the cells carrying nucleic acids, lipids, and metabolites and helps in the exchange of these materials among the cells. Because of this activity, EVs are considered responsible for cancer development as they have the ability for altering the tumor microenvironment [49]. EVs have a dual lipid membranous coat with double-stranded DNA in tumor exomes which act as a biomarker for the detection of cancer and they are released in several biological fluids, such as plasma, saliva, urine, PE, and bronchoalveolar lavage fluid [50]. A number of studies have reported the presence of the double-stranded DNA in the tumor-derived extracellular vesicles. In a study, Jong et al. demonstrated that DNA derived from EV is considerably very efficient for EGFR genotyping compared to cfDNA in lung malignancy. This suggests that the exosomal or extracellular DNA can act as an alternative biomarker for the detection and diagnosis of cancers [50]. However, only a few studies have demonstrated the translational value of exosomal DNA for the potential benefit as a biomarker in cancer [51,52,53]. The fragments carrying mutations of kirsten rat sarcoma (KRAS) and p53 have been detected in the exosomes from pancreatic cancers [53]. Moreover, the exosomal DNAs also have been reported to have mutations in EGFR and BRAF (V600E) [51]. Microvesicles (MVs) also known as exosomes are released from tumor cells in the circulation and pleural effusion. Park et al. reported that MVs derived from pleural effusion of NSCLC patients can support the development of new liquid biopsy tools [54].
microRNA
EVs are rich in diverse contents such as DNA, mRNA, miRNA, lipids and proteins [55]. miRNAs are a part of non-coding RNAs that are never expressed as proteins but are crucial for regulating the transcription, translation processes and gene coding for the proteins [56]. miRNAs are able to function as an oncogene or as a suppressor [57]. The oncogenic effect of miRNA is due to deletion or silencing of mRNA expression of oncogene inhibitors, silencing of suppressor gene amplification or up-regulation of expression in malignant cells. The absence or lack of miRNA can lead to tumorigenesis in lungs due to severe cellular abnormalities [58]. 52.5% miRNA genes are located in cancer-associated genomic regions or in fragile sites. Mutations occurring in these fragile sites are associated with oncogenesis. Example, miRNA-15a and miRNA-16/1, which are coded by genes located in a fragile site on chromosome 13 (area 14.2) [59, 60]. Reduced or lack of expression of these miRNAs were detected in patients with mantle cell lymphoma (MCL), multiple myeloma (MM), B-cell lymphoma, chronic lymphocytic leukemia (CLL), and prostate cancer [60]. This shows a significant association between abnormal miRNA expression and cancer prognosis [59].
miRNAs are observed to be exclusively expressed in most of the cancers including lung cancer. These have also been reported to be circulating in the blood and serum of patients with lung cancers [61,62,63]. Studies have reported the expression of miRNA from pleural effusion of NSCLC patients [55, 64]. NSCLC patients were found to have a decline in levels of let-7 miRNA. This down-regulation of let-7 expression was reported to be suggestively associated with venous invasion, cancer metastasis, advanced TNM stages, worse prognosis and lower post-operative survival in NSCLC patients [65]. miRNA-520b also plays a role as tumor suppressor gene in the progress of NSCLC. Its expression is downregulated in lung cancer tissue and was negatively associated with tumor development, TNM staging and lymph node metastasis [66]. miRNAs can be used in the differential diagnosis of MPEs and non-PEs. A recent study has shown that miRNAs were found to be altered in MPE and significantly associated with overall survival. In a study done by Wang et al. the low expression of miRNA-93, miRNA-134, miRNA-151, miRNA-345 and high expression of miRNA-100 were associated with poor survival [67]. Other studies have reported that significantly lower expression of miRNA-22, miRNA-134, and miRNA-185, were present in patients with MPE as compared to those with non-MPE [57]. Another study has shown reduced expression of miRNA-198 in MPE-associated lung adenocarcinoma compare to non-malignant effusions [68].
It is found that, 12 miRNAs were overexpressed (miRNA-17, miRNA-21, miR-106, miR-146, miR-155, miR-191, miR-192, miRNA-203, miRNA-205, miRNA-210, miRNA-212, and miRNA-214) between the tumor and normal specimens [69]. A study by Rabinowits et al. concluded that there is potential for diagnosis of lung cancer based on the differences in the sequences of miRNA expression. This study also suggested that circulating exosomal miRNA could be useful as a screening test for lung adenocarcinoma [70]. The upregulation of multiple miRNAs (including miRNA-484, miRNA-320, let-7a, and miRNA-125a-5p) was found in malignant pleural mesothelioma compared to benign asbestos-related pleural effusion, providing a potential role of miRNA in diagnosis and management [71]. Furthermore, the divergent expression levels of miRNA-182 and miRNA-210 are seen in MPEs which is associated with lung adenocarcinoma compared to benign pleural effusion [72].
