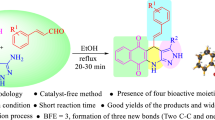
Abstract
A series of pyrano-fused benzophenazines were synthesized using a bifunctional thiourea-based organocatalyst from the one-pot four-component reaction of 2-hydroxy-1,4-naphthoquinone, benzene-1,2-diamines, malononitrile or its derivatives and isatins or aromatic aldehydes in aqueous medium. Metal-free reaction condition, water as solvent, high bond forming efficiency (five new bonds formed in one step), good yields and easy purification process are the notable features of this methodology.
Graphical Abstract

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Organocatalysis has emerged as a powerful tool in organic synthesis, especially for the synthesis of various biologically active heterocyclic compounds [1–6] due to their improved stability, lesser toxicity and metal-free reaction conditions. Among organocatalysts, thiourea-based organocatalysts [7, 8] have numerous applications in organic synthesis due to their ability to activate carbonyl compounds by double hydrogen bonding. Jacobsen [9], Schreiner [10] and Takemoto [11] catalysts are some of the most well-explored thiourea-based organocatalysts. Bifunctional organocatalysts bearing a thiourea moiety and an amine group exhibit better catalytic activity [12–14] due to their dual activation of both electrophile and nucleophile by double-hydrogen-bonding interactions of the thiourea moiety and the basic amine moiety (Fig. 1).
Multicomponent reactions (MCRs) have gained considerable attention in recent years for the easy access of diverse classes of compounds. They are considered ecofriendly and cost-effective tools because of their pot, atom and step economic approach [15–19]. Therefore, the design of novel MCRs for the synthesis of diverse heterocycles has remained as an important topic for medicinal and organic chemists. Heterocyclic compounds, especially functionalized nitrogen and oxygen heterocycles, play important roles in medicinal chemistry [20, 21]. Benzo[a]phenazines and pyrans are structural subunits in a variety of important natural products and show a variety of pharmaceutical activities, such as antimalarial, anticancer activities and are also employed as useful substrates to prepare dyestuffs, pesticides, antibiotics and antitumor agents [22–30]. Some of the pharmaceutical active pyrano-fused benzophenazine derivatives [31–33] are shown in Fig. 2.
Considering the importance of benzophenazine and pyran derivatives, only very few methods [34–38] are known in the literature for the synthesis of pyrano-fused benzophenazines. In continuation of our research on MCRs for the synthesis of biologically important heterocycles [39–44], we wish to report the synthesis of some thiourea-based organocatalysts and their application in the synthesis of pyrano-fused benzophenazines via the one-pot four-component reaction of 2-hydroxy-1,4-naphthoquinone, o-phenylenediamines, malononitriles and substituted isatins or aldehydes under reflux condition in aqueous medium (Scheme 1).
Results and discussion
We recently reported the three-component reaction of aldehydes, 1,3-dimethyl-6-aminouracil and 2-hydroxy-1,4-naphthoquinone/4-hydroxycoumarin for the synthesis of tri-substituted methane derivatives in the presence of a bifunctional thiourea-based organocatalyst in aqueous medium [39]. In continuation of our research on multicomponent reactions using thiourea-based organocatalyst, we initially prepared organocatalysts I–IV (Fig. 3) by the reaction of 2-piperidinoethyl isothiocyanate and the corresponding primary amines in DCM at room temperature.
Next, we screened these thiourea-based organocatalysts I–IV for the synthesis of pyrano benzophenazine-fused spirooxindoles 5. To find the best reaction conditions, we studied the reaction of 2-hydroxy-1,4-naphthoquinone 1, 1,2-diamines 2, malononitrile 3 and 5-chloroisatin 4a as our model reaction in the presence of some readily available organocatalysts as well as with our synthesized organocatalysts I–IV, and the results are summarized in Table 1.
Among them, organocatalyst IV gave the best results in terms of reaction time and yield obtained (92 %, entry 9). Next, the same reaction was performed in the presence of various amounts (5, 10, 15, 20, 30 mol%) of organocatalyst IV (entries 9–13), and the best result was obtained using 20 mol% of IV (Table 1, entry 9).
Then, various solvents, such as DMSO, \(\hbox {H}_{2}\hbox {O}\), EtOH, \(\hbox {CH}_{3}\hbox {CN}\), THF, DMF and DCM (Table 2, entries 1–7), were screened, and water was found to be the best solvent for the reaction in terms of yield and reaction time (92 %, entry 3).
Next, a variety of pyrano benzophenazine-fused spirooxindoles 5 were synthesized by varying 1,2-diamines 2, malononitrile derivatives 3 and isatin derivatives 4 under the optimized reaction conditions, and the results are shown in Table 3. All the reactions proceeded smoothly to yield the corresponding products (5a–o) in good yields (75–92 %).
Considering the importance of the molecular skeleton having benzophenazine and pyran moieties, the scope of the reaction was explored using a variety of aldehydes in place of isatin derivatives under the optimized reaction conditions to afford the corresponding products, and the results are summarized in Table 4.
The proposed mechanism for the synthesis of pyrano-fused benzophenazines in the presence of organocatalyst IV is shown in Scheme 2. Based on literature reports [34–38], we believe that the condensation reaction between 2-hydroxy-1,4-naphthoquinone and the benzene 1,2-diamine leading to the corresponding benzo[a]phenazin-5-ol A does not need any catalyst. However, our organocatalyst plays significant role in other steps, and it activates both the electrophile and nucleophile through its thiourea moiety and basic amine moiety, respectively. The Knoevenagel condensation of isatin or aldehyde with malononitrile affords B, which undergoes a Michael addition with A to form intermediate C in the presence of organocatalyst IV. A subsequent cyclization leads to the formation of D which undergoes tautomerization to form the corresponding final product 5 or 7.
Conclusions
We have synthesized a series of pyrano-fused benzophenazines in the presence of thiourea-based organocatalysts by the one-pot four-component reaction of 2-hydroxy-1,4-naphthoquinone, benzene-1,2-diamines, malononitriles and isatin/aldehydes in aqueous medium. The attractive features of this methodology are metal-free reaction conditions, use of a bifunctional organocatalyst, water as solvent, easy purification and good yields.
