Abstract
Adiponectin is an adipokine hormone that influences glucose utilization, insulin sensitivity and energy homeostasis. To investigate the effect of adiponectin on lipids deposition in broilers, rosiglitazone and dexamethasone were used to treat broilers. A total of 120 twenty-three-day-old male Cobb broilers were randomly divided into 3 groups for 3 weeks of drug treatment. Serum adiponectin level and fatty acid composition in muscles were measured. Adiponectin, adiponectin receptors (adipoR1, adipoR2) and lipid metabolism-related genes expression levels in muscles were measured using real-time PCR. Western blot was used to measure the expression levels of lipid metabolism-related proteins and the phosphorylation of p38 mitogen-activated protein kinase (p38 MAPK)/activating transcription factor 2 (ATF2) pathway marker proteins. Rosiglitazone increased serum adiponectin concentration and the expression levels of adiponectin and adipoR1 (P < 0.05), while dexamethasone had the opposite effect. Intramuscular fat content, total fatty acid, saturated fatty acid and monounsaturated fatty acid reduced in the rosiglitazone treatment group (P < 0.05). In the rosiglitazone treatment group, the expression levels of lipogenic genes and proteins decreased in the muscles, whereas the expression levels of lipolysis genes increased. Meanwhile, the phosphorylation levels of p38MAPK and ATF2 increased with supplementation of rosiglitazone and decreased in the dexamethasone treatment group (P < 0.01). These results indicated that rosiglitazone and dexamethasone could regulate adiponectin expression in muscle of broilers and adiponectin had an anti-lipogenic effect by p38 MAPK/ATF2 signaling pathway.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Adiponectin—also termed adipocyte complement-related protein of 30 kDa (ACRP30), adipose most abundant gene transcript 1 (apM1), gelatin-binding protein of 28 kDa (GBP28) or AdipoQ—is a 30 kDa adipokine hormone that is secreted primarily from adipose tissue. Adiponectin consists of an N-terminal collagenous domain and a C-terminal globular domain that acts as the receptor binding effector [1, 2]. Adiponectin works by binding to two specific receptors, adipoR1 and adipoR2 [3]. Although adiponectin was once thought to be exclusively secreted from adipose tissue, the expression of adiponectin and its receptors have recently been detected in multiple tissues of broilers, such as the liver, adenohypophysis, kidney, skeletal muscle and adipose tissue [4].
Intramuscular fat (IMF) content is an important indicator of meat quality. Study has shown that a higher IMF content had a positive effect on the juiciness, palatability and tenderness of meat [5]. In addition, the fatty acid composition of chicken meat had received increasing attention owing to its implications in human health and product quality [6]. Recent studies have shown that the concentration of adiponectin in plasma was inversely related to the deposition of chicken belly fat [7, 8]. Adiponectin could also increase fatty acid oxidation in skeletal muscle cells [9]. However, there has been no evidence demonstrating the relationship between adiponectin and fatty acid composition in muscle.
p38 mitogen-activated protein kinase (MAPK) is a major kinase in the MAPK family and plays essential role in regulating cell proliferation, inflammation and immune responses. Recent studies suggested that p38 MAPK acted as an essential mediator in regulating adiponectin-induced glucose uptake and fatty acid oxidation in C2C12 myotubes and played a negative role in adipogenesis via inhibition of CCAAT/enhancer binding protein beta (C/EBPβ) and peroxisome proliferator-activated receptor gamma (PPARγ) transcriptional activities [3, 10, 11]. Activating transcription factor 2 (ATF2), a ubiquitously expressed member of the cyclic adenosine monophosphate (cAMP)-responsive element binding protein family of basic region leucine-zipper transcription factors, has been implicated in multiple responses in mammalian cells by regulating the expression of a broad spectrum of target genes [12]. Maekawa et al. [13] (2007) indicated that ATF-2 family transcription factors play a role in adipocyte differentiation and fat storage. It is still not clear, however, whether the p38 MAPK signaling pathway involved in the process of adiponectin induced lipids deposition in muscle of broilers.
