Abstract
Nain-e Havandi (Andrographis paniculata Nees.) (AP) is an annual herbaceous plant belonging to the family Acanthacea. Only a few species of Andrographis genus out of 28 are medicinally concerned of which AP is the most important. Knowledge about the arrival of AP to Iran is extremely lacking but most probably it has been imported from India. However, evidence implies the familiarity of Iran’s folkloric medicine with this plant, but it has been disappeared from contemporary medicine for unknown reasons. Presence of active ingredients from diterpenoids group such as andrographolide, neoandrographolide and 14-deoxy-11,12-didehydroandrographolide has given incredible unique medicinal properties to the plant. Traditionally, Nain-e Havandi has been used in the role of a non-farm plant as a remedy for skin problems, flu, respiratory disease, and snakebite in East and Southeast Asia for centuries. Recently, it has been utilized as a treatment for HIV, hepatitis, diabetes, cancer and kidney disorders. Intensive cultivation of the herb started only in the past decade in countries such as China, India, Thailand, Indonesia, West Indies, Mauritius and to some extent, in Malaysia. Availability of different ecological zones in Iran complies with reestablishment of AP in tropical and temperate regions of the country. This is killing two birds with one stone, supporting the conservational and economic aspects.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Medicinal herbs are moving from margin to mainstream use with a greater number of people seeking remedies and health approaches free from side effects caused by synthetic chemicals [211]. Recently, considerable attention has been paid to utilize eco-friendly and bio-friendly plant-based products for the prevention and cure of different human diseases. It has been recorded that 80% of the world’s population has fidelity in traditional medicine, particularly plant based drugs for their primary healthcare [1]. Medicinal plants have been applied to treat diseases for thousands of years. An estimate suggests that around 25% of the commonly used medicines contain compounds obtained from plants, and a few plants are proposed as rich reservoirs for drug discoveries to treat infectious diseases [2, 3]. The rising market demand for its products led the plant AP to become an alternative crop. Good-quality dried leaves of AP could be sold for US$5 per kg while the purified active ingredients, such as andrographolide and its derivatives, cost as much as US$100,000/kg from specialist chemical suppliers [4]. As a highlighted point the United States and China as two main super powers put their efforts to develop commercial products from the herb for different usages.
Historical background
According to references, Indians have used the leaves of AP as anti-venom against snakebite since ancient times [5]. Nain-e Havandi has been employed historically in epidemics, including the global flu epidemic of 1919 [6]. This pandemic is recognized as the most fatal infectious disease outbreak in human history and even more virulent than the Black Death of the fourteenth century, and AP was credited during that period as a wonder drug for arresting the spread of the contagious illness [7] as no country escaped its attack unless India. Evidence about the circumstances of this plant’s arrival into Iran is exceedingly lacking but the most logical way to achieve the issue is looking at the history of the relationship between Iran and India. The existence of several empires spanning both Persia (preferably Iran) and northern India territories ensured the constant migration of people between the two regions [8]. However, relations between India and Iran date back to the Neolithic era, but the answer will specifically cover a period of time that officially commenced from 2,600 years ago during the Achaemenian dynasty [9], at the time that Indian emissaries were present at the courts of Cyrus (Kurush) the Great (590 BCE–529 BCE). Afterwards, within the Afsharid dynasty when Nader Shah or King Nader (1,688–1,747) campaigned in India during the eighteenth century [10] and until recent. Records showed that this medicinal herb has been extensively used in traditional herbal medicine in China, Southeast Asia and the Persian Gulf area for the treatment of several diseases, including inflammatory diseases [11].
Botany
Andrographis paniculata (Burm. f.) Wallich ex Nees. also known as the King of Bitters in English is a medicinal herb from the family Acanthaceae. Perhaps different ecological and climatic conditions caused the plant to be introduced as a perennial plant [12], while most of the references present another botanical definition of the herb as an annual plant [7, 13]. A brittle-branched stem, herbaceous plant erecting to a height of 30–110 cm with glabrous, simple, opposite styled leaves [7, 13] and white flowers with rose–purple spots on the petals [14]. Even though AP is known as a hermaphroditic, self-compatible and a habitual inbreeding plant [15], there is an assumed rate of 28% cross pollination for it [16]. Inflorescence pattern extends axillary with terminal panicle or raceme. AP has a fibrous or adventitious root system (Fig. 1).
a Andrographis paniculata herb in pod stage with terminal or axillary panicles. b Young plant: aerial and underground parts of AP. c Fruit of AP in capsule form. d Opened capsule. e Rugose seeds of AP inside the fruit. f flowering plant with a small flower. g A flower with opened anther and pollen grains
Taxonomic position
Taxonomically Andrographis paniculata is classified as below:
- Kingdom:
-
Plantae, plants;
- Subkingdom:
-
Tracheobionta, vascular plants;
- Superdivision:
-
Spermatophyta, seed plants;
- Division:
-
Angiosperma
- Class:
-
Dicotyledonae
- Sub-class:
-
Gamopetalae
- Series:
-
Bicarpellatae
- Order:
-
Personales
- Tribe:
-
Justicieae
- Family:
-
Acanthaceae
- Genus:
-
Andrographis
- Species:
-
Paniculata (Burm. f) Nees [13]
Geographical distribution and preferred habitats
The genus Andrographis is composed of roughly 40 species several members of which enjoy a reputation in traditional medicine [17]. AP populations are distributed over a broad eco-geographical range in tropical Asian countries often in isolated patches. It grows abundantly in Southern and Southeastern Asia, including India, Sri Lanka, Pakistan and Indonesia [13]. So-called “native populations” occur only in the Indian subcontinent and especially South India and Sri Lanka, which perhaps represent the centre of origin and diversity of the species [18, 19]. Sabu [16], stated that the herb is an introduced species in the northern areas of India, Thailand, Brunei, Malaysia, Indonesia, the West Indies such as Jamaica, Barbados and Bahamas, Hong Kong and in the tropical areas of the Americas [18, 20–22]. This plant is also currently cultivated in southwestern Nigeria [23]. AP can be stemmed in a variety of habitats, for instance; plains, hill slopes, wastelands, farms, dry or wet lands, seashores, and even roadsides, but it has a preferred tendency to grow in moist shady places, forests, and wastelands [18, 20–22].
Terminology
We believe that the most probable reason for naming the plant as AP (Fig. 2a) is the presence of a diterpene lactone in the leaves namely andrographolide and also the existence of two flavones viz., andrographin and panicolin in the roots. Regardless of the above-mentioned case, the botanical name of the plant consisted of Andrographis and paniculata carrying other meanings as well. Andro is a place name in India. In fact, Andro is a town in Imphal East district, Manipur state, North East of India. Andro also is a prefix in the Greek language meaning “male” or “masculine” can refer to a number of things. Graphis (Fig. 2b) is a genus of lichens from the family Graphidaceae [24, 25] and in the Latin language means pencil, paintbrush or writing style, and paniculata performs a Latin pronunciation of paniculate or panicle, which refers to the inflorescence of the plant.
Synonyms
Normally, most of the useful plants got a vernacular name in various languages. Hence, as a matter of fact, giving a specific Persian name to AP could be meaningful because it means that either the physicians of ancient Iran were quite knowledgeable about its curative effects in treating their patients or the ordinary people were familiar with the plant as a traditional folk remedy. Anyway, it is necessary to remind that Nain is a name of town in the central part of Iran. Possibly, herbalists and traditional apothecaries values AP due to its miraculous ability in treating ailments and this made it to possess different names from East to West. Azerbaijani and Turkish names of this plant have been given by the first author of the present article which has been inspired from the English name of AP (Table 1).
Cytology
To date, five conducted cytological studies on AP in the early 1980s, concur on 25 pairs of chromosomes (2n = 50) as the chromosome number for the species [26–30].
Genetic diversity
Initial steps to investigate the genetic diversity of AP using molecular approaches were taken only in the late 1990s. The first try was given by employing RAPD (randomly amplified polymorphic DNA) markers and resulted in a moderate level of variation engaged with intra-specific study of accessions collected from India and Southeast Asia. Additionally, the clustering analysis using unweighted pair group method with arithmetic mean (UPGMA) was limited to five major groups based on geographical distributions that generally reflected the expected trends among the genotypes [31]. Isozymes were the second molecular tool used to elucidate the genetic variations among AP genotypes while quantitative variations in content of the plant’s major active component (andrographolide), were used to indicate phytochemical diversity [32]. AP has been exposed to ISSR (inter simple sequence repeat), SSCP (single strand conformation polymorphism) and PCR-RFLP (polymerase chain reaction-restriction fragment length polymorphism) markers in different cases [7, 33, 36, 38, 40–43]. A summary of several conducted studies using various marker systems on AP to ascertain its genetic variation is presented in Table 2.
Agrotechnology and tissue culture of AP
Despite numerous studies conducted on the pharmacology, phytochemical composition, and therapeutic features of AP, it has not been subjected to detailed agronomic studies worldwide except in a few Asian countries like India, China, Thailand and to a lesser extent in Malaysia.
