Abstract
A variety of organocatalysts has been screened for the synthesis of arylaminonaphthols. It has been shown that (N,N-dimethylethanolamine) is a highly efficient organocatalyst for the direct synthesis of a novel class of arylaminonaphthols via three-component condensation of 2-naphthol, aldehydes, and arylamines under solvent-free conditions. Mild, one-pot, and green reaction conditions, relatively short reaction times and good yields make this protocol highly significant. 25 new compounds have been synthesized by this method.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Compounds bearing 1,3-amino-oxygenated functional groups are frequently found in various biologically active natural products [1] and are used as drugs [1–3]. One important class of such compounds is the aminonaphthols, so-called ‘Betti bases’ [4]. In light of these compounds possesing beneficial biological properties, such as antipain, antibacterial, hypotensive, and bradycardiac activities [5–8] (Fig. 1), the catalytic and synthetic applications of Betti bases, as well as the synthesis of substituted Betti base derivatives have become an important area in synthetic organic chemistry [4, 9].
Since Betti’s first report in 1900, several methods for the synthesis of aminonaphthols have been developed. The most popular method for the preparation of aminonaphthols is the reaction of an aldehyde, 2-naphthol and an aliphatic amine (Scheme 1) [4]. Another approach is the hydrolysis of preformed amidoalkyl naphthols. For this reason, the synthesis of amidoalkyl naphthols has received a lot of attention (Scheme 1) [10–19]. A literature survey shows that most of the reported methods suffer from disadvantages, such as low product yields, expensive catalysts, long-reaction times and laborious workup [4, 9]. Furthermore, most of these methods are unsuccessful [20–22] or inefficient [23, 24] for the synthesis of arylaminonaphthols. However, some hetroarylaminonaphthols have been synthesized when using electron-rich hetroaromatic amines as amine sources [25–27].
Organocatalysts have shown immense promise in multicomponent reactions [28]. The choice of organic catalyst has some advantages including (i) easy preparation or availability, (ii) performing reactions under milder reaction conditions, (iii) selectivity, and (iv) compatibility with numerous functional groups [29]. Recently, various organocatalysts including derivatives of \(N\),\(N\)-dialkylethanolamine have been used to catalyze Michael addition [30] and Friedel–Crafts alkylation reactions [31, 32]. Nevertheless, to the best of our knowledge, there are no reports for the synthesis of Betti bases using organocatalysts. Therefore, we selected several commercially available amines and aminoalcohols to catalyze the Betti reaction, taking advantage of their hydrogen bond donor and/or acceptor attributes.
As a part of our ongoing interest in the study of MCRs [33–37], herein we describe the use of bifunctional organocatalysts as catalysts for the direct and efficient synthesis of alkyl- and arylaminonaphthol derivatives via the one-pot and solvent-free three-component condensation of \(\beta \)-naphthol, aldehydes, and arylamines.
Results and discussion
As a starting point for our studies, the reaction of 2-naphthol (3a) with benzaldehyde (12a) and aniline (13a) was chosen as our model reaction to investigate. For this, a variety of organocatalysts (Fig. 2) was tested using various organic solvents and solvent-free condition. The results are summarized in Table 1.
As shown in Table 1, different conventional organic solvents, such as EtOH, \(\hbox {CHCl}_{3}\) and \(\hbox {CH}_{2}\hbox {Cl}_{2}\) afforded low to moderate yields at r.t. (15–50 %, entries 1–15). Interestingly, the best results were achieved under solvent-free conditions. Organocatalyst IX, with one H-bond donor and one H-bond acceptor gave a promising result (70 % yield, entry 26). Although organocatalysts V, VI, and VII possess two or three H-bond donors and one H-bond acceptor, they produced lower yields (55–62 %, entries 22–24), and organocatalysts without a H-bond donor (I–IV) provided poor results (15–25 % yield, entries 18–21). These results show the importance of H-bond donor and H-bond acceptor interactions of the catalyst with substrates in these reactions. Notably, there was no need to carry out the reactions in an inert atmosphere.
To optimize the reaction conditions, the model reaction was exposed to different quantities of organocatalyst IX (5, 7.5, and 10 mol %) at various temperatures. As shown in Table 2, the best result (90 % yield) was obtained when the reaction was performed using 7.5 mol% of \(N\),\(N\)-dimethylethanolamine (DMEA) at 50 \(^{\circ }\hbox {C}\) (entry 6).
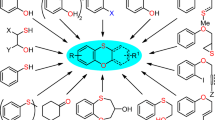
To explore the scope and generality of this procedure, the condensation of 2-naphthols with a variety of aromatic aldehydes and aromatic amines (Fig. 3) was examined in the presence of DMEA (7.5 mol%) at 50 \(^{\circ }\hbox {C}\) under solvent-free condition (Table 3).
As shown in Table 3, the reactions were carried out efficiently within 30–45 min, and the desired products were produced in good to excellent yields (Fig. 4). Different substituted benzaldehydes successfully reacted with aniline and 2-naphthol giving the corresponding products 14a–i in 81–95 % yields. Reacting substituted anilines with benzaldehyde and 2-naphthol afforded their corresponding products 14j–n in 80–95 % yields. Also, when both of benzaldehyde and aniline were substituted the desired products 14o–q obtained in 92–95 % yields.
The reaction of heteroaromatic aldehydes with heteroarylamines in the presence of catalytic amounts of DMEA also afforded desired products 14r–14t in high yields (92–95 %).
To rationalize the beneficial role of DMEA, we offer two plausible interactions of bifunctional organocatalyst in this transformation (Fig. 5).
After obtaining satisfactory results for the formation of arylaminonaphthols (Fig. 4), we extended this method to the synthesis of the corresponding alkylaminonaphthol analogs under similar conditions and, as expected, the products were also obtained in good to high yields (Table 4).
A plausible mechanistic rationalization of the DMEA-catalyzed reaction is shown in Scheme 2. It is expected that the reaction proceeds via an iminium salt intermediate which is generated by the condensation of a secondary aliphatic amine with aldehyde followed by dehydration. In the next step, the nucleophilic attack of 2-naphthol to the iminium salt generates an alkylaminonaphthol. In a similar manner, corresponding arylaminonaphthols are synthesized via imine intermediates when using primary aromatic amines.
To highlight the applicability and the efficiency of our methodology, we compared the results of two DMEA-catalyzed reactions with other recently reported methodologies. As shown in Table 5, DMEA has a significant impact in the performance of the reaction proving to be convincingly superior to other catalytic methods.
Conclusions
In conclusion, we have developed a high-yielding protocol for the synthesis of aminonaphthols using DMEA as organocatalyst. A novel series of arylaminonaphthols were synthesized via the one-pot three-component reaction of an aldehyde, an aromatic amine, and 2-naphthol in the presence of DMEA under solvent-free conditions. Our methodology proved to also be useful for the synthesis of alkylaminonaphthols. The operational simplicity of our procedure, short-reaction times, easy workup, extremely mild reaction conditions, and environmental friendliness (non-corrosive catalyst) make our method very attractive.
Experimental
All chemicals were purchased from Merck or Aldrich chemical companies and used without further purification. Melting points were determined on a MEL-TEMP model 1202D and are uncorrected. FT-IR spectra were recorded on a Bruker Tensor 27 spectrometer as KBr disks. The \(^{1}\hbox {H NMR}\) spectra were recorded on a Bruker Spectrospin Advance 400 spectrometer. \(^{13}\hbox {C NMR}\) spectra were also recorded on the same instrument at 100 MHz. All chemical shifts were reported as \(\delta \,\hbox {(ppm)}\) relative to solvent’s signals as internal standards and coupling constants \((J)\) are given in Hz. Multiplicities are indicated as the following: s, singlet; d, doublet; t, triplet; q, quartet; m, multiplet; dd, doubled doublet. Preparative layer chromatography was done using silica gel (Merk Kieselgel 60 HF254, no. 7739).
General procedure for the synthesis of aminonaphthols
To a mixture of 2-naphthol (1 mmol), aldehyde (1 mmol) and amine (1.2 mmol) was added DMEA (7.5 mol%). The mixture was stirred at 50 \(^{\circ }\hbox {C}\) under solvent-free conditions in an oil bath for an appropriate time (Tables 3, 4), and the reaction was monitored by TLC (acetone/chloroform/n-hexane: 1/2/5). After completion of the reaction, the mixture was cooled to room temperature, ethanol (10 mL) was added, and the mixture was stirred for 10 min. The resulting solid was collected by filtration. Crude products (14l, 14m, 14q, 14r, and 14t) were purified by preparative layer chromatography on silica gel using (EtOAc / \(n\)-hexane, 1:5). Compounds (14a-k, 14n, 14o, 14p, 14s, and 16a-j) were purified by recrystallization (EtOH/acetone, 4:1). All compounds were characterized by melting point (mp), IR, \(^{1}\hbox {H NMR},\,^{13}\hbox {C NMR}\) and elemental (C, H, N) analysis (Fig. 5).
