Abstract
In the present work we measured blood levels of total homocysteine (tHcy), vitamin B12 and folic acid in patients with Parkinson´s disease (PD) and in age-matched controls and searched for possible associations between these levels with smoking, alcohol consumption, L-DOPA treatment and disease duration in PD patients. We initially observed that plasma tHcy levels were increased by around 30 % in patients affected by PD compared to controls. Linear correlation, multiple regression and comparative analyses revealed that the major determinant of the increased plasma concentrations of tHcy in PD patients was folic acid deficiency, whereas in controls tHcy levels were mainly determined by plasma vitamin B12 concentrations. We also observed that alcohol consumption, gender and L-DOPA treatment did not significantly alter plasma tHcy, folic acid and vitamin B12 levels in parkinsonians. Furthermore, disease duration was positively associated with tHcy levels and smoking was linked with a deficit of folic acid in PD patients. Considering the potential synergistic deleterious effects of Hcy increase and folate deficiency on the central nervous system, we postulate that folic acid should be supplemented to patients affected by PD in order to normalize blood Hcy and folate levels, therefore potentially avoiding these risk factors for neurologic deterioration in this disorder.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Parkinson´s disease (PD) is one of the most common neurodegenerative disorders. It is characterized by progressive loss of dopaminergic neurons in the substancia nigra. Cognitive deterioration is found in approximately one-firth of individuals affected by PD who progressively develop dementia (Aarsland et al. 2001). However, the pathogenesis and the variability of the motor and cognitive deterioration are still unknown and there is a need to define the modifiable risk factors to prevent the progression of this disease and their evolution to neurodegeneration.
However, although the exact causes of PD are not fully established, genetic and environmental factors leading to oxidative stress, mitochondrial dysfunction and apoptosis have been associated with its pathogenesis (Jenner and Olanow 1996; de Rijk et al. 1997; Langston 1998; Duan and Mattson 1999; Taylor et al. 1999; Jenner 1998; Silva et al. 2000; Tatton 2000; Lau et al. 2005; Hancock et al. 2007).
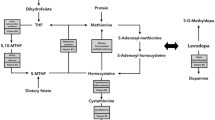
A few studies have demonstrated that homocysteine (Hcy) levels are increased in blood of PD patients and some investigators emphasized the role of levodopa (L-DOPA), the mainstay of the therapy for PD patients, causing hyperhomocysteinemia in these patients (Kuhn et al. 1998; Yasui et al. 2000; Rogers et al. 2003; Isobe et al. 2005). In contrast, other studies did not confirm this association (Todorovic et al. 2006). Hcy is an amino acid arising from methylation of methionine that does not participate in protein synthesis. Recently, it has received especial attention because of its association with pathogenesis of various cerebrovascular, cardiovascular and neurological disorders, including Alzheimer´s disease and PD (Clarke et al. 1998; Kuhn et al. 1998; Hankey and Eikelboom 1999; Muller et al. 1999; Diaz-Arrastia 2000; O’ Suilleabhain et al. 2004).
The major causes of hyperhomocysteinemia in the general population are deficiencies of folic acid, vitamin B12, and vitamin B6 that are necessary for Hcy metabolism (Jacques et al. 2001; de Bree et al. 2002; Castro et al. 2006; Selhub 2006). Furthermore, deficiencies of folic acid and vitamin B12 are associated with degeneration of neurons (Scott et al. 1981; Bottiglieri 1996) and lead to increased Hcy blood levels, which may induce neurotoxic effects through oxidative stress and excitotoxicity (Sachdev 2005; Obeid and Herrman 2006). On the other hand, clinical studies have demonstrated that plasma Hcy levels are positively correlated with alcohol levels (Bleich et al. 2000). Hcy rise may result from reduced activity of the enzyme methionine synthase caused by acetaldehyde, a by-product of ethanol (Barack et al. 2001). Smoking has been also associated with high levels of Hcy, but this is still on debate (Jacques et al. 2001; Quik 2004; Siniscalchi et al. 2006).
Given the inconsistencies across the literature on the relevance of possible factors inducing Hcy increase in blood of PD patients (Jacques et al. 2001; Kuhn et al. 2001; Quik 2004; Siniscalchi et al. 2006; Todorovic et al. 2006), the initial objective of the present investigation was to compare Hcy levels in blood of PD patients with those of age-matched healthy subjects. We also searched for the interplay between Hcy, folic acid and vitamin B12 levels with smoking, alcohol intake, L-DOPA treatment and disease duration in PD patients in order to clarify the major determinant factors of blood Hcy levels.
