Abstract
Transgenerational inheritance of various diseases and phenotypes has been demonstrated in diverse species and involves various epigenetic markers. Obesity and malnourishment are nutritional stresses that have effects on offspring through increasing their risk of diabetes and/or obesity. Obesity and malnourishment both affect glucose metabolism and alter oxidative stress parameters in key organs. We induced obesity and malnutrition in F0 female rats by the use of obesogenic diet and protein-deficient diet, respectively. F0 obese and malnourished females were mated with control males and their offspring (F1 generation) were maintained on control diets. The male and female F1 offspring were mated with controls and the resultant offspring (F2 generation) were maintained on control diet. Glucose-sensing markers, glucose metabolism, indicators of insulin resistance and oxidative stress parameters were assessed during fetal development and till the adulthood of the offspring. Glucose-sensing genes were significantly over-expressed in distinct fetal tissues of F2 offspring of malnourished F1 females (F2-MF1F), specifically in fetal pancreas, liver, and adipose tissue. Nuclear and mitochondrial 8-oxo-dG DNA content was significantly elevated in F2-MF1F fetal pancreas. Maternal FBG was significantly elevated in F2-MF1F and F2 offspring of obese F1 females (F2-OF1F) during pregnancy. Males and females offspring of F2-OF1 exhibited significantly elevated FBG and impaired OGTT. Offspring of F2-MF1F showed similar results, while that of F2-MF1M did not significantly deviate from controls. F2-OF1F and F2-MF1F offspring exhibited significant deviation in insulin levels and HOMA-IR levels from controls. Malnourishment has a stronger transgenerational effect through maternal line compared to obesity and malnourishment through paternal line in increasing risk of diabetes in F2 generation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Transgenerational inheritance of diseases and phenotypes has been demonstrated across diverse species and organisms of various evolutionary backgrounds [1]. Diabetes risk is not only determined by genetic and environmental factors, but in utero environment plays a significant role in the risk of diabetes as well [2]. Barker and Hales proposed the “thrifty phenotype” hypothesis, which maintains that the fetus responds to maternal malnutrition by decreasing insulin secretion and sensitivity. When these offspring are faced by surplus of nutrients, they fail to adapt efficiently leading to catchup growth and obesity, which increase the risk of adult-onset diabetes [3]. Manifold mechanisms have been suggested to mediate fetal programming effects observed in epidemiological studies, such as tissue remodeling [4], feto-placental endocrine exchange [5], and epigenetic mechanisms [6].
Nutritional stress during gestation—manifested as malnourishment or obesity—is correlated with increased risk of obesity in offspring. Epidemiological studies showed that obesity increases the risk of diabetes in the affected organism and in its offspring [7]. Total weight gain during pregnancy should not exceed 20 pounds for obese females regardless of their ethnicity or age [8]. Obesity during pregnancy is a major risk factor for diabetes in offspring and increases the risk of distinct pregnancy complications [9]. Malnourishment effects are pronounced and their transgenerational effect has been demonstrated in numerous studies conducted on humans [10, 11].
Obesity and malnourishment (protein-deficient diet) affect glucose metabolism and alter its homeostasis [12, 13]. Maternal glucose concentration during gestation affects various aspects of pregnancy as well as fetal growth in humans [14]. Obesity and malnourishment both affect glucose metabolism during gestation in rats [15], and a number of parameters were exploited to evaluate this effect. Glucose transporter 2 (GLUT2) is a major glucose transporter expressed in various tissues most notably liver and pancreas, and plays an important role in glucose sensing. GLUT2 plays a seminal role in controlling insulin secretion by pancreatic β-cells in mice [16]. Glucose utilization by cells is initiated by its phosphorylation by the enzyme glucokinase (GK), which is the rate-limiting step in glycolysis [17]. The impairment in glucose metabolism and the subsequent hyperglycemia may mediate the increased oxidative stress and its associated cellular damage. Moreover, defined parameters are used to evaluate oxidative stress by assessing oxidized protein and nucleic acid content. Increased oxidative stress can damage DNA through modification of bases such as formation of 8-oxo-guanine [18].