Rosiglitazone is an antidiabetic drug in the thiazolidinedione class of drugs. Many studies in animal and human populations have shown that peroxisome proliferatoractivated receptor (PPAR) agonists could increase the expression of adiponectin and its receptors [14, 15]. Dexamethasone is a potent synthetic member of the glucocorticoid class of steroid drugs. Previous studies have demonstrated that glucocorticoids inhibited adiponectin in humans, both when administered exogenously to healthy subjects and in cases of endogenous cortisol hyperproduction [16].
The aim of the present study was to verify the effect of rosiglitazone and dexamethasone on adiponectin expression, and to determine the molecular mechanisms that adiponectin regulate lipids deposition in muscle of broilers.
Materials and methods
Experimental animals and diets
The experiment was conducted using a total of 120 commercial male Cobb broiler chicks. The chicks were exposed to light for 24 h on the first day of the experiment, 23 h on the second and third days, then 16 h thereafter. The temperature was initially set at 32 °C on the first day and was progressively reduced by 1.7 °C each week to reach 23 °C at 35 days of age. The chicks were allowed ad libitum access to the experimental diets and water. The broilers were managed according to the Cobb broiler management guide. Body weight and feed consumption were recorded at the end of each week. The basal diets were formulated to meet or exceed NRC (1994) requirements for broilers. All animal researches were approved by the Institutional Animal Care and Use Committee of Northwest A&F University.
Treatment and sample collection
23 days after hatching, the broilers were randomly allotted by body weight to one of three groups (24 replicate cages of 5 chicks per cage) in a completely randomized design. One group of broilers (control group, CON) was given normal saline solution through an intraperitoneal injection, one group (rosiglitazone group, ROS) received a 0.5 mg/kg BW day dose of rosiglitazone by oral administration and was injected with normal saline solution and one group (dexamethasone group, DEX) was injected with a 1 mg/kg BW day dose of dexamethasone.
On day 44 of the trial, two broilers from each cage were selected according to average body weight within the cage following a 12-h fasting, weighed individually, killed by cervical dislocation, and then immediately bled. Fresh blood was collected into 10 ml plastic centrifuge tubes, left overnight at 4 °C, and centrifuged at 2,500×g at 4 °C for 10 min. Serum was collected for assessment of the concentration of adiponectin. Muscle from the breast and leg were immediately separated and stored at −80 °C for assessment of gene expression, protein level and IMF content.
Serum adiponectin
Serum adiponectin concentration was measured at absorption at 450 nm by use of an avian adiponectin ELISA kit (USCN Life Corporation, USA).
IMF determination
Intramuscular fat determination was determined on duplicate 1.5 g samples of breast and leg muscle. After thawing at 4 °C for 48 h, adhering adipose and connective tissue were removed from the muscle then freeze-dried overnight. Petroleum ether fat extractions were conducted for 8 h on the resultant dried product using a Soxtec Extraction System, and then the extracted fat was dried for 1 h at 105 °C. The IMF content was showed on freeze-dried basis.
Fatty acid (FA) composition in muscle
In preparation for the analysis of FA composition, samples of breast and leg meat (5 g each) were freeze-dried and then ground. Methyl heptadecanoate was dissolved into n-hexane (1 mg/ml) as an internal standard. Methylesters of the FA were prepared [17]; samples were analyzed according to Qi et al. [6]. Composition was expressed as the absolute weight in the freeze-dried sample.