Liquid biopsy using plasma vs pleural effusion fluid in lung cancer
Many studies have demonstrated that the yield of cfDNA in the supernatant of pleural effusion fluid is comparatively higher than pleural effusion sediment and plasma samples. A study conducted by Lin Tong et al. showed that concentrations of cfDNA in PE supernatants were higher than plasma, which is median: 278.1 ng/ml PE and median: 20.4 ng/ml plasma respectively. PE supernatants (98.4%) are rich in tumor DNA than plasma (87%) and PE sediments (90.5%) [73]. About 93% of the tissue determined driver mutations were observed in PE-cfDNA while only 62% were observed in plasma cfDNA. PE-cfDNA also reported the highest detection of EGFR mutations (71%) whereas it was comparatively as low as 59% in plasma cfDNA [73]. On the other hand, another study reported the level of plasma cfDNA was particularly higher in non-small cell lung carcinoma when associated to benign lung tumors, which acted as the basis to discriminate between NSCLC and benign lung tumors showing 91% sensitivity and 68% specificity [74]. It has also been demonstrated the sensitivity of detection of ctDNA during the early stages of lung cancer was about 50% [74]. Studies have shown that PE-cfDNA can robustly detect EGFR, KRAS and EGFR resistance mutations [75, 76]. A study conducted by Song Z et al. support the potential utility of PE-exoDNA, which can be used as an alternative source for genetic testing [77].
The plasma protein has also been reported for early diagnosis of lung malignancy. On the contrary, the pleural effusion is observed in advance stages of cancer, which makes it unavailable for the detection of lung cancer in an early stage [73]. A combination of 16 driver gene mutations in the ctDNA and 8 circulation plasma proteins called CancerSEEK was evaluated in a study for the early diagnosis of various cancers like lung cancer [30]. This combination included cancer antigen 125 (CA-125), myeloperoxidase (MPO), carcinoembryonic antigen (CEA), cancer antigen 19-9 (CA19-9), hepatocyte growth factor (HGF), pro-lactin (PRL), tissue inhibitor of metalloproteinases 1 (TIMP-1) protein levels, and osteopontin (OPN). The study reported that the sensitivity of CancerSEEK in detecting the presence of early cancers was really high with 70% for median and 60% for lung cancer. This method also distinguished the original organ of cancer effectively [30, 78].
Discussion
Pleural effusion is an extra fluid buildup in the space between the lungs and the chest wall. It is observed in cases of lymphoma and cancers of breast, lung, and ovary [79]. However pleural effusion is also seen in cases other than cancer, such as in tuberculosis. As both the type of pleural effusion fluids (malignant and tuberculous) have similar biochemical profiles, differentiating them is quite difficult. A study reported that adenosine deaminase (ADA) of pleural fluid, which is an enzyme of macrophages and activated T lymphocytes, is sensitive biomarker in discriminating between MPE and tuberculous pleural effusion (TPE). Also, the percentage of polynuclear leukocyte in MPE was reported to be higher than TPE [80]. Out of 5888 MPE samples, the most common cause of MPE was reported to be in lung cancer (35.6%) [81].
Identifying biomarkers in pleural effusion can be a non-invasive approach to screening lung cancers. The identification of makers like EGFR, Kirsten rat sarcoma (KRAS), v- Raf murine sarcoma viral oncogene homolog B (BRAF) and translocation in gene rearrangement rat osteosarcoma (ROS1), anaplastic lymphoma kinase (ALK), can predict the nature of malignancy and assess the suitability of patient for gene-targeted therapy [79]. The importance of EGFR mutations has been known for the indicators for better clinical outcomes in NSCLC patients receiving EGFR TKIs. Interestingly, it is reported that 30–50% of patients developed T790M mutation (threonine 790 with methionine) which is a resistant mutation to EGFR TKIs [48].
Currently, ctDNA is the target of liquid biopsy methods. As noted earlier ctDNA are highly fragmented pieces of DNA from the tumors and their concentration makes about less than 0.1% to over 10% of the total cfDNA [9]. The detection of such a low concentration of DNA is quite challenging, particularly at an early stage. To solve this problem, many recent strategies have been emerged [16]. Several PCR based technologies such as ddPCR and BEAMing (beads, emulsion, amplification, and magnetic) have been developed which show high sensitivity ranging between 1% and 0.001% [16, 82, 83]. However digital PCR techniques are not suitable for larger studies because it may miss substantial information [16]. NGS based platforms such as CAPP-Seq (cancer personalized profiling by deep sequencing, TAm-Seq (tagged amplicon deep sequencing), AmpliSeq, and Safe-Seq (safe sequencing system) could help resolve this issue and allow wide ranging application of liquid biopsy [13, 16, 83, 84]. As discussed in this review, MPE supernatants could be used as an excellent source for biomarkers in lung cancer patients. The investigation of these circulating biomarkers may help in detect early disease progression which could be a vital step towards decreased mortality due to lung cancer.
