Abstract
Early life stress increases the risk of developing psychopathology accompanied by reduced cognitive function in later life. Maternal separation induces anxiety-like behaviours and is associated with impaired memory. On the other hand, exercise has been shown to diminish anxiety-like behaviours and improve cognitive function. The effects of maternal separation and exercise on anxiety, memory and hippocampal proteins were investigated in male Sprague–Dawley rats. Maternal separation produced anxiety-like behaviours which were reversed by exercise. Maternal separation also enhanced object location memory which was not affected by exercise. Exercise did, however, increase synaptophysin and phospho-extracellular signal-regulated kinase (p-ERK) in the hippocampus of non-separated rats and this effect was not observed in maternally separated rats. These findings show that maternal separation selectively enhanced n memory and prevented activation of the MAPK/ERK signalling pathway in the adult rat hippocampus.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Early life stressors are known to increase an individual’s risk of developing a psychological disorder in later life (Lupien et al. 2009). Perinatal stressors occur at a time when the brain is undergoing rapid development and consequently influence the individual’s response to stress in adulthood (Champagne et al. 2009; Daskalakis et al. 2011; Kaplan et al. 2001).
Maternal separation is a well-established protocol that involves repeated separation of the pups from the dam during the early postnatal period which disturbs the hypothalamus-pituitary-adrenal (HPA) axis and alters brain function (Banihashemi and Rinaman 2010; Kaplan et al. 2001; Lupien et al. 2009). Regulators of the HPA axis such as the hippocampus and amygdala are also affected by maternal separation and may thus contribute to altered HPA axis activity and cognitive function (Schmolck et al. 2002).
On the other hand, exercise has been shown to have opposite effects to maternal separation on anxiety-like behaviours and cognitive function (Duman et al. 2008; Mello et al. 2009). Exercise stimulates neurogenesis (Lou et al. 2008) and enhances long-term potentiation (LTP) required for learning and memory formation (O’Callaghan et al. 2007). At the cellular and molecular level memory has been shown to depend on glutamate receptor activation (Barker et al. 2006; Sanderson et al. 2007) and brain-derived neurotrophic factor (BDNF) (Bekinschtein et al. 2008; Vaynman et al. 2004), among other factors, resulting in activation of calcium-dependent (Tinsley et al. 2009) and mitogen-activated protein kinase (MAPK) signalling pathways (Purcell et al. 2003) These pathways do not act independently (Scimeca et al. 1997). Evidence suggests that they may be interdependent (Li and Keifer 2009). It appears that specific components of these pathways are necessary for certain types of learning and memory to occur (Izquierdo et al. 2006). For instance, extracellular signal-regulated kinase (ERK), a member of the MAPK pathway, is essential for retention of memories acquired in aversive contexts (Cestari et al. 2006; Huang et al. 2010; Runyan and Dash 2004; Tarpley et al. 2009)
This study was performed with the aim of extending a previous finding that exercise had beneficial effects on spatial learning in maternally separated juvenile rats (Grace et al. 2009). In the present study the effects of exercise on recognition memory were tested in adulthood. Non-aversive, hippocampus-dependent memory tasks were chosen. In addition to the behavioural study, biochemical analyses of synaptophysin, a marker of neuronal plasticity, as well as phospho-ERK (p-ERK), ERK and MAPK phosphatase-1 (MKP-1) were evaluated to determine how the MAPK pathway was affected by maternal separation and exercise. It was found that maternal separation improved object location memory whereas exercise activated the MAPK/ERK signalling pathway in the hippocampus of non-separated rats.
Materials and methods
Animals
Sprague–Dawley rats were purchased from the University of Stellenbosch. The rats were housed under standard laboratory conditions in a 12 h:12 h light/dark cycle with lights on at 06:00. At postnatal day 47 (P47) rats were transferred to a 12 h:12 h light/dark cycle with lights on at 21:00 to allow memory testing during the rat’s dark phase. The temperature was maintained at 21°C ± 2°C. The rats had free access to water and rat chow. All protocols were approved by the Faculty of Health Sciences Animal Ethics Committee of the University of Cape Town (009/007). The study was carried out according to international guidelines (South African National Standard: The Care and Use of Animals for Scientific Purposes, 2008).
Experimental groups
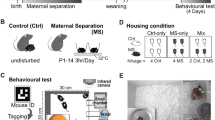
Whole litters were randomly assigned to maternally separated or non-separated groups at birth (postnatal day zero or P0). Maternal separation commenced from P2 to P14. Rats were weaned at P21. Half of the maternally separated rats and half of the non-separated rats were randomly selected for voluntary exercise at P50. A total of four groups of rats were tested in this study, namely, non-separated non-runners (NSnR, n = 11); non-separated runners (NSR, n = 11); maternally separated non-runners (MSnR, n = 11) and maternally separated runners (MSR, n = 11). Behavioural tests were performed at P28 and between P75- P81. At P88 animals were killed and hippocampal tissue was collected for Western blot analysis of protein content. Rats were weighed every second day from P71.
