Abstract
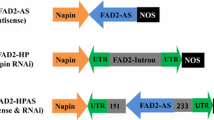
Seed lipids of oilseed rape (Brassica napus) usually contain small proportions (<3%) of stearic acid. The objective of this study was to increase the content of stearic fatty␣acid in rapeseed oil. An antisense down-regulation of the endogenous stearoyl-ACP desaturase (SAD) catalysing the reaction step from stearic to oleic acid in two different genetic backgrounds was studied. The result of down-regulation of the SAD yielded an about 10-fold increase of stearic acid from 3.7% up to 32% in single seeds of transgenic low-erucic acid rapeseed (LEAR), while high-erucic acid rapeseed (HEAR) showed a 4-fold increase of C18:0 from 1% up to 4%. It could be shown in pooled T2 seed material of LEAR rapeseed, that the stearic acid content is highly correlated with the down-regulation of SAD as indicated by the␣stearate desaturation proportion (SDP). The importance of the promoter strength for the alteration of a trait was confirmed in this study as no change in the fatty acid composition of transgenic plants was achieved with gene constructs controlled by the weak FatB4 seed-specific promoter from Cuphea lanceolata.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Barret P, Delourme R, Renard M, Domergue F, Lessire R, Delseny M, Roscoe TJ (1998). A rapeseed FAE1 gene is linked to the E1 locus associated with variation in the content of erucic acid. Theor Appl Genet 96:177–186
De Block M, De Brouwer D, Tenning P (1989) Transformation of Brassica napus and Brassica oleracea using Agrobacterium tumefaciens and the expression of the bar and neo genes in transgenic plants. Plant Physiol 91:694–701
Domergue F, Chevalier S, Santarelli X, Cassagne C, Lessire R, (1999) Evidence that oleoyl-CoA and ATP-dependent elongations coexist in rapeseed (Brassica napus L.). Eur J Biochem 263:464–470
Dower WJ, Miller JF, Ragsdale CW (1988) High efficiency transformation of E. coli by high voltage electroporation. Nucl Acids Res 16:6127–6145
Downey RK, Taylor DC (1996) Diversification of canola/rapeseed fatty acid supply for the year 2000. Oleagineux Corps Gras Lipids 3:9–13
Doyle JF, Doyle JL (1990) Isolation of plant DNA from fresh tissue. Focus 12:13–15
Drexler HS, Spiekermann P, Meyer A, Domergue F, Zank T, Sperling P, Abbadi A, Heinz E (2003a) Metabolic engineering of fatty acids for breeding of new oilseed crops: stratagies, problems and first results. J Plant Physiol 8:779–802
Drexler HS, Scheffler JA, Heinz E (2003b) Evaluation of putative seed-specific promoters for Linum usitatissimum. Mol Breeding 11:149–158
Facciotti MT, Bertain PB, Yuan L (1999) Improved stearate phenotype in transgenic canola expressing a modified acyl-acyl carrier protein thioesterase. Nature Biotechnol 17:593–597
Fourmann M, Barret P, Renard M, Pelletier G, Delourme R, Brunel D (1998) The two genes homologous to Arabidopsis FAE1 co-segregate with the two loci governing erucic acid content in Brassica napus. Theor Appl Genet 96:852–858
Grundy SM (1994) Influence of stearic acid on cholesterol metabolism relative to other long-chain fatty acids. Am J Clin Nutr 60:986–990
Han J, Lühs W, Sonntag K, Zähringer U, Borchardt DS, Wolter FP, Heinz E, Frentzen M (2001) Functional characterisation of β-ketoacyl-CoA synthase genes from B. napus L. Plant Mol Biol 46:229–239
Hawkins DJ, Kridl JC (1998) Characterisation of acyl-ACP thioesterases of mangosteen (Garcinia mangostana) seed and high levels of stearate production in transgenic canola. Plant J 13:743–752
Hlousek-Radojcic A, Imai H, Jaworski JG (1995) Oleoyl-CoA is not an immediate substrate for fatty acid elongation in developing seeds of Brassica napus. Plant J 8:803–809
Knutzon DS, Thompson GA, Radke SE, Johnson WB, Knauf VC, Kridl JC (1992) Modification of Brassica seed oil by antisense expression of a stearoyl-acyl carrier protein desaturase gene. Proc Natl Acad Sci USA. 89:2624–2628
Koncz C, Schell J (1986) The promoter of T-DNA gene 5 controls the tissue-specific expression of chimaeric genes carried by a novel type of Agrobacterium binary vector. Mol Gen Genet 204:383–396
