Abstract
Objective To investigate the anti-inflammatory or immunomodulatory effect of shikonin on early stage and established murine collagen-induced arthritis (CIA). Methods Mouse were injected intraperitoneally with shikonin (5 mg/kg) for 10 days along before or after the onset of CIA. The arthritis response was monitored visually by macroscopic scoring. Reverse transcription-polymerase chain reaction and western blotting were employed to determine the mRNA and protein expression of cytokine in patella with adjacent synovium in CIA mouse. Histology of knee was used to assess the occurrence of cartilage destruction and bone erosion. Results Shikonin (5 mg/kg) treatment along had no effect on macroscopic score and incidence of arthritis on early stage of CIA. However, a pronounced amelioration of macroscopic score and cartilage destruction was found in mouse treated with shikonin on established CIA for 10 days. Moreover, The mRNA levels of Th1 cytokines [tumor necrosis factor-α and interleukin (IL)-12] was significantly inhibited both in the synovial tissue and in the articular cartilage in treated groups compared with those in control groups, whereas the mRNA and protein levels of Th2 cytokines (IL-10 and IL-4) remained elevated throughout the treatment period. Moreover, the inflammatory cytokine, the mRNA and protein levels of IL-6 was down-regulated in mice with established CIA after treatment with shikonin. T-box expressed in T cells (T-bet) mRNA levels were decreased in shikonin compared with control group, and GATA-3 mRNA levels were higher than that in control group. Conclusion Shikonin treatment on established CIA can inhibit Th1 cytokines expression and induce Th2 cytokines expression in mice with established CIA. The inhibited effect of shikonin on Th1 cytokines expression may be mediated not only by inhibiting Th1 responses through T-bet mechanism, but also by inducing anti-inflammatory mediators such as IL-10 and IL-4 through a GATA-3 dependent mechanism.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Rheumatoid arthritis (RA) is an autoimmune disease characterized by synovial inflammation that leads to the destruction of cartilage and bone. In the last decade, numerous studies have demonstrated that pro- and anti-inflammatory cytokines, derived predominantely from cells of macrophage lineage, play a major role in the initiation and perpetuation of the chronic inflammatory process in the RA synovial membrane. The involvement of pro-inflammatory cytokines, particularly interleukin (IL)-1 and tumor necrosis factor (TNF)-α, in the pathogenesis of RA is well accepted. It has become clear in the last few years that T cell-derived cytokines expressed preferentially by Th1 cells contribute to joint destruction and inflammation response in RA, whereas Th2 cell-associated cytokines may be protective [1].
The Th1 subset has been defined by the specific production of IFN-γ, IL-1, IL-12, IL-17, and IL-18, and by the stimulation of cell-mediated immunity, whereas the Th2 subset specifically produces IL-4, IL-10 and stimulates humoral immunity [2].
Due to the inhibitory role on the synthesis of IL-1, TNF-α, IL-6, IL-8, IL-12, and IL-17 in macrophages and monocytes, IL-4 was found to be able to effectively antagonize the inflammatory and destructive mediators in RA [3]. Cytokines are considered to be the main determining factors in the initial differentiation of precursor T cells into these distinct subsets. A wealth of studies have shown that, IL-12 is capable to drives Th1 cells differentiation, whereas IL-4 plays a very important role in the promotion of the Th2 cells differentiation [2]. IL-10 is also known as a potent anti-inflammatory cytokine which acts by inhibiting the synthesis of pro-inflammatory cytokines such as IL-1, TNF-α, IL-6 by macrophages and monocytes [4–6]. The GATA-3 and T-box expressed in T cells (T-bet) are both important transcription factors [7]. GATA-3 is expressed during Th2 differentiation via pathways that probably involve the IL-4-dependent activation of the signal transducer and the expression of GATA-3 is critical for Th2 development [8, 9]. By contrast, T-bet is expressed exclusively in Th1 cells. Retroviral expression of T-bet in developing and developed Th2 cells not only induces the production of but also suppresses the production of IL-4 and IL-5 [9]. Thus, a balance between GATA-3 and T-bet is believed to control Th2/Th1 polarization.