Material and methods
Fifty PD patients under L-DOPA treatment and 19 with distinct therapy, as well as 52 age-matched healthy individuals were initially recruited from the outpatient clinics of Hospital de Clínicas de Porto Alegre and Hospital São Lucas. All subjects gave their consent to participate in the study. The Ethics Committee of Hospital de Clínicas de Porto Alegre approved this investigation. The diagnosis of PD was established by a neurologist based on the presence of at least two cardinal signs plus unequivocal response to L-DOPA. Unified Parkinson´s Disease Rating Scale (UPDRS)-motor score; Hoen and Yahr Straging (HY) and Mini Mental State Examination were used. We excluded the subjects with conditions that could affect tHcy levels, such as cancer, renal failure, vascular disease, diabetes mellitus, hypertension, malnutrition, use of anticonvulsants or antifolate drugs, neurological and psychiatric diseases other than PD and individuals with clinical signs of deficiencies of vitamins B6, B12 or folic acid.
L-DOPA was administered to some patients at a daily dose ranging from 250–1250 mg/day (mean ± S.E.M: 683 ± 162 mg/day).
Analyses of total serum level of homocysteine (tHcy), folic acid and vitamin B12 were performed in all individuals (PD patients and controls). Each participant was fasting and off medication for at least 6 h. Thus, we avoided influence of acute L-DOPA/dopaminergic drugs intake.
tHcy levels were estimated by stable isotope dilution and electrospray tandem mass spectrometry (LC-MS-MS) according to Magera et al. (1999). Briefly, plasma (100 µl) was mixed with 20 µl homocystine-d8 (2 nmol). Homocystine-d8 was reduced to homocysteine-d4 –(Hcy−d4) by the addition of 20 µl of 500 mmol/L dithiotreitol and proteins were precipitated by addition of 200 µl of a solution of 1 mL/L formic acid and 0.5 mL/L trifluoroacetic acid in acetonitrile. After centrifugation, 100 µl of the supernatant were transferred to a vial. Calibration curve was prepared in plasma of known tHcy concentration by the addition of 200 µmol/L working solution corresponding to tHcy concentrations of 0–50 µmol/L. A bench top triple quadrupole mass spectrometer (Quatro LC Micromass) was used and the analysis was performed in the multiple reaction monitoring mode in which tHcy and Hcy-d4 were detected through the transition from the precursor to the product ion (m/z 136 to m/z 90 and m/z 140 to m/z 94, respectively). The retention time of tHcy and Hcy-d4 was 5.0 min in a 7.0 min analysis. Plasma reference values ranged from 5 to 15 µmol/L.
Serum folic acid levels were determined using the folate assay kit (Folate II) in a cobas analyser that employs a competitive test principle using natural folate binding protein (FBP) specific for folate. Folate present in the sample competes with the added folate (labeled with biotin) for the binding sites on FBP (labeled with ruthenium complex). Normal ranges were from 3.1 to 17.5 ng/mL.
Vitamin B12 serum levels were measured by using the electrochemiluminescence immunoassay ECLIA in a cobas analyzer. The assay employs a competitive test principle using intrinsic factor specific for vitamin B12. Vitamin B12 in the sample competes with the added vitamin B12 labeled with biotin for the binding sites on the ruthenium-labeled intrinsic factor complex. Normal values range from 156 to 698 pmol/L.
One-way analysis of variance (ANOVA) with post-hoc Duncan multiple comparison and two-tailed t-test for unpaired samples were used in analysis of parametric data and chi-square for nonparametric data. In addition, parametric Pearson linear correlation and multiple regression analysis were also used to detect associations between variables. All analyses were performed using the Statistical Package for the Social Sciences (SPSS) software in a PC-compatible computer. Significance was considered when P < 0.05.
Results
Table 1 shows some characteristics of the PD patients and healthy controls regarding age, sex, smoking, alcohol consumption and L-DOPA treatment. We observed that there were more males in PD patients than in healthy control subjects (chi square = 6.557, P < 0.001) and that alcohol consumption was higher in the control group (chi square = 5.223, P < 0.05). In contrast, age and smoking habit did not differ between controls and PD patients.