Mitochondria are the major source of ATP in most cell types. Mitochondria generate reactive oxygen species through oxidative phosphorylation reactions that play a significant role in increasing cellular oxidative stress [19]. Biogenesis of mitochondria is independent of cellular replication. Mitochondrial Transcription factor A (mtTFA) is a nuclear protein that binds mitochondrial DNA (mtDNA) and mediates its replication. mtTFA is expressed in response to various cellular milieus such as glucose level and oxidative stress in human cells and rats [20]. mtDNA copy number changes spatially and temporally depending on the type of tissue involved. Although the exact mechanism of mtDNA copy number regulation is unknown, mild cellular damage as well as glucose levels may affect mtDNA copy number in mammalian cells [21]. Mitochondrial uncoupling protein 2 (UCP2) is a mitochondrial glucose-sensing molecule that is expressed in diverse tissues in humans and has a role in controlling insulin secretion by pancreatic β-cells as well [22]. UCP2 expression is also associated with increased oxidative stress and has a role in mediating mice pancreatic β-cell dysfunction [23].
Previously, we documented that maternal obesity and malnutrition affect glucose sensing and tolerance, mtDNA copy number, and the expression of genes involved in the mitochondrial biogenesis and function in the muscles and adipose tissues of the F1 offspring. Also, we found that male F1 offspring appear to be more sensitive for fetal diabetogenic programming than female rat offspring [24]. In this study, we investigate the transgenerational effects of parental diet of three groups of rats (obese on obesogenic diet, malnourished on protein-deficient diet, and controls on chow diet) on glucose-sensing genes, glucose metabolism parameters, insulin levels, and pancreatic β-cell development in F2 generation of rats. In addition, we investigate the impact of paternal and maternal line on this transgenerational effect.
Materials and methods
Animals
The Institutional Animal Care and Use Committee at the Medical Research Institute—Alexandria University has approved the animal protocol. Female Wistar rats were divided into three groups: obese, malnourished, and controls. Obesity and malnourishment were induced through maintaining neonate rats on obesogenic and low-protein diets, respectively, for 2 months after weaning. The details of diet composition are shown in Table 1 and methods of induction were previously published [24]. We sampled 10 male and 10 female rats for each group that developed obesity or malnourishment. The female rats belonging to each group were mated with healthy males to generate the F1 generation. The F1 generation males of each group (F1 control CF1, F1 obese OF1, and F1 malnourished MF1) were mated with healthy control females, and the females of each group were mated with healthy control males. Pregnancy was confirmed by the presence of vaginal mucus plug next morning. Following mating, all pregnant female rats received control diets (CD). Body weights and fasting blood glucose (FBG) levels were monitored three times a week. All pregnancies (except 10 of each group used for prenatal experiments) were allowed to give birth naturally. Numbers and sex of pups, birth weight, and nasoanal length were measured for each pup on postnatal day (PND) 1. Pups remained with dams until they were weaned and all dams were fed CD ad libitum during lactation. Pups were divided into 5 groups according to the group of the F1 that generated it: F2 offspring of control pregnancy (F2-CF1), F2 offspring of OF1 males (F2-OF1M), F2 offspring of OF1 females (F2-OF1F), F2 offspring of MF1 males (F2-MF1M), and F2 offspring of MF1 females (F2-MF1F). Body weights of the F2 offspring were monitored weekly until PND 210 (30 weeks). The glucose tolerance curve was constructed for each subgroup at 30 weeks postnatally. Eight pups (4 males and 4 females) of each subgroup were used for the construction of OGTT. The plasma was obtained for the assessment of glucose, insulin, and HOMA-IR.
Methods
Isolation of pancreatic β-cells
Pancreas was isolated and placed in a sterile ice-cold HBSS under aseptic conditions followed by removal of gut and spleen residues. The pancreas was chopped into fine pieces and washed twice with HBSS supplemented with penicillin (100 U/ml), streptomycin (0.1 mg/ml), and 5 % HEPES, and digested in 4 ml collagenase solution (2.5 mg/ml in 10 mM HEPES, 7.5 CaCl2, 5 % (v/v) glycerol, pH 7.5) for 40 min at 37 °C. Then, adding 10 ml of ice-cold HBSS stopped the digestion. Digested tissue was filtered through stainless-steel tea strainer (0.5 mm mesh pore size) after washing with ice-cold HBSS. The sample was centrifuged for 2 min at 4 °C at 300×g. The supernatant was discarded and the pellet was re-suspended in Ficoll 25 % with 10 mM HEPES. This was layered over by various concentrations of Ficoll 23, 20.5, and 11 % to form a density gradient. After centrifugation at 300×g for 15 min, the islets were collected at the interface between density 11 and 20.5 %. The islets were separated by centrifugation and then washed three times with HBSS. Then, cells were suspended in RPMI 1640 medium. The purity of the islets was examined by staining with dithoizone [25].