Experimental
General
Starting materials and solvents are commercially available and used without further purification. The purity of the synthesized compounds were ascertained by thin-layer chromatography on silica gel GF 254 in ethyl acetate using iodine vapours as the developing agent. Melting points were determined by the melting point apparatus using capillary tube method and uncorrected. IR spectra were recorded on a Shimadzu FTIR spectrophotometer. \(^{1}\hbox {H}\) NMR and \(^{13}\hbox {C}\) NMR spectra were recorded in \(\hbox {CDCl}_{3}\) and DMSO-\(\hbox {d}_{6}\) and were expressed in parts per million (\(\delta \), ppm) downfield using \(\hbox {Me}_{4}\hbox {Si}\) as internal standard on a Bruker Avance II 400 MHz spectrophotometer. Spin multiplicities are given as s (singlet), d (doublet), t (triplet) and m (multiplet) as well as brs (broad singlet). Coupling constants (J) are given in hertz. Elemental analyses were carried out in a Perkin Elmer 2400 automatic carbon, hydrogen, nitrogen analyzer. Optical rotation was measured on a Jasco P-2000 digital polarimeter with \([\alpha ]_\mathrm{D}\) values reported in degrees; concentration (c) is in g/100 mL.
General procedure for the synthesis of thiourea-based organocatalysts (I–IV)
First, amines (0.5 mmol) were dissolved in DCM and allowed to cool at \(0\,^{\circ }\hbox {C}\). Then, 2-piperidinoethyl isothiocyanate (0.5 mmol, 0.082 mL) was added to the reaction mixture and stirred at room temperature until the reaction was complete as determined by TLC. The reaction mixture was cooled, and the solid was filtered off and washed with ethanol to afford the desired product.
1-((1R,2S)-2,3-Dihydro-2-hydroxy-1H-inden-1-yl)-3-(2-(piperidin-1-yl)ethyl)thiourea (I)
White solid (0.104 g, 33 %): mp 185–187\(\,^{\circ }\hbox {C}\). IR (KBr) \(\hbox {cm}^{-1}\): 3456, 3210, 3050, 2923, 1592, 1230, 755. \([\alpha ]_\mathrm{D}^\mathrm{r.t}= -75.0\) (\(c = 0.5\), \(\hbox {CHCl}_{3}\)), \(^{1}\hbox {H}\) NMR (400 MHz, \(\hbox {CDCl}_{3})\,\delta \): 1.80–0.80 (m, 6H, \(\hbox {CH}_{2}\)), 2.41–2.32 (m, 5H, \(\hbox {CH}_{2})\), 2.65 (brs, 1H, NH), 2.96–2.91 (m, 1H, CH), 3.17–3.12 (m, 1H, CH), 3.44–3.40 (s, 2H, \(\hbox {CH}_{2}\)), 4.30–4.10 (m, 1H, \(\hbox {CH}_{2}\)), 4.70–4.50 (m, 1H, \(\hbox {CH}_{2}\)), 5.90–5.60 (m, 1H, \(\hbox {CH}_{2}\)), 7.27–7.02 (m, 3H, Ar-H), 7.32 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 8.04 (brs, 1H, NH), 8.99 (brs, 1H, OH) ppm. \(^{13}\hbox {C}\) (100 MHz, \(\hbox {CDCl}_{3}\)) \(\delta \): 23.9, 25.0, 39.4, 41.5, 54.8, 62.8, 73.1, 125.2, 126.9, 128.2, 140.5, 182.8 ppm. Anal. Calcd for \(\hbox {C}_{17}\hbox {H}_{25}\hbox {N}_{3}\hbox {OS}\) (319.46): C, 63.91; H, 7.89; N, 13.15; Found: C, 63.84; H, 7.86; N, 13.02.
1-((S)-1-Hydroxy-3-phenylpropan-2-yl)-3-(2-(piperidin-1-yl)ethyl) thiourea (II)
White needle-like solid (0.112 g, 32 %): mp 191–\(194\,^{\circ }\hbox {C}\). IR (KBr) \(\hbox {cm}^{-1}\): 3518, 3239, 2927, 1559, 1455, 1366, 1236, 1118, 1075, 978, 768. \([\alpha ]_\mathrm{D}^\mathrm{r.t} = -3.0\) (\(c=0.5\), \(\hbox {CHCl}_{3}\)), \(^{1}\hbox {H}\) NMR (400 MHz, DMSO-\(\hbox {d}_{6}\)) \(\delta \): 1.41–1.40 (m, 2H, \(\hbox {CH}_{2}\)), 1.55–1.49 (m, 4H, \(\hbox {CH}_{2}\)), 2.39–2.36 (m, 6H, \(\hbox {CH}_{2}\)), 2.88–2.77 (m, 2H, \(\hbox {CH}_{2}\)), 3.45–3.35 (m, 4H, \(\hbox {CH}_{2}\)), 4.41 (brs, 1H, NH), 4.82 (brs, 1H, NH), 7.20–7.15 (m, 1H, CH), 7.32–7.24 (m, 5H, Ar-H), 7.59 (brs, 1H, OH) ppm. \(^{13}\hbox {C}\) (100 MHz, DMSO-\(\hbox {d}_{6}\)) \(\delta \): 23.8, 25.0, 30.2, 33.9, 42.2, 54.5, 60.6, 124.2, 124.7, 126.7, 127.8, 143.2, 182.4 ppm. Anal. Calcd for \(\hbox {C}_{17}\hbox {H}_{27}\hbox {N}_{3}\hbox {OS}\) (321.48): C, 63.51; H, 8.47; N, 13.07; Found: C, 63.59; H, 8.50; N, 13.20.