Seed dormancy is a problem in AP propagation [39, 45]. Hormonal solutions and hot-water treatment are suggested to overcome the case [46, 47]. For herbs, where a leaf is the major plant part used for the medicinal purpose, promoting vegetative growth is the priority. Efforts to improve foliar production through proper management of fertilizer application should be of the main concern for commercial production [48]. Herbage yield of AP is influenced by date of planting and harvest [49, 50]. AP is mostly sown with spacing of 30 × 15 cm between May and July which result to a plant density of 222,222 plant/ha and average biomass of 3 t/ha [55, 56]. Conventional vegetative propagation of AP (through seed) is very difficult and too slow to meet the commercial quantities required. Hence, non-conventional biological strategies such as tissue culture are alternative methods for improving the propagation, yield and phytochemical content in AP [61]. Tissue culture has been successfully used as a technique for the formation of new flavones by differentiating callus cultures of the plant [62, 63]. Tissue culture is successfully used as a technique for formation of new flavones by differentiating callus cultures of the plant [64]. A list of agronomic field trials on AP is presented in Table 3.
Breeding and physiology of AP
Classical genetic scrutiny and breeding of AP using conventional schemes such as intra-specific hybridization have been neglected due to its troublesome procedure. On the other hand, the plant’s weak response to manual crossing, acted as an obstacle to this approach.
Literatures about physiologic characreistics of the plant are extremely lacking. However, physiologic aspects of the plant, like relative growth rate (RGR) has been taken into consideration very recently [59]. However, initial researches suggests a critical model for leaf area index (LAI). This means that LAI increasing associated with increasing crop growth rate (CGR) during growth stage and CGR reaches the maximum at a specific level of LAI, beyond which it remains the same despite the increases in LAI. The LAI at which CGR reaches the utmost may be called the critical LAI [204]. Specific leaf area (SLA) increases up to 90 days after sowing (DAS) and there is gradual decreasing there after up to 120 DAS. This is the maturity sign of the herb [55].
Phytochemistry investigations and major constituents
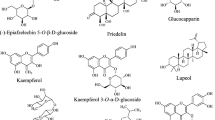
Chemical explorations of AP began by the end of the nineteenth century when Boorsma [65] isolated an extremely bitter lactone diterpene in 1896 for the first time. Subsequently, Gorter [66], was the first scientist to name the above-mentioned constituent as andrographolide (ANDRO or AG or AP1). 41 years later, the second non-bitter component namely neoandrographolide (NAG or AP4) was discovered by Kleipool [67]. Isolation of the third relatively minor diterpene, 14-deoxy-11,12-didehydroandrographolide or 11,12-didehydro-14-deoxyandrographolide (DDAG or AP3) was reported by Balmain and Connolly [68], along with three other diterpenes, including andrographiside, 14-deoxyandrographolide and 14-deoxyandrographiside. Studies were also done in order to unravel the chemical structures of andrographolide [69], and neoandrographolide [70]. The important compounds isolated from different parts of the plant according to Rastogi and Mehrotra [71], were apigenin-7,4/-di-O-methyl ether, carvacrol, eugenol, myristic acid, hentriacontane, tritriacontane, oroxylon A, wogonin, and diterpenoids like andrograpanin, andropanoside, andrographolide and neoandrographolide. A few other active components, including 14-deoxy-11,12-dihydroandrographolide (andrographolide D), homoandrographolide, andrographosterin and stigasterol were subsequently reported [72]. Nevertheless, it is usually agreed that three diterpenic compounds namely andrographolide (AG), neoandrographolide (NAG), and 14-deoxy-11, 12-didehydroandrographolide (DDAG) are the major constituents of the plant’s extract (Fig. 3) responsible for its pharmacological activities [73–210].
In addition, leaves of AP contain andrographosterol and andrographone while flavones-apigenin-7,4-dio-O-methyl ether, 5-hydroxy-7,8,2′,3′-tetramethoxyflavone, andrographin, panicolin and a-sitosterol are accumulated in the roots [78, 79]. Series of newly reported semi-synthetic andrographolides are given below [80]. 14-Deoxy-11,14-didehydroandrographolide (AP10), 8,17-Epoxyandrographolide, 12,13-Dihydroandrographolide, 14-deoxy-12,13-dihydroandrographolide, 13,14-Didehydro-12-hydroxy-3,19-methene-O-andrographolide, 3,19-(p-methanoxylphenylmethene-O-)-andrographolide,14-deoxy-11,12-didehydro-15-isopropylideneandrographolide, 14-Deoxy-11,12-didehydro-15-(2,8-dimethanyl-2,7-octanedieneylidene)-andrographolide, 15-Benzylidene-14-deoxy-11,12-didehydroandrographolide, 14-Deoxy-11,12-didehydro-15-p-fluorobenzylideneandrographolide, 14-Deoxy-11,12-didehydro-15-trimethoxylbenzylideneandrographolide, 14-Deoxy-11,12-didehydro-15-p-chlorobenzylideneandrographolide, 15-m-Bromoacylbenzylidene-14-deoxy-11,12-didehydroandrographolide, 14-Deoxy-15-(p-(dimethanylamino)-benzylidene)-11,12-didehydroandrographolide, 14-Deoxy-11,12-didehydro-15-p-methoxylbenzylidene andrographolide, 15-(Benzo[1, 3]dioxole-5-methanylidene)-14-deoxy-11,12-didehydroandrographolide, 14-Deoxy-11,12-didehydro-15-(-2-furanmethanylidene)-andrographolide, 14-Deoxy-11,12-didehydro-3,19-dinicotinateandrographolide, 14-Deoxy-11,12-didehydro-3,19-dinicotinate-15-(2,8-dimethanyl-2,7-octanedieneylidene)-andrographolide, 15-Benzylidene14-deoxy-11,12-didehydro-3,19-dinicotinateandrographolide, 15-(p-Chlorinebenzylidene)-14-deoxy-11,12-didehydro-3,19-dinicotinateandrographolide, 15-(m-Bromoacylbenzylidene)-14-deoxy-11,12-didehydro-3,19-dinicotinateandrographolide, 14-Deoxy-11,12-didehydro-3,19-dinicotinate-15-(p(-dimethylamino)benzylidene)-andrographolide, 14-Deoxy-11,12-didehydro-3,19-dinicotinate-15-(2-furanmethanylidene)-andrographolide, and 14-Deoxy-11,12-didehydro-3,19-dinicotinate-15-p-methoxylbenzylideneandrographolide.
Pharmacology and remedies
Conducted researches in the past three decades have confirmed that AP, properly administered, has a surprisingly broad range of pharmacological effects such as Inhibitory impact of its active diterpenoids on platelet aggregation [81] and lots of other extremely beneficial effects [82]. Almost all the review papers about AP have extensively focused on its medicinal properties [13, 83, 92, 120, 121] thus, we have prepared a comprehensive table concerning medicinal and curative effects of AP (Table 4).
Posology and dosage in clinical trials
Available instructions are varied due to the type of disorder, type of the formulation (crude extract, solid or liquid formulation or purified active ingredients), and nature of the study (clinical trials, in vitro or in vivo studies on animals or human). Nevertheless, most of the clinical tests used products standardized to 4–5 mg/kg of andrographolide with a typical dosage of a 400 mg tablet, 3 times a day for the first few days of flu [152]. Some of the clinical trials have chosen direct use of powdered crude material with 1,200 mg/day of AP extracts standardized to 5% andrographolide or a placebo for a period of 5 days [6, 153, 154]. A decoction of 3 g of crude drug twice daily is proposed for pyrexia treatment [123, 155, 156]. For the common cold 1.5–3.0 g of powdered crude drug 3 times daily after meals and at bedtime [155, 156]. For diarrhea a decoction from 3–9 g crude drug as a single dose is needed [123, 155], or two tablets of 500 mg 4 times daily, after meals and at bedtime [123, 156]. Recommended daily dose for AP powder ~850 mg or 1–2 capsules (500 mg/capsule) and intake 4 times/day [156]. Coon and Ernst [120], have reviewed that a daily dose of andrographolide up to 60–380 mg/day could be efficient in healing of upper respiratory tract infections. However, the treatment period can take a range from three to 8 days in different studies [153, 154, 157–161]. 200–300 mg of andrographolide daily in 2–3 times is recommended for relieving cold and fever symptoms. Doses in a range between 1,000 and 2,000 mg 3 times daily have been applied in some studies and a typical standardization for content of andrographolide in AP is 4–6% [153]. An advisory from the Langone Medical Center noted that virtually all the published studies of AP have involved a single proprietary product. It is not clear that the results of these studies apply to products using different AP sources, or different methods of extraction [162].