Conclusion
cfDNA, miRNAs and exosomes offers a better alternative for genomic profiling in malignant pleural effusion when tumor tissues are not available for assessing tumor genomics in advanced lung cancer patients. Moreover, PE-cfDNA could possibly be used for EGFR mutation detection and treatment response as an alternative to plasma-cfDNA in advanced malignancy stage, which suggests that malignant pleural effusion could be used as a potential alternative for liquid biopsy.
Data availability
No datasets were generated or analysed during the current study.
References
Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, Parkin DM, Forman D, Bray F (2015) Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer 136(5):E359–E386. https://doi.org/10.1002/ijc.29210
Vos T, Abajobir AA, Abate KH, Abbafati C, Abbas KM, Abd-Allah F, Abdulkader RS, Abdulle AM, Abebo TA, Abera SF (2017) Global, regional, and national incidence, prevalence, and years lived with disability for 328 diseases and injuries for 195 countries, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet 390(10100):1211–1259. https://doi.org/10.1016/S0140-6736(17)32154-2
Edwards BK, Noone AM, Mariotto AB, Simard EP, Boscoe FP, Henley SJ, Jemal A, Cho H, Anderson RN, Kohler BA (2014) Annual Report to the Nation on the status of cancer, 1975–2010, featuring prevalence of comorbidity and impact on survival among persons with lung, colorectal, breast, or prostate cancer. Cancer 120(9):1290–1314. https://doi.org/10.1002/cncr.28509
Siegel RL, Miller KD, Jemal A (2018) Cancer statistics. Ca Cancer J Clin 68(1):7–30. https://doi.org/10.3322/caac.21442
Sazon D, Santiago SM, Soo Hoo G, Khonsary A, Brown C, Mandelkern M, Blahd W, Williams AJ (1996) Fluorodeoxyglucose-positron emission tomography in the detection and staging of lung cancer. Am J Respir Crit Care Med 153(1):417–421. https://doi.org/10.1164/ajrccm.153.1.8542152
Wender R, Fontham ET, Barrera E Jr, Colditz GA, Church TR, Ettinger DS, Etzioni R, Flowers CR, Scott Gazelle G, Kelsey DK (2013) American cancer society lung cancer screening guidelines. CA Cancer J Clin 63(2):106–17. https://doi.org/10.3322/caac.21172
Tang JH, Chia D (2015) Liquid biopsies in the screening of oncogenic mutations in NSCLC and its application in targeted therapy. Crit Rev Oncog 20(5–6):351–71. https://doi.org/10.1615/CritRevOncog.v20.i5-6.90
Lim M, Kim C-J, Sunkara V, Kim M-H, Cho Y-K (2018) Liquid biopsy in lung cancer: clinical applications of circulating biomarkers (CTCs and ctDNA). Micromachines (Basel) 9(3):100. https://doi.org/10.3390/mi9030100
Mathai RA, Vidya RVS, Reddy BS, Thomas L, Udupa K, Kolesar J, Rao M (2019) Potential utility of liquid biopsy as a diagnostic and prognostic tool for the assessment of solid tumors: implications in the precision oncology. J Clin Med 8(3):373. https://doi.org/10.3390/jcm8030373
Domínguez-Vigil IG, Moreno-Martínez AK, Wang JY, Roehrl MH, Barrera-Saldaña HA (2018) The dawn of the liquid biopsy in the fight against cancer. Oncotarget 9(2):2912–2922. https://doi.org/https://doi.org/10.18632/oncotarget.23131
Pasini L, Ulivi P (2019) Liquid biopsy for the detection of resistance mechanisms in NSCLC: comparison of different blood biomarkers. J Clin Med 8(7):998. https://doi.org/10.3390/jcm8070998
Alix-Panabières C, Pantel K (2016) Clinical applications of circulating tumor cells and circulating tumor DNA as liquid biopsy. Cancer Discov 6(5):479–491. https://doi.org/10.1158/2159-8290.CD-15-1483
Siravegna G, Marsoni S, Siena S, Bardelli A (2017) Integrating liquid biopsies into the management of cancer. Nat Rev Clin Oncol 14(9):531–48. https://doi.org/10.1038/nrclinonc.2017.14
