Abstract
Glioma-associated oncogene homolog-1 (Gli-1) is considered a marker of Hedgehog pathway activation and is associated with the progression of several cancers. We have previously reported that Gli-1 was correlated with invasion and metastasis in hepatocellular carcinoma (HCC). However, the exact roles and mechanisms of Gli-1 in HCC invasion are unclear. In this study, we found that small interfering RNA mediated down-regulation of Gli-1 expression significantly suppressed adhesion, motility, migration, and invasion of both SMMC-7721 and SK-Hep1 cells. Furthermore, down-regulation of Gli-1 significantly reduced expressions and activities of both matrix metalloproteinase (MMP)-2 and MMP-9. In addition, we found that down-regulation of Gli-1 resulted in up-regulation of E-cadherin and concomitant down-regulation of Snail and Vimentin, consistent with inhibition of epithelial-mesenchymal transition (EMT). Taken together, our results suggest that down-regulation of Gli-1 suppresses HCC cell migration and invasion likely through inhibiting expressions and activations of MMP-2, 9 and blocking EMT.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Hepatocellular carcinoma (HCC) is one of the most prevalent cancers and the third most common cause of cancer-related death worldwide [1]. Invasion and metastasis are characteristic features of HCC and the main cause of death in patients with HCC [2]. However, the underlying mechanisms of HCC invasion and metastasis are still not fully understood. To further elucidate the molecular mechanisms underlying invasion and metastasis of HCC will help to identify novel potential therapeutic targets and consequently improve its prognosis.
Hedgehog (Hh) is a secretory signaling protein that was initially isolated from Drosophila [3]. Hh signaling is initiated when Hh binds to the transmembrane receptor Patched (Ptch), which in turn relieves Ptch-mediated repression of Smoothened (Smo), a seven-transmembrane protein. The derepression of Smo triggers intracellular signal transduction cascade that activates the transcription of Hh target gene through activation of a family of zinc-finger transcription factors, the glioma-associated oncogenes (Gli) [4, 5]. Constitutive activation of the Hh signaling has been implicated in various human malignancies [5–7]. In our earlier report, we showed that Hh signaling pathway induced cell migration and invasion of HCC [8].
In vertebrates, there are three Gli proteins: glioma-associated oncogene homolog-1 (Gli-1), Gli-2, and Gli-3. Among these Gli, Gli-1 acts as a strong activator to mediate and/or amplify the Hh response and is itself a transcriptional target of Hh pathway [9, 10]. Therefore, Gli-1 is considered to be a reliable marker of Hh pathway activation [10–14]. Accumulating evidences have shown that Gli-1 is sufficient to induce basal cell carcinoma (BCC)-like skin tumors and hyperproliferation of central nervous system (CNS) [15, 16]. Furthermore, Gli-1 has been implicated in the progression of several cancers including pancreatic cancer [17], esophageal squamous cell cancer [18], oral squamous cell carcinoma [19], and ovarian cancer [20]. In addition, we have found that Gli-1 overexpression is correlated with capsular invasion, advanced tumor stage, vascular invasion, and intrahepatic metastasis in HCC [8]. However, the exact roles and underlying mechanisms of Gli-1 in invasion and metastasis of HCC remain to be elucidated.
In this study, we studied the effect of small interfering RNA (siRNA) mediated down-regulation of Gli-1 on HCC cell adhesion, migration, and invasion. Furthermore, the underlying mechanisms of down-regulation of Gli-1 were investigated. Our results showed that Gli-1 siRNA suppressed HCC cell migration and invasion by down-regulating matrix metalloprotease (MMP)-2, 9 expressions and activities and blocking epithelial- mesenchymal transition (EMT).
Materials and methods
Cell culture
Human hepatoma cell lines, SMMC-7721 and SK-Hep1, were maintained in Dulbecco’s modified Eagle’s medium (DMEM) (Gibco, CA, USA) supplemented with 10 % fetal bovine serum (Gibco, CA, USA) and 100 U/ml penicillin and 100 µg/ml streptomycin in a humidified atmosphere of 5 % CO2 at 37 °C.