Adverse reactions and safety issues
Even though in general, AP has not been implicated with any serious side effects in the relevant experiments on humans, the main complain about this plant is that it may affect the fertility of the person receiving the drug. Nonetheless, several studies have proven its safety. No side effects were observed when participants were monitored for changes in liver and kidney functions, blood counts, and other laboratory measures when treated with 1,200 mg/day of AP for 5 days [6]. This regime has been mentioned as a short-term treatment with 15 times the clinical dosage of andrographolide [163]. Rare and minor side effects from AP in two cases of urticaria had been reported [157]. In another claim, large oral doses of AP may cause gastric discomfort, vomiting, and loss of appetite. These side effects emerged due to the bitter taste of andrographolide [164]. Intra-arterial or retrograde intravenous injections of AP’s crude extract may cause anaphylactic reactions [83, 164, 165]. Limitations in direct testing of the drug on humans could be compensated by in vivo analysis on animals. There have been conflicting with results from such animal studies. Studies on mice had raised concerns that AP may reduce fertility in male rats when fed with 20 mg/kg/day of AP powder daily [166]. In a toxicity survey on mice, the minimal lethal dose (MLD) of the main active components of AP, andrographolide (AG) and 14-deoxy-11,12-didehydroandrographolideos (DDAG) was 20 g/kg of body weight by mouth feeding, which was about more than 100 times higher than the recommended daily dosage [167]. No testicular toxicity was found in vivo with treatment of 20, 200 and 1,000 mg/kg during 60 days in rats [168]. However, degeneration of anatomical structures in testicles and testicular toxicity in male rats was reported when rats were treated with dried extracts of AP up to 1 g/1 kg of body weight daily for 2 months. One group of female mice also did not fare well on high dosages of AP when fed 2 g/kg of body weight daily for 6 weeks (1,000 times higher than the usual human dose). All female mice failed to get pregnant when mated with fertile males. Concurrently, 95.2% of the control females got pregnant when mated with a similar group of male mice [84]. Another study established a potential explanation that AP relaxes the uterus [169]. These results raised concerns regarding the use of AP by pregnant women, in case they take an overdose. Hence, the conclusion is that the anti-fertility effects of AP cannot occur in the normal situations. Related studies comply with no side effect as well as acceptable tolerance for Kan Jang treatment [170, 171]. One of the latest investigations in a human trial using the widely tested Andrographis–Eleutherococcu’s combination discovered no negative effect on male semen quality and fertility. However, rather a positive trend with respect to the number of spermatozoids in the whole ejaculates, the percentage of active (normokinetic) forms of spermatozoids, and fertility indices, together with a decrease in the percentage of inactive (diskinetic) forms of spermatozoids were observed [172]. Gallbladder contraction could be stimulated by taking AP [173], so the drug must be avoided by persons with gallbladder disease, unless with physician’s prescription.
Formulations and commercialization
Our review showed that AP as a medicine can be formulated solely or mixed with other species in different configurations such as a powder, pill, tablet, capsule, decoction and crude extract [174]. A crude mixture of AP along with five other species, including Momardica charantia, Phyllanthus niruri, Terminallia chebula, Glycyrrhiza glabra and Punica granatum in powder form was authenticated by Choudhari et al. [126] for treating HIV. Both fresh and dried AP leaves, as well as the fresh juice of the whole plant, have been widely used in traditional medicines and advanced biomedical studies. In research on diabetes, AP decoction was used in combination with Momordica charantia fruit juice [175]. Crude chloroformic, ethanolic and methanolic AP extracts have been administrated by numerous scientists as the most common form of application for different aims [102, 112, 137, 139, 146, 149, 176–178]. However, sometimes chloroform extracts in vitro appeared more competent than the methanol extract in controlling malaria [137]. Pharmaceutical products of AP are available in drop and syrup forms as well, while leaf juice of the plant is used as syrup for the treatment of liver diseases [179]. The syrup also is a healer of cold symptoms like coughs [153, 180]. The commercialization and branding of AP are proceeding swiftly in trade formulations for pharmaceutical products such as Liver tonics with hepatoprotective effects [181].
Wonderful application of AP in special fields
Although, an enormous proportion of AP’s reputation is because of its medicinal properties but, in fact, this herb has been subjected to all sorts of studies in the twentieth century for possible applications in various fields such as agriculture, forestry, poultry and animal production, veterinary medicine and even for industrial applications.
Herbicidal capacity
Phytotoxic and allelopathic effects of AP are confirmed as its ground adversely affects the metabolism of the host plant (Parthenium hysterophorus L.) by allelochemical interference with photosynthesis and decreasing the total chlorophylls, polyphenols, amino acids, proteins, carbohydrates, starch and lipid contents. So, residues of Pathenium hysterophorus L. could be used as a potent bio-herbicide [182].
Insecticidal function
Novel findings reveales that AP has the potential role in eco-friendly mosquito control programs. AP exerted a mosquitocide influence against the malaria vector, Anopheles stephensi Liston. The larvicidal, pupicidal, adulticidal and ovicidal properties of whole plant ethanolic extract were evaluated under laboratory conditions and the most effective results were obtained for larvicidal and pupicidal activities [183].
Alternative antibiotic in poultry
AP can be used as an alternative to antibiotics as a growth promoter of broiler chicken. Besides this, the FCR (total feed consumed by a bird/total weight gain) was significantly better in the group treated with a combination of basal diet and AP’s shredded dried leaves when compared to virginiamycin treated, and panchagavya treated groups [184, 185].
Animal production, aquaculture, and veterinarian utilization
AP has antimicrobial and growth promoting function and may be used as an alternative to antibiotics and tonic [186]. The nature of AP supports its capable role in acting as an antioxidant supplement along with vitamin E and Curcuma longa L. This can improve goat meat quality by preventing lipid oxidation, color stability, increasing drip loss of the longissimus dorsi (LD) muscle as well as the chevon tenderness particularly in post-mortem aging [187, 188]. A blend of AP extract and dry Indian almond leaf (Terminalia catappa) positively affected tail growth and hematocrit value in Fancy carp (Cyprinus carpio Linn.) [189]. Mix of AP and Noni (Morinda citrifolia) promotes pigmentation and phagocytosis in goldfish (Carasius auratus) [190].
AP leaves decoction in vitro appeared with promising impacts by killing the microfilaria of Dipetalonema reconditum in a short time in dog. Three subcutaneous injections of the extract into infected dogs and rabbits at 0.06 ml/kg body-weight decreased the number of microfilariae in blood by more than 85%. Although no toxic effect of the extract was observed in rabbits, but the treated dogs became lethargic initially for a week, probably due to the mass killing of microfilariae [191]. Microfilaricidal effect of AP is detected by controlling Canine dirofilariasis as a common tropical parasitic animal disease caused by Dirofilaria immitis. l, 10, 100 μg/ml and 1 mg/ml concentrations of dried aqueous extracts of each plant dissolved in dimethyl sulfoxide (DMSO), have been tested on infected samples of sheltered dogs [203].
Forestry (a protector element against fungal disease)
The upshot of a literature on subtropical jungles verified the ability of AP to control fungal diseases in plants together in line with its antifungal efficacy in humans. A protein extracted from AP leaves with molecular mass of 39.5 kDa can inhibit the spore germination and hyphal extension of Trichosporium vesiculosum fungus [192]. This fungus is the pathogenic agent that causes blister bark within a species of She-Oak tree (Casuarina equisetifolia).
Industrial application
Different techniques such as weight loss, electrochemical impedance spectroscopy, linear polarization, and potentio-dynamic polarization assays revealed that the leaves extract of AP plant can serve as a powerful inhibitor of the corrosion of mild steel in hydrochloric acid media [193].
Conclusion
Despite AP’s price decline in India’s regional market [194] due to the global economic crisis demands for herbal medicinal products are boosted in general yet AP has been listed among prioritized plants for development and cultivation under the scheme of the National Medicinal Plants Board of India [195]. AP has been presented in the list of highly traded Indian medicinal plants [195]. In another survey, AP has been positioned as the 17th crop among the 32 prioritized medicinal plants of India with a demand of 2197.3 tons in year 2005–2006 and annual growth of 3.1% [7, 205, 206]. Priority of AP is regarded by herbal industries of developing countries such as Malaysia [207], Thailand [208] and Nigeria [23]. Furthermore, the profitable exploitation of biochemical compounds of AP is hampered due to their limited availability [196]. AP has taken the sixth place due to its role in attaining the United Nations millennium development goals (MDGs) target, which is fighting against HIV/AIDS, malaria and other diseases [197–199]. All these results imply that the absence of this plant in Iran’s pharmaceutical market must be remedied by the implementation of precise programs to develop the cultivation of the herb as a new alternative crop in different agricultural ecosystems. Commencing such a plan in Iran will support the conservation and biodiversity of AP in this country since many experts have warned that the Indian AP’s population is decreasing considerably [200, 209]. Besides, the government of Sri Lanka has prohibited the export of certain protected medicinal plant products such as Coscinium fenestratum, Salacia reticulata and even declared AP as an endangered species [201]. These circumstances provide an excellent opportunity for Iran to take its share in producing the plant and its commercial derivatives. Conducive natural conditions, availability of educated and skillful human resources and technicalities are supportive factors which can realize the dream of Nain-e Havandi’s return into the plateau of Iran. Adding AP to the treasury of available medicinal plants will play a major role in Iran’s herbal pharmaceutical industry.