Guo Z, Xie Z, Shi H, Du W, Peng L, Han W, Duan F, Zhang X, Chen M, Duan J (2019) Malignant pleural effusion supernatant is an alternative liquid biopsy specimen for comprehensive mutational profiling. Thorac Cancer 10(4):823–831. https://doi.org/10.1111/1759-7714.13006
Ellison G, Zhu G, Moulis A, Dearden S, Speake G, McCormack R (2013) EGFR mutation testing in lung cancer: a review of available methods and their use for analysis of tumour tissue and cytology samples. J Clin Pathol 66(2):79–89. https://doi.org/10.1136/jclinpath-2012-201194
Palmirotta R, Lovero D, Cafforio P, Felici C, Mannavola F, Pellè E, Quaresmini D, Tucci M, Silvestris F (2018) Liquid biopsy of cancer: a multimodal diagnostic tool in clinical oncology. Ther Adv Med Oncol 10:1758835918794630. https://doi.org/10.1177/1758835918794630
Huang M-J, Lim K-H, Tzen C-Y, Hsu H-S, Yen Y, Huang B-S (2005) EGFR mutations in malignant pleural effusion of non-small cell lung cancer: a case report. Lung Cancer 49(3):413–415. https://doi.org/10.1016/j.lungcan.2005.02.016
Gridelli C, Rossi A, Carbone DP, Guarize J, Karachaliou N, Mok T, Petrella F, Spaggiari L, Rosell R (2015) Non-small-cell lung cancer. Nat Rev Dis Primers 1:15009. https://doi.org/10.1038/nrdp.2015.9
Brown P (2016) The cobas® EGFR mutation test v2 assay. Future Oncol 12(4):451–2. https://doi.org/10.2217/fon.15.311
Keshishyan S, Harris K (2017) Asymptomatic malignant pleural effusion to observe or to manage. J Thorac Dis 9(Suppl 10):S1146-S1147. https://doi.org/https://doi.org/10.21037/jtd.2017.06.52
Wu S, Gow C-H, Yu C, Chang Y, Yang C-H, Hsu Y, Shih J, Lee Y, Yang P (2008) Frequent epidermal growth factor receptor gene mutations in malignant pleural effusion of lung adenocarcinoma. Eur Respir J 32(4):924–30. https://doi.org/10.1183/09031936.00167407
Morgensztern D, Waqar S, Subramanian J, Trinkaus K, Govindan R (2012) Prognostic impact of malignant pleural effusion at presentation in patients with metastatic non–small-cell lung cancer. J Thorac Oncol 7(10):1485–9. https://doi.org/10.1097/JTO.0b013e318267223a
Yeo CD, Kim JW, Kim KH, Ha JH, Rhee CK, Kim SJ, Kim YK, Park CK, Lee SH, Park MS (2013) Detection and comparison of EGFR mutations in matched tumor tissues, cell blocks, pleural effusions, and sera from patients with NSCLC with malignant pleural effusion, by PNA clamping and direct sequencing. Lung Cancer 81(2):207–212. https://doi.org/10.1016/j.lungcan.2013.04.023
Blackmore CC, Black WC, Dallas RV, Crow HC (1996) Pleural fluid volume estimation: a chest radiograph prediction rule. Acad Radiol 3(2):103–9. https://doi.org/10.1016/S1076-6332(05)80373-3
Kocijančič I, Vidmar K, Ivanovi-Herceg Z (2003) Chest sonography versus lateral decubitus radiography in the diagnosis of small pleural effusions. J Clin Ultrasound 31(2):69–74. https://doi.org/10.1002/jcu.10141
Qureshi NR, Gleeson FV (2006) Imaging of pleural disease. Clin Chest Med 27(2):193–213. https://doi.org/10.1016/j.ccm.2006.02.001
Hierholzer J, Luo L, Bittner RC, Stroszczynski C, Schoenfeld N, Dorow P, Loddenkemper R, Grassot A (2000) MRI and CT in the differential diagnosis of pleural disease. Chest 118(3):604–609. https://doi.org/10.1378/chest.118.3.604
Heffner JE, Klein JS (2008) Recent advances in the diagnosis and management of malignant pleural effusions. Mayo Clin Proc. https://doi.org/10.4065/83.2.235
Ilié M, Hofman P (2016) Pros: can tissue biopsy be replaced by liquid biopsy? Transl Lung Cancer Res 5(4):420. https://doi.org/https://doi.org/10.21037/tlcr.2016.08.06
Wu Z, Yang Z, Dai Y, Zhu Q, Chen L-A (2019) Update on liquid biopsy in clinical management of non-small cell lung cancer. Onco Targets Ther 12:5097–9. https://doi.org/10.2147/OTT.S203070