Small interfering RNA transfection
Small interfering RNA (siRNA) duplex designed to target Gli-1 (sense: 5′-CCAGU GUCCUCGACUUGAAdTdT-3′; antisense: 5′-UUCAAGUCGAGGACACUGGTdTd-3′) and control siRNA (scrambled siRNA) were purchased from Ribobio Co., Ltd (Guangzhou, China). Transfection was performed using the LipofectAMINE2000 reagent (Invitrogen, Carlsbad, CA, USA) according to the manufacturer’s instruction as described previously [21].
Real-time PCR
Total RNA was extracted from cells using TRIzol reagent (Invitrogen, Carlsbad, CA, USA) following the manufacturer’s protocol. Complementary DNA (cDNA) was synthesized and real-time polymerase chain reaction (PCR) was performed as previously described [22]. The primers for human Snail (forward primer: 5′-CCCCAATCGGAAGCCTAACT-3′, reverse primer: 5′-AGATGAGCATTGGCAGCGA-3′), E-cadherin (forward primer: 5′-ACAACGCCCCCATACCAGA-3′, reverse primer: 5′-CACTCGCCCCGTGTGTTAGT-3′), and Vimentin (forward primer: 5′-TTGACAATGCGTCTCTGGCA-3′, reverse primer: 5′-TCCTTGTCGTACAGGTTTAGC-3′) were used in the real-time PCR. The primers specific for human MMP-2, MMP-9, Gli-1, and β-actin were reported previously [8, 21, 22].
Western blot
Western blot was done as described previously [8, 21]. In brief, total protein was extracted from cells and protein concentrations were then measured. Equal amounts of protein were separated by SDS–polyacrylamide (PAGE) gels and transferred to polyvinylidene difluoride (PVDF) membranes (Millipore, Bedford, MA, USA). The membranes were incubated with the primary antibodies against Gli-1 (Santa Cruz Biotechnology, Santa Cruz, CA, USA) at 1:400, MMP-2 (Cell Signaling Technology, Beverly, MA, USA) at 1:1,000, MMP-9 (Cell Signaling Technology) at 1:1,000, Snail (Cell Signaling Technology) at 1:800, E-cadherin (Cell Signaling Technology) at 1:800, Vimentin (Santa Cruz) at 1:500, and GAPDH (Santa Cruz) at 1:2,000 and then with horseradish peroxidase (HRP)-conjugated secondary antibodies. The bands were detected with enhanced chemiluminescence (ECL) kit (Pierce, Rockford, IL, USA) and exposed to X-ray film.
Cell adhesion
Cell adhesion assay was performed as described previously [8, 21]. In brief, the cells (1 × 105/well) were plated into 96-well plates precoated with fibronectin (Becton–Dickinson, Bedford, MA, USA). After incubation for 1 h at 37 °C in 5 % CO2 incubator, the cells were washed with PBS to remove the unattached cells. The attached cells were analyzed by MTT assay.
Wound-healing assay
Wound-healing assay was performed as described previously [21].
Cell migration and invasion assay
Cell migration and invasion assay were done as previously described [8, 21]. Briefly, 20 h after transfection, the cells (1 × 105/well) with serum-free medium were plated onto the upper chamber of 24-well Transwell inserts (Corning, NY, USA). The lower chamber was filled with 500 μL DMEM with 20 % fetal bovine serum. After 24 h of incubation, the cells on the upper surface of the filter were wiped out with a cotton swab and the cells that had migrated to the lower surface of the filter were fixed, stained, and counted. The cellular invasion potential was determined by the same procedures described above, except that the Matrigel-coated Transwell inserts were used.
Cell viability assay
Cell viability was examined by 3-[4,5-dimethylthiazol-2-yl]-2, 5-diphenyltetrazolium bromide (MTT) assay. In brief, after 20 h of transfection, MTT (Sigma, St. Louis, MO, USA) solution was added into each well. After 4 h, the medium was removed and 100 μL dimethyl sulfoxide (DMSO) was added into each well. The plates were read at 570 nm on an enzyme-linked immunity implement. Each experiment was repeated at least thrice in quadruplicate wells.
MMP gelatinase activity assay
The activities of MMP-2 and MMP-9 in the conditioned media were determined by MMP gelatinase activity assay as described previously [8, 21]. Briefly, after 20 h of transfection, cells were incubated in serum-free DMEM for 18 h. The conditioned media were then collected and centrifuged. The activities of MMP-2 and MMP-9 in the conditioned media were detected using CHEMICON Gelatinase Activity Assay Kit (Cat. No. ECM700) (Chemicon, Temecula, CA, USA) following manufacturer’s protocols.
