Abstract
Visfatin, a newly identified proinflammatory adipokine, has been linked to coronary artery disease (CAD). The −1535C>T polymorphism (rs61330082) located in the visfatin gene promoter is reportedly associated with proinflammatory status. However, it is unclear whether this polymorphism correlates with plasma levels of inflammatory markers including visfatin, hs-CRP, IL-6 and TNF-α in CAD patients. The present study was to investigate the potential association of the −1535C>T polymorphism with plasma levels of visfatin, IL-6, C reactive protein (hs-CRP) and TNF-α in patients with CAD. We conducted a hospital based study with 171 CAD patients to examine the association between the −1535C>T polymorphism and plasma levels of visfatin, hs-CRP, IL-6 and TNF-α. Plasma visfatin levels were markedly different between patients with stable angina pectoris (SAP, 11.91 ± 0.70 ng/l) and those with unstable angina pectoris (UAP, 17.49 ± 0.20 ng/l) or acute myocardial infarction (AMI, 16.63 ± 0.22 ng/l; SAP versus UAP or AMI, P < 0.05). Compared with the CC genotype, variant genotypes CT and TT correlated with significantly lower levels of visfatin, hs-CRP, IL-6 and TNF-α in the SAP group (P < 0.05), with lower levels of hs-CRP and IL-6 in the UAP group (P < 0.05), and with lower levels of visfatin in the AMI group (P < 0.05) after adjustment for age, gender, smoking, hypertension, diabetes, dyslipidemia and medication. Our results suggest that the −1535C>T polymorphism is associated with decreased plasma levels of inflammatory markers in CAD patients, reflecting that this polymorphism might provide a useful marker for predicting the development of CAD events.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Coronary artery disease (CAD) is a major cause of mortality worldwide [1]. Many studies have revealed the central position of inflammation in the pathogenesis of atherosclerosis, which is the main cause of CAD [2]. The mechanisms of atheroma plaque instability are a new focus of interest to understand the pathophysiology of development of CAD [3]. Visfatin (also known as pre-B cell colony enhancing factor), was originally cloned as a growth factor that enhances differentiation of pre-B cells in synergy with interleukin 7 (IL-7) and stem cell factor [4]. Recent studies have reported that serum visfatin levels are correlated with the serum levels of CRP, IL-6 and TNF-α, indicating that circulating visfatin may reflect inflammatory status [5, 6]. Visfatin has also been shown to potentially play a role in plaque destabilization [7]. In addition, one recent study showed that visfatin might be an independent risk factor for acute coronary syndrome (ACS) [8].
It has been reported that a visfatin promoter polymorphism is linked to acute and low-grade inflammation [9, 10]. The −1535C>T polymorphism (rs61330082), was shown to be associated with the regulation of visfatin gene expression and serum lipid level [10–12], although some conflicting results were also reported [10, 11]. However, our previous study demonstrated that this genetic variant is associated with a decreased risk of CAD in a Chinese population [13]. The aim of the present hospital-based study was to investigate the potential association of the −1535C>T polymorphism with plasma levels of visfatin, IL-6, hs-CRP, and TNF-α in patients with CAD.
Materials and methods
Study population
From May 2008 to December 2008, the study enrolled 171 consecutive unrelated patients with CAD admitted to the First Affiliated Hospital of Nanjing Medical University. The diagnosis of CAD was certified by coronary angiography performed with the Judkins technique via a quantitative coronary angiographic system [14]. CAD was defined as luminal narrowing of more than 50% in one or more main coronary arteries. Two cardiologists who assessed the angiograms were blinded to the patients’ histories. Patients were divided into three subgroups: stable angina pectoris (SAP 41 patients), unstable angina pectoris (UAP, 96 patients), and acute myocardial infarction (AMI, 32 patients), which were defined as previously described [15]. All subjects enrolled were of Han Chinese origin and resided in or near Jiangsu Province. Patients were excluded if they had a history of significant concomitant diseases, including cardiomyopathy, bleeding disorders, renal failure, previous thoracic irradiation therapy, or malignant diseases. Hypertension was defined as resting systolic blood pressure >140 mmHg and/or diastolic blood pressure >90 mmHg or if the patient was actively treated with antihypertensive agents. Diabetes was defined as fasting blood glucose >7.0 mmol/l or a diagnosis of diabetes needing diet or antidiabetic drug therapy. Dyslipidemia was defined as total cholesterol level of >5.72 mmol/l or if the patient was being treated with cholesterol-lowering medication. Information on smoking status (current smoker versus nonsmoker) was obtained from a questionnaire. Individuals who formerly or currently smoked ≥10 cigarettes per day for at least 2 years were defined as smokers. This study was approved by the First Affiliated Hospital of Nanjing Medical University Ethics Committee, and informed consent was obtained from each participant.