Maternal separation
Sexually experienced males were paired with virgin females for breeding. The studs were removed from the cages as soon as the females had given birth. The day of birth was designated post-natal day zero, P0. Litters were randomly assigned to the maternal separation group, MS, or the non-separated group, NS. On P2 litters were culled to 8 pups with preference for male pups. Only male pups were used. The maternally separated pups were separated from the dams for three hours each day between 08:30 and 13:00 (Aisa et al. 2007). The pups were taken to a different room and placed under infrared lamps (temperature = 32 ±1°C) to prevent hypothermia. The maternal separation dams and the non-separated rats were left in their home cages in the animal facility. From P15 the pups and dams were left undisturbed except for regular cleaning of cages until weaning at P21 (Gaszner et al. 2009).
Voluntary exercise
At P47 rats were transferred to the exercise room which had a shifted 12 h:12 h light–dark cycle (lights off at 09:00). The rats were housed in the exercise room for 4 days before being placed, individually, in cages with attached running-wheels. The wheels were connected to a computer that recorded the number of revolutions. The non-runners (exercise controls) were placed in identical cages with immobilized wheels. At P70, the rats were taken out of the running-wheel cages and were housed communally in groups of 3–5 rats per cage.
Behavioural assessment
Rats were allowed to habituate in the behaviour suite for at least 1 h before behavioural testing. The behaviour room had a light intensity of 50 lux. All the tests were performed between 11:00 and 17:00. All of the behavioural equipment was cleaned with a soapy solution followed by 10% ethanol before the first trial and also in between trials. Animal behaviour was recorded on a Sony Handycam video camera affixed ± 1 m above the behavioural apparatus and analysed using Ethovision software v7.0 XT (Noldus Information Technology, Netherlands).
Open field test
Rats were tested in the open field on P28 and P75. The open field arena comprised a wooden box (100 cm × 100 cm × 60 cm) with a black interior. A white line was used to demarcate the inner and outer zones on the floor of the arena. The inner zone measured 70 cm × 70 cm and was 15 cm from the walls of the box. Each rat was placed in the arena facing a corner and allowed to explore the arena freely for a 5-min period. The parameters measured were mean velocity, total distance travelled, time spent (duration) in the outer zone, duration in the inner zone, and frequency of transitions to the inner and outer zones.
Elevated plus maze
The elevated plus maze was used to assess the level of anxiety-like behaviour in the rats (Walf and Frye 2007). The apparatus was raised 50 cm above the floor. The open and closed arms measured 10 cm × 45 cm with the centre measuring 10 cm × 10 cm. The walls of the closed arms were 40 cm high. The floor and the walls of the closed arms were painted black. The rat was placed in the centre square facing an open arm and allowed to explore the maze for a 5-min period. Rats were placed in the EPM 1 h after completion of the open field test on P75. Parameters that were measured were time spent in the open and closed arms as well as frequency of transitions into the open and closed arms.
Novel object recognition task
The novel object recognition test was used to assess recognition memory in the rats (Becker et al. 2009; Oreland et al. 2010). One hour after completion of the EPM test, the animals were allowed to explore the object recognition arena for 5 min to be familiarised with the box. The object recognition box was similar to the open field box but smaller (65 cm × 65 cm × 60 cm). Objects used in the tests were made from wood and were painted different colours. The objects were placed approximately 5 cm from the walls of the box and screwed into place so that they could not be moved by the rats.
The protocol consisted of a sample phase and test phase. The animals were subjected to 2 test phases: one for short term and another for long term memory (1 h and 24 h after the sample phase). In the sample phase, on P76 the animals were presented with two identical objects to explore for 5 min. In the test phases, one object was switched for a novel one and exploration time was reduced to 3 min. The familiar object was kept the same for both test sessions. The time that the rat spent with each object was recorded. Intact memory was indicated by rats spending more time with the novel object. Rat-object interaction was defined as ‘the animal directing its nose toward the object at a distance of 2 cm or less (Barker et al. 2007) and it excluded sitting or climbing on the object. In all the tests, the rat was placed facing the wall opposite the object.
Object-in-place task
The object-in-place task tested the rat’s ability to discriminate between objects whose location had been swapped (Oreland et al. 2010). In the sample phase the rat was allowed to explore 4 objects that were placed equidistant at the corners of the box. In the test phase, which took place 5 min after sample acquisition, 2 adjacent objects were swapped. The rats with intact memory would have been able to notice the change and thus, spend more time with the objects that had been moved. This test was performed on P78.