ELISA measurements
ELISA kit (Mercodia, Sweden) was used to measure insulin concentration in plasma.
Nuclear DNA isolation
The nuclear DNA was isolated from fetal tissues using GenJet genomic DNA isolation kit (Fermentas, EU), according to the manufacturer instructions.
Mitochondrial DNA isolation
The mitochondrial DNA was isolated from the pancreatic islets using mitochondrial DNA isolation kit (Bio-Vision, Canada), according to the manufacturer instructions.
Total RNA isolation
Total RNA was isolated from different tissues using GenJet RNA isolation kit (Fermentas, EU) according to the manufacturer’s instructions.
Assay of 8-oxo-deoxy guanine content in nuclear and mitochondrial DNA
Isolated DNA was washed twice with 70 % ethanol, dried, and dissolved in 200 μl of 10 mM Tris/HCl, 0.1 mM EDTA, and 100 mM NaCl (pH 7.0). For enzymatic digestion, 100 units of DNase I was added per 200 μg DNA at 37 °C for 1 h. Thereafter, 5 units of nuclease P1 and 30 μl of 10 mM ZnSO4 were added, and the mixture was incubated for 1 h. After re-adjusting the pH with 100 μl of 0.4 M Tris/HCl (pH 7.8) followed by the addition of 3 units of alkaline phosphatase, the samples were incubated for 30 min. Enzymes were precipitated with acetone, the precipitates were removed by centrifugation, and the supernatant was evaporated to dryness. The DNA hydrolysates were assessed for 8-oxo-dG content using HT 8-oxo-dG ELISA Kit II (Trevigen, USA).
RT-PCR analysis
Gene expression of mtTFA [26], GLUT2, Glucokinase [27], and UCP2 [28] was determined semi-quantitatively by the One-step RT-PCR Master Mix Gold Beads (Bioron, Germany). Primers of UCP2, GLUT2, glucokinase, and mtTFA were purchased from Bioneer Co (Bioneer, USA) (Table 2).
To standardize the amount of mRNA in each sample, RT-PCR of glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was performed in parallel (Table 2). Thereafter, the PCR reaction product was run on 1.5 % agarose and stained with ethidium bromide. The bands were visualized using a UV plate. The bands were scanned, and the data were analyzed via UVP DOC-ITLS™ Image Acquisition and Analysis software (Ultra-Violet product, Ltd. Cambridge, UK) that analyzes the band density relative to the GAPDH band (as internal control).
Mitochondrial DNA copy number
mtDNA content relative to the nuclear gene, peroxisome proliferator-activated receptor-γ coactivator 1α (PGC1α), was measured using real-time PCR as described previously [29] (Table 2). Reactions were carried out using SYBR Green PCR Master Mix (Applied Biosystems), 0.5 μM of each forward and reverse primer, and 50 ng genomic DNA with the following conditions: 95 °C for 10 min followed by 40 cycles of 95 °C for 15 s, 60 °C for 30 s, and 72 °C for 30 s. The relative mtDNA copy number was calculated using the ΔΔCt method as fully described previously [30].
Statistical analysis
All statistical analyses were performed using SPSS statistical software version 18 (SPSS, Chicago, IL). All data are presented as mean ± SD or SEM. The Kolmogorov–Smirnov test was used to verify the normal distribution of the studied variables. One-way analysis of variance (ANOVA) with post hoc test (LSD) was performed on each normally distributed variable to compare the mean values from the offspring of obese and malnourished mothers with the offspring of control mothers. Mann–Whitney U test was used to evaluate the statistical significance of not normally distributed variables (mTFA and UCP2 gene expression and mtDNA copy number) between the different groups. t test was used to compare the mean values of females and males of the same group at the same age. Differences were considered significant at p < 0.05.
Results
FBG level during gestation
The pregnancies of female F1 offspring of maternal obesity (OF1F) and of female F1 offspring of maternal malnutrition (MF1F) mated with control males have a significantly elevated FBG during gestation (Fig. 1). On the other hand, the pregnancies of control females mated with males of F1 offspring of maternal obesity (OF1M) and with males of F1 offspring of maternal malnutrition (MF1M) showed no significant changes in FBG. The number of pregnancy that developed hyperglycemia was null in all groups except the MF1F pregnancies in which 6 out of 15 pregnancies have developed hyperglycemia (Table 3).