1-((S)-2,3-Dihydro-1H-inden-1-yl)-3-(2-(piperidin-1-yl)ethyl) thiourea (III)
White crystalline solid (0.215 g, 70 %): mp 215–\(217\,^{\circ }\hbox {C}\). IR (KBr) \(\hbox {cm}^{-1}\): 3244, 3078, 2924, 1601, 1512, 1454, 1238, 964, 752. \([\alpha ]_\mathrm{D}^\mathrm{r.t} = -31.0\) (\(c = 0.5\), \(\hbox {CHCl}_{3}\)), \(^{1}\hbox {H}\) NMR (400 MHz, \(\hbox {CDCl}_{3}\)) \(\delta \): 1.28–1.20 (m, 6H, \(\hbox {CH}_{2}\)), 1.96–1.89 (m, 1H, CH), 2.40–2.10 (m, 4H, \(\hbox {CH}_{2}\)), 2.45–2.43 (m, 2H, \(\hbox {CH}_{2}\)), 2.75–2.74 (m, 1H, CH), 3.03–2.85 (m, 3H, \(\hbox {CH}_{2}\)), 3.47–3.32 (m, 2H, \(\hbox {CH}_{2}\)), 6.73 (brs, 1H, NH), 7.22–7.19 (m, 3H, Ar-H), 7.36 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 9.82 (brs, 1H, NH) ppm. \(^{13}\hbox {C}\) NMR (100 MHz, \(\hbox {CDCl}_{3}\)) \(\delta \): 23.8, 25.1, 30.2, 33.9, 42.3, 54.6, 60.8, 124.3, 124.8, 126.7, 127.9, 143.3, 180.4 ppm. Anal. Calcd for \(\hbox {C}_{17}\hbox {H}_{25}\hbox {N}_{3}\hbox {S}\) (303.47): C, 67.28; H, 8.30; N, 13.85; Found: C, 67.34; H, 8.33; N, 13.97.
1-(1-Benzylpiperidin-4-yl)-3-(2-(piperidin-1-yl)ethyl) thiourea (IV)
White powdery solid (0.304 g, 84 %): mp 240–\(242\,^{\circ }\hbox {C}\). IR (KBr) \(\hbox {cm}^{-1}\): 3273, 3151, 3054, 2929, 1574, 1495, 1240, 961, 860, 747. \(^{1}\hbox {H}\) NMR (400 MHz, \(\hbox {CDCl}_{3}\)) \(\delta \): 1.48–1.47 (m, 2H, \(\hbox {CH}_{2}\)), 1.59–1.51 (m, 6H, \(\hbox {CH}_{2}\)), 2.05–2.02 (m, 2H, \(\hbox {CH}_{2}\)), 2.15–2.09 (m, 2H, \(\hbox {CH}_{2}\)), 2.48–2.42 (m, 7H, \(\hbox {CH}_{2}\), CH), 2.88–2.85 (m, 2H, \(\hbox {CH}_{2}\)), 3.45–3.38 (m, 2H, \(\hbox {CH}_{2}\)), 3.51 (s, 2H, \(\hbox {CH}_{2}\)), 4.02 (brs, 1H, NH), 6.72 (brs, 1H, NH), 7.33–7.22 (m, 5H, Ar-H) ppm. \(^{13}\hbox {C}\) (100 MHz, \(\hbox {CDCl}_{3}\)) \(\delta \): 24.1, 25.8, 30.9, 32.2, 41.8, 52.4, 54.5, 62.9, 127.9, 128.4, 129.3, 138.3, 181.1 ppm. Anal. Calcd for \(\hbox {C}_{20}\hbox {H}_{32}\hbox {N}_{4}\hbox {S}\) (360.56): C, 66.62; H, 8.95; N, 15.54; Found: C, 66.71; H, 8.98; N, 15.68.
General procedure for the synthesis of compounds (5a–o & 7a–j)
First 2-hydroxy-1,4-naphthoquinone (1 mmol) and benzene-1,2-diamine (1 mmol) were refluxed in water (3 mL) for 5 minutes in the presence of 20 mol% thiourea-based organocatalyst IV. Afterwards, isatin or an aldehyde (1 mmol) followed by malononitrile (1 mmol) was added and the reaction was continued until the reaction was complete as determined by TLC. Then, the reaction mixture was allowed to cool to room temperature, and the resulting precipitate was filtered, washed first with water and then with ethanol (5 mL), dried, and recrystallized from an ethanol, ethyl acetate (2:1) mixture to afford the pure product.
3-Amino-11-methyl-2’-oxospiro[benzo[c]pyrano[3,2-a]phenazine-1,3-indoline]-2-carbonitrile (5b)
Yellow solid. mp 345–\(346\,^{\circ }\hbox {C}\). IR (KBr) \(\hbox {cm}^{-1}\): 3364, 3218, 3182, 1697, 1713, 1658, 1605, 1473, 1373, 1165, 1064, 952, 813, 756. \(^{1}\hbox {H}\) NMR (400 MHz, DMSO-\(\hbox {d}_{6}\)) \(\delta \): 2.56 (s, 3H, \(\hbox {CH}_{3}\)), 6.75 (t, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 6.98 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 7.05 (t, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 7.18 (t, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 7.50 (d, \(J = 8.0\,\hbox {Hz}\), 3H, Ar-H, \(\hbox {NH}_{2}\)), 7.61 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 7.99 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 8.03 (t, \(J = 8.0\,\hbox {Hz}\), 2H, Ar-H), 8.51 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 9.24 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 10.78 (s, 1H, NH) ppm. \(^{13}\hbox {C}\) NMR (100 MHz, DMSO-\(\hbox {d}_{6}\)) \(\delta \): 21.6, 49.3, 56.4, 109.2, 110.4, 117.8, 121.6, 122.4, 123.4, 124.7, 125.4, 126.3, 127.3, 127.5, 128.6, 129.7, 130.3, 130.6, 133.3, 135.7, 138.7, 139.8, 140.9, 141.2, 142.8, 147.0, 159.4, 179.1 ppm. Anal. Calcd. For \(\hbox {C}_{28}\hbox {H}_{17}\hbox {N}_{5}\hbox {O}_{2}\) (455.47): C, 73.84; H, 3.76; N, 15.38; Found: C, 73.93; H, 3.79; N, 15.42.