1-[Phenyl(phenylamino)methyl]naphthalen-2-ol (14a)
White solid; Yield 90 %; mp: 132–133 \(^{\circ }\hbox {C};^{1}\hbox {H NMR}\) (400 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 4.15 (1H, bs, N–H, disappeared in the presence of \(\hbox {D}_{2}\hbox {O}\)), 6.17 (1H, s, methine-H), 6.76 (2H, d, \(J = 7.6\, \hbox {Hz}\), Ar–H), 6.92 (1H, t, \(J = 7.3\, \hbox {Hz}\), Ar–H), 7.13–7.49 (10H, m, Ar–H), 7.76–7.82 (3H, m, Ar–H), 11.54 (1H, bs, OH, disappeared in the presence of \(\hbox {D}_{2}\hbox {O}\)) ppm; \(^{13}\hbox {C NMR}\) (100 MHz, \(\hbox {CDCl}_{3}\)): \(\updelta \) 61.7, 115.2, 118.9, 120.4, 120.8, 121.8, 125.5, 125.8, 126.9, 127.0, 127.6, 127.9, 128.2, 128.3, 128.4, 128.9, 139.9, 145.6, 156.7 ppm.
1-[(2-Chlorophenyl)(phenylamino)methyl]naphthalen-2-ol (14b)
Colorless crystals; Yield 90 %; mp: 150–152 \(^{\circ }\hbox {C}\); FT-IR (KBr) \(\nu \) 3418, 3301, 3059, 2906, 1604, 1490, 1229, 754 \(\hbox {cm}^{-1};^{1}\hbox {H NMR}\) (400 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 4.07 (1H, bs, N–H), 6.54 (1H, s, methine-H), 6.80 (2H, d, \(J = 8\, \hbox {Hz}\), Ar–H), 6.93 (1H, t, \(J = 7.3\, \hbox {Hz}\), Ar–H), 7.08–7.18 (5H, m, Ar–H), 7.22–7.38 (3H, m, Ar–H), 7.49 (1H, d, \(J\) = 8 Hz, Ar–H), 7.58 (1H, d, \(J = 8.4\, \hbox {Hz}\), Ar–H), 7.77–7.79 (2H, m, Ar–H), 11.64 (1H, bs, OH) ppm; \(^{13}\hbox {C NMR}\) (100 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 58.4, 111.7, 115.5, 118.9, 120.3, 121.1, 121.9, 126.0, 126.7, 127.9, 128.0, 128.3, 129.0, 129.1, 129.2, 129.3, 130.4, 132.8, 136.4, 145.9, 156.0 ppm. Anal. Calcd. For \(\hbox {C}_{23}\hbox {H}_{18}\hbox {ClNO}\): C, 76.77; H, 5.04; N, 3.89; Found: C, 76.51; H, 5.12; N, 3.75 %.
1-[(3-Chlorophenyl)(phenylamino)methyl]naphthalen-2-ol (14c)
Colorless crystals; Yield 90 %; mp: 138–140 \(^{\circ }\hbox {C}\); FT-IR (KBr) \(\nu \) 3378, 3337, 3054, 2902, 1601, 1496, 1231, 754 \(\hbox {cm}^{-1};^{1}\hbox {H NMR}\) (400 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 4.12 (1H, bs, N–H), 6.12 (1H, s, methine-H), 6.74 (2H, d, \(J = 8\, \hbox {Hz}\), Ar–H), 6.92 (1H, t, \(J = 7.3\, \hbox {Hz}\), Ar–H), 7.12–7.20 (4H, m, Ar–H), 7.31 (1H, t, \(J = 7.3\, \hbox {Hz}\), Ar–H), 7.37–7.43 (3H, m, Ar–H), 7.64 (1H, s, Ar–H), 7.73–7.79 (3H, m, Ar–H), 11.36 (1H, bs, OH) ppm; \(^{13}\hbox {C NMR}\) (100 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 60.9, 112.1, 115.2, 119.0, 120.1, 120.9, 121.9, 122.2, 125.6, 125.9, 128.1, 128.4, 129.3, 129.9, 130.0, 130.3, 130.7, 142.1, 145.3, 155.1 ppm. Anal. Calcd. For \(\hbox {C}_{23}\hbox {H}_{18}\hbox {ClNO}\): C, 76.77; H, 5.04; N, 3.89; Found: C, 76.48; H, 5.14; N, 3.78 %.
1-[(4-Chlorophenyl)(phenylamino)methyl]naphthalen-2-ol (14d)
Colorless crystals; Yield 92 %; mp: 121–123 \(^{\circ }\hbox {C}\); FT-IR (KBr) \(\nu \) 3412, 3348, 3060, 2962, 1623, 1599, 1491, 1220, 753 \(\hbox {cm}^{-1};^{1}\hbox {H NMR}\) (400 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 4.04 (1H, bs, N–H), 6.11 (1H, s, methine-H), 6.75 (1H, d, \(J = 8\, \hbox {Hz}\), Ar–H), 6.85–6.92 (1H, m, Ar–H), 7.05 (1H, t, \(J = 8\, \hbox {Hz}\), Ar–H), 7.13–7.21 (3H, m, Ar–H), 7.29–7.46 (7H, m, Ar–H), 7.72–7.83 (2H, m, Ar–H), 11.41 (1H, bs, OH) ppm; \(^{13}\hbox {C NMR}\) (100 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 60.9, 111.7, 115.2, 118.0, 121.8, 122.7, 123.2, 124.6, 125.8, 127.2, 127.6, 128.4, 128.5, 129.6, 132.5, 140.1, 145.5, 151.6, 155.0 ppm. Anal. Calcd. For \(\hbox {C}_{23}\hbox {H}_{18}\hbox {ClNO}\): C, 76.77; H, 5.04; N, 3.89; Found: C, 76.54; H, 5.09; N, 3.81 %.
1-[(Phenylamino)(\(p\)-tolyl)methyl]naphthalen-2-ol (14e)
Colorless crystals; Yield 92 %; mp: 130–132 \(^{\circ }\hbox {C}\); FT-IR (KBr) \(\nu \) 3419, 3342, 3052, 2966, 1602, 1497, 1230 \(\hbox {cm}^{-1};^{1}\hbox {H NMR}\) (400 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 2.29 (3H, s, \(\hbox {--CH}_{3}\)), 4.17 (1H, bs, N–H), 6.12 (1H, s, methine-H), 6.73 (2H, d, \(J = 8\, \hbox {Hz}\), Ar–H), 6.89 (1H, t, \(J = 7.2\, \hbox {Hz}\), Ar–H), 7.11–7.13 (5H, m, Ar–H), 7.25–7.38 (4H, m, Ar–H), 7.71–7.78 (3H, m, Ar–H), 11.61 (1H, bs, OH) ppm; \(^{13}\hbox {C NMR}\) (100 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 20.1, 61.4, 112.9, 115.2, 118.9, 120.4, 120.7, 121.7, 125.7, 126.8, 127.9, 128.0, 128.4, 128.8, 128.9, 130.4, 137.0, 137.3, 145.7, 155.0 ppm. Anal. Calcd. For \(\hbox {C}_{24}\hbox {H}_{21}\hbox {NO}\): C, 84.92; H, 6.24; N, 4.13; Found: C, 84.64; H, 6.31; N, 4.06 %.
1-[(3,4-Dimethoxyphenyl)(phenylamino)methyl]naphthalen-2-ol (14f)
Light brown solid; Yield 95 %; mp: 139–141 \(^{\circ }\hbox {C}\); FT-IR (KBr) \(\nu \) 3398, 3318, 3016, 2959, 1598, 1510, 1227, 1027 \(\hbox {cm}^{-1};^{1}\hbox {H NMR}\) (400 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 3.79 (3H, s, \(\hbox {--OCH}_{3}\)), 3.81 (3H, s, \(\hbox {--OCH}_{3}\)), 4.19 (1H, bs, N–H), 6.11 (1H, s, methine-H), 6.75–6.79 (3H, m, Ar–H), 6.90 (1H, t, \(J = 7.3\, \hbox {Hz}\), Ar–H), 6.98–7.00 (2H, m, Ar–H), 7.10-7.16 (3H, m, Ar–H), 7.29 (1H, t, \(J = 7.3\, \hbox {Hz}\), Ar–H), 7.39 (1H, m, Ar–H), 7.72–7.81 (3H, m, Ar–H), 11.55 (1H, bs, OH) ppm; \(^{13}\hbox {C NMR}\) (100 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 54.8, 61.5, 109.7, 110.4, 113.0, 115.2, 118.9, 119.3, 120.4, 120.7, 121.8, 125.7, 127.9, 128.1, 128.4, 128.9, 130.4, 132.5, 145.7, 148.1, 148.5, 155.0 ppm. Anal. Calcd. For \(\hbox {C}_{25}\hbox {H}_{23}\hbox {NO}_{3}\): C, 77.90; H, 6.01; N, 3.63; Found: C, 77.63; H, 6.07; N, 3.58 %.