Lassner MW, Lardizabal K, Metz JG (1996) A jojoba ß-ketoacyl-CoA synthase cDNA complements the canola fatty acid elongation mutation in transgenic plants. Plant Cell 8:281–292
Liu Q, Singh S, Green A (2002) High-stearic and high-oleic cottonseed oils produced by hpRNA-mediated posttranscriptional gene silencing. Plant Physiol 129:1732–1743
Lühs W, Friedt W (1994) Stand und Perspektiven der Züchtung von Raps (Brassica napus L.) mit hohem Erucasäure-Gehalt im Öl für industrielle Nutzungszwecke. Fat Sci Technol 96:137–146
Millar A, Kunst L (1997) Very-long-chain fatty acid biosynthesis is controlled through the expression and specificity of the condensing enzyme. Plant J 12:121–131
Puyaubert J, Garbay B, Costaglioli P, Dieryck W, Roscoe TJ, Renard M, Cassagne C, Lessire R (2001) Acyl-CoA elongase expression during seed development in␣Brassica napus. Biochim Biophys Acta 1533:141–152
Puyaubert J, Garcia C, Chevalier S, Lessire R (2005) Acyl-CoA elongase, a key enzyme in the development of high-erucic acid rapeseed? Eur J Lipid Sci Technol 107:263–267
Reed KC, Mann DA (1985). Rapid transfer of DNA from agarose gels to nylon membranes. Nucl Acid Res 13:7207–7221
Roscoe TJ, Lessire R, Puyaubert J, Renard M, Delseny M (2001) Mutations in the fatty acid elongation 1 gene are associated with a loss of β-ketoacyl-CoA synthase activity in low erucic acid rapeseed. FEBS Lett 492:107–111
Singh SP, Zhou XR, Liu Q, Stymne S, Green AG (2005) Metabolic engineering of new fatty acids in plants. Curr Opin Plant Biol 8:197–203
Slocombe SP, Piffanelli P, Fairbairn D, Bowra S, Hatzopoulos P, Tsiantis M, Murphy DJ (1994) Temporal and Tissue-Specific Regulation of a Brassica napus Stearoyl-Acyl Carrier Protein Desaturase Gene. Plant Physiol 104:1167–1176
Smith NA, Singh SP, Wang MB, Stoutjesdijk PA, Green AG, Waterhouse PM (2000) Total silencing by intron-spliced hairpin RNAs. Nature 407:319–320
Stoutjesdijk PA, Singh SP, Liu Q, Hurlstone CJ, Waterhouse PA, Green AG (2002) hpRNA-mediated targeting of the Arabidopsis FAD2 gene gives highly efficient and stable silencing. Plant Physiol 129:1723–1731
Syring-Ehemann A (2001) Isolierung und Charakterisierung von Acyl-ACP-Desaturasegenen aus dem Fettsäurebiosyntheseweg von Lindera obtusiloba. PhD Thesis, Universität Karlsruhe
Thelen JJ, Ohlrogge JB (2002) Metabolic engineering of fatty acid biosynthesis in plants. Metab Eng 4:12–21
Töpfer R, Martini N, Schell J (1995) Modification of plant lipid synthesis. Science 268:681–686
Voelker TA, Jones A, Cranmer AM, Davies HM, Knutzon DS (1997) Broad-range and binary-range suggest an alternative mechanism for medium-chain production in seeds. Plant Physiol 114:669–667
Weier D, Hanke C, Eickelkamp A, Lühs W, Dettendorfer J, Schaffert E, Möllers C, Friedt W, Wolter FP, Frentzen M (1997) Trierucoylglycerol biosynthesis in transgenic plants of rapeseed (Brassica napus L.). Fett/Lipid 99:160–165
Yu S, Derr J, Etherton TD, Kris-Etherton PM (1995) Plasma cholesterol-predictive equations demonstrate that stearic acid is neutral and monounsaturated fatty acids are hypocholesterolemic. Am J Clin Nutr 61:1129–1139
Zarhloul MK, Friedt W, Khoschkhoi, Yazdi MR, Lühs W (1999) Genetic transformation and shoot regeneration ability of re-synthesised Brassica napus line ‘RS 306’. Cruciferae Newsl 21:59–60
Acknowledgements
This research was jointly funded by the German Federal Ministry of Consumer Protection, Food and Agriculture (BMVEL), the Fachagentur für Nachwachsende Rohstoffe e.V. (FNR, Project No. 97 NR 079-F), Guelzow, and Norddeutsche Pflanzenzucht Hans-Georg Lembke KG, Hohenlieth, Germany. The authors thank Sonja Weber, Stavros Tzigos, Petra Degen and Marinela-Gabriela Ciobanu for their excellent technical assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
Karim Zarhloul and Christof Stoll have contributed in equal parts to the present work
Rights and permissions
About this article
Cite this article
Karim Zarhloul, M., Stoll, C., Lühs, W. et al. Breeding high-stearic oilseed rape (Brassica napus) with high- and low-erucic background using optimised promoter-gene constructs. Mol Breeding 18, 241–251 (2006). https://doi.org/10.1007/s11032-006-9032-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11032-006-9032-3