Statistical analysis
All data were expressed as mean ± standard deviation (SD) and were analyzed by two-tailed Student’s t test. Statistical analyses were performed using SPSS 13.0 for Windows (SPSS Inc., Chicago, IL, USA). P < 0.05 was considered significant.
Results
Gli-1 siRNA inhibited Gli-1 expression in hepatoma cells
Our previous study has shown that Gli-1 is overexpressed in SMMC-7721 and SK-Hep1 cells [8]. siRNA-mediated RNA interference has proven to be a powerful means for investigating the function of gene. Therefore, the specific siRNA against Gli-1 was used to inhibit the Gli-1 expression. Our results demonstrated that Gli-1 mRNA levels were significantly down-regulated in a concentration-dependent manner, and the greatest knockdowns were achieved at 100 nM in both SMMC-7721 and SK-Hep1 cells transfected with Gli-1 siRNA for 20 h (Fig. 1a). Furthermore, the effectiveness of the siRNA to inhibit Gli-1 expression was confirmed by western blot (Fig. 1b). In contrast, control siRNA had little effect on Gli-1 mRNA and protein expressions (Fig. 1). These data indicated that Gli-1 siRNA effectively suppressed mRNA and protein expressions of Gli-1 in hepatoma cells.
siRNA-mediated down-regulation of Gli-1 in hepatoma cells. After SMMC-7721 and SK-Hep1 cells were transfected with control siRNA or various concentrations of Gli-1 siRNA for 20 h, the mRNA and protein levels of Gli-1 were detected by real-time PCR and western blot, respectively. a Specific siRNA against Gli-1 significantly down-regulated Gli-1 mRNA expression, whereas control siRNA did not affect Gli-1 mRNA expression. *P < 0.01, compared with controls. b Western blot showed that Gli-1 protein expression was significantly inhibited by Gli-1 siRNA
Down-regulation of Gli-1 inhibited hepatoma cell adhesion, migration, and invasion
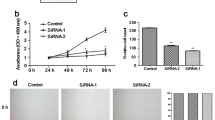
Because we have found that Gli-1 overexpression is closely correlated with invasion and metastasis in human HCC samples [8], we further confirmed whether Gli-1 contributes to hepatoma cell migration and invasion in vitro. To that end, we down-regulated Gli-1 expression by using the RNA interference approach in hepatoma cells and then examined the adhesive, migratory, and invasive potential of these cells. First, we found that the cell adhesive ability of SMMC-7721 and SK-Hep1 cells was inhibited in a dose-dependent manner after transfection with Gli-1 siRNA (Fig. 2a). Next, the results of wound-healing assays demonstrated that Gli-1 siRNA significantly suppressed the motility of both SMMC-7721 and SK-Hep1 cells (Fig. 2b). Then, we found that the migratory potential of Gli-1 siRNA-transfected SMMC-7721 and SK-Hep1 cells was dramatically decreased (Fig. 2c). Finally, down-regulation of Gli-1 expression by siRNA significantly inhibited invasive capability of SMMC-7721 and SK-Hep1 cells (Fig. 2d). However, control siRNA did not affect adhesion, migration, and invasion of SMMC-7721 and SK-Hep1 cells.
Down-regulation of Gli-1 suppressed cell adhesion, motility, migration, and invasion in hepatoma cells. SMMC-7721 and SK-Hep1 cells were transfected with control siRNA or Gli-1 siRNA for 20 h. a The cells were then seeded into 96-well plates that were precoated with fibronectin and incubated for 1 h. The adhered cells were analyzed by MTT assay. *P < 0.01, compared with controls. b The cell monolayers were wounded with a pipette tip. Representative images were taken at 0 and 24 h after wounds were made. c The cells seeded into the upper chambers of 24-well Transwell inserts and incubated for 24 h. The migrated cells were counted by microscope (×100 magnification). The data were presented as a percentage of control. *P < 0.01, compared with controls. d The invasive potential of the cells was assayed using Matrigel-coated invasion chambers. *P < 0.01, compared with controls
To determine whether the effect of Gli-1 siRNA on cell adhesion, migration, and invasion was dependent on its cellular cytotoxicity, we examined the viability of SMMC-7721 and SK-Hep1 cells transfected with Gli-1siRNA for the same length of time using MTT assay. The results of MTT assay showed that the viability of SMMC-7721 and SK-Hep1 cells was not affected after transfection with Gli-1 siRNA for 20 h at the indicated concentrations (Fig. 3). These results suggest that the effect of down-regulation of Gli-1 expression by siRNA on hepatoma cell adhesion, migration, and invasion is independent of its cellular cytotoxicity. Since 100 nM Gli-1 siRNA caused the greatest suppression in hepatoma cell adhesion, migration, and invasion, we used 100 nM Gli-1 siRNA for subsequent experiments.