Collagen-induced arthritis (CIA) is a widely used experimental model of poly-arthritis. It can be induced in susceptible strains of mice and rats by immunization with type II collagen, the major component of articular cartilage, and has histopathologic features in common with RA [10].
Zicao (purple gromwell), the dried root of Lithospermum erythrorhizon Sieb, et Zucc, Arnebia euchroma (Royle) Johnst, or Arnebia guttata Bunge, is a commonly used herbal medicine in China and other countries. Shikonin is a major active chemical component isolated from zicao with a molecular weight of 288. Like its genitor plant, shikonin possesses numerous pharmacological properties, including anti-inflammatory and anti-tumor properties and the ability to mediate cellular and humoral immunity [11, 12].
Recent datas have demonstrated that pretreatment with arnebinone (a component from shikonin derivatives) significantly inhibited the carrageenan-induced paw edema and also suppressed the development of chronic arthritis induced by Freund’s complete adjuvant (FCA) [13], but little is known of the effect of shikonin in CIA. The pharmacological profile of zicao led us to study the potential effect of shikonin on RA. Hence, in this paper, we investigated the role of shikonin in the pathogenesis of inflammatory joint disease by determining the effect of administering shikonin on mice with type II CIA either before the onset of clinical symptoms or after the establishment of full-blown disease. The analysis included histopathology of the joints, with a special emphasis on cartilage destruction, measurement of mRNA levels for cytokines and inhibitors in synovial tissue and cartilage by reverse transcription-polymerase chain reaction (RT-PCR) technology, and protein levels for cytokines in synovial tissue and cartilage by western blot analysis.
Materials and methods
Animals
Male DBA/1 Lac/J mice were originally purchased from Slac Laboratories (Shanghai, China). Mice were housed in cages, and water and food were provided ad libitum. DBA/l mice were immunized at the age of 8–10 weeks.
Materials
LPS (Escherichia coli 01 11:84), bovine type II collagen, and FCA were purchased from Sigma (St Louis, MO, USA). RNA PCR Kit (AMV, Version 3.0) was obtained from TaKaRa Biotechnology Co., Ltd (Dalian, China). TRIzol reagent was purchased from Invitrogen Corporation (CA, USA). Marker DNA ladder was obtained from Solarbio Science & Technology Co., Ltd. Agarose was obtained from Life Technologies (Beijing, China), and primers were purchased from Invitrogen Corporation (Beijing, China). Shikonin was purchased from National Institute for the Control Pharmaceutical and Biological Products (Beijing, China). Antibodies against IL-6, IL-10, TNF-α, IL-12, and IL-4 were purchased from Santa Cruz Biotechnology (CA, USA).Goat anti-rat IgG and rabbit anti-goat IgG were purchased from Golden Bridge Biotechnology Co., Ltd (Beijing, China).
Induction of CIA
Bovine type II collagen was diluted with 0.05 M acetic acid to a concentration of 2 mg/ml and thereafter was emulsified in an equal volume of FCA (2 mg/ml of MTH37Ra). The mice were immunized intradermally at the base of the tail with 100 μl of emulsion (100 μg of collagen). On day 21, the animals were given booster injections intraperitoneally with 100 μg of bovine type II collagen dissolved in phosphate buffered saline (PBS). This resulted in the onset of arthritis around day 28 in 20–40% of the mice. Mice without arthritis at this time point were selected to be used in studies of “classic” CIA [10].
Acceleration of CIA
Mice without any macroscopic signs of arthritis on day 28 were used for LPS-accelerated CIA. Arthritis onset was initiated by a single intraperitoneal injection of 40 μg of LPS. Thus, cytokine treatments were performed in two CIA models: classic and accelerated. As described previously, administration of 40 μg of LPS to non-immunized DBA/1 mice did not result in any macroscopic or histologic abnormalities [14].