Pregnancy outcome
The number of pups per litter was 12 ± 1 for pregnancies of control F1 offspring (CF1), 12 ± 2 for OF1M pregnancies, 11 ± 1 for OF1F pregnancies, 11 ± 2 for MF1M pregnancies, and 10 ± 2 for MF1F pregnancies (Table 3). We did not detect malformations in the offspring of all groups.
Prenatally, fetal glucose-sensing gene expression and 8-oxo-dG DNA content are significantly elevated in F2-MF1F
Fetal pancreas expression of GLUT2 was not changed from controls in F2-OF1M and F2-OF1F fetuses. Moreover, F2-MF1M fetuses did not exhibit significant deviation from GLUT2 expression levels in F2-CF1. However, in F2-MF1F fetuses, GLUT2 was significantly over-expressed. A similar pattern was observed in fetal liver expression of GLUT2, which was significantly upregulated in F2-MF1F fetuses solely (Fig. 2). GK and UCP2 expression results mirror those observed for GLUT2 expression. GK and UCP2 were significantly over-expressed in fetal pancreas of F2-MF1F fetuses. GK upregulation was also observed in fetal liver, while UCP2 over-expression was observed in fetal adipose tissue of F2-MF1F fetuses. In addition, mtTFA and mtDNA copy numbers were not affected in F2-MF1M and F2-OF1M as well as F2-OF1F fetuses. However, mtTFA expression and mtDNA copy number were significantly reduced in fetal pancreas of F2-MF1F fetuses, while in fetal liver and muscle only mtTFA expression was reduced significantly (Fig. 3). Nuclear and mitochondrial 8-oxo-dG levels were significantly increased in fetal pancreas of F2-MF1 fetuses (Fig. 3).
Glucose-sensing gene expression. Data presented as mean ± SEM (n = 10). *Significant difference from F2-CF1 by ANOVA (p < 0.05). Gene expression was assessed semi-quantitatively relative to house-keeping gene GAPDH. a Bar chart showing GLUT2 gene expression in fetal pancreas and fetal liver, b bar chart showing GK gene expression in fetal liver and pancreas of different rat groups, and c bar chart showing UCP2 gene expression in certain fetal tissues
Oxidative stress parameters in various fetal tissues. Data presented as mean ± SEM (n = 15). *Significant difference from F2-CF1 by ANOVA (p < 0.05). a Bar chart showing mtTFA expression in fetal liver, pancreas, skeletal muscle, and adipose tissue of fetuses of the several rat groups, b bar chart showing mtDNA copy number in fetal pancreas of gestation from different rat groups, c bar chart showing nuclear and mitochondrial 8-oxo-dG content per million dG base in fetal pancreas across the different study groups, and d bar chart showing nuclear 8-oxo-dG content per million dG bases in fetal liver, muscle, and adipose tissue in different study groups
Postnatally, F2-MF1F and F2-OF1F offspring have significantly elevated FBG and impaired OGTT
The body weight of the offspring was significantly heavier in males compared to females of the same group, but this effect was evident in rats of all age groups of F2-OF1M and from the age of 10–30 weeks in F2-OF1F as well as F2-MF1F. Moreover, in F2-MF1M males were significantly heavier than females from the age of 20–30 weeks. The only group that showed a significant increase in body weight compared to control regardless of age and gender was the F2-MF1F offspring (Fig. 4).
Changes in body weight with age. Each data point represents mean ± SEM (n = 50). *Significant difference from corresponding rats in F2-CF1 offspring group by ANOVA (p < 0.05). All data points in each curve are significantly different from the opposite sex in the same group by t test (p < 0.05) except if the symbol (#) is present. a Curve showing change in body weight in male and female offspring of F2-CF1 rats, b curve showing change in body weight in male and female offspring of F2-OF1M rats, c curve showing change in body weight in male and female offspring of F2-OF1F rats, d curve showing change in body weight in male and female offspring of F2-MF1M rats, and e curve showing change in body weight in male and female offspring of F2-MF1F rats
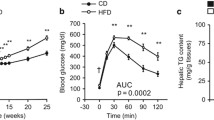
FBG levels were elevated in F2-OF1M and F2-OF1F offspring, but it did not reach statistical significance except in female F2-OF1F offspring at the age of 20 weeks. Moreover, in F2-MF1F FBG was significantly elevated in both male and female rats from the age of 20 weeks till the last measurement (30 weeks) (Table 4). OGTT results measured at the age of 30 weeks were significantly elevated from controls in F2-OF1M, F2-OF1F, and F2-MF1F. However, the deviation from control OGTT values was maintained even at 120 min after glucose administration in F2-OF1F and F2-MF1F, but in F2-OF1M OGTT values were significantly elevated only in male offspring from 30 to 90 min after glucose administration (Fig. 5).