Down-regulation of Gli-1 decreased MMP-2 and MMP-9 expressions and activities
MMP-2 and MMP-9 are known to play critical roles in the extracellular matrix (ECM) degradation and be correlated with HCC invasion and metastasis [22]. Therefore, we studied whether decreased migratory and invasive potentials of Gli-1 siRNA-transfected cells were associated with MMP-2 and MMP-9. First, real-time PCR and western blot were conducted to detect the alteration in the expressions of MMP-2 and MMP-9. The results showed that down-regulation of Gli-1 significantly decreased MMP-2 and MMP-9 mRNA and protein expressions in SMMC-7721 and SK-Hep1 cells (Fig. 4a, b). Next, we determined whether down-regulation of Gli-1 could decrease MMP-2 and MMP-9 activities. We found that MMP-2 and MMP-9 activities were significantly inhibited in the conditioned media from Gli-1 siRNA-transfected SMMC-7721 and SK-Hep1 cells (Fig. 4c). However, control siRNA had negligible effect on the expressions and activities of MMP-2 and MMP-9 (Fig. 4).
Down-regulation of Gli-1 inhibited expressions and activities of MMP-2 and MMP-9. SMMC-7721 and SK-Hep1 cells were transfected with control siRNA or 100nM Gli-1 siRNA for 20 h. a MMP-2 and MMP-9 mRNA expressions in the cells were detected using real-time PCR. *P < 0.001, compared with controls. b MMP-2 and MMP-9 protein expression levels were determined by western blot. c Conditioned media were collected and activities of MMP-2 and MMP-9 were analyzed by the MMP gelatinase activity assay. *P < 0.001, compared with controls
Gli-1 silencing up-regulated E-cadherin expression and down-regulated Snail and Vimentin expressions
Epithelial-mesenchymal transition (EMT) is a key event in HCC invasion [23, 24]. Therefore, we further investigated whether Gli-1 has any effect on EMT. Real-time PCR and western blot were done to detect the expressions of E-cadherin, Snail, and Vimentin. Our results showed that down-regulation of Gli-1 mediated by siRNA led to significant up-regulation of E-cadherin mRNA and protein levels and concomitant down-regulation of mRNA and protein levels of Snail and Vimentin in SMMC-7721 and SK-Hep1 cells, whereas control siRNA showed no effect (Fig. 5), suggesting that down-regulation of Gli-1 blocks EMT.
Knock-down of Gli-1 up-regulated E-cadherin expression and down- regulated Snail and Vimentin expressions. SMMC-7721 and SK-Hep1 cells were transfected with control siRNA or 100 nM Gli-1 siRNA for 20 h. a Real-time PCR analysis of E-cadherin, Snail, and Vimentin mRNA expressions in transfected cells. *P < 0.001, compared with controls. b Western blot analysis of E-cadherin, Snail, and Vimentin protein expressions in transfected cells
Discussion
Hedgehog (Hh) signaling pathway is known to play important roles in embryonic development and the formation and development of several human cancers [5–7, 25]. Gli-1 is a core member of Hh signaling pathway and is induced in cells that receive Hh signaling [26]. Gli-1 functions as an activator of transcription in Hh pathway. Therefore, Gli-1 expression, especially nuclear translocation of Gli-1, is considered a marker of Hh pathway activation [10–14]. Accumulating evidences have shown that Gli-1 is implicated in the progression of several cancers including HCC [8, 17–20]. However, the precise roles and mechanisms of Gli-1 in tumor invasion are still undefined. In this study, our results demonstrated that down-regulation of Gli-1 by siRNA significantly inhibited hepatoma cell adhesion, migration, and invasion, suggesting that Gli-1 plays an important role in HCC invasion and metastasis and that Gli-1 might be a potential therapeutic target for HCC. Furthermore, our results showed that down-regulation of Gli-1 reduced MMP-2 and MMP-9 expressions and activities. In addition, we found that down-regulation of Gli-1 increased E-cadherin expression and decreased expressions of Snail and Vimentin. Taken together, these results indicate that down-regulation of Gli-1 suppresses HCC migration and invasion likely through inhibiting expressions and activations of MMP-2, 9 and blocking EMT.