References
Dubey NK, Kumar R, Tripathi P (2004) Global promotion of herbal medicine: India’s opportunity. Curr Sci 86(1):37–41
Tavili A, Farajollahi A, Pouzesh H, Bandak E (2010) Treatment induced germination improvement in medicinal species of Foeniculum vulgare Miller and Cuscuta epithymum L. Mod Appl Sci 4(7):163–169
Mukhtar M, Arshad M, Ahmad M, Pomerantz RJ, Wigdahl B, Parveen Z (2008) Antiviral potentials of medicinal plants. Virus Res 131(2):111–120
DP UN (2002) Examples of the development of pharmaceutical products from medicinal plants. Promoting herbal drugs. Thailand 10:45–56
Meenatchisundaram S, Parameswari G, Michael A (2009) Studies on antivenom activity of Andrographis paniculata and Aristolochia indica plant extracts against Daboia russelli venom by in vivo and in vitro methods. Indian J Sci Technol 2(4):76–79
Hancke J, Burgos R, Caceres D, Wikman G (1995) A double blind study with a new monodrug Kan Jang: decrease of symptoms and improvement in the recovery from common colds. Phytother Res 9(8):559–562
Sharma SN, Sinha RK, Sharma DK, Jha Z (2009) Assessment of intra-specific variability at morphological, molecular and biochemical level of Andrographis paniculata (Kalmegh). Curr Sci 96(3):402–408
Chopra V (2003) New trends in Indo-Russian relations. Gyan Books, New Delhi, p 309
Sacks D, Murray O, Brody LR (2005) Encyclopedia of the ancient Greek world. Facts on File, New York, p 412
Durand SHM (1908) Nadir Shah. London, A Constable and co ltd 352 Pages
Abu-Ghefreh AA, Canatan H, Ezeamuzie CI (2009) In vitro and in vivo anti-inflammatory effects of andrographolide. Int Immunopharmacol 9(3):313–318
Prathanturarug S, Soonthornchareonnon N, Chuakul W, Saralamp P (2007) Variation in growth and diterpene lactones among field-cultivated Andrographis paniculata. J Nat Med 61:159–163
Mishra SK, Sangwan NS, Sangwan RS (2007) Andrographis paniculata (Kalmegh): a review. Phcog Rev 1(2):283–298
Chiramel T, Bagyaraj DJ, Patil CSP (2006) Response of Andrographis paniculata to different arbuscular mycorrhizal fungi. J Agric Technol 2:221–228
Lattoo SK, Khan S, Dhar AK, Choudhary D, Gupta KK, Sharma PR (2006) Genetics and mechanism of induced male sterility in Andrographis paniculata (Burm. f.) Nees and its significance. Curr Sci 91(4):515–519
Sabu KK (2002) Intraspecific variations in Andrographis paniculata Nees. PhD Thesis, Kerala University,Thiruvananthapuram, India
Rao YK, Vimalamma G, Rao CV, Tzeng YM (2004) Flavonoids and andrographolides from Andrographis paniculata. Phytochemistry 65(16):2317–2321
Hooker JD (1885) The flora of British India, Vol. IV. p.501. Henrietta Street, Convent Garden, London
Bhat VS, Nanavati DD (1977) Andrographis paniculata (Burm. f.) Wall. ex Nees (kalmegh). Indian Drugs 15:187–190
Ridley HN (1925) The Flora of the Malay Peninsula, Vol V. The Flora of the Malay Peninsula, Vol V
Backer CA, Van Den Brink Jr RCB (1965) Flora of Java, vol 2. Wolter-Noordhoff, Gronigen
Correll DS, Correll HB (1982) The flora of the bahama archipelago. J Cramer, Vaduz, Liechtenstein, p 1692
Fasola TR, Ayodele AE, Odetola AA, Umotok NE (2010) Foliar epidermal morphology and anti-diabetic property of Andrographis paniculata (Burm. f.) Wall ex. Nees. Ethnobot Leaflets 14:593–598
Staiger B, Kalb K, Grube M (2006) Phylogeny and phenotypic variation in the lichen family Graphidaceae (Ostropomycetidae, Ascomycota). Mycol Res 110(7):765–772
Archer AW, Elix JA (2007) New species and new reports in the Australian Graphidaceae. Telopea 11(4):451–462
Cheng YC, Liu BK, Jiang ZS, Duan YC (1991) Observations of chromosome numbers of several medical plants publication. J Hunan Agric Cytol Coll 11(2):166–170
Saggoo MI (1983) Cytomorphological studies on plants of economic importance of Bicarpellatae from India Publication. None listed Collation 259 Pages
Saggoo MI, Bir S (1986) Meiotic studies in certain members of family Acanthaceae from South India. J Indian Bot Soc 65(3):310–315
Govindarajan T, Subramanian D (1983) Karyomorphological studies in south Indian Acanthaceae. Cytologia 48(3):491–504
Roy SK, Datta PC (1988) Chromosomal biotypes of Andrographis paniculata in India and Bangladesh publication. Cytol Coll 53:369–378
Padmesh P, Sabu KK, Seeni S, Pushpangadan P (1999) The use of RAPD in assessing genetic variability in Andrographis paniculata Nees, a hepatoprotective drug. Curr Sci 76:833–835
Sabu KK, Padmesh P, Seeni S (2001) Intraspecific variation in active principle content and isozymes of Andrographis paniculata Nees (Kalmegh): a traditional hepatoprotective medicinal herb of India. J Med Aroma Pl Sci 23:637–647
Jebril AA, Saad MS, Stanslas J, Mihdzar AK, Ramisah M, Yong A (2003) Genetic variation and antitumor activity of Andrographis paniculata. Fifth National Genetic Congress, Kuala Lumpur
Melaku A, Saad MS, Yunus AG, Stanslas J (2003) Genetic diversity of Andrographis Paniculata Nees. germplasm in Malaysia. Fifth National Genetic Congress, Kuala Lumpur
Ng KT (2004) Morphological characterization of the Hempedu Bumi (Andrographis paniculata) germplasm from eastern part of Malaysia. Bachelor Thesis, Universiti Putra Malaysia, Malaysia
Maison T, Volkaert H, Boonprakob U, Paisooksantivatana Y (2005) Genetic diversity of Andrographis paniculata wall. ex Nees as revealed by morphological characters and molecular markers. Kasetsart J (Nat Sci) 39:388–399
Saad MS, Chia SH, Melaku A, Ng KT, Jebril A, Stanslas J, Norlia Y, Yunusa AG (2006) Genetic diversity of Hempedu Bumi (Andrographis paniculata) germplasm in peninsular Malaysia as revealed by morphological character’s variability. Proceeding: National Conference on Agrobiodiversity Conservation and Sustainable Utilization. Kuching, Sarawak, Malaysia
Saad MS, Chia SH, A. Jebril MS, Ramisah H, Suharni N, Milan. AR (2006) Genetic diversity in Hempedu Bumi (Andrographis paniculata) germplasm in Malaysia as revealed by RAPD polymorphism. Agriculture congress. University Putra Malaysia 227-229
Talei D, Mihdzar AK, Yusop MK, Valdiani A, Puad MA (2012) Seed dormancy and germination in King of Bitters (Andrographis paniculata Nees.). J Agric Sci 4(5) (Under publication)
Lattoo SK, Dhar RS, Khan S, Bamotra S, Bhan MK, Dhar AK, Gupta KK (2008) Comparative analysis of genetic diversity using molecular and morphometric markers in Andrographis paniculata (Burm. f.) Nees. Genet Resour Crop Evol 55(1):33–43
Sakuanrungsirikul S, Jetana A, Buddanoi P, Dithachaiyawong J (2008) Intraspecific variability assessment of Andrographis paniculata collections using molecular markers. Acta Hort (ISHS) 786:283–286
Chua KH, Li CG, Xue Charlie CL, Pang ECK (2009) Genetic diversity of Andrographis Paniculata (Burm. f.) Nees of Brunei Darussalam determined by RAPD and PCR-RFLP analyses (Chapter 11) In: De Silva T, Bahorun T, Sahu M, Huong LM (eds) Treditional and Alternative Medicine Research and Policy. Centre for Science and Technology of the Non-Aligned and Other Developing Countries. 594 pages
Chia SH (2009) Reproductive System and Genetic Diversity of Hempedu Bumi (Andrographis Paniculata) Germplasm in Peninsular Malaysia. Master Thesis, Universiti Putra Malaysia, Malaysia
Pandey AK, Mandal AK (2010) Variation in morphological characteristics and andrographolide content in Andrographis paniculata (Burm. f.) Nees of Central India. Iranica J Energy Environ (IJEE) 1(2):165–169
Saraswathy S, Manavalan RSA, Vadivel E, Manian K, Subramanian S (2004) Studies on seed germination in kalmegh (Andrographis paniculata Nees.). South Indian Hortic 52(1/6):286–290
Kumar RN, Chakraborty S, Nirmal KJI (2011) Methods to break seed dormancy of Andrographis paniculata (Burm.f.Nees): an important medicinal herb of tropical Asia. Asian J Exp Biol Sci 2(1):143–146
Kumar B, Verma SK, Singh HP (2011) Effect of temperature on seed germination parameters in Kalmegh (Andrographis paniculata Wall. ex Nees.). Ind Crops Prod 34:1241–1244