We first observed that tHcy levels were significantly higher (30 %) in PD patients compared to healthy controls (t (78) = 2.688; P < 0.01] (Fig. 1). Considering that the percentage of males was higher among PD patients (Table 1), we tested whether sex influenced plasma tHcy, folic acid and vitamin B12 serum levels in our sample of PD patients. We found no significant difference between males and females regarding blood concentrations of tHcy, vitamin B12 and folic acid (Table 2). Next, we verified that alcohol consumption did not affect tHcy, folic acid or vitamin B12 serum concentrations in our PD sample (Table 3). However, it can be observed in the tables that males and PD patients using alcohol tend to have higher plasma levels of tHcy with no alterations of serum levels of vitamin B12 and folic acid. Taken together, it was assumed that the increase of plasma tHcy concentrations in our sample of PD patients could not be mainly attributed to gender or alcohol intake differences between these patients.
We also investigated whether smoking influenced blood concentrations of tHcy, vitamin B12 and folic acid in PD patients. We verified that PD smokers presented a marked reduction of folic acid (t (41) = 2.504; P < 0.05), as well as a nonsignificant increase of tHcy (Table 4).
We then examined in more details the influence of folic acid on tHcy levels in PD patients and controls. For this experiment, we divided patients and controls into two groups (with serum levels of folic acid below and above13 ng/mL), and then compared tHcy levels between patients and controls. We observed that PD patients presented tHcy levels significantly higher than controls when folic acid levels were below 13 ng/mL (t (40) = 2.713; P < 0.0001). In contrast, no difference between plasma tHcy levels in controls and patients occurred when folic acid concentrations were higher than 13 ng/mL (Table 5). These data indicate that folic acid deficiency is associated with the increased levels of tHcy in PD patients.
We also studied the influence of L-DOPA treatment on blood levels of tHcy, vitamin B12 and folic acid in PD patients. We observed no significant difference between these levels in L-DOPA-treated and non-treated patients [(Hcy: (t (41) = 0.290; p > 0.05); vitamin B12: (t (41) = 0.043; p > 0.05); folic acid: (t (41) = 0.040; p > 0.05)]. In addition, plasma tHcy levels were not significantly correlated with L-DOPA doses [r = 0.108, P > 0.05]. In contrast, disease duration was significantly correlated with plasma tHcy levels [r = 0.289; P < 0.05], implying that L-DOPA intake for long periods may induce tHcy increase.
In the next set of the analysis we searched for possible associations between blood tHcy, vitamin B12 and folic acid levels in PD patients and healthy controls. A significant inverse correlation between tHcy and folic acid concentrations, but not with vitamin B12 levels, was observed in PD patients (Fig. 2). Furthermore, controls presented a significant inverse correlation between tHcy and vitamin B12 (Fig. 3) and no correlation with folic acid levels.
Finally, in order to better clarify the relative influence of folic acid and vitamin B12 on tHcy levels in PD patients and controls, we carried out multiple regression analysis adjusted for age and gender with tHcy as the dependent variable and folic acid and vitamin B12 as the independent variables. We observed that tHcy levels were negatively associated to folic acid levels [β = −0.550; P < 0.05], but not with vitamin B12 levels in PD patients, reinforcing previous findings. In addition, tHcy levels were negatively associated with vitamin B12 levels (β = −0.428; P < 0.05), but not with folic acid concentrations in controls. Taken together, these data support the assumption that tHcy levels were mainly determined by folic acid in our sample of PD patients and by vitamin B12 levels in healthy controls.
Discussion
Parkinson´s disease (PD) is characterized by dysfunction and degeneration of the dopaminergic neurons of substantia nigra, resulting in bradykinesia, tremor and rigidity (Marsden 1994). Although not fully established, PD neuropathology has been related to oxidative stress, mitochondrial dysfunction and apoptosis (Jenner and Olanow 1996; Jenner 1998; Tatton 2000). Furthermore, genetic and environmental factors are involved in the pathogenesis of PD (de Rijk et al. 1997; Langston 1998; Duan and Mattson 1999; Taylor et al. 1999; Silva et al. 2000; Lau et al. 2005; Hancock et al. 2007). However, the exact mechanisms involved in most cases are still a mystery.