OGTT results in F2 offspring of different study groups conducted at the age of 30 weeks. Each data point represents mean ± SEM (n = 10). *Color coded and indicates significant difference from corresponding rats in F2-CF1 by ANOVA (p < 0.05). a OGTT curve of F2-CF1 male and female offspring, b OGTT curve of F2-OF1M male and female offspring, c OGTT curve of F2-OF1F male and female offspring, d OGTT curve of F2-MF1F male and female offspring, and e OGTT curve of F2-MF1F male and female offspring
F2-OF1F and F2-MF1F offspring have high insulin levels and are insulin resistant
Insulin levels were significantly elevated in females compared to males F2-OF1F, an effect that was observed in all age categories of this group and in some age categories (5, 15, and 25 weeks) in F2-MF1F. Moreover, insulin levels were significantly reduced in male F2-OF1M (age 15 and 20 weeks) and female F2-OF1M (age 10 and 15 weeks). However, in male F2-MF1F insulin levels were significantly elevated from the age of 10 weeks onwards to age of 30 weeks and in female F2-MF1F from the age of 25 weeks till the last measurement (30 weeks) (Table 4). As expected, HOMA-IR levels did not differ between male and female rats of the same group in most offspring categories studied. However, female F2-OF1F had HOMA-IR levels that were significantly higher compared to their male counterparts and F2-CF1 of the same gender. In F2-MF1F, both male and female rats had significantly higher HOMA-IR compared to F2-CF1 at the age of 10 and 15 weeks, respectively. F2-OF1M and F2-MF1M HOMA-IR levels did not exhibit any particular pattern of increase or decrease compared to controls in the majority of measurements (Table 4).
Discussion
Our results indicate that in utero programming effects of maternal diet are transmitted mainly through maternal line rather than paternal line. The effect of maternal malnutrition seems to elicit more profound effect on several glucose sensing and oxidative stress parameters. Although we cannot extend the results of this study to humans, these ideas could be used to guide further clinical studies. Prenatally, the glucose-sensing genes GLUT2, GK, and UCP2 were significantly upregulated in fetal pancreas of F2-MF1F, which indicates impaired glucose levels in gestation. In fact, studies conducted on cultured pancreatic cells have shown that high glucose concentrations induce expression of GLUT2 [31], GK [32], and UCP2 [33] genes. We previously demonstrated similar changes in the F1 offspring of both maternal malnutrition and obesity [24]. The observed prenatal changes in gene expression could be explained by different mechanisms. First, intra-uterine malnutrition or obesity microenvironment during the embryonic development of F1 mothers may induce epigenetic modifications of these genes that pass through ova into the next generation (F2). Second, the gestational diabetes observed in the pregnancies of female F1 offspring of maternal malnutrition and obesity (MF1F and OF1F) could induce these changes in gene expression of glucose-sensing genes. Similar pattern of changes were observed in the F1 fetuses of maternal diabetes [15]. Third, oxidative stress observed in the fetal pancreas (as increased level of 8-oxo-dG) may be a consequence of increased gestational glucose level and may play a role in the disturbed epigenetic regulation.
Female offspring of F2-OF1F exhibited significantly elevated FBG during gestation, which suggests that the transgenerational effect was subtle, and under appropriate stress—such as gestation—impairment of glucose metabolism is observed. Moreover, markers of oxidative stress (nuclear and mitochondrial 8-oxo-dG) were significantly elevated in fetal pancreas of F2-MF1F indicating increased oxidative stress in fetal pancreas. The increase in oxidative stress is known to reduce mtTFA expression [20], which was observed in fetal pancreas of F2-MF1F. mtTFA is involved in mitochondrial DNA replication [21], and thus reduction in mtTFA expression may explain the reduction in mitochondrial DNA copy number. Intriguingly, insulin levels were elevated in F2-MF1F male and female offspring, which suggests that the pancreas was not severely affected in adult rats despite the increased oxidative stress observed during gestation.
In peripheral fetal tissues, similar pattern was observed. Glucose-sensing genes GK and GLUT2 were over-expressed in fetal liver, and UCP2 was over-expressed in fetal adipose tissue of F2-MF1F; both findings indicate that the abnormal intra-uterine environment had an effect on peripheral tissues and might predispose to diabetes. Studies conducted on cultured rat hepatocytes have shown that glucose induces GLUT2 expression in hepatocytes [34]. In addition, UCP2 expression in adipose tissue is induced by an increase in free fatty acids [35] and some studies have shown that UCP2 expression in adipose tissue is instrumental for the metabolic abnormalities observed in diabetes [36]. Liver and adipose tissues play a major role in insulin resistance [37]. In adult F2-MF1F offspring, indicators of insulin resistance were significantly elevated in both males and females. Also insulin levels were significantly elevated in both male and female F2-MF1F offspring.