Assessment of CIA
Mice were examined visually for the appearance of arthritis in the peripheral joints, and scores for arthritis severity were given (macroscopic score) as previously described [15, 16]. Mice were considered to have arthritis when significant changes in redness and or swelling were noted in the digits or in other parts of at least two paws. At later time points, ankylosis was also included in the macroscopic scoring. Cumulative scoring depending on redness, swelling and in later stadium ankylosis was as follows; 0—no changes; 0.25–1 to 2 toes red or swollen; 0.5–3 to 5 toes red or swollen; 0.5—swollen ankle; 0.5—swollen footpad; 0.5—severe swelling and ankylosis. The macroscopic score was assessed by two independent, blinded observers [17].
Histology
Mice were sacrificed by ether anesthesia; ankle and knee joints were removed and fixed for 4 days in 4% formalin. After decalcification in 5% formic acid, the specimens were processed for paraffin embedding. Tissue sections (7 pan) were stained with hematoxylin and eosin. Histopathologic changes were scored according to the following parameters. Infiltration of cells was scored on a scale of 0–3, depending on the amount of inflammatory cells in the synovial cavity and synovial tissues. A characteristic parameter in CIA is the progressive loss of articular cartilage. This destruction was separately graded on a scale of 0–3, ranging from the appearance of dead chondrocytes (empty lacunae) to a complete loss of articular cartilage. Histopathologic changes in the knee joints were scored in the patellofemoral region on five semi-serial sections of the joint. For the ankle joint, we scored the calcaneous region. Scoring was done in a blinded manner by two observers, as described earlier [15, 16].
Treatment protocol of shikonin in CIA arthritis
To investigate the effect of shikonin on the murine CIA arthritis. DBA/1 Lac/J mice without signs of arthritis on day 28 were injected intraperitoneally with shikonin (5 mg/kg) for 10 days, and with PBS as a control. DBA/1 Lac/J mice with established CIA on day 35 were selected and divided into two groups of at least ten mice with similar arthritis scores. Thereafter, mice were injected intraperitoneally daily with shikonin (5 mg/kg), starting on day 35 up to day 44. Mice were scored every other day for erythema and swelling by two independent observers.
RNA isolation
Mice were killed by cervical dislocation, immediately followed by dissection of the patella with adjacent synovium [15, 16]. The synovium samples were immediately frozen in liquid nitrogen. Five separate synovium samples were ground into powder using a grinder. Total RNA was extracted in 1 ml TRIzol reagent in a procedure similar to that used for cartilage samples [15, 16].
Polymerase chain reaction amplification
One microgram of synovial RNA and cartilage RNA were used for RT-PCR. Messenger RNA was reverse-transcribed to complementary DNA (cDNA) using oligo-dT primers and 3 μl cDNA was used in each PCR amplification. PCR was performed at a final concentration of 0.15 μl of each primer, and 0.25 μl of Ex Taq HS polymerase (TaKaRa Biotechnology Co., Ltd) in standard PCR buffer. Primers for IL-12, IL-4, IL-6, IL-10, T-bet, GATA-3, and TNF-α genes were designed using PRIMER 5.0, and synthesized by Invitrogen Corporation. Primers sequences and PCR condition are shown in Table 1.
Western blot analysis
Total proteins were isolated from the dissected synovium. Protein concentration was measured by the Lowry method. Twenty micrograms of protein was mixed with 4× gel loading buffer [4× = 250 mmol/l Tris–HCl, pH6.8, 8% sodium dodecyl sulfate (SDS), 20% glycerol, 0.2% bromophenol blue, and 5% β-mercaptoethanol], separated on 12% SDS-polyacrylamide gel under reducing conditions, and transferred onto Nitroplus-2000 membrane (Micron Separations Inc., Westborough, MA, USA). Nonspecific antibody binding was blocked by pre-incubation of the membranes in 1 × Tris-buffered-saline (TBS) containing 5% skim milk for 1 h at room temperature. Membranes were then incubated overnight at 4°C with appropriated antibodies at different dilutions in 1 × TBS containing 2% skim milk. After washing, they were incubated with goat anti-rat IgG and rabbit anti-goat IgG at 1:1,000 dilutions for 1 h at room temperature. Bands were visualized by employing the enhanced chemiluminescence kit per the manufacturer’s instruction [18–20].