1- [(2-Bromophenyl)(phenylamino)methyl]naphthalen-2-ol (14g)
Light brown crystals; Yield 81 %; mp: 150–152 \(^{\circ }\hbox {C}\); FT-IR (KBr) \(\nu \) 3425, 3344, 3057, 3018, 2960, 1621, 1601, 1494, 1224, 694 \(\hbox {cm}^{-1};^{1}\hbox {H NMR}\) (400 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 4.07 (1H, bs, N–H), 6.49 (1H, s, methine-H), 6.79 (2H, d,\( J\) = 8.0 Hz, Ar–H), 6.91 (1H, t, \(J = 8.0\, \hbox {Hz}\), Ar–H), 7.12–7.17 (6H, m, Ar–H), 7.27–7.38 (2H, m, Ar–H), 7.57 (1H, d, \(J = 8.4\, \hbox {Hz}\), Ar–H), 7.66–7.69 (1H, m, Ar–H), 7.78 (2H, d, \(J=8.8\, \hbox {Hz}\), Ar–H), 11.62 (1H, bs, OH) ppm; \(^{13}\hbox {C NMR}\) (100 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 60.8, 111.8, 115.4, 118.9, 120.4, 121.0, 121.9, 123.5, 126.0, 127.3, 127.9, 128.0, 128.3, 129.3, 129.4, 130.4, 132.4, 138.0, 145.9, 155.9 ppm. Anal. Calcd. For \(\hbox {C}_{23}\hbox {H}_{18}\hbox {BrNO}\): C, 68.33; H, 4.49; N, 3.46; Found: C, 68.06; H, 4.54; N, 3.51 %.
6-Bromo-1-[(2-bromophenyl)(phenylamino)methyl]naphthalen-2-ol (14h)
Pale yellow solid; Yield 85 %; mp: 163–165 \(^{\circ }\hbox {C}\); FT-IR (KBr) \(\nu \) 3391, 3343, 3053, 2950, 1649, 1597, 1494, 1027, 755 \(\hbox {cm}^{-1};^{1}\hbox {H NMR}\) (400 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 4.06 (1H, bs, N–H), 6.42 (1H, s, methine-H), 6.79 (2H, d, \(J = 8.0\, \hbox {Hz}\), Ar–H), 6.95 (1H, t, \(J = 8.0\, \hbox {Hz}\), Ar–H), 7.12–7.22 (6H, m, Ar–H), 7.43 (2H, m, Ar–H), 7.68–7.71 (2H, m, Ar–H), 7.94 (1H, s, Ar–H), 11.66 (1H, bs, OH) ppm; \(^{13}\hbox {C NMR}\) (100 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 60.9, 112.3, 115.5, 115.7, 120.1, 121.3, 122.2, 123.4, 127.4, 128.4, 129.0, 129.23, 129.27, 129.4, 129.5, 129.8, 132.6, 137.8, 145.8, 156.3 ppm. Anal. Calcd. For \(\hbox {C}_{23}\hbox {H}_{17}\hbox {Br}_{2}\hbox {NO}\): C, 57.17; H, 3.55; N, 2.90; Found: C, 56.91; H, 3.61; N, 2.94 %.
1-[(4-Methoxyphenyl)(phenylamino)methyl]naphthalen-2-ol (14i)
Light brown solid; Yield 92 %; mp: 270 \(^{\circ }\hbox {C}\) (dec.); FT-IR (KBr) \(\nu \) 3412, 3337, 3008, 2951, 1619, 1508, 1241, 821 \(\hbox {cm}^{-1};^{1}\hbox {H NMR}\) (400 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 3.68 (3H, s, \(\hbox {--OCH}_{3}\)), 4.12 (1H, bs, N–H), 6.10 (1H, s, methine-H), 6.71 (2H, d, \(J = 8.2\, \hbox {Hz}\), Ar–H), 6.80 (2H, d, \(J = 8.5\, \hbox {Hz}\), Ar–H), 6.88 (1H, t, \(J =7.3\, \hbox {Hz}\), Ar–H), 7.08–7.14 (3H, m, Ar–H), 7.24–7.37 (4H, m, Ar–H), 7.69–7.77 (3H, m, Ar–H), 11.54 (1H, bs, OH) ppm; \(^{13}\hbox {C NMR}\) (100 MHz, \(\hbox {CDCl}_{3})\): \(\delta \) 54.2, 61.0, 113.1, 113.5, 115.2, 118.9, 120.4, 120.7, 121.7, 125.7, 127.9, 128.0, 128.2, 128.3, 128.8, 130.4, 132.1, 145.7, 154.9, 158.5 ppm. Anal. Calcd. For \(\hbox {C}_{24}\hbox {H}_{21}\hbox {NO}_{2}\): C, 81.10; H, 5.96; N, 3.94; Found: C, 80.87; H, 5.98; N, 3.90 %.
1-[(3-Chlorophenylamino)(phenyl)methyl]naphthalen-2-ol (14j)
Colorless crystals; Yield 90 %; mp: 112–114 \(^{\circ }\hbox {C}\); FT-IR (KBr) \(\nu \) 3375, 3337, 3062, 2925, 1595, 1476, 752 \(\hbox {cm}^{-1};^{ 1}\hbox {H NMR}\) (400 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 4.13 (1H, bs, N–H), 6.16 (1H, s, methine-H), 6.62 (1H, dd, \(J = 8.0\, \hbox {Hz},\,J=1.2 \hbox {Hz}\), Ar–H), 6.78 (1H, s, Ar–H), 6.88 (1H, d, \(J = 8.0\, \hbox {Hz}\), Ar–H), 7.05 (1H, t, \(J=8.0\, \hbox {Hz}\), Ar–H), 7.14 (1H, d, \(J=8.8\, \hbox {Hz}\), Ar–H), 7.29–7.41 (5H, m, Ar–H), 7.45 (2H, d, \(J=7.0\, \hbox {Hz}\), Ar–H), 7.75–7.80 (3H, m, Ar–H), 10.88 (1H, bs, OH) ppm; \(^{13}\hbox {C NMR}\) (100 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 61.4, 112.3, 113.0, 115.6, 118.9, 120.4, 120.8, 122.0, 125.9, 126.9, 127.7, 128.1, 128.2, 128.5, 129.2, 129.6, 130.4, 134.0, 139.6, 146.9, 154.8 ppm. Anal. Calcd. For \(\hbox {C}_{23}\hbox {H}_{18}\hbox {ClNO}\): C, 76.77; H, 5.04; N, 3.89; Found: C 76.52; H, 5.11; N, 3.85 %.
1-[(4-Methoxyphenylamino)(phenyl)methyl]naphthalen-2-ol (14k)
Light Brown solid; Yield 80 %; mp: 117–119 \(^{\circ }\hbox {C}\); FT-IR (KBr) \(\nu \) 3397, 3327, 3025, 2927, 1621, 1509, 1235, 821 \(\hbox {cm}^{-1};^{1}\hbox {H NMR}\) (400 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 3.70 (3H, s, \(\hbox {OCH}_{3}\)), 4.23 (1H, bs, N–H), 6.11 (1H, s, methine-H), 6.70–6.76 (4H, m, Ar–H), 7.15 (1H, d, \(J=8.8 \hbox {Hz}\), Ar–H), 7.28–7.39 (5H, m, Ar–H), 7.48 (2H, d, \(J=7.2\, \hbox {Hz}\), Ar–H), 7.73–7.78 (3H, m, Ar–H), 11.98 (1H, bs, OH) ppm; \(^{13}\hbox {C NMR}\) (100 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 54.5, 62.5, 112.7, 113.8, 116.8, 119.1, 120.3, 121.7, 125.7, 126.9, 127.5, 127.9, 128.0, 128.4, 128.9, 130.5, 139.1, 140.1, 153.9, 155.3 ppm. Anal. Calcd. For \(\hbox {C}_{24}\hbox {H}_{21}\hbox {NO}_{2}\): C, 81.10; H, 5.96; N, 3.94; Found: C, 80.91; H, 5.92; N, 3.89 %.