Khairul AR, H. SA, Asiah AM (2007) Growth performance of Hempedu Bumi (Andrographis paniculata) as affected by organic fertilizer rates. J Int Soc Southeast Asia Agric Sci 13(2):126-149
Nemade S, Mohod N, Wankhade SG, Paturde JT (2003) Effect of planting and harvesting dates on yield and quality of kalmegh (Andrographis paniculata). J Med Arom Plant Sci 25(4):981–983
Bhan MK, Dhar AK, Khan S, Lattoo SK, Gupta KK, Choudhary DK (2006) Screening and optimization of Andrographis paniculata (Burm. f.) Nees for total andrographolide content, yield and its components. Sci Hortic 107(4):386–391
Prasad PP, Joseph B (1997) Oushada Sasyemgalum Avayude Krishireethikalum (Malayalam). Nagarjuna Research Foundation, Thodupuzha, p 171
Joy PP, Thomas J, Mathew S, Skaria BP (2001) Medicinal Plants. In: Bose TK, Kabir J, Das P, Joy PP (eds) Tropical horticulture, vol 2. Naya Prokash, Calcutta, pp 449–632
Joshi DN, Bhojvaid PP, Dobriyal MJR (2003) Cultivation techniques for satavar (Asparagus racemosus) & kalmegh (Andrographis paniculata). Int J For Usufructs Manage (IJFUM) 4(2):70–72
Maibangsa S, Baruah BP, Mahanta TK, Maibangsa M, Singh KD, Sharma NN, Deka N, Borah RC, Talukdar A, Kataky JCS (2004) Rauwolfia serpentina, Andrographis paniculata and Asparagus racemosus-cultivation practices in hills zone of Assam, Proceedings of the national symposium on Biochemical approaches for utilization and exploitation of commercially important plants. Jorhat, India. 273-281
Parashar R, Upadhyay A, Singh J, Diwedi SK, Khan NA (2011) Morpho-physiological evaluation of Andrographis paniculata at different growth stages. World J Agric Sci 7(2):124–127
Man S, Singh A, Tripathi RS, Verma RK, Gupta MM (2011) Growth behavior, biomass and diterpenoid lactones production in Kalmegh (Andrographis paniculata Nees.) strains at different population densities. Agric J 6(3):115–118
Sanjutha S, Subramanian S, Rani CI, Maheswari J (2008) Integrated nutrient management in Andrographis paniculata. Res J Agric Biol Sci 4(2):141–145
Wankhade SG, Khode PP, Wanjari SS, Paturde JT, Sakure SM (2005) Herb yield and quauty of kalmegh (Andrographis paniculata) as influenced by time of planting and harvesting. Indian J Agric Res 39(4):303–306
Talei D, Mihdzar AK, Yusop MK, Valdiani A, Puad MA (2012) Growth indices alteration in medicinal plant of Andrographis paniculata (Burm.f.) Ness. under salinity stress. J Agric Sci 4(4) (Under publication)
Ramesh G, Shivanna MB, Santa Ram A (2011) Interactive influence of organic manures and inorganic fertilizers on growth and yield of kalmegh. (Andrographis paniculata Nees.). Int Res J Pl Sci 2(1):16–21
Martin KP (2004) Plant regeneration protocol of medicinally important Andrographis paniculata (Burm. F.) Wallich ex Nees via somatic embryogenesis. In Vitro Cell Dev Biol Pl 40(2):204–209
Bowes BG (1969) The fine structure of wall modifications and associated structures in callus tissue of Andrographis paniculata Nees. New Phytol 68(3):619–626
Behera PR, Nayak P, Barik DP, Rautray TR, Thirunavoukkarasu M, Chand PK (2010) ED-XRF spectrometric analysis of comparative elemental composition of in vivo and in vitro roots of Andrographis paniculata (Burm. f.) Wall. ex Nees: a multi-medicinal herb. Appl Radiat Isot 68(12):2229–2236
Jalal MAF, Overton KH, Rycroft DS (1979) Formation of three new flavones by differentiating callus cultures of Andrographis paniculata. Phytochemistry 18(1):149–151
Boorsma WA (1896) Constituents of Andrographis paniculata. Med’s Lands Plant 18:63
Gorter MK (1911) The bitter constituent of Andrographis paniculata Nees. Ree Trav Chim 30:151–160
Kleipool RJC (1952) Constituents of Andrographis paniculata Nees. Nature 169:33–34
Balmain A, Connolly JD (1973) Minor diterpenoid constituents of Andrographis paniculata Nees. J Chem Soc Perkin Trans 1:1247–1251
Smith AB, Toder BH, Carroll PJ, Donohue J (1982) Andrographolide: an X-ray crystallographic analysis. J Chem Crystallogr 12(4):309–319
Chan WR, Taylor DR, Willis CR, Bodden HW (1971) The structure and stereochemistry of neoandrographolide, a diterpene glucoside from Andrographis paniculata Nees. Tetrahedron 27(21):5081–5091
Rastogi RP, Mehrotra BN (1990) Compendium of Indian medicinal plants, vol 3. CDRI Lucknow and PID, New Delhi, pp 441–443
Siripong P, Kongkathip B, Preechanukool K, Picha P, Tunsuwan K, Taylor WC (1992) Cytotoxic diterpenoid constituents from Andrographis paniculata Nees leaves. J Sci Soc Thailand 18(4):187–194
Sagineedu SR, Ng SL, Razak IA, Fun HK, Lajis NH, Stanslas J (2006) 3,19-(2,6-Dimethoxybenzylidene) andrographolide. Acta Cryst 62(12):5806–5808
Srivastava A, Misra H, Verma RK, Gupta MM (2004) Chemical fingerprinting of Andrographis paniculata using HPLC, HPTLC and densitometry. Phytochem Anal 15(5):280–285
Matsuda T, Kuroyanagi M, Sugiyama S, Umehara K, Ueno A, Nishi K (1994) Cell differentiation-inducing diterpenes from Andrographis paniculata Nees. Chem Pharm Bull 42(6):1216–1225
Basak A, Cooper S, Roberge AG, Banik UK, Chretien M, Seidah NG (1999) Inhibition of proprotein convertases-1, 7 and furin by diterpines of Andrographis paniculata and their succinoyl esters. Biochem J 338(1):107–113
Jarukamjorn K, Nemoto N (2008) Pharmacological aspects of Andrographis paniculata on health and its major diterpenoid constituent andrographolide. J Health Sci 54(4):370–381
Ali ME, Biswas KM, Chowdhury SA (1972) Andrographis paniculata. VI. Root flavones and their sturcture. Pak J Sci Ind Res 15:33–36
Govindachari TR, Pal BR, Srinivasa M, Kalyanaram PS (1969) Investigations on Andrographis paniculata. Indian J chem 7:306–310
Xu HW, Dai GF, Liu GZ, Wang JF, Liu HM (2007) Synthesis of andrographolide derivatives: a new family of [alpha]-glucosidase inhibitors. Bioorg Med Chem 15(12):4247–4255
Thisoda P, Rangkadilok N, Pholphana N, Worasuttayangkurn L, Ruchirawat S, Satayavivad J (2006) Inhibitory effect of Andrographis paniculata extract and its active diterpenoids on platelet aggregation. Eur J Pharmacol 553(1–3):39–45
Kataky A, Handique PJ (2010) A brief overview on Andrographis paniculata (Burm. f) Nees., a high valued medicinal plant: boon over synthetic drugs. Asian J Sci Technol 6:113–118
Shahid A (2011) Andrographis paniculata: a review of pharmacological activities and clinical effects. Altern Med Rev 16(1):66–77
Zoha MS, Hussain AH, Choudhuryand SA (1989) Antifertility effects of Andrographis paniculata in mice. Bangladesh Med Res Counc Bull 15:34–37
Liu J, Wang ZT, Ge BX (2008) Andrograpanin, isolated from Andrographis paniculata, exhibits anti-inflammatory property in lipopolysaccharide-induced macrophage cells through down-regulating the p38 MAPKs signaling pathways. Int immunopharm 8(7):951–958
Suebsasana S, Pongnaratorn P, Sattayasai J, Arkaravichien T, Tiamkao S, Aromdee C (2009) Analgesic, antipyretic, anti-inflammatory and toxic effects of andrographolide derivatives in experimental animals. Arch Pharm Res 32(9):1191–1200
Chandra R, Kumarappan CT, Kumar J, Mandal SC (2010) Antipyretic activity of JURU-01-a polyherbal formulation. Global J Pharmacol (GJP) 4(1):45–47
Chandrasekaran CV, Gupta A, Agarwal A (2010) Effect of an extract of Andrographis paniculata leaves on inflammatory and allergic mediators in vitro. J Ethnopharmacol 129(2):203–207
Zhao HY, Fang WY (1991) Antithrombotic effects of Andrographis paniculata nees in preventing myocardial infarction. Chin Med J 104(9):770–775
Mamatha A, Kalpana SP, Purnima A, Kushal., Soujanya., Gokul (2010) Effect of gamma irradiation on pharmacological activity of Andrographis paniculata J Pharm Res 3(11):2638-2639
Sheeja K, Guruvayoorappan C, Kuttan G (2007) Antiangiogenic activity of Andrographis paniculata extract and andrographolide. Int Immunopharmacol 7(2):211–221
Niranjan A, Tewari SK, Lehri A (2010) Biological activities of kalmegh (Andrographis paniculata Nees) and its active principles: a review. Indian J Nat Prod Resour 1(2):125–135