More recently, it has been shown that Hcy levels are increased in blood and CSF of PD patients (Kuhn et al. 1998; Yasui et al. 2000; Isobe et al. 2005), but it is not yet known the relevant factors inducing hyperhomocysteinemia in this disease. In this context, recent reports have emphasized the role of L-DOPA treatment increasing blood Hcy levels (Yasui et al. 2000; Rogers et al. 2003), although this is still on debate (Todorovic et al. 2006). A possible mechanisms for this increase was that the biotransformation of L-DOPA to dopamine leads to a depletion of S-adenosylmethionine needed for Hcy conversion to methionine. There is also some data showing that alcohol consumption and tobacco habit induce Hcy increase in the general population, but the data on these findings are rather scarce and also confusing (Bleich et al. 2000; Barack et al. 2001; Jacques et al. 2001; Quik 2004; Siniscalchi et al. 2006). Thus, we aimed here to clarify the interplay of some of these factors in the induction of hyperhomocysteinemia in parkinsonian patients.
Therefore, our investigation initially compared the blood levels of tHcy, between PD patients and age-matched healthy individuals. We found a significant elevation (over 30 %) of plasma tHcy levels in PD patients in comparison with controls, as previously found by others (Kuhn et al. 1998; Muller et al. 1999). Furthermore, we observed that plasma tHcy increased levels were not due to age, gender or reduced glomerular filtration. Since Hcy promptly crosses the BBB (Grieve et al. 1992), it is feasible that high brain Hcy concentrations were achieved in our recruited PD patients, as previously observed (Isobe et al. 2005). Since neurotoxic properties have been attributed to Hcy (Wall et al. 1980; Blount et al. 1997; Lipton et al. 1997; Upchurch et al. 1997; Cher et al. 2001; Sachdev et al. 2002; Streck et al. 2002, 2003; Lazarewicz et al. 2003), it is conceivable that high Hcy concentrations may be deleterious to the brain of PD patients potentially accelerating neurodegeneration in this disease. However, previous experimental evidence has shown that elevated blood or brain concentrations of Hcy are not sufficient to cause neuronal damage (Duan et al. 2002; Kruman et al. 2002) and also does not predict a worse progression to neurologic deterioration in early PD (O’Suilleabhain et al. 2004).
We also demonstrated by linear correlation, multiple regression and by comparison of tHcy levels between controls and PD patients with low and high blood folic acid concentrations that folate deficiency was an important determinant of hyperhomocysteinemia in our PD patients, distinctly from vitamin B12 which was inversely related to tHcy concentrations in healthy subjects.
To our knowledge the present work demonstrates for the first time that folate deficiency and hyperhomocysteinemia are associated in PD patients. With respect to the pathophysiological importance of these data, it has been previously demonstrated that folate deficiency and elevated levels of Hcy are both necessary for the degeneration of dopaminergic neurons in an experimental model of PD (Duan et al. 2002). Furthermore, considering that low folate concentrations together with high tHcy levels were associated with decline of cognition in old people (Mooijaart et al. 2005), it is conceivable that high Hcy levels may be a risk factor enhancing neuronal susceptibility to other neurotoxic insults, such as folate deficiency in parkinsonian patients. Further studies are however necessary to better clarify this point.
It has been shown that smoking is associated with high tHcy levels (Jacques et al. 2001; Bazzano et al. 2003; Lau et al. 2005), but other investigators have shown that nicotine may be (Jansoon and Jankovic 1985; Kirch et al. 1988; Fowler et al. 1996). Our present data revealed a nonsignificant increase of tHcy in PD patients that actively smoked compared to nonsmokers, but more important folic acid levels were markedly decreased in PD smokers. Saw and colleagues (2001) elegantly demonstrated that the increase of plasma Hcy levels in smokers disappeared after correction for plasma folate indicating that tobacco-induced folate deficiency rather than tobacco per se is a more important cause of hyperhomocysteinemia. Thus, the observations of a significant decrease of folic acid concentrations especially in PD smokers probably contributed to the elevated serum tHcy concentrations in these patients.
Chronic alcoholism is also able to induce high Hcy levels (Van der Gaag et al. 2000; Blasco et al. 2005), although moderate alcohol consumption does not alter blood Hcy concentrations (Beulens et al. 2005) and tHcy levels become normal after two weeks of alcohol abstinence (Hultberg et al. 1993). In our study, we observed a moderate nonsignificant increase of tHcy levels in patients that had lately consumed alcohol (less than a week of blood collection). This moderate increase was not due to hepatic damage, as revealed by normal liver tests, but could possibly be due to the reduction of methionine synthase activity caused by acetaldehyde, a by-product of ethanol (Barack et al. 2001).