DNA extraction and genotyping
Peripheral venous blood was drawn from each subject. Genomic DNA was extracted with the AxyPrep DNA Blood kit (Axygen Scientific Inc, Union City, CA, USA). The −1535C>T (rs61330082) polymorphism was genotyped by the polymerase chain reaction–ligase detection reaction (PCR–LDR) sequencing method, as reported previously [13]. Additionally, about 10% of the samples were randomly selected and retested, and the results were 100% concordant.
Laboratory measurements
A fasting venous blood was drawn from each subject and anticoagulated with EDTA (dipotassium salt). Samples were separated immediately after collection by centrifugation at 2,000 × g for 15 min and stored at −70°C until analysis. The plasma levels of total cholesterol (TC), triglyceride (TG), high-density lipoprotein cholesterol (HDL-C), low-density lipoprotein cholesterol (LDL-C), fasting blood glucose, and high-sensitivity CRP (hs-CRP) were measured as described previously [16]. Plasma visfatin levels were determined by ELISA method [Human visfatin ELISA kit, Phoenix Pharmaceuticals, Belmont, CA, USA; sensitivity (minimum detectable concentration), 2.30 ng/ml; IntraCV < 5% and InterCV < 12%]. Plasma IL-6 levels were measured by ELISA method (Human IL-6 ELISA kit, R&D Systems Inc. Minneapolis, MN, USA; range, 4.7–300 pg/ml; sensitivity < 0.5 pg/ml). Plasma TNF-α levels were assessed by ELISA method (Human TNF-α ELISA kit, R&D Systems Inc.; range, 15.6–1000 pg/ml; sensitivity < 2 pg/ml). To eliminate inter-assay variability, all samples were tested in a single assay.
Statistical analysis
Statistical analyses were carried out with Stata ver. 8.0 (STATA Corp., College Station, TX) and SPSS 13.0 (SPSS Inc., Chicago, IL). Non-normally distributed variables were expressed as median (range), and normally distributed variables were expressed as mean ± SEM as appropriate. To normalize the skewed distributions, log-transformations were applied when needed (hs-CRP, TNF-α, IL-6 and visfatin). Normality was tested using the Kolmogorov–Smirnov test. The Student’s t-test was used to estimate the differences in continuous variables without skew (presented as mean ± SEM). Differences in continuous variables departing from the normal distribution even after transformation (presented as median and interquartile range) were analyzed by Mann–Whitney U-test. Pearson χ 2-test was used to compare allele distribution and qualitative variables represented as frequencies. Hardy–Weinberg equilibrium was assessed by a χ 2 goodness-of-fit test. Pearson’s correlation was performed to examine association of visfatin with hs-CRP, TNF-α and IL-6. Multivariate analysis of variance (MANOVA) using general linear model was performed to test correlations between variant genotypes and plasma levels of hs-CRP, IL-6, TNF-alpha and visfatin (log-transformation) after adjustment for age, sex, smoking, hypertension, diabetes, dyslipidemia and medication. A two-tailed P-value <0.05 was considered to be statistically significant.
Results
Demographic information
The baseline characteristics of study subjects are shown in Table 1. Patients with UAP (65 ± 1 year) were older than those with SAP (61 ± 2 year). Patients with AMI had higher TG and circulating inflammatory marker levels, such as hs-CRP, TNF-α and IL-6 than those patients with SAP and UAP. In particular, plasma visfatin levels were markedly lower in patients in the SAP subgroup compared with the levels in patients in the UAP and AMI subgroups (SAP, 11.91 ± 0.70 ng/l; UAP, 17.49 ± 0.20 ng/l; AMI, 16.63 ± 0.22 ng/l, SAP versus UAP or AMI; P < 0.05). No significant differences were observed in gender, body mass index (BMI), hypertension, diabetes mellitus, TC, HDL-C, LDL-C, or smoking status among the three subgroups. In addition, no differences were evident among the three subgroups regarding use of medications, including beta-blockers, calcium channel blockers, statins, angiotensin converting enzyme inhibitors, and angiotensin receptor blockers.
Distribution of the visfatin genotype
The genotype distribution in our study subjects showed no deviation from Hardy–Weinberg equilibrium (P > 0.05). The distribution of the CC genotype among patients with AMI was significantly different than that in the SAP and UAP subgroups (AMI, 59.4%; SAP, 31.7%; UAP, 33.4%; AMI and UAP versus SAP, P < 0.05; AMI versus UAP, P < 0.05) as shown in Table 1.