Burgos RA, Hancke JL, Bertoglio JC, Aguirre V, Arriagada S, Calvo M, Cáceres DD (2009) Efficacy of an Andrographis paniculata composition for the relief of rheumatoid arthritis symptoms: a prospective randomized placebo-controlled trial. Clin Rheumatol 28(8):931–946
Rifa’i M (2010) Andrographolide ameliorate rheumatoid arthritis by promoting the development of regulatory T cells. J Trop Life Sci 1(1):5–8
Limsong J, Benjavongkulchai E, Kuvatanasuchati J (2004) Inhibitory effect of some herbal extracts on adherence of Streptococcus mutans. J Ethnopharmacol 92(2–3):281–289
Abubacker MN, Vasantha S (2010) Antibacterial activity of ethanolic leaf extract of Andrographis paniculata Nees (Acanthaceae) and its bioactive compound andrographolide. Drug Inv Today (DIT) 2:440–442
Aromdee C, Sriubolmas N, Wiyakrutta S, Suebsasna S, Khunkitti W (2011) Effect of the derivatives of andrographolide on the morphology of Bacillus subtilis. Arch Pharm Res 34(1):71–77
Xu Y, Chen A, Fry S, Barrow RA, Marshall RL, Mukkur T (2007) Modulation of immune response in mice immunised with an inactivated Salmonella vaccine and gavaged with Andrographis paniculata extract or andrographolide. Int Immunopharmacol 7(4):515–523
Sheeja K, Kuttan G (2007) Modulation of natural killer cell activity, antibody-dependent cellular cytotoxicity, and antibody-dependent complement-mediated cytotoxicity by andrographolide in normal and Ehrlich ascites carcinoma-bearing mice. Integr Cancer Ther 6(1):66–73
Jada SR, Subur GS, Matthews C, Hamzah AS, Lajis NH, Saad MS, Stevens MFG, Stanslas J (2007) Semisynthesis and in vitro anticancer activities of andrographolide analogues. Phytochemistry 68(6):904–912
Woo AYH, Waye MMY, Tsui SKW, Yeung STW, Cheng CHK (2008) Andrographolide up-regulates cellular-reduced glutathione level and protects cardiomyocytes against hypoxia/reoxygenation injury. J Pharmacol Exp Ther 325(1):226–235
Zhang CY, Tan BKH (1997) Mechanisms of cardiovascular activity of Andrographis paniculata in the anaesthetized rat. J Ethnopharmacol 56(2):97–101
Borhanuddin M, Shamsuzzoha M, Hussain A (1994) Hypoglycaemic effects of Andrographis paniculata Nees on non-diabetic rabbits. Bangladesh Med Res Counc Bull 20(1):24–26
Subramanian R, Asmawi MZ, Sadikun A (2008) Effect of ethanolic extract of Andrographis paniculata (Burm. F.) Nees on a combination of fat-fed diet and low dose streptozotocin induced chronic insulin resistance in rats. Diabetol Croatica 37(1):13–22
Lee MJ, Rao YK, Chen K, Lee YC, Chung YS, Tzeng YM (2010) Andrographolide and 14-deoxy-11,12-didehydroandrographolide from Andrographis paniculata attenuate high glucose-induced fibrosis and apoptosis in murine renal mesangeal cell lines. J Ethnopharmacol 132(2):497–505
Shukla B, Visen PKS, Patnaik GK, Dhawan BN (1992) Choleretic effect of andrographolide in rats and guinea pigs. Planta Med 58(2):146–149
Tripathi GS, Tripathi YB (1991) Choleretic action of andrographolide obtained from Andrographis paniculata in rats. Phytother Res 5(4):176–178
Gupta S, Yadava JNS, Tandon JS (1993) Antisecretory (antidiarrhoeal) activity of Indian medicinal plants against Escherichia coli enterotoxin-induced secretion in rabbit and guinea pig ileal loop models. Pharm Biol 31(3):198–204
Gupta S, Choudhry MA, Yadava JNS, Srivastava V, Tandon JS (1990) Antidiarrhoeal activity of diterpenes of Andrographis paniculata (Kal-Megh) against Escherichia coli enterotoxin in in vivo models. Pharm Biol 28(4):273–283
Farnsworth NR, Bunyapraphatsara N (1992) Thai medicinal plants (recommended for primary health care system), 1st edn. Prachachon Press, Bangkok, pp 130–142
Khalsa Singh KP, Tierra M (2008) The way of ayurvedic herbs. Lotus Press, Twin Lakes, p 380
Ko HC, Wei BL, Chiou WF (2006) The effect of medicinal plants used in Chinese folk medicine on RANTES secretion by virus-infected human epithelial cells. J Ethnopharmacol 107(2):205–210
Khare CP (2004) Indian herbal remedies: rational western therapy, ayurvedic, and other traditional usage, botany. Springer Verlag, Berlin, p 525
Choudhury BR, Poddar MK (1984) Andrographolide and kalmegh (Andrographis paniculata) extract: in vivo and in vitro effect on hepatic lipid peroxidation. Methods Find Exp Clin Pharmacol 6(9):481–485
Mehrotra R, Rawat S, Kulshreshtha DK, Patnaik GK, Dhawan BN (1990) In vitro studies on the effect of certain natural products against hepatitis B virus. Indian J Med Res 92:133–138
Trivedi NP, Rawal UM, Patel BP (2007) Hepatoprotective effect of andrographolide against hexachlorocyclohexane-induced oxidative injury. Integr Cancer Ther 6(3):271–280
Kapil A, Koul IB, Banerjee SK, Gupta BD (1993) Antihepatotoxic effects of major diterpenoid constituents of Andrographis paniculata. Biochem Pharmacol 46(1):182–185
Trivedi N, Rawal UM (2000) Hepatoprotective and toxicological evaluation of Andrographis paniculata on severe liver damage. Indian J Pharmacol 32(5):288–293
Dumrongsak P, Nadege B, Catherine A, Georges M, Alail B, Helene M (2009) Effect of Andrographis paninculata extract and Andrographolide on hepatic cyt P450 mRNA expression and monoxygenase activity after in vivo administration to rats and in vitro in rat and human hepatic cultures. Chem Biol Interact 179(2–3):247–255
Coon JT, Ernst E (2004) Andrographis paniculata in the treatment of upper respiratory tract infections: a systematic review of safety and efficacy. Planta Med 70(4):293–298
Gairola S, Gupta V, Bansal P, Singh R, Maithani M (2010) Herbal antitussives and expectorants: a review. Int J Pharm Sci Rev Res 5(2):5–9
Kokate CK, Gokhale MSB, Gokhale AS (2007) Cultivation of medicinal plants, 5th edn. Nirali Prakashan, Pune, pp 11–79
World Health Organization (1990) Medicinal plants in Vietnam. WHO Regional publication, Western pacific series. Manila 3:11–90
Wiart C, Kumar K, Yusof M, Hamimah H, Fauzi Z, Sulaiman M (2005) Antiviral properties of ENT labdene diterpenes of Andrographis paniculata Nees, inhibitors of herpes simplex virus type 1. Phytother Res 19(12):1069–1070
Puri A, Saxena R, Saxena RP, Saxena KC, Srivastava V, Tandon JS (1993) Immunostimulant agents from Andrographis paniculata. J Nat Prod 56(7):995–999
Choudhari AB, Rangari VD, Darvekar VMI (2011) Formulation development for treatment and management of HIV-Aids. Int J Pharm Pharm Sci 3(1):105–108
Chang RS, Ding L, Gai-Qing C, Smith KM (1991) Dehydroandrographolide succinic acid monoester as an inhibitor against human immunodeficiency virus (HIV). Proc Soc Exp Biol Med 197:59–66
Basak A, Banik UK, Basak S, Seidah NG, Li S (2006) Evaluation of Anti-Proprotein Convertase Activity of Diterpene Andrographolid Derived Products. In: Khatib A-Majid (ed) Regulation of carcinogenesis, angiogenesis and metastasis by the proprotein convertases (PCs), a new potential strategy in cancer therapy. Springer, The Netherlands, pp 137–154
Reddy VLN, Reddy SM, Ravikanth V, Krishnaiah P, Goud TV, Rao T, Ram TS, Gonnade RG, Bhadbhade M, Venkateswarlu Y (2005) A new bis-andrographolide ether from Andrographis paniculata nees and evaluation of anti-HIV activity. Nat Prod Res 19(3):223–230
Calabrese C, Berman SH, Babish JG, Ma X, Shinto L, Dorr M, Wells K, Wenner CA, Standish LJ (2000) A phase I trial of andrographolide in HIV positive patients and normal volunteers. Phytother Res 14(5):333–338
Lü W (1995) Prospect for study on treatment of AIDS with traditional Chinese medicine. J Tradit Chin Med 15:3–9
Liu J, Wang ZT, Ji LL (2007) In vivo and in vitro anti-inflammatory activities of neoandrographolide. Am J Chin Med 35(2):317–328
Chao WW, Kuo YH, Lin BF (2010) Anti-inflammatory Activity of New Compounds from Andrographis paniculata by NF-KB Transactivation Inhibition. J Agric Food Chem (58):2505-2512
Yi-Feng X, Bu-Qing Y, Yi-Dan L, Jian-Guo W, Xiang-Jiu H, Xianfeng L, Xinsheng Y, Dawei M, Arne S, Robert PH, Nigel SK, Jian-Guo G (2004) Andrographolide Attenuates Inflammation by Inhibition of NF-B Activation through Covalent Modification of Reduced Cysteine 62 of p501. J Immunol (22):4207-4217