Tumor invasion and metastasis are a complex multistep process involving destruction of basement membranes and proteolysis of extracellular matrix (ECM) as an early event in the metastatic cascade [27]. Matrix metalloproteinases (MMPs), are a family of zinc-dependent endoproteinases that play an important role in proteolysis of ECM [28, 29]. Among the MMPs, MMP-2 and MMP-9 are especially important for tumor invasion and metastasis [27–29]. Our previous studies have revealed that MMP-2 and MMP-9 play important roles in HCC invasion and metastasis [8, 21, 22]. In this study, we found down-regulation of Gli-1 by siRNA could significantly suppress the expressions and activities of MMP-2 and MMP-9, suggesting that Gli-1 is involved in HCC migration and invasion through regulating expressions and activations of both MMP-2 and MMP-9.
Epithelial-mesenchymal transition (EMT) is a process by which epithelial cells lose fundamental epithelial characteristics and acquire a mesenchymal phenotype. EMT endows epithelial cells with enhanced migratory and invasive potential [30]. EMT is critical for normal development, tissue repair and tumor invasion, and metastasis [24, 30, 31]. Studies have suggested that EMT plays a pivotal role HCC invasion and metastasis [23, 24].
A hallmark of EMT is loss of E-cadherin expression [30, 32]. E-cadherin is a calcium-dependent cell adhesion molecule that participates in cell–cell adhesion. The loss of E-cadherin expression is associated with invasion and metastasis of tumor including HCC [33]. Several mechanisms are responsible for loss of E-cadherin in cancer, including transcriptional repression by EMT-inducing factors. Among the EMT-inducing factors, zinc-finger transcription factor Snail is the most prominent one [34–36]. Snail can bind to E-cadherin promoter and repress its transcription, which has been implicated in the triggering of EMT [34, 35]. Snail has been found to be correlated with invasion, metastasis, and poor prognosis in several tumors including HCC [36, 37]. Vimentin is a marker for EMT because Vimentin is characteristically up-regulated in cells undergoing EMT [38, 39]. Vimentin is overexpressed in a variety of epithelial cancers and its overexpression is correlated with tumor growth, invasion, and poor prognosis [39]. Therefore, we studied whether down-regulation of Gli-1 inhibited EMT by detecting the alteration in expressions of E-cadherin, Snail, and Vimentin. Our results demonstrated that down-regulation of Gli-1 significantly increased E-cadherin expression and concomitantly decreased Snail and Vimentin expressions in hepatoma cells, indicating that down-regulation of Gli-1 might be an effective approach for blocking EMT and that down-regulation of Gli-1 suppresses HCC cell migration and invasion partly through blocking EMT.
In summary, our results show that Gli-1 plays a critical role in HCC cell migration and invasion. Furthermore, down-regulation of Gli-1 significantly inhibits HCC cell migration and invasion through down-regulating MMP-2, 9 and blocking EMT. These data not only suggest that down-regulation of Gli-1 expression may provide a useful therapeutic option for HCC invasion and metastasis, but also provide new insights into the molecular mechanisms of HCC invasion and metastasis. However, the roles for Gli-1 in invasion of HCC cells in vitro merit further investigation in vivo. Further researches on other molecular mechanisms underlying Gli-1-mediated HCC invasion and metastasis are also warranted.