Association of visfatin with hs-CRP, TNF-α, and IL-6
Plasma visfatin level was positively correlated with plasma levels of hs-CRP (r = 0.663, P < 0.001), IL-6 (r = 0.410, P = 0.008), and TNF-α (r = 0.279, P = 0.077) in the SAP subgroup (Fig. 1). However, no significant correlation between plasma visfatin concentration and plasma levels of hs-CRP, IL-6 or TNF-α was found in the UAP and AMI subgroups (data not shown).
Association of the −1535C>T polymorphism with plasma levels of inflammatory markers
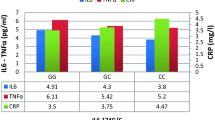
Figure 2 presents the association of the −1535C>T variant with the concentrations of visfatin, hs-CRP, IL-6, and TNF-α. Compared to the CC genotype, variant genotypes CT and TT were independently correlated with lower levels of visfatin, hs-CRP, IL-6, and TNF-α in the SAP subgroup (P < 0.05), but only with levels of hs-CRP and IL-6 in the UAP subgroup (P < 0.05), and only with visfatin in the AMI subgroup (P < 0.05).
Levels of hs-CRP, IL-6, TNF-alpha and visfatin in SAP (a), UAP (b) and AMI (c) according to visfatin genotypes. Data are presented as means ± SEM. Multivariate analysis of variance (MANOVA) using general linear model was performed to test correlations between variant genotypes and plasma levels of hs-CRP, IL-6, TNF-alpha and visfatin (log-transformation) after adjustment for age, sex, smoking, hypertension, diabetes, dyslipidemia and medication (* P < 0.05, ** P < 0.01 for CT+TT versus CC)
Discussion
In the present study, we analyzed the association between the −1535C>T polymorphism and plasma levels of inflammatory mediators, including visfatin, hs-CRP, IL-6 and TNF-α in patients with CAD. According to our data, the −1535 C to T variant was associated with reduced plasma levels of visfatin, hs-CRP, IL-6 and TNF-α in subgroups of SAP, UAP and AMI in a Chinese population. In addition, plasma levels of visfatin, hs-CRP and IL-6 were significantly elevated in the UAP and AMI subgroups compared with those in the SAP subgroup (P < 0.05).
Mounting evidence shows that CAD and ACS are diseases with multifactorial causes. Recent studies revealed that proinflammatory cytokines and genetic factors contribute to the development of CAD and ACS [17–19]. Markedly increased concentrations of hs-CRP and IL-6 are also found in patients with ACS [20]. These results support the hypothesis that proinflammatory cytokines might not only play key roles in the development of CAD, but also might provide useful markers for predicting the development of CAD events.
Visfatin, a 52-kDa protein, appears to be predominantly produced by the visceral adipose tissue and seems to have insulin-like actions [21]. Visfatin expression was upregulated in amniotic epithelial cells by cytokines including IL-1b, TNF-a and IL-6 [22]. Conversely, recombinant visfatin increases the expression of IL-6 and IL-8, suggesting a complex interaction between visfatin and other inflammatory cytokines [23]. We found a significantly positive correlation between plasma concentrations of visfatin and hs-CRP (r = 0.663, P < 0.001) and IL-6 (r = 0.410, P = 0.008) in the SAP subgroup. Although not significant, we also observed a trend for an association between plasma levels of visfatin and TNF-α (r = 0.279, P = 0.077) in the SAP subgroup. These findings indicate that visfatin might be associated with proinflammatory response. However, there were no significant correlations between plasma levels of visfatin and hs-CRP, IL-6 and TNF-α in the subgroups of UAP and AMI (data not shown). The precise reason for that is yet to be known.
Visfatin also induces endothelial vascular endothelial growth factor (VEGF) and matrix metalloproteinase (MMP) production and activity [24], suggesting that it has a vital role in the pathogenesis of vascular inflammation. Moreover, visfatin also has been shown to be localized to foam cell macrophages within unstable atherosclerotic lesions [7]. Liu et al. [8] also reported that plasma visfatin levels were significantly correlated with CAD independent of other traditional CAD risk factors. Thereby, visfatin could potentially play a role in plaque destabilization, which is the most frequent cause of ACS from a pathologic and angioscopic perspective [25, 26]. In our study, plasma visfatin levels were markedly different between patients with SAP and patients with UAP or AMI, demonstrating that visfatin potentially plays an important role in the development of CAD.