RNA isolation, cDNA synthesis and Quantitative PCR
RNA was extracted using a TRIpure Reagent kit (Takara), and first strand cDNA was synthesized using a TM First Strand cDNA Synthesis Kit (Fermentas). All PCR primers were synthesized by Shanghai Sangon Biological Engineering Technology and Service Co., Ltd. Primer sequences for peroxisome proliferator activated receptor (PPARγ), acetyl-CoA carboxylase (ACC), fatty acid synthase (FAS), lipoprotein lipase (LPL), adipose triglyceride lipase (ATGL) and adipocyte fatty acid-binding protein (A-FABP) genes are shown in Table 1. Glyceraldehyde 3-phosphate de-hydrogenase (GAPDH) and 18S RNA were used as internal control for PCR amplification. PCR amplification was performed using LineGene (Bioer). Quantitative PCR was performed in 25 μl reactions, each containing 12.5 μl of SYBR Premix EX Taq (Takara), 0.5 μl each of upstream and downstream primer, 1.0 μl of cDNA, and 10.5 μl of deionized water. The reaction mixtures were incubated for the initial denaturation at 95 °C for 10 min, followed by 40 cycles, with each cycle consisting of 95 °C for 15 s and 60 °C for 1 min. The levels of mRNAs expression were normalized to 18S RNA and GAPDH. The expression of genes was analyzed using the 2−ΔΔCt method [18].
Western blot
Muscle extracts were prepared by placing muscle in 500 μl of lysis buffer for 30 min at 4 °C. The lysates were clarified by centrifugation at 10,000×g for 10 min at 4 °C, and the supernatants were used to assay protein levels. Total protein extracts (0.05 g) were separated by electrophoresis on 12 and 5 % SDS-PAGE gels using a slab gel apparatus and then transferred to PVDF nitrocellulose membranes. After transfer, the membranes were blocked in blocking buffer for 2 h at room temperature. The membranes were then incubated with primary antibodies against GAPDH, PPARγ, C/EBPα, FAS, ATGL (purchased from Santa Cruz) and p38MAPK, phospho-p38MAPK (pT180/pY182), ATF2, phospho-ATF2 (pT71) (purchased from Epitomic) at 4 °C overnight, washed 3 times with TBST buffer, and incubated with the appropriate HRP-conjugated secondary antibodies from Santa Cruz for 2 h at room temperature. Proteins were visualized using chemiluminescent peroxidase substrate (Millipore), and then the blots were quantified using a ChemiDoc XRS system (Bio-Rad) and the analysis software Quantity One (Bio-Rad).
Statistical analysis
Statistical calculations were performed in SAS v8.0 (SAS Institute, Cary, NC, USA). The statistical significance of a relationship was determined using the ANOVA regression table. Comparisons among individual means were made by Duncan’s multiple comparison after ANOVA. The data were presented as the mean ± SD, P < 0.05 was considered to be statistically significant, and P < 0.01 was considered to be extremely significant.
Results
Serum adiponectin and the expression levels of adiponectin and its receptors in muscles
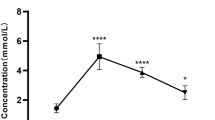
As shown in Fig. 1a, serum adiponectin increased significantly (from 10.85 ± 1.85 to 15.25 ± 2.76 μg/ml, P < 0.01) after 3 weeks of treatment with rosiglitazone and decreased significantly (from 10.85 ± 1.81 to 7.50 ± 0.83 μg/ml, P < 0.01) with dexamethasone injections. The expression level of adiponectin in both breast and leg muscles showed the same trend, with expression increasing significantly (P < 0.01) when supplemented with rosiglitazone and decreasing significantly (P < 0.01) in broilers treated with dexamethasone (Fig. 1b). The expression level of adipoR1 increased significantly in the rosiglitazone treatment group (P < 0.01), while remarkably decreasing in the dexamethasone treatment group (P < 0.01) (Fig. 1c). AdipoR2 was not significantly changed in breast muscle of treated broilers (P > 0.05), and only significantly increased in leg muscle in the rosiglitazone treatment group (P < 0.05) (Fig. 1d).