Shin S, Kim J, Kim Y, Cho S-M, Lee K-A (2017) Assessment of real-time PCR method for detection of EGFR mutation using both supernatant and cell pellet of malignant pleural effusion samples from non-small-cell lung cancer patients. Clin Chem Lab Med 55(12):1962–9. https://doi.org/10.1515/cclm-2016-0851
Liu X, Lu Y, Zhu G, Lei Y, Zheng L, Qin H, Tang C, Ellison G, McCormack R, Ji Q (2013) The diagnostic accuracy of pleural effusion and plasma samples versus tumour tissue for detection of EGFR mutation in patients with advanced non-small cell lung cancer: comparison of methodologies. J Clin Pathol 66(12):1065–9. https://doi.org/10.1136/jclinpath-2013-201728
Akamatsu H, Koh Y, Kenmotsu H, Naito T, Serizawa M, Kimura M, Mori K, Imai H, Ono A, Shukuya T (2014) Multiplexed molecular profiling of lung cancer using pleural effusion. J Thorac Oncol 9(7):1048–52. https://doi.org/10.1097/JTO.0000000000000203
Liu D, Lu Y, Hu Z, Wu N, Nie X, Xia Y, Han Y, Li Q, Zhu G, Bai C (2014) Malignant pleural effusion supernatants are substitutes for metastatic pleural tumor tissues in EGFR mutation test in patients with advanced lung adenocarcinoma. PloS One 9(2):e89946. https://doi.org/10.1371/journal.pone.0089946
Buttitta F, Felicioni L, Del Grammastro M, Filice G, Di Lorito A, Malatesta S, Viola P, Centi I, D'Antuono T, Zappacosta R (2013) Effective assessment of egfr mutation status in bronchoalveolar lavage and pleural fluids by next-generation sequencing. Clin Cancer Res 19(3):691–8. https://doi.org/10.1158/1078-0432.CCR-12-1958
Carter J, Miller JA, Feller-Kopman D, Ettinger D, Sidransky D, Maleki Z (2017) Molecular profiling of malignant pleural effusion in metastatic non–small-cell lung carcinoma. The effect of preanalytical factors. Ann Am Thorac Soc 14(7):1169–1176. https://doi.org/10.1513/AnnalsATS.201609-709OC
Liu L, Shao D, Deng Q, Tang H, Wang J, Liu J, Guo F, Lin Y, Peng Z, Mao M (2018) Next generation sequencing-based molecular profiling of lung adenocarcinoma using pleural effusion specimens. J Thorac Dis 10(5):2631–2637. https://doi.org/https://doi.org/10.21037/jtd.2018.04.125
Yang S-R, Lin C-Y, Stehr H, Long SR, Kong CS, Berry GJ, Zehnder JL, Kunder CA (2018) Comprehensive genomic profiling of malignant effusions in patients with metastatic lung adenocarcinoma. J Mol Diagn 20(2):184–94. https://doi.org/10.1016/j.jmoldx.2017.10.007
Kimura H, Fujiwara Y, Sone T, Kunitoh H, Tamura T, Kasahara K, Nishio K (2006) EGFR mutation status in tumour-derived DNA from pleural effusion fluid is a practical basis for predicting the response to gefitinib. Br J Cancer 95(10):1390–95. https://doi.org/10.1038/sj.bjc.6603428
Diaz LA Jr, Bardelli A (2014) Liquid biopsies: genotyping circulating tumor DNA. J Clin Oncol 32(6):579–86. https://doi.org/10.1200/JCO.2012.45.2011
Stroun M, Maurice P, Vasioukhin V, Lyautey J, Lederrey C, Lefort F, Rossier A, Chen XQ, Anker P (2000) The origin and mechanism of circulating DNA. Ann N Y Acad Sci 906(1):161–8. https://doi.org/10.1111/j.1749-6632.2000.tb06608.x
Schwarzenbach H, Hoon DS, Pantel K (2011) Cell-free nucleic acids as biomarkers in cancer patients. Nat Rev Cancer 11(6):426–37. https://doi.org/10.1038/nrc3066
Cheng F, Su L, Qian C (2016) Circulating tumor DNA: a promising biomarker in the liquid biopsy of cancer. Oncotarget 7(30):48832-48841. https://doi.org/https://doi.org/10.18632/oncotarget.9453
Li J, Dittmar RL, Xia S, Zhang H, Du M, Huang CC, Druliner BR, Boardman L, Wang L (2017) Cell-free DNA copy number variations in plasma from colorectal cancer patients. Mol Oncol 11(8):1099–111. https://doi.org/10.1002/1878-0261.12077