We also found that L-DOPA treatment was not associated with hyperhomocysteinemia in the PD patients, which is in accordance with some studies (Bladini et al. 2001; O’Suileabhain et al. 2004; Todorovic et al. 2006) and disagree with other investigations (Yasui et al. 2000; Rogers et al. 2003; Hassin-Bauer et al. 2006; Religa et al. 2006). These conflicting results may be due to the variable daily amount of L-DOPA intake. Our patients took an average daily dose of L-DOPA of about 600 mg, which is low compared to other studies. It is conceivable that L-DOPA treatment is not the main cause of hyperhomocysteinemia in PD, but rather L-DOPA treatment allied to other intrinsic and exogenous factors, including folate deficiency, as here demonstrated (Rogers et al. 2003; Lamberti et al. 2005; Zoccolella et al. 2005). Another contributing factor to high plasma tHcy levels in PD could be the duration of treatment and/or disease, being hypothesized that tHcy higher levels occur because patients ingest L-DOPA for longer periods (Religa et al. 2006). In this context, we found a positive correlation between blood tHcy levels and duration of the disease.
To conclude, our study shows that tHcy levels are increased in PD patients and that this increase if strongly associated with a reduction of folic acid levels. Keeping normal folic acid levels through intake of food rich in this vitamin or direct folate supplementation in PD patients may prevent the increase of blood tHcy levels, potentially avoiding the cognitive symptoms and neurodegeneration resulting from hyperhomocysteinemia and folate deficiency in the PD population (Bottiglieri 1996; Kruman et al. 2002; O’Suileabhain et al. 2004; Kado et al. 2005). It should be emphasized that folic acid is actively transported through the blood-brain barrier and is present at higher concentrations in the CSF than in serum, implying an important functional role in the brain (Herbert and Zalusky 1962). This is a simple, cheap intervention free of side effects that could hypothetically also reduce the hazard for both ischaemic heart and cerebrovascular diseases in PD patients (Muller et al. 2003). However, the evaluation of such anti-hyperhomocysteinemia strategies in prevention of dementia and vascular episodes in PD requires additional studies.
References
Aarsland D, Andersen K, Larsen JP, Lolk A, Nielsen H, Kragh-Sorensen P (2001) Risk of dementia in Parkinson’s disease: a community-based, prospective study. Neurology 56:730–736
Barack AJ, Beckenhauer HC, Kharbanda KK, Tuma DJ (2001) Chronic ethanol consumption increases homocysteine accumulation in hepatocytes. Alcohol 2:77–81
Bazzano LA, He J, Munter P, Vupputuri S, Whelton PK (2003) Relationship between cigarette smoking and novel risk factors for cardiovascular disease in United Stated. Ann Intern Med 138:891–897
Beulens JW, Sierkasma A, Schoafsma G, Kok FJ, Struys EA, Jakobs C, Hendriks HF (2005) Kinetics of Hcy metabolism after moderate alcohol consumption. Alcohol Clin Exp Res 29:739–745
Bladini F, Fancellu R, Martignonim E, Magiagalli A, Pacchetti C, Samuele A, Nappi G (2001) Plasma homocysteine and l-dopa metabolism in patients with Parkinson disease. Clin Chem 47:1102–1104
Blasco C, Caballeria J, Deulofen R, Lligona A, Pares A, Lluis JM, Gual A, Rodes J (2005) Prevalence and mechanisms of hyperhomocysteinemia in chronic alcoholics. Alcohol Clin. Exp. 29:739–745
Bleich S, Degner D, Bandelow B, Von Ahsen N, Ruther E, Kornhuber J (2000) Plasma homocysteine is a predictor of alcohol withdrawal seizures. NeuroReport 11:2749–2752
Blount BC, Mack MM, Wehr CM, Macgregor JT, Hiatt RA, Wang G, Wickramasinghe SN, Everson RB, Ames BN (1997) Folate deficiency causes uracil misincorporation into human DNA and chromosome breakage: implication for cancer and neuronal damage. Proc Natl Acad Sci USA 94:3290–3295