Our results are in concordance with results of retrospective studies in humans [38]. Maternal malnourishment was correlated with increased mortality from diabetes in grandsons and granddaughters. Moreover, in studies conducted on rats, similar results were observed, where malnutrition during gestation and/or lactation resulted in impaired glucose metabolism and induced insulin resistance in F2 generation [39]. However, other studies have shown that obesity in F1 causes abnormal pancreatic function and insulin resistance in F2 generation [40]. Possible reason for this discrepancy is that in our experiments obesity was induced by a different obesogenic diet fed through a different regimen [24]. High-fat diet per se is a known inducer of diabetes in mice [41], and thus diabetes may confound the effects of obesity alone.
Transgenerational inheritance observed in this study was mainly evident through the maternal and not the paternal line. A possible explanation for this is that a defect may have occurred in mitochondrial DNA of primordial germ cells of the females during pregnancy due to the abnormal intra-uterine environment. Subsequently, this will lead to an alteration in the oocytes leading to an abnormal phenotype in offspring. To confirm this hypothesis, further analysis must be done on mitochondrial DNA changes and their subsequent effects. Another possible hypothesis is that the fetal reprogramming of primordial germ cells that starts in utero may be altered in the female in a manner that prevents insulin-like growth factor II (IGF-II) normal imprinting and causes IGF-II over-expression, which is known to be a major contributor to diabetes acquisition later on in adulthood [42].
Our observations suggest that maternal malnourishment has a stronger transgenerational effect on F2 generation. Maternal malnourishment significantly alters glucose homeostasis in F2 generation. Obesity transgenerational effect was subtle and was observed mainly through maternal line as elevated FBG during gestation, low insulin level, and increased insulin resistance.
Abbreviations
- CD:
-
Control diets
- CF1:
-
Control F1 generation
- FBG:
-
Fasting blood glucose
- F2-CF1:
-
F2 offspring of control pregnancy
- F2-OF1M:
-
F2 offspring of OF1 males
- F2-OF1F:
-
F2 offspring of OF1 females
- F2-MF1M:
-
F2 offspring of MF1 males
- F2-MF1F:
-
F2 offspring of MF1 females
- GLUT2:
-
Glucose transporter 2
- GK:
-
Glucokinase
- HOMA-IR:
-
Homeostatic model assessment of insulin resistance
- IGF-II:
-
Insulin-like growth factor II
- MF1:
-
Malnourished F1 generation
- mtTFA:
-
Mitochondrial transcription factor A
- mtDNA:
-
Mitochondrial DNA
- OF1:
-
Obese F1 generation
- OGTT:
-
Oral glucose tolerance test
- 8-Oxo-dG:
-
8-Oxo-deoxy guanine
- PND:
-
Postnatal day
- UCP2:
-
Uncoupling protein 2
References
Heard E, Martienssen RA (2014) Transgenerational epigenetic inheritance: myths and mechanisms. Cell 157:95–109
Hales CN, Barker DJ (1992) Type 2 (non-insulin-dependent) diabetes mellitus: the thrifty phenotype hypothesis. Diabetologia 35:595–601
Hales CN, Barker DJ (2001) The thrifty phenotype hypothesis. Br Med Bull 60:5–20
Plagemann A, Harder T, Rake A, Melchior K, Rohde W, Dorner G (2000) Hypothalamic nuclei are malformed in weanling offspring of low protein malnourished rat dams. J Nutr 130:2582–2589
Godfrey KM (2002) The role of the placenta in fetal programming—a review. Placenta 23(Suppl A):S20–S27. doi:10.1053/plac.2002.0773
Lillycrop KA, Phillips ES, Jackson AA, Hanson MA, Burdge GC (2005) Dietary protein restriction of pregnant rats induces and folic acid supplementation prevents epigenetic modification of hepatic gene expression in the offspring. J Nutr 135:1382–1386
Dabelea D, Crume T (2011) Maternal environment and the transgenerational cycle of obesity and diabetes. Diabetes 60:1849–1855