Soave A, Chun FK-H, Hillebrand T, Rink M, Weisbach L, Steinbach B, Fisch M, Pantel K, Schwarzenbach H (2017) Copy number variations of circulating, cell-free DNA in urothelial carcinoma of the bladder patients treated with radical cystectomy: a prospective study. Oncotarget 8(34):56398–56407. https://doi.org/10.18632/oncotarget.17657
Husain H, Nykin D, Bui N, Quan D, Gomez G, Woodward B, Venkatapathy S, Duttagupta R, Fung E, Lippman SM (2017) Cell-free DNA from ascites and pleural effusions: molecular insights into genomic aberrations and disease biology. Mol Cancer Ther 16(5):948–55. https://doi.org/10.1158/1535-7163.MCT-16-0436
Li Z, Guo X, Tang L, Peng L, Chen M, Luo X, Wang S, Xiao Z, Deng Z, Dai L (2016) Methylation analysis of plasma cell-free DNA for breast cancer early detection using bisulfite next-generation sequencing. Tumour Biol 37(10):13111–9. https://doi.org/10.1007/s13277-016-5190-z
Kawahara A, Fukumitsu C, Taira T, Abe H, Takase Y, Murata K, Yamaguchi T, Azuma K, Ishii H, Takamori S (2015) Epidermal growth factor receptor mutation status in cell-free DNA supernatant of bronchial washings and brushings. Cancer Cytopathol 123(10):620–8. https://doi.org/10.1002/cncy.21583
Pathan M, Fonseka P, Chitti SV, Kang T, Sanwlani R, Van Deun J, Hendrix A, Mathivanan S (2019) Vesiclepedia 2019: a compendium of RNA, proteins, lipids and metabolites in extracellular vesicles. Nucleic Acids Res 47(D1):D516–D9. https://doi.org/10.1093/nar/gky1029
Lee JS, Hur JY, Kim IA, Kim HJ, Choi CM, Lee JC, Kim WS, Lee KY (2018) Liquid biopsy using the supernatant of a pleural effusion for EGFR genotyping in pulmonary adenocarcinoma patients: a comparison between cell-free DNA and extracellular vesicle-derived DNA. BMC cancer 18(1):1236. https://doi.org/10.1186/s12885-018-5138-3
Thakur BK, Zhang H, Becker A, Matei I, Huang Y, Costa-Silva B, Zheng Y, Hoshino A, Brazier H, Xiang J (2014) Double-stranded DNA in exosomes: a novel biomarker in cancer detection. Cell Res 24(6):766–9. https://doi.org/10.1038/cr.2014.44
Vanni I, Alama A, Grossi F, Dal Bello MG, Coco S (2017) Exosomes: a new horizon in lung cancer. Drug Discov Today 22(6):927–36. https://doi.org/10.1016/j.drudis.2017.03.004
Kahlert C, Melo SA, Protopopov A, Tang J, Seth S, Koch M, Zhang J, Weitz J, Chin L, Futreal A (2014) Identification of double-stranded genomic DNA spanning all chromosomes with mutated KRAS and p53 DNA in the serum exosomes of patients with pancreatic cancer. J Biol Chem 289(7):3869–75. https://doi.org/10.1074/jbc.C113.532267
Park JO, Choi DY, Choi DS, Kim HJ, Kang JW, Jung JH, Lee JH, Kim J, Freeman MR, Lee KY (2013) Identification and characterization of proteins isolated from microvesicles derived from human lung cancer pleural effusions. Proteomics 13(14):2125–2134. https://doi.org/10.1002/pmic.201200323
Lin J, Wang Y, Zou Y-Q, Chen X, Huang B, Liu J, Xu Y-M, Li J, Zhang J, Yang W-M (2016) Differential miRNA expression in pleural effusions derived from extracellular vesicles of patients with lung cancer, pulmonary tuberculosis, or pneumonia. Tumor Biol 37(12):15835–45. https://doi.org/10.1007/s13277-016-5410-6
Ling H, Fabbri M, Calin GA (2013) MicroRNAs and other non-coding RNAs as targets for anticancer drug development. Nat Rev Drug Discov 12(11):847–65. https://doi.org/10.1038/nrd4140
Wojczakowski W, Kobylarek D, Lindner J, Limphaibool N, Kaczmarek M (2019) MicroRNAs–novel biomarkers for malignant pleural effusions. Contemp Oncol 23(3):133–140. https://doi.org/10.5114/wo.2019.89241