Bottiglieri T (1996) Folate, vitamin B12 and neuropsychiatric disorders. Nutr Rev 54:382–390
Castro R, Rivera I, Blom HJ, Jakobs C, Tavares de Almeida I (2006) Homocysteine metabolism, hyperhomocysteinaemia and vascular disease: an overview. J Inherit Metab Dis 29:3–20
Cher CL, Huanh RF, Chen YH, Cheng JT, Liu TZ (2001) Folate deficiency-induced oxidative stress and apoptosis are cognitive mediated via homocysteine-dependent overproduction of hydrogen peroxide and enhenced activation of NF-kappa B in human Hep G2 cells. Biomed Pharmacother 55:434–442
Clarke R, Smith AD, Jobst KA, Refsum H, Sutton L, Ueland PM (1998) Folate, vitamin B12, and serum total homocysteine levels in confirmed Alzheimer disease. Arch Neurol 55:1449–1455
de Bree A, Verscuren WM, Kromhout D, Kluijtmans LA, Blom HJ (2002) Homocysteine determinants and the evidence to what extent homocysteine determinants the risk of coronary heart disease. Pharmacol Rev 54:599–618
de Rijk MC, Breteler MM, den Breeijen JH, Launer LJ, Grobbee DE, van der Meché FG, Hofman A (1997) Dietary antioxidants and Parkinson disease. The Rotterdam Study. Arch Neurol 54:762–765
Diaz-Arrastia R (2000) Homocysteine and neurologic disease. Arch Neurol 57:1422–1427
Duan W, Mattson MP (1999) Dietary restriction and 2-deoxy-glucose administration improve behavioral outcome and reduce degeneration of dopaminergic neurons in models of Parkinson’s disease. J Neurosci Res 57:195–206
Duan W, Ladenhein B, Culter RG, Kruman II, Cadet JL, Mattson MP (2002) Dietary folate deficiency and elevated homocysteine levels endanger dopaminergicneurons in models of Parkinson’s disease. J Neurochem. 80:101–110
Fowler JS, Volkow ND, Wang GJ, Pappas N, Logan J, Macgregor R, Alexoff D, Shea C, Schlyer D, Wolf AP, Warner D, Zezulkova I, Cilento R (1996) Inhibition of Monoamine-Oxidase-B in the Brains of Smokers. Nature 379:733–736
Grieve A, Butcher SP, Griffiths R (1992) Symaptosomal plasma membrane transport of excitatory sulphur amino acid transmitter candidates: kinetic characterisation and analysis of carrier specificity. J Neurosci Res 32:60–68
Hancock DB, Martin ER, Stajich JM, Jewett R, Stacy MA, Scott BL, Vance JM, Scott WK (2007) Smoking, caffeine, and nonsteroidal anti-inflammatory drugs in families with Parkinson disease. Arch Neurol 64:576–580
Hankey GJ, Eikelboom JW (1999) Homocysteine and vascular disease. Lancet 354:407–413
Hassin-Bauer S, Coben O, Vakil E, Sela B, Nitan Z, Schwartz R, Chapman J, Tanne D (2006) Plasma homocysteine levels and Parkinson’s disease: disease progression, carotid intima-media thickness and neuropsychiatric complications. Clin Neuropharmacol 29:305–311
Herbert V, Zalusky R (1962) Interrrelation of vitamin B12 and folic metabolism: folic acid clearance studies. J Clin Invest 41:1263–1276
Hultberg B, Berdlung M, Aandersson A, Frank A (1993) Elevated plasma homocysteine in alcoholics. Alcohol Clin Exp Res 17:687–689
Isobe C, Murata T, Sato C, Terayama Y (2005) Increase of total homocysteine concentration in cereprospinal fluid in patients with Alzheimer’s disease and Parkinson’s disease. Life Sci 15:1836–1843
Jacques PF, Bostom AG, Wilsin PW, Rich S, Rosenberg IH, Selhub J (2001) Determinants of plasma total homocysteine in the Framingham Offspring cohort. Am J Clin Nutr 73:613–621
Jansson B, Jankovic J (1985) Low Cancer Rates among Patients with Parkinson’s Disease. Ann Neurol 17:505–509
Jenner P (1998) Oxidative Mechanisms in Nigral Cell Death in Parkinson’s Disease. Mov Disord 13:24–34
Jenner P, Olanow CW (1996) Oxidative Stress and the Pathogenesis of Parkinson’s Disease. Neurology 47:161–170