1-[(4-Nitrophenylamino)(phenyl)methyl]naphthalen-2-ol (14l)
Yellow solid; Yield 95 %; mp: 154–156 \(^{\circ }\hbox {C}\); FT-IR (KBr) \(\nu \) 3396, 3354, 3024, 2955, 1597, 1509, 1308, 1112, 768 \(\hbox {cm}^{-1};^{1}\hbox {H NMR}\) (400 MHz, \(\hbox {CDCl}_{3})\): \(\delta \) 5.07 (1H, bs, N–H, disappeared in the presence of \(\hbox {D}_{2}\hbox {O}\)), 6.41 (1H, s, methine-H), 6.71 (2H, d, \(J=8.8\, \hbox {Hz}\), Ar–H), 7.10 (1H, d, \(J = 8.8\, \hbox {Hz}\), Ar–H), 7.29–7.37 (4H, m, Ar–H), 7.43–7.46 (3H, m, Ar–H), 7.76–7.86 (3H, m, Ar–H), 8.02 (2H, d, \(J=8.8\, \hbox {Hz}\), Ar–H), 11.67 (1H, bs, OH, disappeared in the presence of D\(_{2}\)O) ppm; \(^{13}\hbox {C NMR}\) (100 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 58.1, 113.1, 113.7, 118.4, 120.6, 122.5, 125.0, 126.3, 126.5, 127.5, 128.2, 128.4, 128.5, 129.6, 130.6, 138.9, 139.7, 151.4, 153.1 ppm. Anal. Calcd. For \(\hbox {C}_{23}\hbox {H}_{18}\hbox {N}_{2}\hbox {O}_{3}\): C, 74.58; H, 4.90; N, 7.56; Found: C, 74.32; H, 4.95; N, 7.49 %.
1-[(4-Fluorophenylamino)(phenyl)methyl]naphthalen-2-ol (14m)
White solid; Yield 90 %; mp: 110–112 \(^{\circ }\hbox {C}\); FT-IR (KBr) \(\nu \) 3397, 3339, 3023, 2962, 1602, 1503, 1229, 815, 752 \(\hbox {cm}^{-1}\); \(^{1}\hbox {H NMR}\) (400 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 4.14 (1H, bs, N–H), 6.11 (1H, s, methine-H), 6.69–6.72 (2H, m, Ar–H), 6.80–6.85 (2H, m, Ar–H), 7.14 (1H, d, \(J = 8.8\, \hbox {Hz}\), Ar–H), 7.27–7.39 (5H, m, Ar–H), 7.46–7.48 (2H, m, Ar–H), 7.73–7.78 (3H, m, Ar–H), 11.68 (1H, bs, OH) ppm; \(^{13}\hbox {C NMR}\) (100 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 61.5, 112.9, 114.9, 115.3, 118.9, 120.4, 120.8, 121.8, 125.7, 126.9, 127.2, 127.4, 127.9, 128.1, 128.4, 128.9, 129.0, 130.5, 137.1, 137.4, 145.7, 155.1, 157.1, 159.6 ppm. Anal. Calcd. For \(\hbox {C}_{23}\hbox {H}_{18}\hbox {FNO}\): C, 80.45; H, 5.28; N, 4.08; Found: C, 80.24; H, 5.26; N, 4.05 %.
1-[(3,4-Dichlorophenylamino)(phenyl)methyl]naphthalen-2-ol (14n)
Brown solid; Yield 90 %; mp: 123–125 \(^{\circ }\hbox {C}\); FT-IR (KBr) \(\nu \) 3402, 3352, 3061, 2953, 1596, 1474, 1024, 749 \(\hbox {cm}^{-1};^{ 1}\hbox {H NMR}\) (400 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 4.27 (1H, bs, N–H), 6.13 (1H, s, methine-H), 6.52 (1H, dd, \(J = 8.8\, \hbox {Hz},\, J=2.4\, \hbox {Hz}\), Ar–H), 6.82 (1H, d, \(J = 2.4\, \hbox {Hz}\), Ar–H), 7.09–7.13 (2H, m, Ar–H), 7.29–7.33 (4H, m, Ar–H), 7.35–7.43 (3H, m, Ar–H), 7.72–7.77 (3H, m, Ar–H), 10.54 (1H, bs, OH) ppm; \(^{13}\hbox {C NMR}\) (100 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 60.9, 114.2, 116.9, 118.7, 120.3, 122.1, 122.4, 126.0, 126.7, 126.8, 127.7, 128.1, 128.2, 128.4, 129.3, 129.9, 130.4, 131.9, 139.3, 145.2, 154.4 ppm. Anal. Calcd. For \(\hbox {C}_{23}\hbox {H}_{17}\hbox {Cl}_{2}\hbox {NO}\): C, 70.06; H, 4.35; N, 3.55; Found: C, 69.85; H, 4.38; N, 3.53 %.
1-[(4-Chlorophenyl)(3-chlorophenylamino)methyl]naphthalen-2-ol (14o)
Brown solid; Yield 92 %; mp: 121–123 \(^{\circ }\hbox {C}\); FT-IR (KBr) \(\nu \) 3398, 3345, 3063, 2964, 1597, 1484, 750 \(\hbox {cm}^{-1};^{1}\hbox {H NMR}\) (400 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 4.25 (1H, bs, N–H), 6.12 (1H, s, methine-H), 6.56 (1H, d, \(J = 8.0\, \hbox {Hz}\), Ar–H), 6.72 (1H, s, Ar–H), 6.84 (1H, d, \(J=8.0\, \hbox {Hz}\), Ar–H), 6.97 (1H, t, \(J=8.0\, \hbox {Hz}\), Ar–H), 7.10 (1H, d, \(J=8.8\, \hbox {Hz}\), Ar–H), 7.24 (2H, d, \(J=8.4\, \hbox {Hz}\), Ar–H), 7.28–7.40 (4H, m, Ar–H), 7.71 (2H, d, \(J=8.4\, \hbox {Hz}\), Ar–H), 7.75 (1H, d, \(J\) = 8.0 Hz, Ar–H), 10.69 (1H, bs, OH) ppm; \(^{13}\hbox {C NMR}\) (100 MHz, \(\hbox {CDCl}_{3})\): \(\delta \) 65.9, 112.8, 115.3, 118.7, 120.2, 120.7, 122.1, 122.3, 126.0, 128.0, 128.1, 128.2, 128.4, 128.6, 129.4, 129.5, 130.2, 133.3, 133.9, 137.9, 146.6, 154.4 ppm. Anal. Calcd. For \(\hbox {C}_{23}\hbox {H}_{17}\hbox {Cl}_{2}\hbox {NO}\): C, 70.06; H, 4.35; N, 3.55; Found: C, 69.81; H, 4.39; N, 3.53 %.
1-[(4-Methoxyphenylamino)(\(p\)-tolyl)methyl]naphthalen-2-ol (14p)
Brown solid; Yield 95 %; mp: 114–116 \(^{\circ }\hbox {C}\); FT-IR (KBr) \(\nu \) 3400, 3338, 3012, 2958, 1604, 1508, 1251, 753 \(\hbox {cm}^{-1};^{1}\hbox {H NMR}\) (400 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 2.40 (3H, s, \(\hbox {--CH}_{3}\)), 3.82 (3H, s, \(\hbox {--OCH}_{3}\)), 4.10 (1H, bs, N–H), 6.07 (1H, s, methine-H), 6.69–6.75 (2H, m, Ar–H), 6.92 (2H, d, \(J=8.8\, \hbox {Hz}\), Ar–H), 7.14 (2H, d, \(J = 8.0\, \hbox {Hz}\), Ar–H), 7.24–7.27 (3H, m, Ar–H), 7.35 (2H, d, \(J\) = 8.0 Hz, Ar–H), 7.74–7.79 (3H, m, Ar–H), 11.67 (1H, bs, OH) ppm; \(^{13}\hbox {C NMR}\) (100 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 19.6, 64.3, 83.4, 112.3, 115.3, 118.1, 122.1, 122.4, 124.3, 125.6, 126.9, 127.3, 128.1, 128.2, 128.4, 131.3, 132.1, 136.4, 141.8, 143.5, 151.9 ppm. Anal. Calcd. For \(\hbox {C}_{25}\hbox {H}_{23}\hbox {NO}_{2}\): C, 81.27; H, 6.27; N, 3.79; Found: C, 81.09; H, 6.29; N, 3.75 %.