Adiponectin in serum and relative mRNA expression level of the adiponectin gene in leg and breast muscle of broilers treated with rosiglitazone (ROS, 0.5 mg/kg BW day) and dexamethasone (DEX, 1 mg/kg BW day). a Adiponectin content in serum (n = 10). Adiponectin and its receptor gene’s relative mRNA expression levels in muscles (n = 10). b adiponectin, c AdipoR1, and d AdipoR2. Values are mean ± SD. CON control group. In the same tissue, compare to control group, * means significant at P < 0.05 level, ** means significant at P < 0.01 level
Growth performance and fat deposition in muscle
Growth performance was recorded during the experiment. Figure 2 a showed that the average final weight of broiler in dexamethasone treatment group was significantly lower than the other two groups (P < 0.01). In rosiglitazone treatment group, daily weight gain of broiler was more than the control (P > 0.05), while daily weight gain in dexamethasone treatment group was only 46.3 % of the control (P < 0.01) (Fig. 2b). However, there was no significantly different in feed intake among three groups (P > 0.05) (Fig. 2c). As a result, feed conversion in dexamethasone treatment group was significant higher than the control group (P < 0.01) (Fig. 2d). The IMF content in muscle was also detected. As shown in Fig. 2e, rosiglitazone significantly decreased IMF content in both breast muscle and leg muscle (P < 0.01), while IMF content increased in dexamethasone treatment group (P < 0.05).
Performance of broilers treated with rosiglitazone (ROS, 0.5 mg/kg BW day) and dexamethasone (DEX, 1 mg/kg BW day). a Average final weight (n = 40). b Average daily gain (n = 40). c Average feed intake (n = 40). d Feed conversion (n = 40). e IMF (n = 10). Values are mean ± SD. CON control group. In the same tissue, compare to control group, * means significant at P < 0.05 level, ** means significant at P < 0.01 level
Fatty acid composition of breast and leg muscles
Fatty acid compositions of breast and leg muscles were affected by treatment with rosiglitazone and dexamethasone (Table 2). A trend toward decreasing proportions of saturated fatty acid and monounsaturated fatty acid was observed in broilers that were supplemented with rosiglitazone, while increasing proportions were observed in the dexamethasone treatment group. This trend was particularly apparent for palmitic acid (C16:0), stearic acid (C18:0), lignoceric acid methyl ester (C24:0) and oleic acid (C18:1n9).There was no significant difference in polyunsaturated fatty acid in breast or leg muscle among the three treatment groups (P > 0.05). However, the ratio of polyunsaturated fatty acid (PUFA)/saturated fatty acid (SFA) increased in the rosiglitazone treatment group and decreased in the dexamethasone treatment group, in both the breast and leg muscles.
Abundance of mRNA and proteins related to lipid metabolism in muscles
To gain a comprehensive understanding of the molecular mechanisms underlying the effects of adiponectin, the expression levels of PPARγ, ACC, FAS, LPL, ATGL and A-FABP mRNA and part of proteins were detected in breast and leg muscles. As shown in Fig. 3, rosiglitazone significantly increased PPARγ and ATGL mRNA levels, while decreased the expression levels of FAS and A-FABP in both breast and leg muscles (P < 0.01); dexamethasone significantly increased the expression levels of PPARγ (P < 0.05), ACC (P < 0.05) and A-FABP genes (P < 0.01), while significantly decreased the expression level of ATGL mRNA in both breast and leg muscles. Western blot analysis demonstrated that rosiglitazone significantly increased the expression levels of PPARγ and ATGL proteins in muscles, while decreased the expression level of FAS protein (P < 0.01). Dexamethasone had the opposite effect to rosiglitazone on protein expression.
Expression levels of lipid metabolism-related genes and proteins in breast and leg muscles of broilers treated with rosiglitazone (Ros, 0.5 mg/kg BW day) and dexamethasone (Dex, 1 mg/kg BW day). a Expression levels of lipid metabolism-related genes (n = 10). b Lipid metabolism related proteins in breast muscle (n = 6). c Lipid metabolism related proteins in leg muscle (n = 6). Values are mean ± SD. CON control group. In the same tissue, compare to control group, * means significant at P < 0.05 level, ** means significant at P < 0.01 level
Effect of p38 MAPK/ATF2 signaling pathway in broiler muscle
As shown in Fig. 4, the phosphorylation levels of p38 MAPK and its downstream target-ATF2 were activated by rosiglitazone (P < 0.01) both in breast muscle and leg muscle, while dexamethasone reduced the phosphorylation levels of p38 MAPK and ATF2 in muscles (P < 0.01).