It has been universally accepted that ACS, including UAP and AMI, could cause much more serious cardiac events than SAP. In the present study, the distribution of the −1535C>T genotype was significantly different among the three subgroups. Genotypes with variants CT and TT were less frequent in the UAP and AMI groups compared to the SAP group. It is possible that the T variant might present a protective effect, whereas the C allele might increase risk with respect to severity of CAD. This result is consistent with that of our previous study [13].
It has been reported that circulating inflammatory mediator levels are substantially influenced by environmental and genetic factors [27]. In addition, investigators have suggested that inflammatory genetic polymorphisms also contribute to the variation in plasma levels of inflammatory markers such as hs-CRP, IL-6 and TNF-α in patients with CAD [19, 28]. We also noted that variant genotypes CT and TT were more significantly correlated with lower levels of visfatin in SAP and AMI subgroups (P < 0.05) than CC genotype after adjustment for confounding factors including age, gender, BMI, smoking, hypertension, diabetes, dyslipidemia, and medication usage. The fact that patients with genotypes CT and TT had significantly lower plasma visfatin levels in the SAP and AMI subgroups, probably results from this variant having lower transcriptional activity. Ye et al. [10] showed that the T variant in the T-1535C SNP (described as C-1543T in their study) resulted in nearly a twofold decrease in the reporter gene expression and transcription of visfatin was decreased in patients with the T variant. Therefore, the T allele might have lower levels of visfatin gene transcription and enzymatic activity than C allele. Furthermore, Liu et al. [29] also confirmed that, compared to the C allele, the T allele significantly attenuated its binding to an IL-1β induced unknown transcription factor in pulmonary vascular endothelial cells, which might reduce expression of PBEF. In contrast, study by Tokunaga et al. [11] suggested that this variation might be not functional. This discrepancy maybe accounted for by differences in experimental conditions, ethnicity and geography. However, the reason for the lack of association between the −1535C>T polymorphism and plasma visfatin level in the UAP subgroup might be the relatively small number of patients. Additional studies are needed to clarify the relationship.
In this study, we also observed a significant relation between the −1535C>T variant and decreased plasma levels of hs-CRP, IL-6 and TNF-α in the subgroups of SAP and UAP. It is possible that this functional variant produces lower expression of plasma visfatin levels. Accordingly, these lower concentrations might down-regulate the expression of hs-CRP, IL-6, and TNF-α [6, 22, 23]. Anyway, this hypothesis needs to be verified with further investigation.
This study has several limitations. First, the relatively small sample size may underpower the results of our study. Second, selection bias in the present study is likely to affect our results. Third, to evaluate the association between the polymorphism and the CAD prognosis, it is preferable to conduct a prospective study to justify this result. Four, the present study lack the direct biochemical evidence indicating whether the variation could be functional or not. Finally, our study was performed in a Chinese population. Data should be extrapolated to other regions and ethnic groups cautiously. However, these limitations do not detract from the main conclusions.
To the best of our knowledge, this study demonstrates for the first time that the −1535C>T polymorphism correlates with reduced plasma levels of inflammatory markers, including visfatin in CAD patients. To enhance understanding of the relationship between this polymorphism and plasma levels of visfatin, hs-CRP, IL-6 and TNF-α in CAD patients, additional large scale studies are required in other populations.
References
Rosamond W, Flegal K, Furie K et al (2008) Heart disease and stroke statistics—2008 update: a report from the American. Circulation 117(4):e25–e146
Hansson GK (2005) Inflammation, atherosclerosis, and coronary artery disease. N Engl J Med 352:1685–1695
Libby P, Sasiela W (2006) Plaque stabilization: can we turn theory into evidence? Am J Cardiol 98((11A)):26P–33P
Samal B, Sun Y, Stearns G et al (1994) Cloning and characterization of the cDNA encoding a novel human pre-B-cell colony-enhancing factor. Mol Cell Biol 14(2):1431–1437
Oki K, Yamane K, Kamei N et al (2007) Circulating visfatin level is correlated with inflammation, but not with insulin resistance. Clin Endocrinol 67:796–800
Moschen AR, Kaser A, Enrich B et al (2007) Visfatin, an adipocytokine with proinflammatory and immunomodulating properties. J Immunol 178:1748–1758