3-Amino-5’-iodo-11-methyl-2’-oxospiro[benzo[c]pyrano [3,2-a]phenazine-1,3-indoline]-2-carbonitrile (5c)
Yellow solid. mp 324–\(325\,^{\circ }\hbox {C}\). IR (KBr) \(\hbox {cm}^{-1}\): 3305, 3209, 3136, 2985, 1720, 1659, 1605, 1474, 1369, 1161, 1065, 952, 813, 763. \(^{1}\hbox {H}\) NMR (400 MHz, DMSO-\(\hbox {d}_{6}\)) \(\delta \): 2.60 (s, 3H, \(\hbox {CH}_{3}\)), 6.98 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 7.44–7.43 (m, 1H, Ar-H), 7.59–7.54 (m, 1H, Ar-H), 7.60 (s, 2H, \(\hbox {NH}_{2}\)), 7.65 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 7.82 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 8.05 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 8.12–8.08 (m, 2H, Ar-H), 8.55 (d, \(J = 8.0\) Hz, 1H, Ar-H), 9.30 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 11.01 (s, 1H, NH) ppm. \(^{13}\hbox {C}\) NMR (100 MHz, DMSO-\(\hbox {d}_{6}\)) \(\delta \): 21.5, 49.2, 56.5, 84.5, 109.6, 111.7, 122.5, 124.7, 125.5, 127.4, 129.7, 130.3, 130.6, 131.8, 133.4, 136.9, 138.3, 139.3, 139.5, 139.7, 140.2, 140.9, 142.7, 147.3, 159.5, 178.6 ppm. Anal. Calcd for \(\hbox {C}_{28}\hbox {H}_{16}\hbox {IN}_{5}\hbox {O}_{2}\) (581.36): C, 57.85; H, 2.77; N, 12.05; Found: C, 57.93; H, 2.80; N, 12.18.
3-Amino-5’-chloro-11-methyl-2’-oxospiro[benzo[c] pyrano[3,2-a]phenazine-1,3-indoline]-2-carbonitrile (5d)
Yellow solid. mp 337–\(339\,^{\circ }\hbox {C}\). IR (KBr) \(\hbox {cm}^{-1}\): 3435, 3307, 3178, 2984, 1756, 1648, 1603, 1478, 1365, 1163, 1069, 956, 817, 756. \(^{1}\hbox {H}\) NMR (400 MHz, DMSO-\(\hbox {d}_{6}\)) \(\delta \): 2.54 (s, 3H, \(\hbox {CH}_{3}\)), 7.06 (t, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 7.16–7.15 (m, 1H, Ar-H), 7.26–7.22 (m, 1H, Ar-H), 7.62–7.59 (m, 3H, H-Ar, \(\hbox {NH}_{2}\)), 7.79–7.75 (m, 1H, Ar-H), 8.01 (t, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 8.06 (t, \(J = 8.0\,\hbox {Hz}\), 2H, Ar-H), 8.51 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 9.26 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 10.96 (s, 1H, NH) ppm. \(^{13}\hbox {C}\) NMR (100 MHz, DMSO-\(\hbox {d}_{6}\)) \(\delta \): 21.6, 49.5, 56.6, 109.5, 110.5, 117.7, 122.5, 123.7, 124.7, 125.4, 125.5, 127.4, 128.2, 129.7, 130.3, 133.4, 137.7, 138.7, 139.3, 139.5, 139.7, 140.1, 140.9, 141.3, 141.8, 147.3, 159.6, 178.9 ppm. Anal. Calcd for \(\hbox {C}_{28}\hbox {H}_{16}\hbox {C}\hbox {lN}_{5}\hbox {O}_{2}\) (489.91): C, 68.64; H, 3.29; N, 14.30; Found: C, 68.73; H, 3.33; N, 14.44.
3-Amino-5’-nitro-2’-oxospiro[benzo[c]pyrano[3,2-a] phenazine-1,3-indoline]-2-carbonitrile (5e)
Yellow solid. mp 314–\(316\,^{\circ }\hbox {C}\). IR (KBr) \(\hbox {cm}^{-1}\): 3452, 3275, 3167, 2989, 1724, 1631, 1601, 1589, 1489, 1388, 1168, 1080, 956, 829, 759. \(^{1}\hbox {H}\) NMR (400 MHz, DMSO-\(\hbox {d}_{6}\)) \(\delta \): 7.30 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 7.69 (s, 2H, \(\hbox {NH}_{2}\)), 7.75 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 7.91–7.87 (m, 2H, Ar-H), 7.97 (s, 1H, Ar-H), 8.03 (t, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 8.05 (t, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 8.22 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 8.29 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 8.59 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 9.33 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 11.59 (s, 1H, NH) ppm. \(^{13}\hbox {C}\) NMR (100 MHz, DMSO-\(\hbox {d}_{6}\)) \(\delta \): 49.3, 55.7, 108.8, 109.4, 116.8, 117.5, 119.6, 122.7, 124.8, 125.5, 125.9, 127.6, 129.1, 129.9, 130.4, 130.5, 130.8, 131.0, 136.6, 139.6, 140.1, 140.9, 142.4, 148.2, 149.4, 159.7, 179.6 ppm. Anal. Calcd for \(\hbox {C}_{27}\hbox {H}_{14}\hbox {N}_{6}\hbox {O}_{4}\) (486.44): C, 66.67; H, 2.90; N, 17.28; Found: C, 66.74; H, 2.93; N, 17.40.
3-Amino-5’-iodo-2’-oxospiro[benzo[c]pyrano[3,2-a] phenazine-1,3-indoline]-2-carbonitrile (5f)
Yellow solid. mp 365–\(367\,^{\circ }\hbox {C}\). IR (KBr) \(\hbox {cm}^{-1}\): 3450, 3275, 3163, 2985, 1724, 1635, 1489, 1388, 1315, 1168, 1080, 829, 756. \(^{1}\hbox {H}\) NMR (400 MHz, DMSO-\(\hbox {d}_{6})\,\delta \): 6.90 (d, \(J = 8.0\) Hz, 1H, Ar-H), 7.29 (s, 1H, Ar-H), 7.53–7.48 (m, 3H, Ar-H), 7.74 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 7.87 (s, 2H, \(\hbox {NH}_{2}\)), 8.02–7.98 (m, 2H, Ar-H), 8.26 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 8.52 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 9.30 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 10.94 (s, 1H, NH) ppm. \(^{13}\hbox {C}\) NMR (100 MHz, DMSO-\(\hbox {d}_{6}\)) \(\delta \): 49.2, 56.6, 84.5, 109.5, 111.7, 117.7, 122.6, 124.9, 125.6, 127.8, 129.1, 129.8, 130.3, 130.5, 130.8, 130.9, 131.8, 136.9, 138.2, 139.5, 140.1, 140.2, 141.0, 142.7, 147.8, 159.5, 178.6 ppm. Anal. Calcd for \(\hbox {C}_{27}\hbox {H}_{14}\hbox {IN}_{5}\hbox {O}_{2}\) (567.34): C, 57.16; H, 2.49; N, 12.34; Found: C, 57.25; H, 2.52; N, 12.46.