Effect of the phosphorylation levels of the maker proteins in p38 MAPK/ATF2 pathway in breast and leg muscles of broilers treated with rosiglitazone (Ros, 0.5 mg/kg BW day) and dexamethasone (Dex, 1 mg/kg BW day). Total p38MAPK, phospho-p38MAPK (pT180/pY182), total ATF2 and phospho-ATF2 (pT71) were immunoblotted (n = 3). CON, control group. Compare to control group, * means significant at P < 0.05 level, ** means significant at P < 0.01 level
Discussion
Recent studies from several independent laboratories demonstrated that rosiglitazone can promote the secretion of plasma adiponectin in humans and mice [19]. In our study, the data showed that rosiglitazone promoted both the secretion of adiponectin and adiponectin gene expression in the muscles of broilers. Furthermore, rosiglitazone significantly enhanced adipoR1 gene expression but seemed to have no effect on adipoR2 gene expression. Researchers have shown that the PPARγ activator had different effects on adiponectin receptors in various cells or tissues [20, 21]. It has been demonstrated that glucocorticoids cause insulin resistance in vivo [22]. In the present study, we showed that dexamethasone decreased adiponectin gene expression in the muscles of broilers. Dexamethasone significantly inhibited adipoR1 gene expression but had no effect on adipoR2 gene expression.
Body fat is indispensable for maintaining life, growth and production in poultry. A major finding in the present study was that adiponectin was not only inversely related to broiler abdomen and subcutaneous fat deposition (data not shown), but also reduced the IMF content of both breast and leg muscles. The mechanisms which adiponectin exerts such effects involved an increase in fatty acid oxidation in muscle. To further study the function of adiponectin on fat deposition in muscle, we determined the fatty acid composition of breast and leg muscles in broilers treated with rosiglitazone and dexamethasone. Our results demonstrated that total FA, SFAs and MUFAs were all reduced by the rosiglitazone supplement and increased in the dexamethasone treatment group. Although the ratio of PUFA/SFA increased in the rosiglitazone treatment group and decreased in the dexamethasone treatment group in both breast and leg muscles, the absolute content of polyunsaturated fatty acid did not change. In recent years, consumers have become increasingly concerned about poultry meat quality. Our research showed that adiponectin could reduce IMF deposition in both breast and leg muscles. As we know, the content of IMF has become an important indicator of meat quality and plays a critical role in consumer acceptance of fresh meat. Previous studies showed that IMF was associated with flavor and juiciness [23]. So serum adiponectin can be termed as a key candidate factor to regulate muscle quality in chicken.