Object location task
The object location task consisted of 3 phases; the acquisition phase and 2 test phases (short term and long term). In the 5-min sample acquisition phase on P79, the animals were presented with two novel identical objects. The first test phase was carried out 1 h after sampling and the 2nd test was 24 h after the first test. In the test phases the location of one object was changed. The test phases lasted 3 min each. Rats with intact memory spent more time exploring the moved object which was indicated by a positive discrimination index. The discrimination index was calculated as the difference in time exploring the moved and unmoved object divided by the total amount of time spent with both objects. There were no extra-arena cues. Animals had to use the position of the objects in relation to their starting position when placed in the arena.
Temporal ordering task
On P81, rats were presented with 2 sets of identical objects which they could explore over 2 sample phases (1 h apart). Three hours after the 2nd sample phase, the rat’s tendency to explore the earlier object was measured (Barker et al. 2006). An intact temporal ordering memory was indicated by the animal spending more time with the 1st sample phase object and a discrimination index that was positive. The discrimination index was calculated as the difference in time spent interacting with objects from the 1st and 2nd phases divided by the total time spent interacting with all objects.
Tissue collection
At P88 the rats were anaesthetised with halothane and then decapitated. Immediately after decapitation, the head was immersed in liquid nitrogen for ±5 s and the dorsal and ventral hippocampus dissected on ice within 2 min. This method decreased the rate of protein phosphorylation and dephosphorylation so that in vitro protein analysis could reflect the in vivo levels as accurately as possible (Errico et al. 2011). The tissue samples were stored at -80°C until Western blot analysis.
Western blot analysis
The dorsal and ventral hippocampal samples were sonicated in 200 μL of RIPA extraction buffer (5 M NaCl; 100% Triton X-100; 10% SDS; 1 M Tris pH 7.5; 1% Deoxycholate) with 0.1% protease inhibitors (Thermo Scientific) and boiled for 10 min (Errico et al. 2011). Protein concentration was determined by the BCA assay according to the manufacturer’s instructions (Pierce, Thermo-Scientific). SDS-PAGE was used to separate the proteins which were subsequently transferred onto a nitrocellulose membrane (Hybond ECL, Amersham). Brief staining with Ponceau S was used to confirm the transfer of protein onto the membranes. After destaining with phosphate buffered saline (10 mM NaH2PO4, 40 mM Na2HPO4, 100 mM NaCl, PBS), the membranes were blocked in a solution of 5% fat-free milk in PBS-Tween (1% Tween) before incubating with primary antibody overnight at 4°C. The following primary antibodies were used: Rabbit-anti-Phospho-ERK1/2, rabbit-anti-ERK1/2 (both 1:1000, Cell Signalling, USA), mouse-anti-synaptophysin (1:20 000) (Abcam Inc.), rabbit-anti-MKP-1 (1:500) (Santa Cruz) and rabbit-anti-p38 (1:10 000) (BioRad). Primary antibodies for synaptophysin and p38 were diluted in 5% fat-free milk in PBS-Tween. Primary ERK, p-ERK and MKP-1 antibodies were diluted in 1% Tween in Tris-buffered saline (20 mM Tris, 150 mM NaCl). Secondary antibodies (diluted in 5% fat-free milk in PBS-Tween) were goat anti-rabbit-HRP (BioRad) (for ERK, p-ERK, MKP-1 and p38) and Goat anti-mouse-HRP (for synaptophysin) (BioRad). Membranes were stripped with 0.2 M NaOH before being probed for a different protein. Proteins were probed in this order: p-ERK, ERK, synaptophysin then p38. MKP-1 was analysed on separate membranes. Bands on the X-ray film were quantified using UNSCAN-IT software. Proteins were expressed as a ratio of the loading control, p38 which is unchanged by stress or exercise (Leem et al. 2011; Meller et al. 2003).
Statistical analysis
All statistical analyses were performed using Statistica 10 (StatSoft, USA). All data were tested for normality using the Shapiro-Wilk’s test. The Mann–Whitney U-test was applied to the open field data obtained on P28 and distance travelled in the running wheels, since these data were not normally distributed. Multiple analysis of variance (MANOVA) was applied to the protein analyses and to all behavioural assessments that commenced after P75. Where MANOVA revealed significant differences, Duncan’s post-hoc test was applied to compute the level of significance of the differences between group mean values.
Results
Body weight and running activity
The weights of the rats increased steadily post-exercise, across all groups, to an average of 331 g at the end of the experiment when brain tissue was collected for protein analysis (NSnR = 335.4 ± 23.14 g; NSR = 330.0 ± 25.14 g; MSnR = 340.1 ± 26.97 g and MSR = 320.3 ± 29.16 g). A two-way ANOVA of data obtained at P71 revealed a significant effect of maternal separation (F(1,40) = 3.42, p = 0.012) and a tendency for exercised rats to weigh less than non-exercised rats (F(1,40) = 2.43, p = 0.053). Duncan’s post-hoc test revealed that the maternally separated runners had significantly lower masses than maternally separated non-runners (MSnR, p = 0.048) and non-separated runners (NSR, p = 0.009). The masses did not differ across groups from P77 onwards.