Rasmussen KM, Abrams B, Bodnar LM, Butte NF, Catalano PM, Siega-Riz AM (2010) Recommendations for weight gain during pregnancy in the context of the obesity epidemic. Obstet Gynecol 116:1191
Chu SY, Callaghan WM, Kim SY, Schmid CH, Lau J, England LJ, Dietz PM (2007) Maternal obesity and risk of gestational diabetes mellitus. Diabetes Care 30:2070–2076. doi:10.2337/dc06-2559a
Veenendaal MV, Painter RC, Rooij S, Bossuyt PM, Post J, Gluckman PD, Hanson MA, Roseboom TJ (2013) Transgenerational effects of prenatal exposure to the 1944–45 Dutch famine. BJOG 120:548–554
Lagisz M, Blair H, Kenyon P, Uller T, Raubenheimer D, Nakagawa S (2014) Transgenerational effects of caloric restriction on appetite: a meta-analysis. Obes Rev 15:294–309
Okitolonda W, Brichard S, Henquin J-C (1987) Repercussions of chronic protein-calorie malnutrition on glucose homeostasis in the rat. Diabetologia 30:946–951
Singla P, Bardoloi A, Parkash AA (2010) Metabolic effects of obesity: a review. World J Diabetes 1:76
Scholl TO, Sowers M, Chen X, Lenders C (2002) Maternal glucose concentration influences fetal growth, gestation, and pregnancy complications. Obstet Gynecol Surv 57:141–142
Kamel MA (2012) Prenatal effects of different intra-uterine milieus on fetal glucose sensing mechanisms during gestation in rats. J Diabetes Metab 3:181
Thorens B (2015) GLUT2, glucose sensing and glucose homeostasis. Diabetologia 58:221–232
MacDonald PE, Joseph JW, Rorsman P (2005) Glucose-sensing mechanisms in pancreatic β-cells. Philos Trans R Soc Lond B 360:2211–2225
Cooke MS, Evans MD, Dizdaroglu M, Lunec J (2003) Oxidative DNA damage: mechanisms, mutation, and disease. FASEB J 17:1195–1214
James AM, Collins Y, Logan A, Murphy MP (2012) Mitochondrial oxidative stress and the metabolic syndrome. Trends Endocrinol Metab 23:429–434
Choi YS, Kim S, Pak YK (2001) Mitochondrial transcription factor A (mtTFA) and diabetes. Diabetes Res Clin Pract 54:S3–S9
Montier LLC, Deng JJ, Bai Y (2009) Number matters: control of mammalian mitochondrial DNA copy number. J Genet Genomics 36:125–131
Diano S, Horvath TL (2012) Mitochondrial uncoupling protein 2 (UCP2) in glucose and lipid metabolism. Trends Mol Med 18:52–58
Krauss S, Zhang C-Y, Scorrano L, Dalgaard LT, St-Pierre J, Grey ST, Lowell BB (2003) Superoxide-mediated activation of uncoupling protein 2 causes pancreatic β cell dysfunction. J Clin Investig 112:1831
Kamel MA, Helmy MH, Hanafi MY, Mahmoud SA, Elfetooh HA, Badr MS (2014) Maternal obesity and malnutrition in rats differentially affect glucose sensing in the muscles and adipose tissues in the offspring. Int J Biochem Res Rev 4:440–469
Amoli MM, Moosavizadeh R, Larijani B (2005) Optimizing conditions for rat pancreatic islets isolation. Cytotechnology 48:75–78
Piantadosi CA, Suliman HB (2006) Mitochondrial transcription factor A induction by redox activation of nuclear respiratory factor 1. J Biol Chem 281:324–333
Parton LE, Diraison F, Neill SE, Ghosh SK, Rubino MA, Bisi JE, Briscoe CP, Rutter GA (2004) Impact of PPARγ overexpression and activation on pancreatic islet gene expression profile analyzed with oligonucleotide microarrays. Am J Physiol Endocrinol Metab 287:E390–E404
Berraondo B, Marti A, Duncan J, Trayhurn P, Martinez J (2000) Up-regulation of muscle UCP2 gene expression by a new beta3-adrenoceptor agonist, trecadrine, in obese (cafeteria) rodents, but down-regulation in lean animals. Int J Obes Relat Metab Disord 24:156–163
Ciapaite J, Bakker SJ, Van Eikenhorst G, Wagner MJ, Teerlink T, Schalkwijk CG, Fodor M, Ouwens DM, Diamant M, Heine RJ (2007) Functioning of oxidative phosphorylation in liver mitochondria of high-fat diet fed rats. Biochimica et Biophysica Acta (BBA) 1772:307–316