Das S, Gautam N, Dey SK, Maiti T, Roy S (2009) Oxidative stress in the brain of nicotine-induced toxicity: protective role of Andrographis paniculata Nees and vitamin E. Appl Physiol Nutr Metab 34(2):124–135
Chan SJ, Wong W, Wong PTH, Bian JS (2010) Neuroprotective effects of andrographolide in a rat model of permanent cerebral ischaemia. Br J Pharmacol 161(3):668–679
Nik Najib NAR, Furuta T, Kojima S, Takane K, Mohd MA (1999) Antimalarial activity of extracts of Malaysian medicinal plants. J Ethnopharmacol 64(3):249–254
Dua VK, Ojha VP, Roy R, Joshi BC, Valecha N, Devi CU, Bhatnagar MC, Sharma VP, Subbarao SK (2004) Anti-malarial activity of some xanthones isolated from the roots of Andrographis paniculata. J Ethnopharmacol 95(2–3):247–251
Mishra K, Dash AP, Swain BK, Dey N (2009) Anti-malarial activities of Andrographis paniculata and Hedyotis corymbosa extracts and their combination with curcumin. Malaria J 12:8–26
Shen YC, Chen CF, Chiou WF (2002) Andrographolide prevents oxygen radical production by human neutrophils: possible mechanism (s) involved in its anti inflammatory effect. Br J Pharmacol 135(2):399–406
Wang T, Liu B, Zhang W, Wilson B, Hong JS (2004) Andrographolide reduces inflammation-mediated dopaminergic neurodegeneration in mesencephalic neuron-glia cultures by inhibiting microglial activation. J Pharmacol Exp Ther 308(3):975–983
Radhika P, Sastry BS, Harika B (2008) Antimicrobial screening of Andrographis paniculata (Acanthaceae) root extracts. Res J Biotech 3(3):62–63
Singha PK, Roy S, Dey S (2003) Antimicrobial activity of Andrographis paniculata. Fitoterapia 74(7–8):692–694
Rao NK (2006) Anti-hyperglycemic and renal protective activities of Andrographis paniculata roots chloroform extract. Iranian J Pharm Ther (IJPT) 5(1):47–50
Singh P, Srivastava MM, Khemani LD (2009) Renoprotective effects of Andrographis paniculata (Burm. f.) Nees in rats. Ups J Med Sci 114(3):136–139
Lin FL, Wu SJ, Lee SC, Ng LT (2009) Antioxidant, antioedema and analgesic activities of Andrographis paniculata extracts and their active constituent andrographolide. Phytother Res 23(7):958–964
Sulaiman M, Zakaria Z, Abdul Rahman A, Mohamad A, Desa M, Stanslas J, Moin S, Israf D (2010) Antinociceptive and antiedematogenic activities of andrographolide isolated from Andrographis paniculata in animal models. Biol Res Nurs 11(3):293
Deng WL (1978) Outline of current clinical and pharmacological research on Andrographis paniculata in China. Newsletter Chinese Herbal Med 10:27–31
Sheeja K, Shihab PK, Kuttan G (2006) Antioxidant and anti-inflammatory activities of the plant Andrographis paniculata Nees. Immunopharmacol Immunotoxicol 28(1):129–140
Vasu S, Palaniyappan V, Badami S (2010) A novel microwave-assisted extraction for the isolation of andrographolide from Andrographis paniculata and its in vitro antioxidant activity. Nat Prod Res 24(16):1560–1567
Raj RK (1975) Screening of indigenous plants for anthelmintic action against human Ascaris lumbricoides: Part-II. Indian J Physiol Pharmacol 19(1):47–49
Rakel D (2007) Integrative medicine, 2nd edn. Saunders Elsevier, Philadelphia, p 1214
Caceres DD, Hancke JL, Burgos RA, Sandberg F, Wikman GK (1999) Use of visual analogue scale measurements (VAS) to asses the effectiveness of standardized Andrographis paniculata extract SHA-10 in reducing the symptoms of common cold. A randomized double blind-placebo study. Phytomedicine 6(4):217–223
Caceres DD, Hancke JL, Burgos RA, Wikman GK (1997) Prevention of common colds with Andrographis paniculata dried extract: a pilot double blind trial. Phytomedicine 4:101–104
World Health Organization. (1993) ASEAN Countries Standard of ASEAN herbal medicine. Aksara Buana Printing, Jakarta 1:193–206
World Health Organization (2002) WHO Monographs on selected medicinal plants, Geneva, 2:12–24
Melchior J, Palm S, Wikman G (1997) Controlled clinical study of standardized Andrographis paniculata extract in common cold-a pilot trial. Phytomedicine 3:315–318
Melchior J, Spasov AA, Ostrovskij OV, Bulanov AE, Wikman G (2000) Double-blind, placebo-controlled pilot and phase III study of activity of standardized Andrographis paniculata Herba Nees extract fixed combination (Kan jang) in the treatment of uncomplicated upper-respiratory tract infection. Phytomedicine 7(5):341–350
Muangman V, Viseshsindh V, Ratana-Olarn K, Buadilok S (1995) The usage of Andrographis paniculata following extracorporeal shock wave lithotripsy (ESWL). J Med Assoc Thai 78(6):310–312
Gabrielian ES, Shukarian AK, Goukasova GI, Chandanian GL, Panossian AG, Wikman G, Wagner H (2002) A double blind, placebo-controlled study of Andrographis paniculata fixed combination Kan Jang in the treatment of acute upper respiratory tract infections including sinusitis. Phytomedicine 9(7):589–597
Thamlikitkul V, Dechatiwongse T, Theerapong S, Chantrakul C, Boonroj P, Punkrut W, Ekpalakorn W, Boontaeng N, Taechaiya S, Petcharoen S, Riewpaiboon W (1991) Efficacy of Andrographis paniculata, Nees for pharyngotonsillitis in adults. J Med Assoc Thai 74:437–442
Langone Medical Centre (2010) Herbs and suppliments: Andrographis paniculata. EBSCO Publishing. 5 Pages
Roy DN, Sen G, Chowdhury KD, Biswas T (2011) Combination therapy with andrographolide and D-penicillamine enhanced therapeutic advantage over monotherapy with D-penicillamine in attenuating fibrogenic response and cell death in the periportal zone of liver in rats during copper toxicosis. Toxicol Appl Pharmacol 250:54–68
Chang HM, But PPH, Yao SC, Wang LL, Yeung SCS (1986) Pharmacology and applications of Chinese materia medica, Vols 1 & 2. World Scientific Publishing, Singapore, 1:918–928
Xue-jun Y, De-xiang L, Hechuan W, Yu Z (1991) A study on the mutagenicity of 102 raw pharmaceuticals used in Chinese traditional medicine. Mutat Res Genet Toxicol 260(1):73–82
Akbarsha M, Manivannan B, Shahul H (1990) Antifertility effect of Andrographis paniculata (Nees) in male albino rat. Indian J Exp Biol 28(5):421–426
State Administration TCM (1998) Chinese herbal medicine. Shanghai Science and Technology, 2:17–67
Burgos RA, Caballero EE, Sanchez NS, Schroeder RA, Wikman GK, Hancke JL (1997) Testicular toxicity assesment of Andrographis paniculata dried extract in rats. J Ethnopharmacol 58(3):219–224
Burgos RA, Aguila MJ, Santiesteban E (2001) Andrographis paniculata (Ness) induces relaxation of uterus by blocking voltage operated calcium channels and inhibits Ca+2 influx. Phytother Res 15(3):235–239
Spasov AA, Ostrovskij OV, Chernikov MV, Wikman G (2004) Comparative controlled study of Andrographis paniculata fixed combination, Kan Jang and an echinacea preparation as adjuvant, in the treatment of uncomplicated respiratory disease in children. Phytother Res 18(1):47–53
Kulichenko L, Kireyeva L, Malyshkina E, Wikman G (2003) A randomized, controlled study of Kan Jang versus amantadine in the treatment of influenza in Volgograd. J Herb Pharmacother 3(1):77–93
Mkrtchyan A, Panosyan V, Panossian A, Wikman G, Wagner H (2005) A phase I clinical study of Andrographis paniculata fixed combination Kan Jang (TM) versus ginseng and valerian on the semen quality of healthy male subjects. Phytomedicine 12(6–7):403–409
Fauziah S, Putra H, Rasyid L (2009) The effect of combinafios of Curcuma Xanthorrhiza Roxb. extract and Andrographis Paniculate Ness. extract on human gallbladder. Agrobiotechnol 1:58–61
Pawar Pk, Sharma S, Singh KC, Sharma RK (2011) Physico-chemical standardisation and development of HPTLC method for the determination of andrographonin in kalmegh navayasloha: an ayurvedic formulation. Int J Res Ayurveda Pharm (IJRAP) 2(1):295–301