The total distance travelled by the maternally separated runners was not different to the distance travelld by non-separated runners during the 20 days of exercise, p = 0.554 (median = 8.62 km, interquartile range = 3.16–22.36 km versus median =11.78 km, interquartile range =6.24–23.41 km, respectively).
Open field
Figure 1 shows the open field result at P28. The Mann–Whitney U-test revealed that maternally separated rats spent significantly less time in the inner zone (p = 0.049) and made fewer transitions from the outer zone into the inner zone of the open field (p = 0.025) than non-separated rats (n = 22). There were no differences between maternally separated and non-separated rats in the total distance travelled, speed of running, time spent in the outer zone or frequency of entries into the outer zone.
Postnatal day 28, maternal separation caused anxiety-like behaviour in the open field. a maternal separation decreased the amount of time spent by rats in the inner zone. b Maternal separation decreased the number of entries made by rats into the inner zone. *Significantly different from non-separated rats, p < 0.05, n = 22. Results are shown as mean ± SEM
Figure 2 shows the open field result at P75. Two-way ANOVA did not reveal any differences between maternally separated and non-separated rats or between exercised and non-exercised rats, in terms of the number of entries and time spent in the inner zone of the open field. A maternal separation effect on mean velocity was observed (F(1,40) = 9.61, p = 0.003). Duncan’s post-hoc test showed that non-separated runners explored the field with greater speed than maternally separated runners (p = 0.018).
Postnatal day 75, non-separated runners explored the open field with greater velocity than rats that had been subjected to maternal separation. Non-separated runners had significantly higher mean velocities in the open field than maternally separated runners and non-runners. *NSR compared to MSR, p < 0.05, n = 11. Results are shown as mean ± SEM
Elevated plus maze
Figure 3 shows the result for the EPM at P75. Two-way ANOVA showed that there was a separation effect (F(1,40)9.45, p = 0.003) and a tendency towards a separation*exercise interaction (F(1,40) = 4.01, p = 0.051). Exercise affected maternally separated and non-separated rats differently, exercise tended to increase the time spent in open arms by maternally separated rats. Maternally separated non-runners spent less time in the open arms compared to non-separated non-runners (p = 0.004). For open arm entries, a separation effect was observed (F(1,40) = 5.05, p = 0.030), exercise tended to increase the number of entries (F(1,40) = 2.95, p = 0.090), particularly in the maternally separated rats. Maternally separated non-runners made the least transitions into the open arms compared to all other groups (p < 0.050).
Exercise reversed maternal separation-induced anxiety-like behaviour in the elevated plus maze at P75. a Time spent in the open arms was decreased by maternal separation. Maternally separated non-running rats spent significantly less time in the open arms than non-separated non-runners, *p < 0.010. b The number of entries into the open arms was also decreased by maternal separation. Maternally separated non-runners made significantly fewer entries into the open arms, *p < 0.05 compared to all other groups. Results are mean ± SEM
Novel object recognition
Table 1 shows the mean (±SEM) of the discrimination indices for the novel object recognition task. Two-way ANOVA revealed no effect of separation or exercise. In the 1st minute, the maternally separated non-runners and maternally separated runners had negative discrimination indices. This showed that they spent more time exploring the old object and less time with the new object.
Object in place
In the 1st and 3rd minute of the retention trial, there were no significant differences in the discrimination indices. In the 2nd minute, a maternal separation effect was revealed by two-way ANOVA of the object in place discrimination index (F(1,40) = 7.705, p = 0.008). The discrimination indices of the maternally separated rats were negative, showing that they spent less time exploring the moved objects. Maternally separated non-runners tended to have lower discrimination indices than non-maternally separated non-runners (p = 0.064).
Object location
Table 1 shows the discrimination indices for the object location task. Short term memory of object location was not differentially affected by maternal separation or exercise. In the long term, after 24 h, maternal separation increased the time that the rats spent with the moved object. Two-way ANOVA revealed a separation effect in the 1st minute (F(1,40) = 14.76, p = 0.0004); and an exercise effect in the 3rd minute (F(1,40) = 4.88, p = 0.030) for the time spent with the moved object. Duncan’s post-hoc test showed that in the 1st minute maternally separated non-runners spent more time with the moved object than non-separated non-runners (p = 0.030); and maternally separated runners spent more time with the moved object than non-separated runners (p = 0.004).