Dahl TB, Yndestad A, Skjelland M et al (2007) Increased expression of visfatin in macrophages of human unstable carotid and coronary atherosclerosis: possible role in inflammation and plaque destabilization. Circulation 115(8):972–980
Liu SW, Qiao SB, Yuan JS, Liu DQ (2009) Association of plasma visfatin levels with inflammation, atherosclerosis, and acute coronary syndromes in humans. Clin Endocrinol 71(2):202–207
Zhang YY, Gottardo L, Thompson R et al (2006) A visfatin promoter polymorphism is associated with low-grade inflammation and type 2 diabetes. Obesity 14:2119–2126
Ye SQ, Simon BA, Maloney JP et al (2005) Pre-B-cell colony enhancing factor as a potential novel biomarker in acute lung injury. Am J Respir Crit Care Med 171:361–370
Tokunaga A, Miura A, Okauchi Y et al (2008) The −1535 promoter variant of the visfatin gene is associated with serum triglyceride and HDL-cholesterol levels in Japanese subjects. Endocr J 55:205–212
Jian WX, Luo TH, Gu YY et al (2006) The visfatin gene is associated with glucose and lipid metabolism in a Chinese population. Diabet Med 23:967–973
Yan JJ, Tang NP, Tang JJ et al (2010) Genetic variant in visfatin gene promoter is associated with decreased risk of coronary artery disease in a Chinese population. Clin Chim Acta 411(1-2):26–30
Santamore WP, Kahl FR, Kutcher MA et al (1988) A microcomputer based automated, quantitative coronary angiographic analysis system. Ann Biomed Eng 16:367–377
Ehara S, Kobayashi Y, Yoshiyama M et al (2004) Spotty calcification typifies the culprit plaque in patients with acute myocardial infarction: an intravascular ultrasound study. Circulation 110(22):3424–3429
Wang B, Pan J, Wang L et al (2006) Associations of plasma 8-isoprostane levels with the presence and extent of coronary stenosis in patients with coronary artery disease. Atherosclerosis 184:425–430
Chen Z, Qian Q, Ma G, Wang J et al (2009) A common variant on chromosome 9p21 affects the risk of early-onset coronary artery disease. Mol Biol Rep 36(5):889–893
Bressler J, Folsom AR, Couper DJ et al (2010) Genetic variants identified in a European genome-wide association study that were found to predict incident coronary heart disease in the atherosclerosis risk in communities study. Am J Epidemiol. 171(1):14–23
Pasalić D, Marinković N, Grsković B et al (2009) C-reactive protein gene polymorphisms affect plasma CRP and homocysteine concentrations in subjects with and without angiographically confirmed coronary artery disease. Mol Biol Rep 36(4):775–780
Lucas AR, Korol R, Pepine CJ (2006) Inflammation in atherosclerosis: some thoughts about acute coronary syndromes. Circulation 113(17):e728–e732
Fukuhara A, Matsuda M, Nishizawa M et al (2005) Visfatin: a protein secreted by visceral fat that mimics the effects of insulin. Science 307:426–430
Ognjanovic S, Bao S, Yamamoto SY et al (2001) Genomic organization of the gene coding for human pre-B-cell colony enhancing factor and experssion in human fetal membranes. J Mol Endocrinol 26(2):107–117
Ognjanovic S, Bryant-Greenwood GD (2002) Pre-B-cell colony-enhancing factor, a novel cytokine of human fetal membranes. Am J Obstet Gynecol 187(4):1051–1058
Adya R, Tan BK, Punn A, Chen J, Randeva HS (2008) Visfatin induces human endothelial VEGF and MMP-2/9 production via MAPK and PI3K/Akt signalling pathways: novel insights into visfatin-induced angiogenesis. Cardiovasc Res 78(2):356–365
Mizuno K, Satomura K, Miyamoto A et al (1992) Angioscopic evaluation of coronary artery thrombi in acute coronary syndromes. N Engl J Med 326(5):287–291
Fuster V, Badimon L, Badimon JJ et al (1992) The pathologenesis of coronary artery disease and the acute coronary syndrome. N Engl J Med 326(4):242–250
Kuhlmann MK, Yoshino M, Levin NW (2004) Differences in cardiovascular mortality rates among hemodialysis patients in the United States and Japan: the importance of background cardiovascular mortality. Hemodial Int 8(4):394–399
Morange PE, Tregouet DA, Godefroy T et al (2008) Polymorphisms of the tumor necrosis factor-alpha (TNF) and the TNF-alpha converting enzyme (TACE/ADAM17) genes in relation to cardiovascular mortality: the AtheroGene study. J Mol Med 86(10):1153–1161
Liu P, Li H, Cepeda J et al (2009) Critical role of PBEF expression in pulmonary cell inflammation and permeability. Cell Biol Int 33(1):19–30
Acknowledgment
This project was supported by the grant from the National Natural Science Foundation of China (No. 30871078).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, LS., Yan, JJ., Tang, NP. et al. A polymorphism in the visfatin gene promoter is related to decreased plasma levels of inflammatory markers in patients with coronary artery disease. Mol Biol Rep 38, 819–825 (2011). https://doi.org/10.1007/s11033-010-0171-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-010-0171-6