Statistical analysis
Differences between experimental groups were analyzed using the Wilcoxon rank test, unless stated otherwise. Differences with P-values below 0.05 were judged to be significant.
Results
Role of shikonin during the onset of CIA
DBA-1 Lac/J mice developed gradual onset of CIA after immunization with C II in CFA, early expression of CIA can be enhanced with a single i.p. injection of 40 μg of LPS at the time of onset on day 28. To investigate the effect of shikonin in early stage CIA, mice without signs of CIA were selected on day 28 and challenged with LPS. Daily treatment with shikonin (5 mg/kg) was given from day 28 to day 37. Macroscopic score and disease incidence were found no difference compared with the control group (Fig. 1). Histology revealed that no effect was found on the influx of inflammatory cells, cartilage destruction, and bone erosion in joint tissues of shikonin-treated animals when compared with the vehicle-treated animals (Table 2), and mRNA levels of IL-12, TNF-α, IL-10, IL-6, IL-4 were slightly changed compared with the control group (Fig. 2).
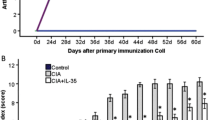
Mean arthritis macroscopic score and incidence over time in mice treated with shikonin after immunization against CII in the CIA model. Immunized DBA/1 Lac/J mice without signs of CIA were selected and divided into two separate groups (n = 10/group). Mice were treated with either 5 mg/kg of shikonin or 0.1 ml PBS for ten consecutive days. Clinical arthritis score and incidence were monitored throughout the experiment. The data represent macroscopic score and mean incidence of arthritis in shikonin-treated or control group
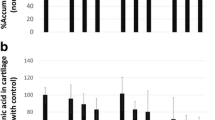
Effect of shikonin on murine collagen-induced arthritis. Mice with accelerated CIA were treated i.p. with 5 mg/kg of shikonin or 0.1 ml PBS for 10 days, starting on day 28. Synovium biopsies of six mice per experimental group were pooled on day 60. Total RNA was extracted as described in Sect. “Materials and methods”. The expression of IL-4, IL-6, IL-10, IL-12, and TNF-α were determined by using RT-PCR analysis. β-Actin was used for loading control. The lower part displays the density histogram data from three-separated RT-PCR analysis (mean ± SE), which represents the relative expression of TNF-α, IL-12, IL-6, IL-10, and IL-4, respectively. M marker, 1 PBS control, 2 shikonin treatment
Role of shikonin in established CIA
To investigate the effect of shikonin in established arthritis, treatment with shikonin (5 mg/kg) was started on day 35 and ended on day 44. A pronounced amelioration of macroscopic score was found when compared with the vehicle-treated animals (Fig. 3). The influx of inflammatory cells, cartilage destruction, and bone erosion revealed amelioration when compared with the control group (Table 2) (Fig. 4).