Temporal ordering
Two-way ANOVA revealed a maternal separation effect in the 2nd minute of the retention trial for time spent with the phase 2 object (F(1,40) = 6.84, p = 0.013). Maternally separated runners spent more time exploring the phase 1 object than the non-separated runners (p < 0.050). The discrimination indices were not significantly different.
Western blot analyses
Figure 4 shows the relative density of synaptophysin in the dorsal hippocampus and ventral hippocampus. Two-way ANOVA revealed an effect of exercise on synaptophysin in the ventral hippocampus (F(1,20) = 5.109, p = 0.035). A similar pattern of protein expression was seen in the dorsal hippocampus. Duncan’s post-hoc test revealed that there were no between group differences.
Figure 5 shows the relative density of p-ERK in the dorsal and ventral hippocampus. Two-way ANOVA revealed a separation*exercise interaction in dorsal hippocampus p-ERK (F(1,40) = 4.83, p = 0.037). Duncan’s post-hoc test showed that p-ERK of non-separated runners was significantly higher than all the other groups (p < 0.05). In the ventral hippocampus, two-way ANOVA revealed a similar separation*exercise interaction (F(1,40) = 0.016). Duncan’s post-hoc test showed that non-separated runners had higher levels of p-ERK than all the other groups (p < 0.05). There were no differences in total Erk levels.
Non-separated runners had higher p-Erk levels in the dorsal and ventral hippocampus than non-runners and maternally separated rats. p-Erk levels were higher in the dorsal hippocampus of non-separated runners than all other groups *p < 0.050. p-Erk levels were higher in the ventral hippocampus of non-separated runners than all other groups # p < 0.01. Results are mean ± SEM
Figure 6 shows the relative density of MKP-1 in the dorsal hippocampus. Two-way ANOVA showed no effect of maternal separation (F(1,20) = 0.004, p = 0.95) or exercise (F(1,20) = 0.159, p = 0.694) on MKP-1 in the dorsal hippocampus. There was a tendency for an interaction between maternal separation and exercise (F(1,20) = 3.32, p = 0.080). Exercise tended to increase MKP-1 in non-stressed rats and to decrease MKP-1 in maternally separated rats.
Discussion
Previously, maternal separation and exercise have been shown to have opposing effects on anxiety-like behaviours and learning and memory (Benetti et al. 2009; Griffin et al. 2009; Liu et al. 2008). The open field and elevated plus maze tests were used to test the state of anxiety in rats (Kazl et al. 2009; Walf and Frye 2007). At P28, the maternally separated rats spent less time in the inner zone of the open field and also made fewer entries into the inner zone than non-separated rats. When retested in the open field at P75, after the exercise regimen, these differences disappeared and the rats’ behaviour in the open field was similar across all groups. However, in the elevated plus maze, maternally separated non-runners made fewer entries and spent less time in the open arms than all other groups which was in agreement with the findings of Aisa et al. (2007) but not in agreement with Benetti et al. (2009), who showed no effect of maternal separation on behaviour in the elevated plus maze. Maternally separated rats that were allowed to exercise voluntarily, spent as much time in the open arms as non-separated rats. These data demonstrated that the maternal separation-induced anxiety-like behaviour was already evident in early adolescence (P28) and persisted into adulthood (P75). Also, as an extension to previous findings, exercise was able to reverse the negative effects of maternal separation on anxiety-like behaviours in the rats (Mabandla and Russell 2011). Of note is that at P75, the differences in anxiety-like behaviours between groups were not observed in the open field which suggests that the elevated plus maze may be a more robust test for anxiety in adult rats. This also highlighted the importance of age when testing rats for anxiety-like behaviours, pubescent rats may not display anxiety-like behaviours in the same way as adult rats.
In the different memory tasks that were carried out, maternal separation had either no effect or a beneficial effect. Maternally separated rats showed improved memory for object location as well as temporal ordering. This finding was contrary to the work of Grace et al. (2009) who showed that exercised rats had an intact memory for objects they had most recently encountered. Maternal separation and exercise did not affect object recognition and object placement which agrees with the finding by Grace et al. (2009) who also showed that maternal separation did not alter memory while exercise improved it in the Morris Water Maze task. This suggested that object recognition and object placement tasks were not affected by maternal separation or exercise. However, there have been studies using Wistar rats that showed that maternal separation impaired object recognition (Aisa et al. 2007) and exercise improved it (García-Capdevila et al. 2009). Taken together, the current evidence suggests that maternal separation may have enhanced the circuitry involved in the memory of object location and temporal ordering which was evident in adult rats but not in juveniles (Grace et al. 2009).