Acetyl-CoA carboxylase is the first committed enzyme in fatty acid synthesis. As a central enzyme in lipogenesis, fatty acid synthase was identified as a candidate gene for determining body fat [24]. Previous studies reported that adiponectin decreased the activities of two key enzymes involved in fatty acid synthesis in fatty liver, including ACC and FAS [25]. Our study detected high levels of ACC gene expression when adiponectin was suppressed by dexamethasone; additionally, FAS gene and protein expression were inhibited by the increase in adiponectin expression that was stimulated in muscles by rosiglitazone. The lipolytic processing of triglyceride-rich lipoproteins by LPL is the central event in plasma lipid metabolism, providing lipids for storage in adipose tissue and fuel for vital organs, such as the heart. Lipoprotein lipase is synthesized and secreted by myocytes and adipocytes, but it migrates into the lumen of capillaries where it hydrolyzes lipoprotein triglycerides [26]. Adipose triglyceride lipase is an important factor involved in fat catabolism in adipose tissue, which catalyzes the first step in adipocyte and muscle triglyceride hydrolysis. Qiao et al. [27] reported that adiponectin suppressed hormone-sensitive lipase activation without altering adipose triglyceride lipase and CGI-58 expression in adipocytes. However, in our results, the expression level of the ATGL protein was positively correlated with adiponectin in muscle. Adipocyte fatty acid-binding protein (FABP4/ap2), a major cytoplasmic protein in adipocytes, plays a central role in the development of obesity and metabolic syndromes [28]. Expression of the activator protein (AP) 2β is closely associated with type-2 diabetes and negatively regulates the expression of the adiponectin gene [29]. In the present study, we have shown down-regulation of the expression of the A-FABP gene by adiponectin, which explains the reduction in total fatty acids in the rosiglitazone treatment group. In short, adiponectin decreases the expression levels of lipogenic genes and increases the expression of lipolysis genes, thereby inhibiting fat deposition in muscle.
The p38 MAPK is a major kinase in the MAPK family and plays essential role in regulating cell proliferation, inflammation, and immune responses [30]. Activated p38 MAPK has been shown to phosphorylate the transcription factors ATF2 [31]. Meanwhile, ATF2 and C/EBPα can form a heterodimeric DNA binding complex in vitro and co-transfection of ATF2 with C/EBPα results in decreased activation of transcription driven from consensus C/EBP-binding sites [32]. Recent studies suggest that p38 MAPK regulates adiponectin-induced glucose uptake and fatty acid oxidation in C2C12 myotubes and primary human hepatocytes [10, 33]. To test the hypothesis that adiponectin mediated the suppression of lipids deposition may through p38 MAPK/ATF2 pathway, we immunoblotted for the phosphorylation of p38 MAPK and ATF2. Data showed that the phosphorylation of p38MAPK and ATF2 increased with the increase of adiponectin, indicating that adiponectin might regulate lipids deposition in broiler muscle by p38 MAPK/ATF2 pathway.
Conclusions
In summary, our data suggested that rosiglitazone could increase the expression of adiponectin and adipoR1 in both breast and leg muscle in broilers, while dexamethasone had the opposite effect. Adiponectin negatively regulated the expression of lipogenic genes and proteins, enhanced lipolysis genes and proteins and, accordingly, inhibited lipids deposition in muscle. Data also showed that adiponectin regulated lipids deposition in broiler muscle by p38 MAPK/ATF2 pathway.
References
Scherer PE, Williams S, Fogliano M, Baldini G, Lodish HF (1995) A novel serum protein similar to C1q, produced exclusively in adipocytes. J Biol Chem 270(45):26746–26749