Amelioration of CIA after shikonin treatment. DBA/1 mice with established CIA were selected on day 35 and divided into two separate groups of at least ten mice per group with roughly the same mean arthritis score. Mice were injected i.p. daily with shikonin (5 mg/kg) or 0.1 ml PBS for 10 days. The macroscopic score for arthritis severity were determined every second day. Arthritis was considered present when 1 or more digits was inflamed. The macroscopic score (mean ± SD) was expressed as the cumulative scores in all paws (from two experiments). Asterisk indicates P < 0.05 between treated and control groups
Representative histological findings in mice with established CIA treated with shikonin. Sections of the knee were collected on day 60 and stained with hematoxylin and eosin. (a) Paws with 0 macroscopic score—no changes; (b) Paws with 0.25 macroscopic score-1 to 2 toes red or swollen (shikonin treatment group); (c) Paws with 0.5 macroscopic score—swollen ankle after PBS treatment on established CIA. (d) PBS-treated mice with collagen II induced arthritis, showing severe cartilage surface disruption, and bone erosion (arrows). (e) Mice with established CIA treated with 5 mg/kg shikonin, showing marked improvements with intact cartilage surface. C cartilage (original magnification, 40×)
Cytokine expression in the arthritic joint
In a repeat experiment, groups of mice were treated with shikonin (5 mg/kg) after the onset of arthritis, and mRNA and protein were extracted from the synovial tissue and patellar cartilage on day 60. As shown in Fig. 5a and b, there was a marked decrease in TNF-α, IL-12, and IL-6 mRNA and protein levels in inflamed synovium and patellar cartilage after treatment with shikonin compared with control group, whereas the mRNA and protein levels of IL-10 and IL-4 was significantly increased. To determine the role of T-bet and GATA-3 in the differentiation of Th1 and Th2 cells in CIA mice, we analyzed T-bet and GATA-3 mRNA levels in synovium and patellar cartilage. T-bet mRNA levels were decreased in shikonin-treated group compared with control group, and GATA-3 mRNA levels were higher than that in control group, which meant the inhibited effect of shikonin on Th1 cytokines expression may be mediated through the inhibition of T-bet expression and acceleration of GATA-3 expression.
Effect of shikonin on murine established collagen-induced arthritis. Mice with established CIA were treated i.p. with 5 mg/kg of shikonin or 0.1 ml PBS for 10 days, starting on day 35. Synovium biopsies of six mice per experimental group were pooled on day 60. (a) Total RNA was extracted as described in Sect. “Materials and methods”. The expression of IL-4, TNF-α, IL-12, IL-6, T-bet, IL-10, and GATA-3 were determined by using RT-PCR analysis. β-Actin was used for loading control. The lower part displays the density histogram data from three-separated RT-PCR analysis (mean ± SE), which represents the relative expression of IL-4, TNF-α, IL-12, IL-6, T-bet, IL-10, and GATA-3, respectively. (b) Expression of IL-10, TNF-α, IL-12, IL-6, and IL-4 were analyzed by western blot as described in Sect. “Materials and methods”. β-Actin was used for loading control. The lower part displays the density histogram data from three-separated western blot analysis (mean ± SE), which represents the relative expression of IL-10, IL-4, IL-6, TNF-α, and IL-12. Asterisk indicates P < 0.05 between treated and control groups. M marker, 1 PBS control group, 2 shikonin treatment group
Discussion
It has become clear in the last few years that T cell-derived cytokines expressed preferentially by Th1 cells contribute to joint destruction and inflammation in RA. However, products from Th2 cells may be protective [1].
This is the first report that demonstrated shikonin treatment on established CIA can inhibit Th1 cytokines expression and reduce the damage of arthritis. In the present study we first investigate the effect of shikonin in accelerated arthritis before the onset, shikonin (5 mg/kg) was given daily from day 28 to day 37.
Our present data clearly demonstrated that shikonin treatment alone was not effective in early arthritis at a dose of 5 mg/kg. However, treatment with shikonin in established arthritis resulted in pronounced amelioration of arthritis, which was confirmed with histology. Recent studies have revealed that shikonin possess anti-inflammatory activities and inhibit the transcriptional activation of human TNF-α in vivo [13, 21–23]. Administration of shikonin has been reported to inhibit ear edema induced by croton oil in mice and paw swelling induced by yeast in rats, and shikonins has been demonstrated to inhibit the transcriptional activation of the human TNF-α promoter through interference with the basal transcription machinery [21]. In inflamed synovium and patellar cartilage of shikonin-treated group on established CIA, there was a marked decrease in TNF-α, IL–12, and IL-6 expression compared with control groups, and enhancement of mRNA and protein levels for IL-10 and IL-4 was observed, which showed shikonin exerted the anti-inflammatory effects through the inhibition of Th1 cytokines and induce Th2 cytokines. CIA is an experimental autoimmune model of human RA that is widely used for studying disease processes as well as for evaluating possible therapeutic agents. Treatment with anti-TNF-α, anti-IL-12, and anti-IL-6 has been reported to be effective in suppressing CIA. Endogenous IL-10 and IL-4 have been shown to partly suppress the production of TNF-α, IL-1, and IL-8 by synovial macrophages and synoviocytes in an animal model of CIA and were confirmed to have beneficial effects in this model [24, 25]. The transcription factors GATA-3 and T-bet have been described as “master switches” in the development of Th1 and Th2 cells, expression of T-bet or GATA-3 determines not only Th differentiation, but can actually override the influence of exogenous polarizing stimuli or previous polarization [26–28]. A significant increase in GATA-3 expression was demonstrated in shikonin-treated group compared with that in control group. The increase in GATA-3 expression in the shikonin-treated group correlated significantly with Th2 cytokines expression. However, T-bet mRNA levels were decreased in shikonin-treated group compared with control group.