Reyes BAS, Bautista ND, Tanquilut NC, Anunciado RV, Leung AB, Sanchez GC, Magtoto RL, Castronuevo P, Tsukamura H, Maeda KI (2006) Anti-diabetic potentials of Momordica charantia and Andrographis paniculata and their effects on estrous cyclicity of alloxan-induced diabetic rats. J Ethnopharmacol 105(1–2):196–200
Mishra US, Mishra A, Kumari R, Murthy PN, Naik BS (2009) Antibacterial activity of ethanol extract of Andrographis paniculata. Indian J Pharm Sci 71(4):436–438
Chao WW, Kuo YH, Hsieh SL, Lin BF (2011) Inhibitory effects of ethyl acetate extract of Andrographis paniculata on NF-B trans-activation activity and LPS-induced acute inflammation in mice. Evid Based Complement Alternat Med 2011:1–9
Rafat A, Philip K, Muniandy S (2010) Antioxidant potential and content of phenolic compounds in ethanolic extracts of selected parts of Andrographis paniculata. J Med Plants Res 4(3):197–202
Silja VP, Samitha Varma K, Mohanan KV (2008) Ethnomedicinal plant knowledge of the Mullu kuruma tribe of Wayanad district, Kerala. Indian J Tradit Know 7(4):604–612
Nesari T, Bhagwat BK, Johnson J, Bhatt NS, Chitre D (2005) Clinical validation of efficacy and safety of herbal Cough Formula. J Herb Pharmacother 4(4):1–12
Patil DA (2011) Ethnomedicine to modern medicine: genesis through ages. J Exp Sci (JES) 2(3):25–29
Nagaraja TG, Deshmukh SM (2009) Phytotoxic effect of Andrographis paniculata Nees on metabolism of Parthenium hysterophorus L. J Biopest 2(2):165–167
Kuppusamy C, Murgan K (2009) Mosquitocidal effect of Andographis paniculata Nees against the malaria vector, Anopheles stephensi Liston (Diptera: culicidae). Int J Integr Biol 5(2):75–81
Mathivanan R, Edwin SC, Amutha R, Viswanathan K (2006) Panchagavya and Andrographis paniculata as alternatives to antibiotic growth promoter on broiler production and carcass characteristics. Int J Poult Sci 5(12):1144–1150
Tipakorn N (2002) Effects of Andrographis paniculata (Burm. F.) Nees on performance, mortality and coccidiosis in broiler chickens. Institute of Animal Physiology and Animal Nutrition. Georg-August Universitat, Gottingen
Chopra RN, Nayer SL, Chopra CI (1992) Glossary of Indian medicinal plants, 3rd edn. Council of Scientific and Industrial Research, New Delhi, p 246
Karami M, Alimon AR, Goh YM (2011) Effect of vitamin E, Andrographis paniculata and turmeric as dietary antioxidant supplementation on lipid and color stability of goat meat. Small Ruminant Res 97:67–71
Karami M, Alimon A, Sazili A, Goh Y, Ivan M (2011) Effects of dietary antioxidants on the quality, fatty acid profile, and lipid oxidation of longissimus muscle in kacang goat with aging time. Meat Sci 88:102–108
Chansue N (2006) Efficacies of dry Indian almond leaf (Terminalia catappa) and Andrographis paniculata (Burn. F) wall. ex nees extract on tail growth and hematocrit of fancy carp (Cyprinus carpio L.). J Thai Vet Med Assoc 57(2):52–62
Tondiew C, Jintasataporn O, Tabthipwon P, Chumkam S (2007) Effect of Noni (Morinda citrifolia) and Fahtalaijons (Andrographis paniculata) on pigmentation and phagocytosis in goldfish (Carasius auratus) Proceedings of the 45th Kasetsart University Annual Conference, Kasetsart University, Thailand. 538-546
Dutta A, Sukul NC (1982) Filaricidal properties of a wild herb, Andrographis paniculata. J Helminthol 56(2):81–84
Ghosh M, Thangaman D, Thapliyal M, Yasodha R, Gurumurth K (2004) Purification of antifungal protein against blister bark pathogen of Casuarina equisetifolia JR Forster et G. Forster. Acta Bot Croat 63:75–81
Singh A, Singh VK, Quraishi MA (2010) Aqueous extract of kalmegh (Andrographis paniculata) leaves as green inhibitor for mild steel in hydrochloric acid solution. Int J Corrosion 2010:1–10
Bhatnagar P, A., Raikwar N, Lodhi S, Mishra A (2008) Marketing Information Service of medicinal plants in Central India. In: Report of the national workshop on sustainable management of Non Timber Forest Products Organized by RCDC and TFRI Jabalpur, India
Sajwan BS (2008) Medicinal plants scenario in india-government initiatives towards resource security and quality assurance for increased trade and commerce. In: International conclave on medicinal plants for ASEAN and BIMSTEC countries Lamphelpat, Imphal, India, pp 11-25
Karuppusamy S, Kalimuthu K (2010) Rapid in vitro multiplication and plant regeneration from nodal explants of Andrographis neesiana: a valuable endemic medicinal plant. Adv Biol Res 4(4):211–216
Colfer CJP, Sheil D, Kaimowitz D, Kishi M (2006) Forests and human health in the tropics: some important connections. Unasylva 224(57):3–10
Hansda R (2009) The outlook for non-wood forest products in Asia and the Pacific. Working Paper No. APFSOS II/WP/2009/18. Asia-Pacific Forestry Sector Outlook Study II Working Paper Series. FAO, Bangkok
FAO (2003) Projections of tobacco production, consumption and trade to the year 2010. Rome, Italy. 101 Pages
Gadgil M (2004) Karnataka state biodiversity strategy and action plan (KBSAP). Environmental Information System (ENVIS), Centre for Ecological Sciences, Indian Institute of Science, Bangalore, p 418
Pushpangadan P, Nair KN, Ahmad MR (2001) Biodiversity and Medicinal Plant Wealth of South Asian Countries. UNESCO sponsored “Regional Training Programme on Biodiversity Systematics: Evaluation and Monitoring with Emphasis on Medicinal Plants” held at NBRI Lucknow, India
Verma N, Vinayak M (2008) Antioxidant action of Andrographis paniculata on lymphoma. Mol Biol Rep 35:535–540
Merawin LT, Arifah AK, Sani RA, Somchit MN, Zuraini A, Ganabadi S, Zakaria ZA (2010) Screening of microfilaricidal effects of plant extracts against Dirofilaria immitis. Res Vet Sci 88:142–147
Yoshida S (1981) Fundamentals of rice crop science. The International Rice Reasearch Institute, Las Banos
Anonymous (2007) National Medicinal Plant Board, Asia-Pacific Forestry Sector Outlook Study II. Country report, Ministry of Environment and Forests, New Delhi, India
Kala CP, Dhyani PP, Sajwan BS (2006) Developing the medicinal plants sector in northern India: challenges and opportunities. J Ethnobiol Ethnomed 2:32
Moideen SVK (2008) Review of medicinal plants status in Malaysia. In: International conclave on medicinal plants for ASEAN and BIMSTEC countries Lamphelpat, Imphal, India, pp 26-29
Chuthaputti A, Chawapradit P (2008) Current situation of medicinal plant cultivation and utilization in Thailand. In: International conclave on medicinal plants for ASEAN and BIMSTEC countries Lamphelpat, Imphal, India, pp 44-51
Natarajan D, Britto SJ, Balaguru B, Nagamurugan N, Soosairaj S, Arockiasamy DI (2003) Identification of conservation priority sites using remote sensing and GIS: a case study from Chitteri hills, Eastern Ghats, Tamil Nadu. Curr Sci 86(9):1316–1323
Pholphana N, Rangkadilok N, Thongnest S, Ruchirawat S, Ruchirawat M, Satayavivad J (2004) Determination and variation of three active diterpenoids in Andrographis paniculata (Burm. f.) Nees. Phytochem Anal 15(6):365–371
Shivaprasad HN, Pharm M (2008) Expectations of natural/herbal products industry. In: International conclave on medicinal plants for ASEAN and BIMSTEC countries Lamphelpat, Imphal, India, pp 78–82
Acknowledgments
We would like to express our cordial thanks to Prof. Dr. Mehdi Tajbakhsh and Prof. Dr. Majid Nojavan from Urmia University (West Azerbaijan-Iran), for their admirable advice. Our appreciation is also extended to Dr. Arash Javanmard and Dr. Vahid Omidvar for their assistance in providing useful papers for our review. The special thank of the authors goes to our labmate Mr. Ahmad Sharifkhani for his valuable helps to capture professional photos (Fig. 1).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Valdiani, A., Kadir, M.A., Tan, S.G. et al. Nain-e Havandi Andrographis paniculata present yesterday, absent today: a plenary review on underutilized herb of Iran’s pharmaceutical plants. Mol Biol Rep 39, 5409–5424 (2012). https://doi.org/10.1007/s11033-011-1341-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-011-1341-x