Kado DM, Karlamangla AS, Huang MH, Troen A, Rowe JW, Selhub J, Seeman TE (2005) Homocysteine versus the vitamins folate, B6 and B12 as predictors of cognitive function and decline in older high-functioning adults: MacArthur Studies of Successful Aging. Am J Med 118:161–167
Kirch DG, Alho AM, Wyatt RJ (1988) Hypothesis: a Nicotine-Dopamine Interaction Linking Smoking with Parkinson’s Disease and Tardive Dyskinesia. Cell Mol Neurobiol 8:285–291
Kruman II, Kumaravel TS, Lohani A, Pedersen WA, Cutler RG, Kruman Y, Haughney N, Lee J, Evans M, Mattson MP (2002) Folic acid deficiency and homocysteine impair DNA repair in hippocampal neurons and sensitize them to amyloid toxicity in animal models of Alzheimer’s disease. J Neurosci. 22:1752–1762
Kuhn W, Roebroek R, Blom H, Van Oppenraaj D, Przuntek H, Kretschmer A, Buttner T, Woitalla D, Muller T (1998) Elevated plasma levels of homocysteine in Parkinson’s disease. Eur Neurol 40:225–227
Kuhn W, Hummel T, Woitalla D, Muller T (2001) Plasma homocysteine and MTHFR C677T genotype in levodopa-trated patients with PD. Neurology 56:281–282
Lamberti P, Zoccolella S, Armenise E, Lamberti SV, Fraddosio A, De Mari M, Iliceto G, Livrea P (2005) Hyperhomocysteinemia in L-dopa treated Parkinson’s disease patients: effect of cobalamin and folate administration. Eur J Neurol 12:365–368
Langston JW (1998) Epidemiology versus genetics in Parkinson’s disease: progress in resolving an age-old debate. Ann Neurol 44:S45–S52
Lau LM, Koudstaal PJ, Van Meurs JBJ, Uitterlinder AG, Hofman A, Breteler MMB (2005) Methylenetetrahydrofolate reductase C677T genotype and PD. Ann Neurol 57:927–930
Lazarewicz JW, Ziembowicz A, Matyja E, Stafiej A, Zieminska E (2003) Homocysteine-evoked 45Ca release in the rabbit hippocampus is mediated by both NMDA and group I metabotropic glutamate receptors: in vivo microdialysis study. Neurochem Res 2:259–269
Lipton SA, Kim WK, Choi YB, Kumar S, D’Emilia DM, Rayudu PV, Arnelle DR, Stamler JS (1997) Neurotoxicity associated with dual actions of homocysteine at the N-methyl-D-aspartate receptor. Proc Natl Acad Sci U.S.A. 94:5923–5928
Magera MJ, Lacey JM, Casetta B, Rinaldo P (1999) Method for the determination of total homocysteine in plasma and urine by stable isotope dilution and electrospray tandem mass spectrometry. Clin Chem 45:1517–1522
Marsden CD (1994) Parkinson’s disease. J Neurol Neurosurg Psychiatry 57:672–681
Mooijaart SP, Gussekloo J, Frolich M, Jolles J, Stott DJ, Westendorp RG, de Craen AJ (2005) Homocysteine, vitamin B12, and folic acid and the risk of cognitive decline in old age: the Leiden 85-Plus study. Am J Clin Nutr 82:866–871
Muller T, Werne B, Fowler B, Kuhn W (1999) Nigral endothelial disfunction, homocysteine, and Parkinson’s disease. Lancet 354:126–127
Muller T, Woitalla D, Kuhn W (2003) Benefit of folic acid supplementation in parkinsonian patients treated qith levodopa. J Neurol Neurosurg Psychiatry 74:549
O’ Suilleabhain PE, Sung V, Hernandez C, Laccritz L, Dewey RB Jr, Bottiglieri T, Diaz-Arrastia R (2004) Elevated plasma homocysteine level in patients with Parkinson disease: motor, affective, and cognitive associations. Arch Neurol 61:865–868
Obeid R, Herrman W (2006) Mechanisms of homocysteine neurotoxixity in neurodegenerative diseases with special reference to dementia. FEBS Lett 580:2994–3005
Quik M (2004) Smoking, nicotine and Parkinson’s disease. Trends Neurosci 27:561–568
Religa D, Czyewski K, Stycznska M, Peplonska B, Lokk J, Chodakowska-Zebrowska M, Stepein K, Winblad B, Barcikowska M (2006) Hyperhomocysteinemia and methylenetetrahydrofolate reductase polymorphism in patients with Parkinson’s disease. Neurosci Lett 404:56–60