Pajvani UB, Du XL, Combs TP, Berg AH, Rajala MW, Schulthess T, Engel J, Brownlee M, Scherer PE (2003) Structure-function studies of the adipocyte-secreted hormone Acrp30/adiponectin: implications for metabolic regulation and bioactivity. J Biol Chem 278(11):9073–9085
Yamauchi T, Kamon J, Ito Y, Tsuchida A, Yokomizo T, Kita S, Sugiyama T, Miyagishi M, Hara K, Tsunoda M, Murakami K, Ohteki T, Uchida S, Takekawa S, Waki H, Tsuno NH, Shibata Y, Terauchi Y, Froguel P, Tobe K, Koyasu S, Taira K, Kitamura T, Shimizu T, Nagai R, Kadowaki T (2003) Cloning of adiponectin receptors that mediate antidiabetic metabolic effects. Nature 423(6941):762–769
Maddineni S, Metzger S, Ocon O, Hendricks G, Ramachandran R (2005) Adiponectin gene is expressed in multiple tissues in the chicken: food deprivation influences adiponectin messenger ribonucleic acid expression. Endocrinology 146(10):4250–4256
Wood JD, Brown SN, Nute GR, Whittington FM, Perry AM, Johnson SP, Enser M (1996) Effects of breed, feed level and conditioning time on the tenderness of pork. Meat Sci 44(1–2):105–112
Qi KK, Chen JL, Zhao GP, Zheng MQ, Wen J (2010) Effect of dietary omega 6/omega 3 on growth performance, carcass traits, meat quality and fatty acid profiles of Beijing-you chicken. J Anim Physiol Anim Nutr (Berl) 94(4):474–485
Ocon-Grove OM, Krzysik-Walker SM, Maddineni SR, Hendricks GL III, Ramachandran R (2008) Adiponectin and its receptors are expressed in the chicken testis: influence of sexual maturation on testicular ADIPOR1 and ADIPOR2 mRNA abundance. Reproduction 136(5):627–638
Tahmoorespur M, Ghazanfari S, Nobari K (2010) Evaluation of adiponectin gene expression in the abdominal adipose tissue of broiler chickens: feed restriction, dietary energy, and protein influences adiponectin messenger ribonucleic acid expression. Poult Sci 89(10):2092–2100
Yoon MJ, Lee GY, Chung J-J, Ahn YH, Hong SH, Kim JB (2006) Adiponectin increases fatty acid oxidation in skeletal muscle cells by sequential activation of AMP-activated protein kinase, p38 mitogen-activated protein kinase, and peroxisome proliferator-activated receptor alpha. Diabetes 55(9):2562–2570
Xin X, Zhou L, Reyes CM, Liu F, Dong LQ (2011) APPL1 mediates adiponectin-stimulated p38 MAPK activation by scaffolding the TAK1-MKK3-p38 MAPK pathway. Am J Physiol Endocrinol Metab 300(1):E103–E110
Aouadi M, Laurent K, Prot M, Le Marchand-Brustel Y, Binetruy B, Bost F (2006) Inhibition of p38MAPK increases adipogenesis from embryonic to adult stages. Diabetes 55(2):281–289
Vlahopoulos SA, Logotheti S, Micas D, Giarika A, Gorgoulis V, Zoumpourlis V (2008) The role of ATF-2 in oncogenesis. BioEssays 30(4):314–327
Maekawa T, Sano Y, Shinagawa T, Rahman Z, Sakuma T, Nomura S, Licht JD, Ishii S (2007) ATF-2 controls transcription of Maspin and GADD45[alpha] genes independently from p53 to suppress mammary tumors. Oncogene 27(8):1045–1054
Long Q, Lei T, Feng B, Yin C, Jin D, Wu Y, Zhu X, Chen X, Gan L, Yang Z (2010) Peroxisome proliferator-activated receptor-gamma increases adiponectin secretion via transcriptional repression of endoplasmic reticulum chaperone protein ERp44. Endocrinology 151(7):3195–3203
Liu BH, Wang YC, Wu SC, Mersmann HJ, Cheng WTK, Ding ST (2008) Insulin regulates the expression of adiponectin and adiponectin receptors in porcine adipocytes. Domest Anim Endocrinol 34(4):352–359
Fallo F, Scarda A, Sonino N, Paoletta A, Boscaro M, Pagano C, Federspil G, Vettor R (2004) Effect of glucocorticoids on adiponectin: a study in healthy subjects and in Cushing’s syndrome. Eur J Endocrinol 150(3):339–344
Sukhija PS, Palmquist DL (1988) Rapid method for determination of total fatty acid content and composition of feedstuffs and feces. J Agric Food Chem 36(6):1202–1206