Szuhai K, van den Ouweland JM, Dirks RW, Lemaître M, Truffert J-C, Janssen GM, Tanke HJ, Holme E, Maassen JA, Raap AK (2001) Simultaneous A8344G heteroplasmy and mitochondrial DNA copy number quantification in myoclonus epilepsy and ragged-red fibers (MERRF) syndrome by a multiplex molecular beacon based real-time fluorescence PCR. Nucleic Acids Res 29:e13
Tal M, Liang Y, Najafi H, Lodish H, Matschinsky F (1992) Expression and function of GLUT-1 and GLUT-2 glucose transporter isoforms in cells of cultured rat pancreatic islets. J Biol Chem 267:17241–17247
Liang Y, Najafi H, Smith RM, Zimmerman EC, Magnuson MA, Tal M, Matschinsky FM (1992) Concordant glucose induction of glucokinase, glucose usage, and glucose-stimulated insulin release in pancreatic islets maintained in organ culture. Diabetes 41:792–806
Patanè G, Anello M, Piro S, Vigneri R, Purrello F, Rabuazzo AM (2002) Role of ATP production and uncoupling protein-2 in the insulin secretory defect induced by chronic exposure to high glucose or free fatty acids and effects of peroxisome proliferator-activated receptor-γ inhibition. Diabetes 51:2749–2756
Rencurel F, Waeber G, Antoine B, Rocchiccioli F, Maulard P, Girard J, Leturque A (1996) Requirement of glucose metabolism for regulation of glucose transporter type 2 (GLUT2) gene expression in liver. Biochem J 314:903–909
Camirand A, Vr Marie, Rr Rabelo, Silva JE (1998) Thiazolidinediones stimulate uncoupling protein-2 expression in cell lines representing white and brown adipose tissues and skeletal muscle. Endocrinology 139:428–431
De Souza CT, Araújo EP, Stoppiglia LF, Pauli JR, Ropelle E, Rocco SA, Marin RM, Franchini KG, Carvalheira JB, Saad MJ (2007) Inhibition of UCP2 expression reverses diet-induced diabetes mellitus by effects on both insulin secretion and action. FASEB J 21:1153–1163
Hajer GR, van Haeften TW, Visseren FL (2008) Adipose tissue dysfunction in obesity, diabetes, and vascular diseases. Eur Heart J 29:2959–2971
Kaati G, Bygren LO, Edvinsson S (2002) Cardiovascular and diabetes mortality determined by nutrition during parents’ and grandparents’ slow growth period. Eur J Hum Genet 10:682–688
Pinheiro A, Salvucci I, Aguila M, Mandarim-de-Lacerda C (2008) Protein restriction during gestation and/or lactation causes adverse transgenerational effects on biometry and glucose metabolism in F1 and F2 progenies of rats. Clin Sci 114:381–392
Graus-Nunes F, Frantz EDC, Lannes WR, da Silva Menezes MC, Mandarim-de-Lacerda CA, Souza-Mello V (2015) Pregestational maternal obesity impairs endocrine pancreas in male F1 and F2 progeny. Nutrition 31:380–387
Winzell MS, Ahrén B (2004) The high-fat diet–fed mouse a model for studying mechanisms and treatment of impaired glucose tolerance and type 2 diabetes. Diabetes 53:S215–S219
Devedjian J-C, George M, Casellas A, Pujol A, Visa J, Pelegrín M, Gros L, Bosch F (2000) Transgenic mice overexpressing insulin-like growth factor-II in β cells develop type 2 diabetes. J Clin Investig 105:731
Acknowledgments
This study is part of a project entitled “Intra-Uterine programming of adult diabetes: an experimental study” supported by the Science and Technology Development Fund (STDF)—Egypt.
Author’s contribution
Maher A. Kamel has conceived and designed the study, carried out the analysis of data, and contributed to the writing and revision of the manuscript. Mervat Y. Hanafi, Moustafa M. Saleh, Mohamed I. Saad, and Taha M. Abdelkhalek have conducted most of the laboratory investigations and contributed to the writing and revision of the manuscript. All authors have read and approved the final manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None declared.
Rights and permissions
About this article
Cite this article
Hanafi, M.Y., Saleh, M.M., Saad, M.I. et al. Transgenerational effects of obesity and malnourishment on diabetes risk in F2 generation. Mol Cell Biochem 412, 269–280 (2016). https://doi.org/10.1007/s11010-015-2633-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-015-2633-6