In this current study, exercise did not improve memory. These findings are supported by earlier work which demonstrates that exercise has no benefit to various forms of object recognition memory (Grace et al. 2009; Aisa et al. 2007). However, there are studies which contradict the current findings, Griffin et al. (2009) and Mello et al. (2009) showed that exercise had a beneficial effect on object recognition memory. The beneficial effect of exercise on memory and learning are dependent on the duration (Stranahan et al. 2006), intensity and frequency of exercise (García-Capdevila et al. 2009). Animals that were more active in running wheels had poorer cognitive function in passive avoidance tasks than animals that were less active (García-Capdevila et al. 2009).
Stress and exercise activate ERK to increase p-ERK levels (Hu et al. 2009; Musazzi et al. 2009). In this current study protein markers of plasticity were differentially affected by maternal separation and exercise. Exercise increased synaptophysin in all groups but failed to increase p-ERK levels in the maternally separated group. This current data shows for the first time, that maternal separation impairs exercise-induced increases in p-ERK in the hippocampus. MKP-1, the regulator of p-ERK was not significantly altered by maternal separation and exercise. which is not consistent with the finding that hippocampal levels of p-ERK are inversely proportional to MKP-1 levels (Duric et al. 2010).
Exercise affected protein levels similarly in the dorsal and ventral hippocampus. The dorsal and ventral hippocampus are distinct functional units each having different roles; the dorsal hippocampus being mainly involved in cognition while the ventral hippocampus played a role in emotionality (Fanselow and Dong 2010). These data have demonstrated that ERK signalling is not required for intact object location memory. This is supported by work which demonstrates that elevated p-ERK is required for aversive-type learning (Huang et al. 2010; Li and Keifer 2009; Mazzucchelli et al. 2002; Tarpley et al. 2009) and calcium-calmodulin dependent protein kinase II is required for object recognition memory (Tinsley et al. 2009).
In conclusion, this work demonstrated for the first time that maternal separation impaires p-ERK signalling in the adult rat hippocampus. In addition, maternal separation improved the memory for object location suggesting that mild chronic stress had a selectively beneficial effect on memory.
Abbreviations
- MS:
-
maternal separation
- MAPK:
-
mitogen-activated protein kinase
- p-ERK:
-
phospho-extracellular signal-regulated protein kinase
- MKP-1:
-
mitogen-activated protein kinase phosphatase
- NS:
-
non-separated
- R:
-
runner
- nR:
-
non-runner
References
Aisa B, Tordera R, Lasheras B, Del Río J, Ramírez MJ (2007) Cognitive impairment associated to HPA axis hyperactivity after maternal separation in rats. Psychoneuroendocrinology 32:256–266. doi:10.1016/j.psyneuen.2006.12.013
Banihashemi L, Rinaman L (2010) Repeated brief postnatal maternal separation enhances hypothalamic gastric autonomic circuits in juvenile rats. Neuroscience 165:265–277. doi:10.1016/j.neuroscience.2009.09.081
Barker GRI, Warburton EC, Koder T et al (2006) The different effects on recognition memory of perirhinal kainate and NMDA glutamate receptor antagonism: implications for underlying plasticity mechanisms. J Neurosci 26:3561–3566. doi:10.1523/JNEUROSCI.3154-05.2006
Barker GRI, Bird F, Alexander V, Warburton EC (2007) Recognition memory for objects, place, and temporal order: a disconnection analysis of the role of the medial prefrontal cortex and perirhinal cortex. J Neurosci 27:2948–2957. doi:10.1523/JNEUROSCI.5289-06.2007
Becker S, Macqueen G, Wojtowicz JM (2009) Computational modeling and empirical studies of hippocampal neurogenesis-dependent memory: Effects of interference, stress and depression. Brain Res 1299:45–54. doi:10.1016/j.brainres.2009.07.095
Bekinschtein P, Cammarota M, Izquierdo I, Medina JH (2008) BDNF and memory formation and storage. Neuroscientist 14:147–156. doi:10.1177/1073858407305850
Benetti F, Mello PB, Bonini JS, Monteiro S, Cammarota M, Izquierdo I (2009) Early postnatal maternal deprivation in rats induces memory deficits in adult life that can be reversed by donepezil and galantamine. Int J Dev Neurosci 27:59–64. doi:10.1016/j.ijdevneu.2008.09.200