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25(4):402–408
Yu JG, Javorschi S, Hevener AL, Kruszynska YT, Norman RA, Sinha M, Olefsky JM (2002) The effect of thiazolidinediones on plasma adiponectin levels in normal, obese, and type 2 diabetic subjects. Diabetes 51(10):2968–2974
Tan GD, Debard C, Funahashi T, Humphreys SM, Matsuzawa Y, Frayn KN, Karpe F, Vidal H (2005) Changes in adiponectin receptor expression in muscle and adipose tissue of type 2 diabetic patients during rosiglitazone therapy. Diabetologia 48(8):1585–1589
Tsuchida AYT, Takekawa S, Hada Y, Ito Y, Maki T, Kadowaki T (2005) Peroxisome proliferator-activated receptor (PPAR)-α activation increases adiponectin receptors and reduces obesity-related inflammation in adipose tissue: comparison of activation of PPAR. Diabetes 54(12):3358–3370
Andrews RC, Walker BR (1999) Glucocorticoids and insulin resistance: old hormones, new targets. Clin Sci 96(5):513–523
Thompson JM (2004) The effects of marbling on flavour and juiciness scores of cooked beef, after adjusting to a constant tenderness. Aust J Exp Agric 44(7):645–652
Berndt J, Kovacs P, Ruschke K, Kloeting N, Fasshauer M, Schoen MR, Koerner A, Stumvoll M, Blueher M (2007) Fatty acid synthase gene expression in human adipose tissue: association with obesity and type 2 diabetes. Diabetologia 50(7):1472–1480
Xu AM, Wang Y, Keshaw H, Xu LY, Lam KSL, Cooper GJS (2003) The fat-derived hormone adiponectin alleviates alcoholic and nonalcoholic fatty liver diseases in mice. J Clin Invest 112(1):91–100
Davies BSJ, Beigneux AP, Barnes Ii RH, Tu Y, Gin P, Weinstein MM, Nobumori C, Nyrén R, Goldberg I, Olivecrona G, Bensadoun A, Young SG, Fong LG (2010) GPIHBP1 is responsible for the entry of lipoprotein lipase into capillaries. Cell Metab 12(1):42–52
Qiao L, Kinney B, Schaack J, Shao J (2011) Adiponectin inhibits lipolysis in mouse adipocytes. Diabetes 60(5):1519–1527
Xu A, Tso AWK, Cheung BMY, Wang Y, Wat NMS, Fong CHY, Yeung DCY, Janus ED, Sham PC, Lam KSL (2007) Circulating adipocyte-fatty acid binding protein levels predict the development of the metabolic syndrome: a 5-year prospective study. Circulation 115(12):1537–1543
Iwamoto N, Yokoyama S (2011) Protein kinase D regulates the adiponectin gene expression through phosphorylation of AP-2: a common pathway to the ABCA1 gene regulation. Atherosclerosis 216(1):90–96
Ono K, Han J (2000) The p38 signal transduction pathway: activation and function. Cell Signal 12(1):1–13
Raingeaud J, Gupta S, Rogers JS, Dickens M, Han J, Ulevitch RJ, Davis RJ (1995) Pro-inflammatory cytokines and environmental stress cause p38 mitogen-activated protein kinase activation by dual phosphorylation on tyrosine and threonine. J Biol Chem 270(13):7420–7426
Shuman JD, Cheong J, Coligan JE (1997) ATF-2 and C/EBPalpha can form a heterodimeric DNA binding complex in vitro. Functional implications for transcriptional regulation. J Biol Chem 272(19):12793–12800
Wanninger J, Neumeier M, Weigert J, Bauer S, Weiss TS, Schaffler A, Krempl C, Bleyl C, Aslanidis C, Scholmerich J, Buechler C (2009) Adiponectin-stimulated CXCL8 release in primary human hepatocytes is regulated by ERK1/ERK2, p38 MAPK, NF-kappaB, and STAT3 signaling pathways. Am J Physiol Gastrointest Liver Physiol 297(3):G611–G618
Acknowledgments
This work was supported by a Grant from The National Nature Science Foundation of China (31172185).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yan, J., Gan, L., Qi, R. et al. Adiponectin decreases lipids deposition by p38 MAPK/ATF2 signaling pathway in muscle of broilers. Mol Biol Rep 40, 7017–7025 (2013). https://doi.org/10.1007/s11033-013-2821-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-013-2821-y