3-Amino-5’-iodo-2’-oxospiro[benzo[c]pyrano[3,2-a]phenazine-1,3-indoline]-2-carboxylate (5g)
Yellow solid. mp 318–\(320\,^{\circ }\hbox {C}\). IR (KBr) \(\hbox {cm}^{-1}\): 3460, 3367, 2978, 1697, 1647, 1604, 1470, 1373, 1172, 1091, 952, 879, 756. \(^{1}\hbox {H}\) NMR (400 MHz, DMSO-\(\hbox {d}_{6}\)) \(\delta \): 0.97 (t, \(J = 8.0\,\hbox {Hz}\), 3H, \(\hbox {CH}_{3}\)), 3.89 (t, \(J = 8.0\,\hbox {Hz}\), 2H, \(\hbox {CH}_{2}\)), 6.80 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 7.22 (s, 1H, Ar-H), 7.42 (t, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 7.88 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 7.94–7.92 (m, 2H, Ar-H), 8.01 (t, \(J = 8.0\) Hz, 1H, Ar-H), 8.06 (t, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 8.23 (s, 2H, \(\hbox {NH}_{2}\)), 8.28 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 8.62 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 9.26 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 10.79 (s, 1H, NH) ppm. \(^{13}\hbox {C}\) NMR (100 MHz, DMSO-\(\hbox {d}_{6}\)) \(\delta \): 13.3, 49.2, 59.1, 75.5, 82.9, 110.6, 111.8, 122.9, 124.7, 125.7, 127.8, 128.9, 129.7, 130.2, 130.4, 130.6, 130.7, 130.9, 135.9, 139.2, 139.6, 139.7, 140.8, 140.9, 145.2, 146.7, 159.7, 167.6, 180.2 ppm. Anal. Calcd for \(\hbox {C}_{29}\hbox {H}_{19}\hbox {IN}_{4}\hbox {O}_{4}\) (614.39): C, 56.69; H, 3.12; N, 9.12; Found: C, 56.76; H, 3.15; N, 9.24.
3-Amino-1-phenyl-1H-benzo[c]pyrano[3,2-a]phenazine-2-carbonitrile (7a)
Yellow powder. mp 301–\(303\,^{\circ }\hbox {C}\). IR (KBr) \(\hbox {cm}^{-1}\): 3457, 3315, 3168, 2193, 1350, 1270. \(^{1}\hbox {H}\hbox {NMR}\) (400 MHz, DMSO-\(d_{6}\)) \(\delta \): 5.37 (s, 1H, CH), 7.08 (t, \(J = 7.4\,\hbox {Hz}\), 1H, Ar-H), 7.21 (t, \(J = 7.6\,\hbox {Hz}\), 2H, Ar-H), 7.36–7.38 (m, 4H Ar-H, \(\hbox {NH}_{2}\)), 7.85–7.86 (m, 3H, Ar-H), 7.86 (t, \(J = 4.8\,\hbox {Hz}\), 1H, Ar-H), 8.00–8.12 (m, 1H, Ar-H), 8.13–8.15 (m, 1H, Ar-H), 8.36 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 9.06 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H) ppm. \(^{13}\hbox {C}\) NMR (100 MHz, DMSO-\(d_{6}\)) \(\delta \): 37.8, 58.5, 114.2, 120.7, 122.4, 125.1, 125.9, 126.9, 128.1, 129.0, 129.3, 129.4, 130.3, 130.5, 130.8, 131.0, 140.0, 140.3, 140.9, 141.8, 145.7, 146.5, 160.2, 160.3 ppm. Anal. Calcd. for \(\hbox {C}_{26}\hbox {H}_{16}\hbox {N}_{4}\hbox {O}\): C, 77.99; H, 4.03; N, 13.99; Found: C, 76.28; H, 4.19; N, 13.57.
3-Amino-1-(4-isopropylphenyl)-1H-benzo[c]pyrano [3,2-a]phenazine-2-carbonitrile (7b)
Yellow solid. mp 352–\(353\,^{\circ }\hbox {C}\). IR (KBr) \(\hbox {cm}^{-1}\): 3348, 3155, 2958, 1662, 1635, 1597, 1496, 1384, 1288, 1153, 1049, 948, 833, 752. \(^{1}\hbox {H}\) NMR (400 MHz, DMSO-\(\hbox {d}_{6}\)) \(\delta \): 1.10 (d, \(J = 8.0\,\hbox {Hz}\), 6H, \(\hbox {CH}(\hbox {CH}_{3})_{2})\), 2.77–2.74 (m, 1H, CH), 5.56 (s, 1H, CH), 7.08 (d, \(J = 8.0\,\hbox {Hz}\), 2H, Ar-H), 7.29 (s, 2H, \(\hbox {NH}_{2}\)), 7.36 (d, \(J = 8.0\,\hbox {Hz}\), 2H, Ar-H), 7.96–7.92 (m, 3H, Ar-H), 7.99 (t, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 8.25–8.21 (m, 1H, Ar-H), 8.31–8.29 (m, 1H, Ar-H), 8.49 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 9.30 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H) ppm. \(^{13}\hbox {C}\) NMR (100 MHz, DMSO-\(\hbox {d}_{6}\)) \(\delta \): 20.7, 23.7, 36.8, 58.0, 114.3, 120.3, 121.9, 122.1, 124.9, 125.5, 125.7, 126.3, 127.4, 128.8, 129.1, 130.3, 130.6, 130.9, 139.6, 139.9, 140.8, 141.6, 142.6, 145.3, 146.2, 146.6, 160.0, 170.3 ppm. Anal. Calcd for \(\hbox {C}_{29}\hbox {H}_{22}\hbox {N}_{4}\hbox {O}\) (442.51): C, 78.71; H, 5.01; N, 12.66; Found: C, 78.80; H, 5.04; N, 12.79.