1-[(4-Chlorophenyl)(4-fluorophenylamino)methyl]naphthalen-2-ol (14q)
Colorless crystals; Yield 92 %; mp: 163–165 \(^{\circ }\hbox {C}\); FT-IR (KBr) \(\nu \) 3431, 3354, 3060, 2960, 1623, 1505, 1220, 815, 771 \(\hbox {cm}^{-1};^{1}\hbox {H NMR}\) (400 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 4.11 (1H, bs, N–H), 6.07 (1H, s, methine-H), 6.73 (2H, t, \(J=8.4\, \hbox {Hz}\), Ar–H), 7.04–7.08 (2H, m, Ar–H), 7.21 (2H, d, \(J=8.4\, \hbox {Hz}\), Ar–H), 7.31–7.35 (3H, m, Ar–H), 7.41 (2H, d, \(J=8.4\, \hbox {Hz}\), Ar–H), 7.88 (3H, d, \(J=8.8\, \hbox {Hz}\), Ar–H), 11.51 (1H, bs, OH) ppm; \(^{13}\hbox {C NMR}\) (100 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 63.3, 111.5, 114.2, 114.5, 117.9, 121.8, 122.8, 125.9, 126.7, 127.1, 127.2, 127.7, 128.4, 128.6, 128.9, 129.5, 132.6, 132.7, 134.4, 140.0, 151.6, 155.0, 157.4, 159.9 ppm. Anal. Calcd. For \(\hbox {C}_{23}\hbox {H}_{17}\hbox {ClFNO}\): C, 73.11; H, 4.54; N, 3.71; Found: C, 72.93; H, 4.57; N, 3.70 %.
1-[(6-Methyl-2-pyridinylamino)(4-nitrophenyl)methyl]naphthalen-2-ol (14r)
Pale yellow solid; Yield 93 %; mp: 160–162 \(^{\circ }\hbox {C}\); FT-IR (KBr) \(\nu \) 3441, 3333, 3064, 2962, 1576, 1514, 1340, 1261, 1098, 802 \(\hbox {cm}^{-1};\, ^{1}\hbox {H NMR}\) (400 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 2.43 (3H, s, \(\hbox {CH}_{3}\)), 5.97 (1H, s, NH, disappeared in the presence of \(\hbox {D}_{2}\hbox {O}\)), 6.46 (1H, d, \(J=8.4\, \hbox {Hz}\), Ar–H), 6.51 (1H, d, \(J=7.2\, \hbox {Hz}\), Ar–H), 7.04 (1H, s, methine-H), 7.22–7.26 (3H, m, Ar–H), 7.34–7.40 (2H, m, Ar–H), 7.54 (2H, d, \( J\) = 8.4 Hz, Ar–H), 7.70 (1H, d, \(J = 8.4\, \hbox {Hz}\), Ar–H), 7.75–7.77 (1H, m, Ar–H), 8.09 (2H, d, \(J\) = 8.4 Hz, Ar–H) ppm; \(^{13}\hbox {C NMR}\) (100 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 28.7, 50.4, 105.6, 112.5, 118.1, 119.5, 121.8, 121.9, 122.6, 125.4, 126.2, 128.3, 128.8, 129.3, 130.5, 138.0, 145.7, 148.9, 153.9, 154.7, 156.0 ppm; Anal. Calcd. For \(\hbox {C}_{23}\hbox {H}_{19}\hbox {N}_{3}\hbox {O}_{3}\): C, 71.67; H, 4.97; N, 10.90; Found: C, 71.48; H, 4.99; N, 10.87 %.
1-[2-Furyl(phenylamino)methyl]naphthalen-2-ol (14s)
White solid; Yield 92 %; mp: 161–163 \(^{\circ }\hbox {C}\); FT-IR (KBr) \(\nu \) 3412, 3364, 3010, 2949, 1509, 1029, 760 \(\hbox {cm}^{-1};\, ^{1}\hbox {H}\) NMR (400 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 4.05 (1H, bs, N–H), 6.81 (1H, s, methine-H), 6.91–6.94 (1H, m, Ar–H), 7.08 (2H, d, \(J=8.4\, \hbox {Hz}\), Ar–H), 7.20 (1H, m, Ar–H), 7.31–7.42 (4H, m, Ar–H), 7.73 (2H, d, \(J=8.4\, \hbox {Hz}\), Ar–H), 7.80 (2H, d, \(J=8.0 \hbox {Hz}\), Ar–H), 7.91 (2H, d, \(J=8.4\, \hbox {Hz}\), Ar–H), 11.08 (1H, bs, OH) ppm; \(^{13}\)C NMR (100 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 58.3, 112.9, 115.4, 119.0, 121.2, 122.4, 125.7, 126.2, 126.4, 127.9, 128.1, 128.6, 129.1, 130.3, 134.7, 139.3, 147.5, 152.1 ppm. Anal. Calcd. For \(\hbox {C}_{21}\hbox {H}_{17}\hbox {NOS}\): C, 76.10; H, 5.17; N, 4.23; Found: C, 75.88; H, 5.16; N, 4.18 %.
1-[(5-(Benzyloxy)-4-oxo-4\(H\)-pyran-2-yl)(phenylamino)methyl]naphthalen-2-ol (14t)
Pale yellow solid; Yield 95 %; mp 123–125 \(^{\circ }\hbox {C}\); FT-IR (KBr) \(\nu \) 3391, 3324, 3061, 2925, 1637, 1601, 1506, 1209, 751 \(\hbox {cm}^{-1}\); \(^{1}\hbox {H NMR}\) (400 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 4.90 (1H, bs, N–H), 5.03 (2H, s, benzylic-H), 6.16 (1H, s, methine-H), 6.48 (1H, s, pyrone-H), 6.74 (2H, d,\( J\) = 8.0 Hz, Ar–H), 6.88 (1H, t, \(J=7.2\, \hbox {Hz}\), Ar–H), 7.14–7.18 (3H, m, Ar–H), 7.33–7.38 (6H, m, Ar–H), 7.50 (1H, t, \(J=8.0\, \hbox {Hz}\), Ar–H), 7.58 (1H, s, pyrone-H), 7.72 (1H, d, \(J=8.8\, \hbox {Hz}\), Ar–H), 7.80 (1H, d, \(J=8.0\, \hbox {Hz}\), Ar–H), 7.86 (1H, d, \(J=8.8\, \hbox {Hz}\), Ar–H), 10.24 (1H, bs, OH) ppm; \(^{13}\hbox {C NMR}\) (100 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 55.4, 70.8, 112.9, 114.4, 118.6, 119.8, 120.1, 122.2, 126.5, 126.6, 127.5, 127.8, 128.0, 128.3, 128.5, 130.1, 130.7, 134.4, 136.2, 140.2, 144.7, 146.2, 154.5, 174.1 ppm. Anal. Calcd. For \(\hbox {C}_{29}\hbox {H}_{23}\hbox {NO}_{4}\): C, 77.49; H, 5.16; N, 3.12; Found: C, 77.28; H, 5.20; N, 3.10 %.
1-(Phenyl(pipyridin-1-yl)methyl)naphthalen-2-ol (16a)
White solid; Yield 87 %; mp: 192–194 \(^{\circ }\hbox {C}\); \(^{1}\hbox {H NMR}\) (400 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 1.21–1.65 (6H, m, \(\hbox {CH}_{2}\)), 1.89–2.07 (2H, m, –NCH), 2.65 (1H, m, –NCH), 3.29 (1H, m, –NCH), 5.06 (1H, s, methine-H), 7.14–7.25 (5H, m, Ar–H), 7.34 (1H, t,\( J\) = 7.6 Hz, Ar–H), 7.54 (2H, m, Ar–H), 7.64 (1H, d, \(J=8.8\, \hbox {Hz}\), Ar–H), 7.67 (1H, d, \(J=8.8\, \hbox {Hz}\), Ar–H), 7.82 (1H, d, \(J=8.4\, \hbox {Hz}\), Ar–H), 14.06 (1H, bs, OH) ppm; \(^{13}\hbox {C NMR}\) (100 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 23.1, 25.0, 50.8, 53.7, 71.0, 115.1, 118.9, 119.9, 121.2, 122.1, 125.3, 126.6, 126.8, 127.5, 127.8, 128.1, 128.3, 131.3, 138.6, 154.4 ppm; Anal. Calcd. For \(\hbox {C}_{22}\hbox {H}_{23}\hbox {NO}\): C, 83.24; H, 7.30; N, 4.41; Found: C, 83.08; H, 7.35; N, 4.36 %.