Hayashita Y, Osada H, Tatematsu Y, Yamada H, Yanagisawa K, Tomida S, Yatabe Y, Kawahara K, Sekido Y, Takahashi T (2005) A polycistronic microRNA cluster, miR-17-92, is overexpressed in human lung cancers and enhances cell proliferation. Cancer Res 65(21):9628–32. https://doi.org/10.1158/0008-5472.CAN-05-2352
Calin GA, Sevignani C, Dumitru CD, Hyslop T, Noch E, Yendamuri S, Shimizu M, Rattan S, Bullrich F, Negrini M (2004) Human microRNA genes are frequently located at fragile sites and genomic regions involved in cancers. Proc Natl Acad Sci USA 101(9):2999–3004. https://doi.org/10.1073/pnas.0307323101
Calin GA, Dumitru CD, Shimizu M, Bichi R, Zupo S, Noch E, Aldler H, Rattan S, Keating M, Rai K (2002) Frequent deletions and down-regulation of micro-RNA genes miR15 and miR16 at 13q14 in chronic lymphocytic leukemia. Proc Natl Acad Sci USA 99(24):15524–9. https://doi.org/10.1073/pnas.242606799
Davis-Dusenbery BN, Hata A (2010) MicroRNA in cancer: the involvement of aberrant microRNA biogenesis regulatory pathways. Genes Cancer 1(11):1100–14. https://doi.org/10.1177/1947601910396213
Etheridge A, Lee I, Hood L, Galas D, Wang K (2011) Extracellular microRNA: a new source of biomarkers. Mutat Res 717(1–2):85–90. https://doi.org/10.1016/j.mrfmmm.2011.03.004
Resnick KE, Alder H, Hagan JP, Richardson DL, Croce CM, Cohn DE (2009) The detection of differentially expressed microRNAs from the serum of ovarian cancer patients using a novel real-time PCR platform. Gynecol Oncol 112(1):55–9. https://doi.org/10.1016/j.ygyno.2008.08.036
Wang Y, Xu Y-M, Zou Y-Q, Lin J, Huang B, Liu J, Li J, Zhang J, Yang W-M, Min Q-H (2017) Identification of differential expressed PE exosomal miRNA in lung adenocarcinoma, tuberculosis, and other benign lesions. Medicine 96(44):e8361. https://doi.org/10.1097/MD.0000000000008361
Takamizawa J, Konishi H, Yanagisawa K, Tomida S, Osada H, Endoh H, Harano T, Yatabe Y, Nagino M, Nimura Y (2004) Reduced expression of the let-7 microRNAs in human lung cancers in association with shortened postoperative survival. Cancer Res 64(11):3753–6. https://doi.org/10.1158/0008-5472.CAN-04-0637
Zhang L, Yu S (2018) Role of miR-520b in non-small cell lung cancer. Exp Ther Med 16(5):3987–95. https://doi.org/10.3892/etm.2018.6732
Wang T, Lv M, Shen S, Zhou S, Wang P, Chen Y, Liu B, Yu L, Hou Y (2012) Cell-free microRNA expression profiles in malignant effusion associated with patient survival in non-small cell lung cancer. PloS One 7(8):e43268. https://doi.org/10.1371/journal.pone.0043268
Han HS, Jo YN, Lee JY, Choi SY, Jeong Y, Yun J, Lee OJ (2014) Identification of suitable reference genes for the relative quantification of microRNAs in pleural effusion. Oncol Lett 8(4):1889–95. https://doi.org/10.3892/ol.2014.2404
Yanaihara N, Caplen N, Bowman E, Seike M, Kumamoto K, Yi M, Stephens RM, Okamoto A, Yokota J, Tanaka T (2006) Unique microRNA molecular profiles in lung cancer diagnosis and prognosis. Cancer Cell 9(3):189–98. https://doi.org/10.1016/j.ccr.2006.01.025
Rabinowits G, Gerçel-Taylor C, Day JM, Taylor DD, Kloecker GH (2009) Exosomal microRNA: a diagnostic marker for lung cancer. Clin Lung Cancer 10(1):42–6. https://doi.org/10.3816/CLC.2009.n.006
Ak G, Tomaszek SC, Kosari F, Metintas M, Jett JR, Metintas S, Yildirim H, Dundar E, Dong J, Aubry MC (2015) MicroRNA and mRNA features of malignant pleural mesothelioma and benign asbestos-related pleural effusion. Biomed Res Int 2015:635748. https://doi.org/10.1155/2015/635748
Tamiya H, Mitani A, Saito A, Ishimori T, Saito M, Isago H, Jo T, Yamauchi Y, Tanaka G, Nagase T (2018) Exosomal MicroRNA expression profiling in patients with lung adenocarcinoma-associated malignant pleural effusion. Anticancer Res 38(12):6707-6714. https://doi.org/10.21873/anticanres.13039.