3-Amino-11-methyl-1-(4-methoxy)-1H-benzo[c] pyrano[3,2-a]phenazine-2-carbonitrile (7c)
Yellow solid. mp 361–\(362\,^{\circ }\hbox {C}\). IR (KBr) \(\hbox {cm}^{-1}\): 3356, 3204, 3162, 2978, 1654, 1607, 1508, 1384, 1242, 1157, 1033, 952, 825, 763. \(^{1}\hbox {H}\) NMR (400 MHz, DMSO-\(\hbox {d}_{6}\)) \(\delta \): 2.63 (s, 3H, \(\hbox {CH}_{3})\), 3.63 (s, 3H, \(\hbox {CH}_{3})\), 5.50 (s, 1H, CH), 6.79 (d, \(J = 8.0\,\hbox {Hz}\), 2H, Ar-H), 7.35–7.33 (m, 4H, Ar-H, \(\hbox {NH}_{2}\)), 7.81 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 7.95 (t, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 8.01 (t, \(J= 8.0\,\hbox {Hz}\), 1H, Ar-H), 8.11 (t, \(J = 8.0\,\hbox {Hz}\), 2H, Ar-H), 8.46 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 9.25 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H) ppm. \(^{13}\hbox {C}\) NMR (100 MHz, DMSO-\(\hbox {d}_{6}\)) \(\delta \): 21.5, 32.9, 36.8, 58.1, 59.7, 114.2, 120.5, 121.8, 121.9, 124.7, 125.5, 126.2, 127.4, 128.6, 128.7, 128.9, 129.9, 130.1, 130.2, 139.6, 139.8, 140.1, 140.4, 142.7, 145.6, 146.5, 159.9, 170.3 ppm. Anal. Calcd for \(\hbox {C}_{28}\hbox {H}_{20}\hbox {N}_{4}\hbox {O}_{2}\) (444.48): C, 75.66; H, 4.54; N, 12.60; Found: C, 75.75; H, 4.57; N, 12.73.
3-Amino-1-(2-chlorophenyl)-1H-benzo[c]pyrano[3,2-a]phenazine-2-carbonitrile (7d)
Yellow solid. mp 301–\(303\,^{\circ }\hbox {C}\). IR (KBr) \(\hbox {cm}^{-1}\): 3336, 3234, 3147, 2984, 1658, 1585, 1477, 1388, 1276, 1157, 1041, 952, 836, 752. \(^{1}\hbox {H}\) NMR (400 MHz, DMSO-\(\hbox {d}_{6}\)) \(\delta \): 6.02 (s, 1H, CH), 7.15–7.11 (m, 2H, Ar-H), 7.25–7.23 (m, 1H, Ar-H), 7.37 (s, 2H, \(\hbox {NH}_{2})\), 7.44–7.42 (m, 1H, Ar-H), 7.95–7.92 (m, 2H, Ar-H), 8.07–7.96 (m, 3H, Ar-H), 8.32–8.29 (m, 1H, Ar-H), 8.50 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 9.29 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H) ppm. \(^{13}\hbox {C}\) NMR (100 MHz, DMSO-\(\hbox {d}_{6}\)) \(\delta \): 36.7, 56.8, 112.9, 119.6, 122.3, 124.9, 125.6, 127.5, 128.1, 128.6, 129.1, 129.2, 129.3, 130.2, 130.3, 130.6, 130.8, 132.3, 140.0, 140.1, 140.8, 141.6, 142.7, 146.7, 159.5, 170.4 ppm. Anal. Calcd for \(\hbox {C}_{26}\hbox {H}_{15}\hbox {C}\hbox {lN}_{4}\hbox {O}\) (434.88): C, 71.81; H, 3.48; N, 12.88; Found: C, 71.90; H, 3.51; N, 13.01.
3-Amino-11-methyl-1-(4-isopropylphenyl)-1H-benzo[c] pyrano[3,2-a]phenazine-2-carbonitrile (7e)
Yellow Solid. mp 321–\(323\,^{\circ }\hbox {C}\). IR (KBr) \(\hbox {cm}^{-1}\): 3333, 3174, 2966, 1658, 1608, 1508, 1384, 1273, 1157, 1049, 948, 817, 759. \(^{1}\hbox {H}\) NMR (400 MHz, DMSO-\(\hbox {d}_{6}\)) \(\delta \): 1.09 (d, \(J = 8.0\) Hz, 6H, \(\hbox {CH}(\hbox {CH}_{3})_{2})\), 2.52 (s, 3H, \(\hbox {CH}_{3}\)), 2.63–2.62 (m, 1H, CH), 5.51 (s, 1H, CH), 7.09–7.06 (m, 2H, Ar-H), 7.25 (s, 2H, \(\hbox {NH}_{2}\)), 7.34 (d, \(J = 8.0\,\hbox {Hz}\), 2H, Ar-H), 7.73 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 7.89 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 7.93 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 8.04 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 8.12 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 8.45 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H), 9.22 (d, \(J = 8.0\,\hbox {Hz}\), 1H, Ar-H) ppm. \(^{13}\hbox {C}\) NMR (100 MHz, DMSO-\(\hbox {d}_{6}\)) \(\delta \): 14.7, 21.5, 23.8, 32.9, 36.8, 58.1, 114.2, 120.4, 121.9, 124.7, 125.6, 126.3, 127.0, 127.4, 128.8, 129.9, 130.1, 133.2, 138.7, 139.6, 139.8, 140.4, 140.5, 141.1, 141.6, 142.7, 145.6, 146.5, 160.0, 171.4, ppm. Anal. Calcd for \(\hbox {C}_{30}\hbox {H}_{24}\hbox {N}_{4}\hbox {O}\) (456.54): C, 78.92; H, 5.30; N, 12.27; Found: C, 79.01; H, 5.33; N, 12.40.