1-[(2-Chlorophenyl)(pipyridin-1-yl)methyl]naphthalen-2-ol (16b)
White solid; Yield 87 %; mp: 150–152 \(^{\circ }\hbox {C}\); FT-IR (KBr) \(\nu \) 3415, 3059, 3011, 2935, 1622, 1470, 1034, 752 \(\hbox {cm}^{-1}\); \(^{1}\hbox {H NMR}\) (400 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 1.21–1.76 (6H, m, \(\hbox {CH}_{2}\)), 2.26–2.36 (2H, m, –NCH), 2.62 (1H, m, –NCH), 3.32 (1H, m, –NCH), 5.83 (1H, s, methine-H), 7.09–7.16 (3H, m, Ar–H), 7.19–7.23 (1H, m, Ar–H), 7.34–7.38 (2H, m, Ar–H), 7.59–7.61 (1H, m, Ar–H), 7.66–7.69 (2H, m, Ar–H), 7.78 (1H, d, \(J = 8.4\, \hbox {Hz}\), Ar–H), 14.30 (1H, bs, OH) ppm; \(^{13}\hbox {C NMR}\) (100 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 22.9, 24.9, 25.3, 48.3, 53.8, 65.2, 114.8, 119.1, 120.2, 121.5, 125.6, 127.0, 127.4, 127.7, 128.1, 128.4, 128.5, 129.9, 131.7, 133.2, 135.8, 155.5 ppm; Anal. Calcd. For \(\hbox {C}_{22}\hbox {H}_{22}\hbox {ClNO}\): C, 75.09; H, 6.30; N, 3.98; Found: C, 74.86; H, 6.32; N, 3.93 %.
1-[(4-Nitrophenyl)(pipyridin-1-yl)methyl]naphthalen-2-ol (16c)
Yellow solid; Yield 88 %; mp: 177–179 \(^{\circ }\hbox {C}\); FT-IR (KBr) \(\nu \) 3387, 3068, 3017, 2937, 1622, 1529, 1349, 753 \(\hbox {cm}^{-1}\); \(^{1}\hbox {H NMR}\) (400 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 1.23–1.69 (6H, m, \(\hbox {CH}_{2}\)), 2.00–2.16 (2H, m, –NCH), 2.61 (1H, m, –NCH), 3.33 (1H, m, –NCH), 5.19 (1H, s, methine-H), 7.15 (1H, d, \(J\) = 8.8 Hz, Ar–H), 7.21–7.25 (1H, m, Ar–H), 7.38 (1H, t, \(J = 8.0\, \hbox {Hz}\), Ar–H), 7.66–7.78 (5H, m, Ar–H), 8.09 (2H, d, \(J = 8.0 \hbox {Hz}\), Ar–H), 13.57 (1H, bs, OH) ppm; \(^{13}\hbox {C NMR}\) (100 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 22.9, 24.9, 51.4, 53.4, 70.1, 113.9, 119.0, 119.4, 121.7, 123.0, 125.7, 127.7, 128.1, 128.7, 129.1, 130.9, 146.2, 146.4, 154.4 ppm; Anal. Calcd. For \(\hbox {C}_{22}\hbox {H}_{22}\hbox {N}_{2}\hbox {O}_{3}\): C, 72.91; H, 6.12; N, 7.73; Found: C, 72.68; H, 6.15; N, 7.66 %.
1-[(3-Nitrophenyl)(pipyridin-1-yl)methyl]naphthalen-2-ol (16d)
Yellow solid; Yield 85 %; mp: 169–171 \(^{\circ }\hbox {C}\); FT-IR (KBr) \(\nu \) 3362, 3061, 3013, 2937, 1619, 1525, 1349, 767 cm\(^{-1}\); \(^{1}\hbox {H NMR}\) (400 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 1.22–1.69 (6H, m, \(\hbox {CH}_{2}\)), 2.13 (2H, m, –NCH), 2.62 (1H, m, –NCH), 3.66 (1H, m, –NCH), 5.19 (1H, s, methine-H), 7.16 (1H, d, \(J=8.8 \hbox {Hz}\), Ar–H), 7.23 (1H, t, \(J=7.2\, \hbox {Hz}\), Ar–H), 7.38 (1H, d, \(J = 7.2\, \hbox {Hz}\), Ar–H), 7.42 (1H, d, \(J = 7.2\, \hbox {Hz}\), Ar–H), 7.69 (2H, t, \(J=8.8\, \hbox {Hz}\), Ar–H), 7.78 (1H, d, \(J = 8.8\, \hbox {Hz}\), Ar–H), 7.93 (1H, m, Ar–H), 8.04 (1H, d, \(J=8.0\, \hbox {Hz}\), Ar–H), 8.40 (1H, s, Ar–H), 13.57 (1H, bs, OH) ppm; \(^{13}\hbox {C NMR}\) (100 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 22.9, 24.9, 51.3, 53.6, 70.1, 114.0, 119.1, 119.4, 121.7, 122.0, 122.9, 125.8, 127.7, 128.1, 129.0, 130.9, 133.9, 141.0, 154.5 ppm; Anal. Calcd. For \(\hbox {C}_{22}\hbox {H}_{22}\hbox {N}_{2}\hbox {O}_{3}\): C, 72.91; H, 6.12; N, 7.73; Found: C, 72.73; H, 6.16; N, 7.68 %.
1-[(Naphthalen-2-yl)(pipyridin-1-yl)methyl]naphthalen-2-ol (16e)
Light brown solid; Yield 88 %; mp 240 \(^{\circ }\hbox {C}\) (dec.); FT-IR (KBr) \(\nu \) 3443, 3014, 2950, 1619, 1384, 1021, 760 \(\hbox {cm}^{-1}\); \(^{1}\hbox {H NMR}\) (400 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 1.25–1.69 (6H, m, \(\hbox {CH}_{2}\)), 2.00 (1H, m, –NCH), 2.21 (1H, m, –NCH), 2.66 (1H, m, –NCH), 3.42 (1H, m, –NCH), 5.26 (1H, s, methine-H), 7.17–7.20 (2H, m, Ar–H), 7.34 (1H, t, \(J = 7.6\, \hbox {Hz}\), Ar–H), 7.41–7.43 (2H, m, Ar–H), 7.65–7.68 (2H, m, Ar–H), 7.73 (3H, m, Ar–H), 7.82 (1H, m, Ar–H), 7.91–7.96 (2H, m, Ar–H), 14.14 (1H, bs, OH) ppm; \(^{13}\hbox {C NMR}\) (100 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 23.1, 25.0, 50.2, 52.9, 70.5, 114.7, 118.7, 120.2, 121.4, 122.3, 124.8, 125.2, 125.4, 126.1, 127.5, 127.6, 127.8, 128.3, 128.7, 128.9, 131.3, 133.6, 134.3, 136.3, 153.9 ppm; Anal. Calcd. For \(\hbox {C}_{26}\hbox {H}_{25}\hbox {NO}\): C, 84.98; H, 6.86; N, 3.81; Found: C, 84.72; H, 6.87; N, 3.77 %.
1-[Morpholino(phenyl)methyl]naphthalen-2-ol (16f)
White solid; Yield 92 %; mp: 174–1766 \(^{\circ }\hbox {C}\); \(^{1}\hbox {H NMR}\) (400 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 2.24–2.41 (3H, m, –NCH), 3.09 (1H, m, –NCH), 3.58–3.81 (4H, m, –OCH), 5.09 (1H, s, methine-H), 7.14–7.27 (5H, m, Ar–H), 7.37 (1H, t, \(J=7.2\, \hbox {Hz}\), Ar–H), 7.55 (2H, m, Ar–H), 7.65–7.69 (2H, m, Ar–H), 7.83 (1H, d, \(J = 8.4 \hbox {Hz}\), Ar–H), 13.18 (1H, bs, OH) ppm; \(^{13}\hbox {C NMR}\) (100 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 50.4, 52.8, 65.8, 70.8, 114.0, 118.7, 199.9, 121.5, 125.5, 127.1, 127.6, 127.8, 128.7, 131.2, 137.5, 153.6 ppm; Anal. Calcd. For \(\hbox {C}_{21}\hbox {H}_{21}\hbox {NO}_{2}\): C, 78.97; H, 6.63; N, 4.39; Found: C, 78.71; H, 6.67; N, 4.36 %.
1-[(2-Chlorophenyl)(morpholino)methyl]naphthalen-2-ol (16g)
White solid; Yield 83 %; mp: 163–165 \(^{\circ }\hbox {C}\); FT-IR (KBr) \(\nu \) 3421, 3054, 2966, 1619, 1460, 1114, 748 \(\hbox {cm}^{-1};\,^{1}\hbox {H NMR}\) (400 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 2.42 (1H, m, –NCH), 2.56–2.74 (2H, m, –NCH), 3.12 (1H, m, –NCH), 3.54-3.89 (4H, m, –OCH), 5.89 (1H, s, methine-H), 7.11–7.17 (3H, m, Ar–H), 7.24 (1H, t, \(J = 7.2\, \hbox {Hz}\), Ar–H), 7.37–7.41 (2H, m, Ar–H), 7.61–7.65 (1H, m, Ar–H), 7.68–7.71 (2H, m, Ar–H), 7.81 (1H, d, \(J = 8.4\,\hbox {Hz}\), Ar–H), 13.38 (1H, bs, OH) ppm; \(^{13}\hbox {C NMR}\) (100 MHz, \(\hbox {CDCl}_{3})\): \(\delta \) 48.1, 52.9, 65.1, 65.7, 66.3, 113.9, 118.9, 120.3, 121.8, 125.8, 127.1, 127.7, 127.8, 128.5, 128.6, 129.0, 129.9, 131.7, 133.4, 134.9, 154.7 ppm; Anal. Calcd. For \(\hbox {C}_{21}\hbox {H}_{20}\hbox {ClNO}_{2}\): C, 71.28; H, 5.70; N, 3.96; Found: C, 71.10; H, 5.73; N, 3.90 %.