Cestari V, Costanzi M, Castellano C, Rossi-Arnaud C (2006) A role for ERK2 in reconsolidation of fear memories in mice. Neurobiol Learn Mem 86:133–143. doi:10.1016/j.nlm.2006.01.003
Champagne DL, de Kloet ER, Joëls M (2009) Fundamental aspects of the impact of glucocorticoids on the (immature) brain. Semin Fetal Neonatal M 14:136–142. doi:10.1016/j.siny.2008.11.006
Daskalakis NP, Claessens SEF, Laboyrie JJL, Enthoven L, Oitzl MS, Champagne DL, de Kloet ER (2011) The newborn rat’s stress system readily habituates to repeated and prolonged maternal separation, while continuing to respond to stressors in context dependent fashion. Horm Behav 60:165–176. doi:10.1016/j.yhbeh.2011.04.003
Duman CH, Schlesinger L, Russell DS, Duman RS (2008) Voluntary exercise produces antidepressant and anxiolytic behavioral effects in mice. Brain Res 1199:148–158. doi:10.1016/j.brainres.2007.12.047
Duric V, Banasr M, Licznerski P et al (2010) A negative regulator of MAP kinase causes depressive behavior. Nat Med 16:1328–1332. doi:10.1038/nm.2219
Errico F, Nisticò R, Napolitano F et al (2011) Persistent increase of D-aspartate in D-aspartate oxidase mutant mice induces a precocious hippocampal age-dependent synaptic plasticity and spatial memory decay. Neurobiol Aging 32:2061–2074. doi:10.1016/j.neurobiolaging.2009.12.007
Fanselow MS, Dong HW (2010) Review are the dorsal and ventral hippocampus functionally distinct structures? Neuron 65:7–19
García-Capdevila S, Portell-Cortés I, Torras-Garcia M, Coll-Andreu M, Costa-Miserachs D (2009) Effects of long-term voluntary exercise on learning and memory processes: dependency of the task and level of exercise. Behav Brain Res 202:162–170. doi:10.1016/j.bbr.2009.03.020
Gaszner B, Jensen KO, Farkas J, Reglodi D, Csernus V, Roubos EW, Kozicz T (2009) Effects of maternal separation on dynamics of urocortin 1 and brain-derived neurotrophic factor in the rat non-preganglionic Edinger-Westphal nucleus. Int J Dev Neurosci 27:439–451. doi:10.1016/j.ijdevneu.2009.05.006
Grace L, Hescham S, Kellaway LA, Bugarith K, Russell VA (2009) Effect of exercise on learning and memory in a rat model of developmental stress. Metab Brain Dis 24:643–657. doi:10.1007/s11011-009-9162-5
Griffin EW, Bechara RG, Birch AM, Kelly AM (2009) Exercise enhances hippocampal-dependent learning in the rat: evidence for a BDNF-related mechanism. Hippocampus 19:973–980. doi:10.1002/hipo.20631
Hu S, Ying Z, Gomez-Pinilla F, Frautschy SA (2009) Exercise can increase small heat shock proteins (sHSP) and pre- and post-synaptic proteins in the hippocampus. Brain Res 1249:191–201. doi:10.1016/j.brainres.2008.10.054
Huang CH, Chiang YW, Liang KC, Thompson RF, Liu IY (2010) Extra-cellular signal-regulated kinase 1/2 (ERK1/2) activated in the hippocampal CA1 neurons is critical for retrieval of auditory trace fear memory. Brain Res 1326:143–151. doi:10.1016/j.brainres.2010.02.033
Izquierdo I et al (2006) Different molecular cascades in different sites of the brain control memory consolidation. Trends Neurosci 29:496–505
Kaplan GA, Turrell G, Lynch JW, Everson SA, Helkala EL, Salonen JT (2001) Childhood socioeconomic position and cognitive function in adulthood. Int J Epidemiol 30:256–263. doi:10.1093/ije/30.2.256
Kazl C, Foote LT, Kim MJ, Koh S (2009) Early-life experience alters response of developing brain to seizures. Brain Res 1285:174–181. doi:10.1016/j.brainres.2009.05.082
Leem et al (2011) Chronic exercise ameliorates the neuroinflammation in mice carrying NSE/htau23. Biochem Bioph Res Co 406:359–365
Li W, Keifer J (2009) BDNF-induced synaptic delivery of AMPAR subunits is differentially dependent on NMDA receptors and requires ERK. Neurobiol Learn Mem 91:243–249. doi:10.1016/j.nlm.2008.10.002
Liu YF, Chen HI, Yu L et al (2008) Upregulation of hippocampal TrkB and synaptotagmin is involved in treadmill exercise-enhanced aversive memory in mice. Neurobiol Learn Mem 90:81–89. doi:10.1016/j.nlm.2008.02.005
Lou S, Liu J, Chang H, Chen P (2008) Hippocampal neurogenesis and gene expression depend on exercise intensity in juvenile rats. Brain Res 1210:48–55. doi:10.1016/j.brainres.2008.02.080
Lupien SJ, McEwen BS, Gunnar MR, Heim C (2009) Effects of stress throughout the lifespan on the brain, behaviour and cognition. Nat Rev Neurosci 10:434–445. doi:10.1038/nrn2639