Tong L, Ding N, Tong X, Li J, Zhang Y, Wang X, Xu X, Ye M, Li C, Wu X (2019) Tumor-derived DNA from pleural effusion supernatant as a promising alternative to tumor tissue in genomic profiling of advanced lung cancer. Theranostics 9(19):5532–41. https://doi.org/10.1097/MD.0000000000008361
Szpechcinski A, Rudzinski P, Kupis W, Langfort R, Orlowski T, Chorostowska-Wynimko J (2016) Plasma cell-free DNA levels and integrity in patients with chest radiological findings: NSCLC versus benign lung nodules. Cancer Lett 374(2):202–7. https://doi.org/10.1016/j.canlet.2016.02.002
Hummelink K, Muller M, Linders TC, van der Noort V, Nederlof PM, Baas P, Burgers S, Smit EF, Meijer GA, van den Heuvel MM (2019) Cell-free DNA in the supernatant of pleural effusion can be used to detect driver and resistance mutations, and can guide tyrosine kinase inhibitor treatment decisions. ERJ Open Res 5(1):00016–2019. https://doi.org/10.1183/23120541.00016-2019
Villatoro S, Mayo-de-las-Casas C, Jordana-Ariza N, Viteri-Ramírez S, Garzón-Ibañez M, Moya-Horno I, García-Peláez B, González-Cao M, Malapelle U, Balada-Bel A (2019) Prospective detection of mutations in cerebrospinal fluid, pleural effusion and ascites of advanced cancer patients to guide treatment decisions. Mol Oncol 13(12):2633–2645. https://doi.org/10.1002/1878-0261.12574
Song Z, Cai Z, Yan J, Shao YW, Zhang Y (2019) Liquid biopsies using pleural effusion-derived exosomal DNA in advanced lung adenocarcinoma. Transl Lung Cancer Res 8(4):392–400. https://doi.org/10.21037/tlcr.2019.08.14
Cohen JD, Li L, Wang Y, Thoburn C, Afsari B, Danilova L, Douville C, Javed AA, Wong F, Mattox A (2018) Detection and localization of surgically resectable cancers with a multi-analyte blood test. Science 359(6378):926–930. https://doi.org/10.1126/science.aar3247
Awadallah SF, Bowling MR, Sharma N, Mohan A (2019) Malignant pleural effusion and cancer of unknown primary site: a review of literature. Ann Transl Med 7(15):353. https://doi.org/10.21037/atm.2019.06.33
Darooei R, Sanadgol G, Gh-Nataj A, Almasnia M, Darivishi A, Eslaminejad A, Raoufy MR (2017) Discriminating tuberculous pleural effusion from malignant pleural effusion based on routine pleural fluid biomarkers, using mathematical methods. Tanaffos 16(2):157–165
Johnston WW (1985) The malignant pleural effusion. A review of cytopathologic diagnoses of 584 specimens from 472 consecutive patients. Cancer 56(4):905-909. https://doi.org/https://doi.org/10.1002/1097-0142(19850815)56:4<905::aid-cncr2820560435>3.0.co;2-u
Diehl F, Schmidt K, Choti MA, Romans K, Goodman S, Li M, Thornton K, Agrawal N, Sokoll L, Szabo SA (2008) Circulating mutant DNA to assess tumor dynamics. Nat Med 14(9):985–990. https://doi.org/10.1038/nm.1789
Perakis S, Speicher MR (2017) Emerging concepts in liquid biopsies. BMC Med 15(1):75. https://doi.org/10.1186/s12916-017-0840-6
Han X, Wang J, Sun Y (2017) Circulating tumor DNA as biomarkers for cancer detection. Genom Proteom Bioinform 15(2):59–72. https://doi.org/10.1016/j.gpb.2016.12.004
Acknowledgements
Gayathri Baburaj would like to acknowledge the support from DST-INSPIRE Fellowship, Department of Science and Technology, Government of India, New Delhi, India [DST/INSPIRE Fellowship/2018/IF180737].
Funding
No sources of funding were used to assist in the preparation of this article.
Author information
Authors and Affiliations
Contributions
The first draft of the manuscript was written by GB, RRD and MR. KSU, PP, MM and JK critically reviewed and edited the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Baburaj, G., Damerla, R.R., Udupa, K.S. et al. Liquid biopsy approaches for pleural effusion in lung cancer patients. Mol Biol Rep 47, 8179–8187 (2020). https://doi.org/10.1007/s11033-020-05869-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-020-05869-7