1-[(3-Chlorophenyl)(morpholino)methyl]naphthalen-2-ol (16h)
Colorless crystals; Yield 82 %; mp: 160–162 \(^{\circ }\hbox {C}\); FT-IR (KBr) \(\nu \) 3430, 3060, 3014, 2964, 1622, 1469, 1119, 752 \(\hbox {cm}^{-1};\, ^{1}\hbox {H NMR}\) (400 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 2.26–2.44 (3H, m, –NCH), 3.07 (1H, m, –NCH), 3.61-3.82 (4H, m, –OCH), 5.05 (1H, s, methine-H), 7.09-7.16 (2H, m, Ar–H), 7.24 (1H, t, \(J=7.6\, \hbox {Hz}\), Ar–H), 7.32 (1H, d, \(J = 8.0\, \hbox {Hz}\), Ar–H), 7.39 (1H, d, \(J=8.0\, \hbox {Hz}\), Ar–H), 7.49 (1H, m, Ar–H), 7.67–7.71 (3H, m, Ar–H), 7.78 (1H, d, \(J=8.4\, \hbox {Hz}\), Ar–H), 12.98 (1H, bs, OH) ppm; \(^{13}\hbox {C NMR}\) (100 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 50.5, 52.7, 65.7, 70.2, 113.3, 118.8, 119.6, 121.7, 125.7, 126.6, 127.7, 127.9, 129.0, 129.5, 130.3, 130.7, 131.0, 139.8, 153.6 ppm; Anal. Calcd. For \(\hbox {C}_{21}\hbox {H}_{20}\hbox {ClNO}_{2}\): C, 71.28; H, 5.70; N, 3.96; Found: C, 71.06; H, 5.75; N, 3.94 %.
1-[(4-Chlorophenyl)(morpholino)methyl]naphthalen-2-ol (16i)
Brown solid; Yield 89 %; mp: 155–157 \(^{\circ }\hbox {C};\,^{1}\hbox {H NMR}\) (400 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 2.16–2.49 (3H, m, –NCH), 3.09 (1H, m, –NCH), 3.61–3.84 (4H, m, –OCH), 5.09 (1H, s, methine-H), 7.14 (1H, d, \(J=8.8\, \hbox {Hz}\), Ar–H), 7.23–7.27 (3H, m, Ar–H), 7.39 (1H, t, \(J=7.6\, \hbox {Hz}\), Ar–H), 7.49–7.51 (2H, m, Ar–H), 7.67–7.74 (2H, m, Ar–H), 7.77 (1H, d, \(J=8.8\, \hbox {Hz}\), Ar–H), 13.05 (1H, bs, OH) ppm; \(^{13}\hbox {C NMR}\) (100 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 50.4, 52.8, 65.7, 70.1, 113.6, 118.7, 119.7, 121.7, 125.7, 127.7, 127.9, 128.7, 128.9, 129.3, 131.0, 132.9, 136.1, 153.5 ppm; Anal. Calcd. For \(\hbox {C}_{21}\hbox {H}_{20}\hbox {ClNO}_{2}\): C, 71.28; H, 5.70; N, 3.96; Found: C, 71.02; H, 5.76; N, 3.92 %.
1-[(4-(Dimethylamino)phenyl)(morpholino)methyl]naphthalen-2-ol (16j)
White solid; Yield 83 %; mp: 135–137 \(^{\circ }\hbox {C}\); FT-IR (KBr) \(\nu \) 3479, 3052, 3006, 2960, 1615, 1521, 1117, 750 \(\hbox {cm}^{-1}\); \(^{1}\hbox {H NMR}\) (400 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 2.25-2.51 (3H, m, –NCH), 2.86 (6H, s, \(\hbox {--CH}_{3}\)), 3.06 (1H, m, –NCH), 3.61–3.82 (4H, m, –OCH), 5.03 (1H, s, methine-H), 6.59 (2H, d, \(J=8.4\, \hbox {Hz}\), Ar–H), 7.13 (1H, d,\( J\) = 8.8 Hz, Ar–H), 7.19–7.24 (1H, m, Ar–H), 7.34–7.39 (3H, m, Ar–H), 7.64–7.69 (2H, m, Ar–H), 7.83 (1H, d, \(J=8.8\, \hbox {Hz}\), Ar–H), 13.30 (1H, bs, OH) ppm; \(^{13}\hbox {C NMR}\) (100 MHz, \(\hbox {CDCl}_{3}\)): \(\delta \) 39.3, 50.2, 52.9, 65.9, 70.3, 111.3, 114.7, 118.6, 120.2, 121.4, 122.3, 124.8, 125.4, 127.8, 128.3, 128.9, 131.3, 149.1, 153.5 ppm; Anal. Calcd. For \(\hbox {C}_{23}\hbox {H}_{26}\hbox {N}_{2}\hbox {O}_{2}\): C, 76.21; H, 7.23; N, 7.73; Found: C, 76.03; H, 7.28; N, 7.69 %.
References
Knapp S (1995) Synthesis of complex nucleoside antibiotics. Chem Rev 95:1859–1876. doi:10.1021/cr00038a006
Mannhold R, Kubinyi H (2006) Nucleic acid drugs. In: Dingermann T, Steinhilber D, Folkers G (eds) Molecular biology in medicinal chemistry. Wiley, Weinheim. doi:10.1002/3527602666
Wang YF, Izawa T, Kobayashi S, Ohno M (1982) Stereocontrolled synthesis of (+)-negamycin from an acyclic homoallylamine by 1,3-asymmetric induction. J Am Chem Soc 104:6465–6466. doi:10.1021/ja00387a060
Cardellicchio C, Capozzi MAM, Naso F (2010) The Betti base: the awakening of a sleeping beauty. Tetrahedron 21:507–517. doi:10.1016/j.tetasy.2010.03.020
Elmendorf HG, Walls CD, Wolf C (2012) Compositions and methods for the treatment of giardiasis. US Patent 20,120,283,267
Gerlach M, Maul C (2007) Substituted 1 and 2-naphthol Mannich bases. US Patent 7,202,242 B2
Gyemant N, Engi H, Schelz Z, Szatmari I, Toth D, Fulop F, Molnar J, de Witte P (2010) In vitro and in vivo multidrug resistance reversal activity by a Betti-base derivative of tylosin. Br J Cancer 103:178–185. doi:10.1038/sj.bjc.6605716
Shen AY, Tsai CT, Chen CL (1999) Synthesis and cardiovascular evaluation of \(N\)-substituted 1-aminomethyl-2-naphthols. Eur J Med Chem 34:877–882. doi: 10.1016/S0223-5234(99)00204-4
Szatmári I, Fülöp F (2013) Syntheses, transformations and applications of aminonaphthol derivatives prepared via modified Mannich reactions. Tetrahedron 69:1255–1278. doi:10.1016/j.tet.2012.11.055
Salama TA (2013) Silicon-induced general, mild, and efficient one-pot, three-component synthesis of amidoalkyl naphthol libraries. Synlett 24:713–718. doi:10.1055/s-0032-1318392
Safari J, Zarnegar Z (2013) A magnetic nanoparticle-supported sulfuric acid as a highly efficient and reusable catalyst for rapid synthesis of amidoalkyl naphthols. J Mol Catal A 379:269–276. doi:10.1016/j.molcata.2013.08.028
Rostamizadeh S, Abdollahi F, Shadjou N, Amani AM (2013) MCM-41-SO\(_{3}\)H: a novel reusable nanocatalyst for synthesis of amidoalkyl naphthols under solvent-free conditions. Monatsh Chem 144:1191–1196. doi: 10.1007/s00706-013-0936-4
Lei ZK, Xiao L, Lu XQ, Huang H, Liu CJ (2013) Graphite supported perchloric acid (HClO\(_{4}\)-C): an efficient and recyclable heterogeneous catalyst for the one-pot synthesis of amidoalkyl naphthols. Molecules 18:1653–1659. doi: 10.3390/molecules18021653
Das VK, Borah M, Thakur AJ (2013) Piper-betle-shaped nano-S-catalyzed synthesis of 1-amidoalkyl-2-naphthols under solvent-free reaction condition: A greener “Nanoparticle-Catalyzed Organic Synthesis Enhancement” approach. J Org Chem 78:3361–3366. doi:10.1021/jo302682k
Csutortoki R, Szatmari I, Fulop F (2013) Syntheses of amido-, carbamido- and carbamatoalkylnaphthols. Curr Org Synth 10:564–583
Mulla SA, Salama TA, Pathan MY, Inamdar SM, Chavan SS (2012) Solvent-free, highly efficient one-pot multi-component synthesis of 1-amido and 1-carbamato-alkyl naphthols/phenols catalyzed by ethylammonium nitrate as reusable ionic liquid under neat reaction condition at ambient temperature. Tetrahedron Lett 54:672–675. doi:10.1016/j.tetlet.2012.12.004
Kotadia DA, Soni SS (2012) Silica gel supported-SO\(_{3}\)H functionalised benzimidazolium based ionic liquid as a mild and effective catalyst for rapid synthesis of 1-amidoalkyl naphthols. J Mol Catal A 353:44–49. doi: 10.1016/j.molcata.2011.11.003