In summary, shikonin treatment alone was not effective in early arthritis at a dose of 5 mg/kg. Shikonin treatment elevated IL-10 and IL-4 expression, whereas reduced IL-12, IL-6, and TNF-α expression in mice with established CIA, the effects of shikonin in this experimental model of arthritis may be mediated not only by inhibiting Th1 responses through T-bet mechanism, but also by inducing anti-inflammatory mediators such as IL-10 and IL-4 through a GATA-3-dependent mechanism. The elucidation of the underlying mechanisms may result in new insights into the late phase of CIA. Moreover, further elucidation of the roles of shikonin in inflammatory synovitis would be helpful for the understanding of the immunopathology of CIA as a model of RA.
References
Vervoordeldonk MJ, Tak PP (2002) Cytokines in rheumatoid arthritis. Curr Rheumatol Rep 4(3):208–217. doi:10.1007/s11926-002-0067-0
Mendoza L (2006) A network model for the control of the differentiation process in Th cells. Biosystems 84(2):101–114. doi:10.1016/j.biosystems.2005.10.004
Okamoto Y, Nishida M (2001) Cytokine balance in the pathogenesis of rheumatoid arthritis. Yakugaku Zasshi 121(2):131–138. doi:10.1248/yakushi.121.131
Moore KW, O’Garra A et al (1993) Interleukin-10. Annu Rev Immunol 11:165–190
Rengarajan J, Szabo SJ et al (2000) Transcriptional regulation of Th1/Th2 polarization. Immunol Today 21:479–483. doi:10.1016/S0167-5699(00)01712-6
Chang HD, Helbig C et al (2007) Expression of IL-10 in Th memory lymphocytes is conditional on IL-12 or IL-4, unless the IL-10 gene is imprinted by GATA-3. Eur J Immunol 37(3):807–817. doi:10.1002/eji.200636385
Szabo SJ, Kim ST et al (2000) A novel transcription factor, T-bet, directs Th1 lineage commitment. Cell 100:655–669. doi:10.1016/S0092-8674(00)80702-3
Sun J, Pearce EJ (2007) Suppression of early IL-4 production underlies the failure of CD4 + T cells activated by TLR-stimulated dendritic cells to differentiate into Th2 cells. J Immunol 178(3):1635–1644
Agnello D, Lankford CS et al (2003) Cytokines and transcription factors that regulate T helper cell differentiation: new players and new insights. J Clin Immunol 23(3):147–161. doi:10.1023/A:1023381027062
Brand DD, Latham KA et al (2007) Collagen-induced arthritis. Nat Protoc 2(5):1269–1275. doi:10.1038/nprot.2007.173
Pietrosiuk A, Skopińska-Rózewska E et al (2004) Immunomodulatory effect of shikonin derivatives isolated from Lithospermum canescens on cellular and humoral immunity in Balb/c mice. Pharmazie 59(8):640–642
Chen X, Yang L et al (2003) Shikonin, a component of Chinese herbal medicine, inhibits chemokine receptor function and suppresses human immunodeficiency virus type 1. Antimicrob Agents Chemother 9(47):2810–2816
Singh B, Sharma MK et al (2003) Anti-inflammatory activity of shikonin derivatives from Arnebia hispidissima. Phytomedicine 10(5):375–380. doi:10.1078/0944-7113-00262
Bernard P, Leung B et al (2004) Combined effects of IL-12 and IL-18 on the induction of collagen-induced arthritis. Clin Exp Immunol 136:440–447. doi:10.1111/j.1365-2249.2004.02430.x
Yamaguchi Y, Fujio K et al (2007) IL–17B and IL–17C are associated with TNF-alpha production and contribute to the exacerbation of inflammatory arthritis. J Immunol 179(10):7128–7136