References
MacMillan DWC (2008) The advent and development of organocatalysis. Nature 455:304–308. doi:10.1038/nature07367
Dondoni A, Massi A (2008) Asymmetric organocatalysis: from infancy to adolescence organocatalysis. Angew Chem Int Ed 47:4638–4660. doi:10.1002/anie.200704684
Yu XH, Wang W (2008) Hydrogen-bond-mediated asymmetric catalysis. Asian J Chem 3:516–532. doi:10.1002/asia.200700415
Dalko PI, Moisan L (2004) In the golden age of organocatalysis. Angew Chem Int Ed 43:5138–5175. doi:10.1002/anie.200400650
Bertelsen S, Jorgensen KA (2009) Organocatalysis-after the gold rush. Chem Soc Rev 38:2178–2189. doi:10.1039/b903816g
List B (2007) Introduction: Organocatalysis. Chem Rev 107:5413–5415. doi:10.1021/cr078412e
Schreiner PR (2003) Metal-free organocatalysis through explicit hydrogen bonding interactions. Chem Soc Rev 32:289–296. doi:10.1039/B107298F
Doyle AG, Jacobsen EN (2007) Small-molecule H-bond donors in asymmetric catalysis. Chem Rev 107:5713–5743. doi:10.1021/cr068373r
Sigman MS, Jacobsen EN (1998) Schiff base catalysts for the asymmetric strecker reaction identified and optimized from parallel synthetic libraries. J Am Chem Soc 120:4901–4902. doi:10.1021/ja980139y
Schreiner PR, Wittkopp A (2002) H-bonding additives act like lewis acid catalysts. Org Lett 4:217–220. doi:10.1021/ol017117s
Okino T, Hoashi Y, Takemoto Y (2003) Enantioselective michael reaction of malonates to nitroolefins catalyzed by bifunctional organocatalysts. J Am Chem Soc 125:12672–12673. doi:10.1021/ja036972z
Takemoto Y (2005) Recognition and activation by ureas and thioureas: stereoselective reactions using ureas and thioureas as hydrogen-bonding donors. Org Biomol Chem 3:4299–4306. doi:10.1039/B511216H
Serdyuk OV, Heckel CM, Tsogoeva SB (2013) Bifunctional primary amine-thioureas in asymmetric organocatalysis. Org Biomol Chem 11:7051–7071. doi:10.1039/C3OB41403E
Fang X, Wang C-J (2015) Recent advances in asymmetric organocatalysis mediated by bifunctional amine-thioureas bearing multiple hydrogen-bonding donors. Chem Commun 51:1185–1197. doi:10.1039/C4CC07909D
Bugaut X, Constantieux T, Coquerel Y, Rodriguez J (2014) In: Zhu J,Wang Q, Wang M-X (eds) Multicomponent reactions in organic synthesis. Chap 5. Wiley, Weinheim, pp 109–158
Choudhury LH, Parvin T (2011) Recent advances in the chemistry of imine-based multicomponent reactions (MCRs). Tetrahedron 67:8213–8228. doi:10.1016/j.tet.2011.07.020
Rotstein BH, Zaretsky S, Rai V, Yudin AK (2014) Small heterocycles in multicomponent reactions. Chem Rev 114:8323–8359. doi:10.1021/cr400615v
Nair V, Rajesh V, Vinod A, Bindu US, Streekenth AR, Mathen JS, Balagopal L (2003) Strategies for heterocyclic construction via novel multicomponent reactions based on isocyanides and nucleophilic carbenes. Acc Chem Res 36:899–907. doi:10.1021/ar020258p
Dömling A (2006) Recent developments in isocyanide based multicomponent reactions in applied chemistry. Chem Rev 106:17–89. doi:10.1021/cr0505728
Das D, Banerjee R, Mitra A (2014) Bioactive and pharmacologically important pyrano[2,3-c]pyrazoles. J Chem Pharmaceut Res 6:108–116
Malladi S, Isloora AM, Peethambar SK, Ganesh BM (2012) Palusa, Goud SK. Der Pharma Chem 4:43–52
Laursen JB, Nielsen J (2004) Phenazine natural products: Biosynthesis, synthetic analogues, and biological activity. J Chem Rev 104:1663–1686. doi:10.1021/cr020473
Hafez HN, Hegab MI, Ahmed-Farag IS, El-Gazzar ABA (2008) A facile regioselective synthesis of novel spiro-thioxanthene and spiro-xanthene-\(9^\prime \),2-[1,3,4]thiadiazole derivatives as potential analgesic and anti-inflammatory agents. Bioorg Med Chem Lett 18:4538–4543. doi:10.1016/j.bmcl.2008.07.042
Mavrodi DV, Blankenfeldt W, Thomashow LS (2006) Phenazine compounds in fluorescent pseudomonas spp. biosynthesis and regulation. Annu Rev Phytopathol 44:417–445. doi:10.1146/annurev.phyto.44.013106.145710
Gamage SA, Spicer JA, Rewcastle GW, Milton J, Sohal S, Dangerfield W, Mistry P, Vicker N, Charlton PA, Denny WA (2002) Structure-activity relationships for pyrido-, imidazo-, pyrazolo-, pyrazino-, and pyrrolophenazinecarboxamides as topoisomerase-targeted anticancer agents. J Med Chem 45:740–743. doi:10.1021/jm010330
Tangmouo JG, Meli AL, Komguem J, Kuete V, Ngounou FN, Lontsi D, Beng VP, Choudhary MI, Sondengam BL (2006) Crassiflorone, a new naphthoquinone from Diospyros crassiflora (Hien). Tetrahedron Lett 47:3067–3070. doi:10.1016/j.tetlet.2006.03.006
Kraus GA, Kim IA (2003) A direct synthesis of \(o\)-methyl claussequinone. J Org Chem 68:4517–4518. doi:10.1021/jo030026j
Vicker N, Burgess L, Chuckowree IS, Dodd R, Folkes AJ, Hardick DJ, Hancox TC, Dangerfield W, Liddle C, Mistry P, Stewart AJ, Denny WA (2002) Novel angular benzophenazines: dual topoisomerase I and topoisomerase II inhibitors as potential anticancer agents. J Med Chem 45:721–739. doi:10.1021/jm010329a