Zolfigol MA, Khazaei A, Moosavi-Zare AR, Zare A, Khakyzadeh V (2011) Rapid synthesis of 1-amidoalkyl-2-naphthols over sulfonic acid functionalized imidazolium salts. Appl Catal A 400:70–81. doi:10.1016/j.apcata.2011.04.013
Tamaddon F, Bistgani JM (2011) \([\text{ MeC(OH) }_{2}]^{+}\text{ ClO }_{4}^{-}\): a new efficient organocatalyst for the preparation of 1-amido-and 1-carbamato-alkyl naphthols. Synlett 2011:2947–2950. doi:10.1055/s-0031-1289906
Mistry SR, Joshi RS, Maheria KC (2011) Zeolite H-BEA catalysed multicomponent reaction: one-pot synthesis of amidoalkyl naphthols—biologically active drug-like molecules. J Chem Sci 123:427–432. doi:10.1007/s12039-011-0095-2
Shaterian HR, Yarahmadi H, Ghashang M (2008) Silica supported perchloric acid (HClO\(_{4}\)-SiO\(_{2})\): an efficient and recyclable heterogeneous catalyst for the one-pot synthesis of amidoalkyl naphthols. Tetrahedron 64:1263–1269. doi: 10.1016/j.tet.2007.11.070
Shaterian HR, Yarahmadi H, Ghashang M (2008) An efficient, simple and expedition synthesis of 1-amidoalkyl-2-naphthols as “drug like” molecules for biological screening. Bioorg Med Chem Lett 18:788–792. doi:10.1016/j.tet.2007.11.070
Karmakar B, Banerji J (2011) A competent pot and atom-efficient synthesis of Betti bases over nanocrystalline MgO involving a modified Mannich type reaction. Tetrahedron Lett 52:4957–4960. doi:10.1016/j.tetlet.2011.07.075
Periasamy M, Anwar S, Reddy MN (2009) Simple and convenient methods for synthesis, resolution and application of aminonaphthols. Indian J Chem B 48:1261–1273
Ghandi M, Olyaei A, Raoufmoghaddam S (2008) One-pot, three-component uncatalyzed quantitative synthesis of new aminonaphthols (Betti bases) in water. Synth Commun 38:4125–4138. doi:10.1080/00397910802279860
Olyaei A, Raoufmoghaddam S, Sadeghpour M, Ebadzadeh B (2010) Convenient and efficient method for the synthesis of \(N\)-heteroaryl aminonaphthols under solvent-free conditions. Chin J Chem 28:825–832. doi: 10.1002/cjoc.201090153
Hosseinian A, Shaterian HR (2012) \(\text{ NaHSO }_{4} \cdot \text{ H }_{2}\text{ O }\) catalyzed multicomponent synthesis of 1-(benzothiazolylamino) methyl2-naphthols under solvent-free conditions. Phosphorus Sulfur Silicon 187:1056–1063. doi: 10.1080/10426507.2012.664221
Buckley BR, Neary SP (2010) Organocatalysis. Annu Rep Prog Chem Sect B Org Chem 106:120–135. doi:10.1039/B927086H
Grondal C, Jeanty M, Enders D (2010) Organocatalytic cascade reactions as a new tool in total synthesis. Nat Chem 2:167–178. doi:10.1038/nchem.539
Marcelli T, van Maarseveen JH, Hiemstra H (2006) Cupreines and cupreidines: an emerging class of bifunctional cinchona organocatalysts. Angew Chem Int Ed 45:7496–7504. doi:10.1002/anie.200602318
Liu G, Zhang S, Li H, Zhang T, Wang W (2011) Organocatalytic enantioselective Friedel–Crafts reactions of 1-naphthols with aldimines. Org Lett 13:828–831. doi:10.1021/ol102987n
Ramachary DB, Babul Reddy G (2007) A new organocatalyst for Friedel–Crafts alkylation of 2-naphthols with isatins: application of an organo-click strategy for the cascade synthesis of highly functionalized molecules. Tetrahedron Lett 48:7618–7623. doi:10.1016/j.tetlet.2007.08.129
Shahrisa A, Safa KD, Esmati S (2014) Synthesis, spectroscopic and DFT studies of novel fluorescent dyes: 3-aminoimidazo[1,2-a]pyridines possessing 4-pyrone moieties. Spectrochim Acta A 117:614–621. doi:10.1016/j.saa.2013.09.056
Shahrisa A, Esmati S, Miri R, Firuzi O, Edraki N, Nejati M (2013) Cytotoxic activity assessment, QSAR and docking study of novel bis-carboxamide derivatives of 4-pyrones synthesized by Ugi four-component reaction. Eur J Med Chem 66:388–399. doi:10.1016/j.ejmech.2013.05.049
Shahrisa A, Esmati S (2013) Three novel sequential reactions for the facile synthesis of a library of bisheterocycles possessing the 3-aminoimidazo [1,2-a] pyridine core catalyzed by bismuth (III) chloride. Synlett 24:595–602. doi:10.1055/s-0032-1318221
Shahrisa A, Miri R, Esmati S, Saraei M, Mehdipour AR, Sharifi M (2012) Synthesis and calcium channel antagonist activity of novel 1,4-dihydropyridine derivatives possessing 4-pyrone moieties. Med Chem Res 21:284–292. doi:10.1007/s00044-010-9534-8
Shahrisa A, Esmati S, Nazari MG (2012) Boric acid as a mild and efficient catalyst for one-pot synthesis of 1-amidoalkyl-2-naphthols under solvent-free conditions. J Chem Sci 124:927–931. doi:10.1007/s12039-012-0285-6
Dindulkar SD, Puranik VG, Jeong YT (2012) Supported copper triflate as an efficient catalytic system for the synthesis of highly functionalized 2-naphthol Mannich bases under solvent free condition. Tetrahedron Lett 53:4376–4380. doi:10.1016/j.tetlet.2012.06.022
Ganesan SS, Rajendran N, Sundarakumar SI, Ganesan A, Pemiah B (2013) \(\beta \)-Naphthol in glycerol: a versatile pair for efficient and convenient synthesis of aminonaphthols, naphtho-1,3-oxazines, and benzoxanthenes. Synthesis 45:1564–1568. doi: 10.1055/s-0033-1338430
Saidi MR, Azizi N, Naimi-Jamal MR (2001) Lithium perchlorate assisted one-pot three-component aminoalkylation of electron-rich aromatic compounds. Tetrahedron Lett 42:8111–8113. doi:10.1016/S0040-4039(01)01732-4
Katritzky AR, Abdel-Fattah AA, Tymoshenko DO, Belyakov SA, Ghiviriga I, Steel PJ (1999) Amino (hetero) arylmethylation of phenols with \(N\)-[\(\alpha \)-amino (hetero) arylmethyl] benzotriazoles. J Org Chem 64:6071–6075. doi:10.1021/jo9903609
Kumar A, Gupta MK, Kumar M (2010) Non-ionic surfactant catalyzed synthesis of Betti base in water. Tetrahedron Lett 51:1582–1584. doi:10.1016/j.tetlet.2010.01.056
Acknowledgments
We thank research affairs of the University of Tabriz for financial support.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Shahrisa, A., Teimuri-Mofrad, R. & Gholamhosseini-Nazari, M. Synthesis of a new class of Betti bases by the Mannich-type reaction: efficient, facile, solvent-free and one-pot protocol. Mol Divers 19, 87–101 (2015). https://doi.org/10.1007/s11030-014-9559-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11030-014-9559-x