Nirmal K, Damian M et al (2003) Prevention of collagen-induced arthritis in mice transgenic for the complement inhibitor complement receptor 1-related gene/protein. J Immunol 171:2109–2115
Smeets RL, van de Loo FAJ et al (2003) Adenoviral delivery of IL-18 binding protein C ameliorates collagen-induced arthritis in mice. Gene Ther 10:1004–1011. doi:10.1038/sj.gt.3301986
Gao B, Fang D et al (2006) The pro-inflammatory cytokines IL–1β and TNF-α induce the expression of Synoviolin, an E3 ubiquitin ligase, in mouse synovial fibroblasts via the Erk1/2-ETS1 pathway. Arthritis Res Ther 172:1186–2011
Dulos J, Verbraak E (2004) Severity of murine collagen-induced arthritis correlates with increased CYP7B activity. Arthritis Rheum 50(10):3346–3353. doi:10.1002/art.20509
Kristine A, Kuhn L et al (2006) Antibodies against citrullinated proteins enhance tissue injury in experimental autoimmune arthritis. J Clin Invest 116:961–973. doi:10.1172/JCI25422
Staniforth V, Wang S-Y et al (2004) Shikonins, phytocompounds from Lithospermum erythrorhizon, inhibit the transcriptional activation of human tumor necrosis factor α promoter in vivo. J Biol Chem 7(279):5877–5885
Chen X, Oppenheim J et al (2001) Shikonin, a component of anti-inflammatory Chinese herbal medicine, selectively blocks chemokine binding to CC chemokine receptor-1. Int Immunopharmacol 1(2):229–236. doi:10.1016/S1567-5769(00)00033-3
Ishida T, Sakaguchi I (2007) Protection of human keratinocytes from UVB-induced inflammation using root extract of Lithospermum erythrorhizon. Biol Pharm Bull 30(5):928–934. doi:10.1248/bpb.30.928
Yu-Hsiang HSU, Hsing-Hui LI et al (2006) Function of interleukin-20 as a proinflammatory molecule in rheumatoid and experimental arthritis. Arthritis Rheum 54(9):2722–2733. doi:10.1002/art.22039
Kageyama Y, Koide Y et al (2004) Plasmid encoding interleukin-4 in the amelioration of murine collagen-induced arthritis. Arthritis Rheum 50(3):968–975. doi:10.1002/art.20107
Murphy KM, Reiner SL (2002) The lineage decisions of helper T cells. Nat Rev Immunol 2:933–944. doi:10.1038/nri954
Szabo SJ, Sullivan BM et al (2003) Molecular mechanisms regulating Th1 immune responses. Annu Rev Immunol 21:713–758. doi:10.1146/annurev.immunol.21.120601.140942
Chakir H, Wang H et al (2003) T-bet/GATA-3 ratio as a measure of the Th1/Th2 cytokine profile in mixed cell populations. predominant role of GATA-3. J Immunol Methods 278:157–169. doi:10.1016/S0022-1759(03)00200-X
Acknowledgments
The authors thank Y. Li, Ph.D. (Department of rheumatology, The Second Affiliated Hospital, Harbin Medical University, Harbin, China) for her help in conducting the study and for reviewing the manuscript. This work were supported by the Science Foundation of Heilongjiang traditional Chinese medicine university and Dr Science Foundation of Harbin Medical University.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dai, Q., Fang, J. & Zhang, Fs. Dual role of shikonin in early and late stages of collagen type II arthritis. Mol Biol Rep 36, 1597–1604 (2009). https://doi.org/10.1007/s11033-008-9356-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-008-9356-7