References
Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D (2011) Global cancer statistics. CA Cancer J Clin 61:69–90
Llovet JM, Burroughs A, Bruix J (2003) Hepatocellular carcinoma. Lancet 362:1907–1917
Nüsslein-Volhard C, Wieschaus E (1980) Mutations affecting segment number and polarity in Drosophila. Nature 287:795–801
Ruiz i Altaba A, Sánchez P, Dahmane N (2002) Gli and Hedgehog in cancer: tumours, embryos and stem cells. Nat Rev Cancer 2:361–372
Rubin LL, de Sauvage FJ (2006) Targeting the Hedgehog pathway in cancer. Nat Rev Drug Discov 5:1026–1033
Amakye D, Jagani Z, Dorsch M (2013) Unraveling the therapeutic potential of the Hedgehog pathway in cancer. Nat Med 19:1410–1422
Briscoe J, Thérond PP (2013) The mechanisms of Hedgehog signalling and its roles in development and disease. Nat Rev Mol Cell Biol 14:416–429
Chen JS, Huang XH, Wang Q, Huang JQ, Zhang LJ, Chen XL, Lei J, Cheng ZX (2013) Sonic hedgehog signaling pathway induces cell migration and invasion through focal adhesion kinase/AKT signaling-mediated activation of matrix metalloproteinase (MMP)-2 and MMP-9 in liver cancer. Carcinogenesis 34:10–19
Mullor JL, Sánchez P, Ruiz i Altaba A (2002) Pathways and consequences: Hedgehog signaling in human disease. Trends Cell Biol 12:562–569
Lee J, Platt KA, Censullo P, Ruiz i Altaba A (1997) Gli1 is a target of Sonic hedgehog that induces ventral neural tube development. Development 124:2537–2552
Ingham PW, McMahon AP (2001) Hedgehog signaling in animal development: paradigms and principles. Genes Dev 15:3059–3087
Karlstrom RO, Tyurina OV, Kawakami A, Nishioka N, Talbot WS, Sasaki H, Schier AF (2003) Genetic analysis of zebrafish gli1 and gli2 reveals divergent requirements for gli genes in vertebrate development. Development 130:1549–1564
Kubo M, Nakamura M, Tasaki A, Yamanaka N, Nakashima H, Nomura M, Kuroki S, Katano M (2004) Hedgehog signaling pathway is a new therapeutic target for patients with breast cancer. Cancer Res 64:6071–6074
Kasper M, Regl G, Frischauf AM, Aberger F (2006) GLI transcription factors: mediators of oncogenic Hedgehog signalling. Eur J Cancer 42:437–445
Dahmane N, Lee J, Robins P, Heller P, Ruiz i Altaba A (1997) Activation of the transcription factor Gli1 and the Sonic Hedgehog signalling pathway in skin tumours. Nature 389:876–881
Dahmane N, Sánchez P, Gitton Y, Palma V, Sun T, Beyna M, Weiner H, Ruiz i Altaba A (2001) The Sonic Hedgehog–Gli pathway regulates dorsal brain growth and tumorigenesis. Development 128:5201–5212
Sheng W, Dong M, Zhou J, Li X, Liu Q, Dong Q, Li F (2014) The clinicopathological significance and relationship of Gli1, MDM2 and p53 expression in resectable pancreatic cancer. Histopathology 64:523–535
Min S, Xiaoyan X, Fanghui P, Yamei W, Xiaoli Y, Feng W (2013) The glioma-associated oncogene homolog 1 promotes epithelial–mesenchymal transition in human esophageal squamous cell cancer by inhibiting E-cadherin via Snail. Cancer Gene Ther 20:379–385
Wang YF, Chang CJ, Lin CP, Chang SY, Chu PY, Tai SK, Li WY, Chao KS, Chen YJ (2012) Expression of hedgehog signaling molecules as a prognostic indicator of oral squamous cell carcinoma. Head Neck 34:1556–1561
Ciucci A, De Stefano I, Vellone VG, Lisi L, Bottoni C, Scambia G, Zannoni GF, Gallo D (2013) Expression of the glioma-associated oncogene homolog 1 (gli1) in advanced serous ovarian cancer is associated with unfavorable overall survival. PLoS ONE 8:e60145
Chen JS, Huang XH, Wang Q, Chen XL, Fu XH, Tan HX, Zhang LJ, Li W, Bi J (2010) FAK is involved in invasion and metastasis of hepatocellular carcinoma. Clin Exp Metastasis 27:71–82