Shahia M, Foroughifar N, Mobinikhaledi A (2015) Synthesis and antimicrobial activity of some tetrahydro quinolonediones and pyrano[2,3-d]pyrimidine derivatives. Iran J Pharm Res 14:757–763
Dar AM, uzzaman Shams (2015) Pathways for the synthesis of pyrimidine and pyran based hetrocyclic derivatives: a concise review. Eur Chem Bull 4:249–259. doi:10.17628/ECB.2015.4.249
de Andrade-Neto VF, Goulart MOF, da Silva Filho JF, da Silva MJ, do Pinto M CFR, Pinto AV, Zalis MG, Carvalho LH, Krettli AU (2004) Antimalarial activity of phenazines from lapachol, beta-lapachone and its derivatives against Plasmodium falciparum in vitro and Plasmodium berghei in vivo. Bioorg Med Chem Lett 14:1145–1149. doi:10.1016/j.bmcl.2003.12.069
Feron O, Riant O, Kiss R, Leclercq J, Chataigne G, Vandelaer N, Lamy C (2013) Novel phenazine derivatives and their use. US Patent 20130289030 A1, 31 Oct 2013
Jardim GAM, Cruz EHG, Valença WO, Resende JM, Rodrigues BL, Ramos DF, Oliveira RN, Silva PEA, da Silva Júnior EN (2015) On the search for potential antimycobacterial drugs: synthesis of naphthoquinoidal, phenazinic and 1,2,3-triazolic compounds and evaluation against mycobacterium tuberculosis. J Braz Chem Soc 26:1013–1027. doi:10.5935/0103-5053.20150067
Hasaninejad A, Firoozi S (2013) One-pot, sequential four-component synthesis of benzo[c]pyrano[3,2-a]phena-zine, bis-benzo[c]pyrano[3,2-a]phenazine and oxospiro benzo[c]pyrano[3,2-a]phenazine derivatives using 1,4-diazabicyclo[2.2.2]octane (DABCO) as an efficient and reusable solid base catalyst. Mol Divers 17:499–513. doi:10.1007/s11030-013-9446-x
Wang SL, Wu F-Y, Cheng C, Zhang G, Liu Y-P, Jiang B, Shi F, Ju S-J (2011) Multicomponent synthesis of poly-substituted benzo[\(a\)]pyrano[2,3-\(c\)]phenazine derivatives under microwave heating. ACS Comb Sci 13:135–139. doi:10.1021/co1000376
Mahdavinia GH, Mirzazadeh M, Notash B (2013) A rapid and simple diversity-oriented synthesis of novel 3-amino-\(2^\prime \)-oxospiro [benzo[\(c\)]pyrano[3,2-\(a\)]phenazine-1,\(3^\prime \)-indoline]-2-carbonitrile/carboxylate derivatives via a one-pot, four-component domino reaction. Tetrahedron Lett 54:3487–3492. doi:10.1016/j.tetlet.2013.04.082
Hasaninejad A, Firoozi S, Mandegani F (2013) An efficient synthesis of novel spiro[benzo[\(c\)]pyrano[3,2-\(a\)]phenazines] via domino multi-component reactions using l-proline as a bifunctional organocatalyst. Tetrahedron Lett 54:2791–2794. doi:10.1016/j.tetlet.2013.03.073
Elah Abadi AY, Maghsoodlou M-T, Heydari R, Mohebat R (2015) PTSA-catalyzed four-component domino reactions for the one-pot synthesis of functionalized 11H-benzo[a]benzo[6,7]chromeno[2,3-c]phenazine-11,16(17H)-diones in PEG. Res Chem Intermed. doi:10.1007/s11164-015-2083-5
Bharti R, Parvin T (2015) Diversity oriented synthesis of tri-substituted methane containing aminouracil and hydroxynaphthoquinone /hydroxycoumarin moiety using organocatalysed multicomponent reactions in aqueous medium. RSC Adv 5:66833–66839. doi:10.1039/c5ra13093j
Bharti R, Parvin T (2015) Molecular Diversity from the L-proline catalyzed, three-component reactions of 4-hydroxycoumarin, aldehyde, and 3-aminopyrazole or 1,3-dimethyl-6-aminouracil. Synth Commun 45:1442–1450. doi:10.1002/chin.201537164
Bharti R, Parvin T (2015) One-pot synthesis of highly functionalized tetrahydropyridines: a camphoresulfonic acid catalyzed multicomponent reaction. J Heterocycl Chem 52:1806–1811. doi:10.1002/jhet.2268
Karamthulla S, Pal S, Parvin T, Choudhury LH (2014) L-proline catalyzed multicomponent reactions: facile access to 2H-benzo[g]pyrazolo[3,4-b]quinoline-5,10(4H,11H)-dione derivatives. RSC Adv 4:15319–15324. doi:10.1039/c4ra00876f
Pal S, Parvin T, Choudhury LH (2012) \(\text{ VCl }_{3}\) catalyzed imine-based multicomponent reactions for the facile access of functionalized tetrahydropyridines and \(\upbeta \)-amino carbonyls. Mol Divers 16:129–143. doi:10.1007/s11030-011-9339-9
Khan AT, Parvin T, Choudhury LH (2008) Effects of substituent in \(\beta \)-position of 1, 3-dicarbonyl compounds in bromodimethylsulfonium bromide catalyzed multicomponent reactions: a facile access to functionalized piperidines. J Org Chem 73:8398–8402. doi:10.1021/jo8014962
Acknowledgments
We are grateful to NIT Patna and the Department of Science and Technology, India for the financial support with Sanction No. SR/FT/CS-008/2010. The authors are grateful to IIT Patna and SAIF-Panjab University for providing the analytical facilities for characterization of products.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bharti, R., Parvin, T. Multicomponent synthesis of diverse pyrano-fused benzophenazines using bifunctional thiourea-based organocatalyst in aqueous medium. Mol Divers 20, 867–876 (2016). https://doi.org/10.1007/s11030-016-9681-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11030-016-9681-z