Mabandla MV, Russell VA (2011) Voluntary exercise reduces the neurotoxic effects of 6- hydroxydopamine in maternally separated rats. Metab Brain Dis 211:16–22. doi:10.1016/j.bbr.2010.02.045.Voluntary
Mazzucchelli C, Vantaggiato C, Ciamei A et al (2002) Knockout of ERK1 MAP kinase enhances synaptic plasticity in the striatum and facilitates striatal-mediated learning and memory. Neuron 34:807–820. doi:10.1016/S0896-6273(02)00716-X
Meller E et al (2003) Region-specific effects of acute and repeated restraint stress on the phosphorylation of mitogen-activated protein kinases. Brain Res 979:57–64
Mello PB, Benetti F, Cammarota M, Izquierdo I (2009) Physical exercise can reverse the deficit in fear memory induced by maternal deprivation. Neurobiol Learn Mem 92:364–369. doi:10.1016/j.nlm.2009.04.004
Musazzi L, Mallei A, Tardito D, Gruber SHM, El Khoury A, Racagni G, Mathé AA et al (2009) Early-life stress and antidepressant treatment involve synaptic signalling and Erk kinases in a gene-environment model of depression. J Psych Res doi:10:1016/j.jpsychires.2009.11.008
O’Callaghan RM, Ohle R, Kelly AM (2007) The effects of forced exercise on hippocampal plasticity in the rat: A comparison of LTP, spatial- and non-spatial learning. Behav Brain Res 176:362–366
Oreland S, Nylander I, Pickering C (2010) Prolonged maternal separation decreases granule cell number in the dentate gyrus of 3-week-old male rats. Int J Dev Neurosci 28:139–144. doi:10.1016/j.ijdevneu.2009.12.005
Purcell AL, Sharma SK, Bagnall MW, Sutton MA, Carew TJ (2003) Activation of a tyrosine kinase-MAPK cascade enhances the induction of long-term synaptic facilitation and long-term memory in Aplysia. Neuron 37:473–484. doi:10.1016/S0896-6273(03)00030-8
Runyan JD, Dash PK (2004) Intra-medial prefrontal administration of SCH-23390 attenuates ERK phosphorylation and long-term memory for trace fear conditioning in rats. Neurobiol Learn Mem 82:65–70. doi:10.1016/j.nlm.2004.04.006
Sanderson DJ, Gray A, Simon A et al (2007) Deletion of glutamate receptor-A (GluR-A) AMPA receptor subunits impairs one-trial spatial memory. Behav Neurosci 121:559–569. doi:10.1037/0735-7044.121.3.559
Schmolck H, Kensinger EA, Corkin S, Squire LR (2002) Semantic knowledge in patient H.M. and other patients with bilateral medial and lateral temporal lobe lesions. Hippocampus 12:520–533. doi:10.1002/hipo.10039
Scimeca JC, Servant MJ, Dyer JO, Meloche S (1997) Essential role of calcium in the regulation of MAP kinase phosphatase-1 expression. Oncogene 15:717–725. doi:10.1038/sj.onc.1201231
Stranahan AM, Khalil D, Gould E (2006) Social isolation delays the positive effects of running on adult neurogenesis. Nat Neurosci 9:526–533. doi:10.1038/nn1668
Tarpley JW, Shlifer IG, Birnbaum MS, Halladay LR, Blair HT (2009) Bilateral phosphorylation of ERK in the lateral and centrolateral amygdala during unilateral storage of fear memories. Neurosci 164:908–917. doi:10.1016/j.neuroscience.2009.08.071
Tinsley CJ, Narduzzo KE, Ho JW, Barker GR, Brown MW, Warburton EC (2009) A role for calcium-calmodulin-dependent protein kinase II in the consolidation of visual object recognition memory. Eur J Neurosci 30:1128–1139. doi:10.1111/j.1460-9568.2009.06917.x
Vaynman S, Ying Z, Gomez-Pinilla F (2004) Hippocampal BDNF mediates the efficacy of exercise on synaptic plasticity and cognition. Eur J Neurosci 20:2580–2590. doi:10.1111/j.1460-19568.2004.03720.x
Walf AA, Frye CA (2007) The use of the elevated plus maze as an assay of anxiety-related behavior in rodents. Nat Protoc 2:322–328. doi:10.1038/nprot.2007.44
Acknowledgments
The authors wish to thank Dr Sharon Prince for technical advice, Mr Charles Harris for design and construction of essential equipment, and Ms Nuraan Ismail and Mr Buyisile Dingalibala, for assistance with animal care. This study was supported by the National Research Foundation and the National Institutes of Health Fogarty International Centre grant R01TW008040 to Michael J Zigmond, principal investigator.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Makena, N., Bugarith, K. & Russell, V.A. Maternal separation enhances object location memory and prevents exercise-induced MAPK/ERK signalling in adult Sprague–Dawley rats. Metab Brain Dis 27, 377–385 (2012). https://doi.org/10.1007/s11011-012-9298-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-012-9298-6