Chen JS, Wang Q, Fu XH, Huang XH, Chen XL, Cao LQ, Chen LZ, Tan HX, Li W, Bi J, Zhang LJ (2009) Involvement of PI3 K/PTEN/AKT/mTOR pathway in invasion and metastasis in hepatocellular carcinoma: association with MMP-9. Hepatol Res 39:177–186
Reichl P, Haider C, Grubinger M, Mikulits W (2012) TGF-β in epithelial to mesenchymal transition and metastasis of liver carcinoma. Curr Pharm Des 18:4135–4147
van Zijl F, Zulehner G, Petz M, Schneller D, Kornauth C, Hau M, Machat G, Grubinger M, Huber H, Mikulits W (2009) Epithelial-mesenchymal transition in hepatocellular carcinoma. Future Oncol 5:1169–1179
Harris LG, Pannell LK, Singh S, Samant RS, Shevde LA (2012) Increased vascularity and spontaneous metastasis of breast cancer by hedgehog signaling mediated upregulation of cyr61. Oncogene 31:3370–3380
Ruiz i Altaba A (1999) Gli proteins and Hedgehog signaling: development and cancer. Trends Genet 15:418–425
Stetler-Stevenson WG (1990) Type IV collagenases in tumor invasion and metastasis. Cancer Metastasis Rev 9:289–303
Hadler-Olsen E, Winberg JO, Uhlin-Hansen L (2013) Matrix metalloproteinases in cancer: their value as diagnostic and prognostic markers and therapeutic targets. Tumour Biol 34:2041–2051
Vihinen P, Kähäri VM (2002) Matrix metalloproteinases in cancer: prognostic markers and therapeutic targets. Int J Cancer 99:157–166
Thiery JP, Acloque H, Huang RY, Nieto MA (2009) Epithelial-mesenchymal transitions in development and disease. Cell 139:871–890
Franco-Chuaire ML, Magda Carolina SC, Chuaire-Noack L (2013) Epithelial-mesenchymal transition (EMT): principles and clinical impact in cancer therapy. Invest Clin 54:186–205
Mallini P, Lennard T, Kirby J, Meeson A (2014) Epithelial-to-mesenchymal transition: what is the impact on breast cancer stem cells and drug resistance. Cancer Treat Rev 40:341–348
Hirohashi S (1998) Inactivation of the E-cadherin-mediated cell adhesion system in human cancers. Am J Pathol 153:333–339
Batlle E, Sancho E, Francí C, Domínguez D, Monfar M, Baulida J, García De Herreros A (2000) The transcription factor snail is a repressor of E-cadherin gene expression in epithelial tumour cells. Nat Cell Biol 2:84–89
Cano A, Pérez-Moreno MA, Rodrigo I, Locascio A, Blanco MJ, del Barrio MG, Portillo F, Nieto MA (2000) The transcription factor snail controls epithelial- mesenchymal transitions by repressing E-cadherin expression. Nat Cell Biol 2:76–83
Wang Y, Shi J, Chai K, Ying X, Zhou BP (2013) The role of Snail in EMT and tumorigenesis. Curr Cancer Drug Targets 13:963–972
Miyoshi A, Kitajima Y, Kido S, Shimonishi T, Matsuyama S, Kitahara K, Miyazaki K (2005) Snail accelerates cancer invasion by upregulating MMP expression and is associated with poor prognosis of hepatocellular carcinoma. Br J Cancer 92:252–258
Ivaska J (2011) Vimentin: central hub in EMT induction? Small GTPases 2:51–53
Satelli A, Li S (2011) Vimentin in cancer and its potential as a molecular target for cancer therapy. Cell Mol Life Sci 68:3033–3046
Acknowledgments
This work was supported by grants from the National Natural Science Foundation of China (81201930, 81101862, 81172079); Research Fund for the Doctoral Program of Higher Education of China (20124423120006); Natural Science Foundation of Guangdong Province (S2012040006803, S2013010016831); Key Project of Guangzhou Municipal Science & Technology Planning (2011J4100053), Doctoral Research Foundation of Guangzhou Medical University (2010C23), and Foundation for Youth Teachers by Sun Yat-Sen University(11ykpy16).
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chen, JS., Li, HS., Huang, JQ. et al. Down-regulation of Gli-1 inhibits hepatocellular carcinoma cell migration and invasion. Mol Cell Biochem 393, 283–291 (2014). https://doi.org/10.1007/s11010-014-2071-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-014-2071-x