Rogers JD, Sanchez-Saffon A, Frol AB, Diaz-Arrastia R (2003) Elevated plasma homocysteine levels in pacients treated with levodopoa: association with vascular disease. Arch Neurol 60:59–64
Sachdev PS (2005) Homocysteine and brain atrophy. Prog Neuropsychopharmacol Biol Psychiatry 29:1152–1161
Sachdev PS, Valenzuela M, Wang XL, Looi JC, Brodaty H (2002) Relationship between plasma homocysteine levels and brain atrophy in healthy elderly individuals. Neurology 58:1539–1541
Saw SM, Yuan JM, Ong CN, Arakawa K, Lee HP, Coetzee GA, Yu MC (2001) Genetic, dietary and other lifestyle determinants of plasma homocysteine concentrations in middle-aged and older Chinese men and woman in Singapore. Am J Clin Nutr 73:232–239
Scott JM, Dinn JJ, Wilson P, Weir DGP (1981) Pathogenesis of subacute combined degeneration: a result of methylgroup deficiency. Lancet 2:334–337
Selhub J (2006) The many facets if hiperhomocysteinemia: studies from the Framingham Cohorts. J Nutr 136:1726–1730
Silva HR, Khan NL, Wood NW (2000) The genetics of Parkinson’s disease. Curr Opin Genet Dev 10:292–298
Siniscalchi A, Gallelli L, Mercuri NB, Ibbadu GF, Sarro G (2006) Role of lifestyle factors on plasma homocysteine levels in Pakinson’s disease patients treated with levodopa. Nutr Neuosci 9:11–6
Streck EL, Zugno AI, Tagliari B, Wannmacher C, Wajner M, Wyse AT (2002) Inhibition of Na+, K+− ATPase activity by the metabolites accumulating in homocystinuria. Metab Brain Dis 17:83–91
Streck EL, Delwing D, Tagliari B, Matte C, Wannmacher CM, Wajner M, Wyse AT (2003) Brain energy metabolism is compromised by the metabolites accumulating in homocystinuria. Neurochem Int 43:597–602
Tatton NA (2000) Increased caspase 3 and bax immunoreactivity accompany nuclear GAPDH translocation and neuronal apoptosis in Parkinson’s disease. Exp Neurol 166:29–43
Taylor CA, Saind-Hilaire MH, Cupples LA, Thomas CA, Burchard AE, Felman RG, Myers RH (1999) Environmental, medical, and family history risk factors for Parkinson’s disease: a New England base control study. Am J Med Genet 88:742–749
Todorovic Z, Dzoljic E, Novakovic I, Mirkovic D, Stojanovic R, Nesic Z, Krajinovic M, Prostran M, Kostci V (2006) Homocysteine serum levels and MTHFH C677T genotyoe in patients with Parkinson’s disease, with and without levopoda therapy. J Neurol Sci 248:56–61
Upchurch GR, Welch GN, Fabian AJ, Freedman JE, Johnson JE, Keany JF, Loscalzo J (1997) Homocysteine decreases bioavailable nitrie oxide by a mechanism involving glutathione peroxidase. J Biol Chem 272:17012–17017
Van der Gaag M, Ubbink JB, Sillanaukee P, Nikkari S, Endriks HFJ (2000) Effect of consumption of red wine, spirits and beer on serum homocysteine. Lancet 355:1522
Wall RT, Harlam JM, Harker LA, Striker GE (1980) Homocysteine induced endothelial cell injury in vitro: a model fot the study of vascular injury. Thromb Res 18:113–121
Yasui K, Kowa H, Nakaso K, Takeshima T, Nakashima K (2000) Plasma homocysteine and MTHFR C6677T genotype in levopoda treated patients with PD. Neurology 55:437–440
Zoccolella S, Lamberti P, Iliceto G, Diroma C, Armenise E, Defazio G, Lamberti SV, Fraddosio A, de Mari M, Livrea P (2005) Plasma homocysteine levels in L-dopa-treated Parkinson’s disease patients with cognitive dysfunctions. Clin Chem Lab Med 43:1107–1110
Acknowledgements
We are grateful by the financial support of CNPq, FAPERGS, PRONEX and the FINEP research grant Rede Instituto Brasileiro de Neurociência (IBN-Net) # 01.06.0842-00.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
dos Santos, E.F., Busanello, E.N.B., Miglioranza, A. et al. Evidence that folic acid deficiency is a major determinant of hyperhomocysteinemia in Parkinson´s disease. Metab Brain Dis 24, 257–269 (2009). https://doi.org/10.1007/s11011-009-9139-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-009-9139-4