Abstract
To investigate the combined effect of aliskiren, a renin inhibitor, and AVE 0991, a Mas-receptor agonist, in experimental hypertension (HT) in rats. HT was produced by administration of deoxycorticosterone acetate (DOCA) and mean arterial blood pressure (MABP) was assessed by tail-cuff method. Treatments were started from 4th week onwards and were continued for 9 days. A significant increase in MABP was noted after 1 week in DOCA control rats, as compared with the base line value. A stable HT developed after 4 weeks of DOCA administration. Treatments with aliskiren and AVE 0991 alone, dose-dependently decreased MABP in DOCA-treated rats. Further, combination of low doses of aliskiren and AVE 0991 significantly reduced MABP, as compared with DOCA control rats and with either drug alone in low doses. It may be concluded that treatment with aliskiren produced down-regulation of both harmful Ang II–AT1-receptor and survival Ang(1–7)/Mas-receptor axis of RAAS. Treatment with combination of low doses of aliskiren and AVE 0991, for the first time, has been shown to produce synergistic blood pressure lowering effect. Therefore, combination of renin inhibitor with Mas-receptor agonist may prove beneficial for the treatment of hypertensive patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Hypertension (HT) is an independent risk factor for various cardiovascular disorders such as arterial aneurysm, strokes, heart failure, and its prevalence is constantly increasing worldwide according to National Health and Nutrition Examination Survey data [1, 2]. The renin–angiotensin–aldosterone system (RAAS) is a very important regulatory system in cardiovascular and blood pressure homeostasis [3], and sustained over-activation of RAAS has been reported to produce end-organ damage in hypertensive patients.
Several preclinical and clinical studies have demonstrated that treatment with either angiotensin-converting enzyme inhibitors (ACEIs) [4] or ANG II type 1-receptor blockers (ARBs) [5] produce significant antihypertensive effect and provide end-organ protection. Furthermore, the beneficial effects of ACEIs and ARBs have been shown to be partly mediated through the activation of Ang1–7/Mas-receptor axis [6–8].
Angiotensin-converting enzyme-2 (ACE-2) is a zinc metallopeptidase which hydrolyses Ang I and II into Ang(1–9) and Ang(1–7) fragments, respectively; Ang(1–9) in turn is converted into Ang(1–7) by the enzymes ACE and endopeptidases [9, 10]. The Ang(1–7) produces vasodilation, antiproliferative, natriuretic activity through activation of Mas-receptor [11, 12]. Further, chronic treatment with angiotensin (1–7) significantly decreases dyslipidemia, a major culprit of cardio-renal dysfunctions in diabetic rats [13, 14]. Moreover, AVE 0991, a nonpeptide Mas-receptor agonist efficiently mimics the effects of angiotensin-(1–7) on the endothelium [15–17].
Renin is the rate-limiting step in the generation of Ang II. Recently, the nonpeptide renin inhibitor, aliskiren (ALK), was approved for the treatment of HT [18]. Animal and clinical studies have revealed striking vasodilator effect of ALK [19]. Furthermore, ALK has proven to be beneficial in several other pathophysiological settings such as cardiac remodeling and fibrosis as well as atherosclerosis [20].
The hypothesis investigated in this study was that whether or not a complete blockade of RAAS with renin inhibitor would down-regulate the beneficial Ang(1–7)/Mas-receptor axis, and if it is so, whether supplementation with AVE-0991, a nonpeptide Mas-receptor agonist would produce an additive or synergistic effect in DOCA-induced HT in rats.
Materials and methods
Age matched Wistar rats of either sex weighing 180–240 g were employed in the present study. The care and the use of these animals were in accordance with the guidelines of the CPCSEA (committee for the purpose of control and supervision of experiments on animals). Experimental protocol was approved by Institutional Animal Ethics Committee (IAEC) (Protocol No. ISF/CPCSEA/IAEC/2010/16).
Deoxycorticosterone acetate-induced (DOCA–salt) experimental HT
DOCA-treated rats were developed HT as previously reported [21]. In brief, a midline abdominal incision was made under anesthesia with a ketamine solution and the left kidney was removed. After 15 days of surgery, administration of DOCA was initiated, twice weekly by subcutaneous injection in a suspension of 0.2 ml of olive oil and drinking water was replaced with 1 % NaCl and 0.2 % KCl [22]. Stable HT develops after 4 week of DOCA–salt administration. The animals were restrained and conditioned for 5 days before commencing blood pressure measurements. The rat tail was heated with 200 W bulb heat lamp for 3–5 min and mean arterial blood pressure (MABP) was recorded using noninvasive blood pressure measurement technique (NIBP-Biopac-MP 100; Goleta, CA, USA). At least three separate indirect pressures were averaged for each animal. The animals having MABP ≥180 mmHg were considered as a hypertensive [23].
Experimental protocol
This study consists of seven groups and each group comprises of 5–6 rats of either sex. ALK and AVE 0991 were dissolved in normal saline solution (0.9 %).
Group I (Sham control): rats were underwent sham surgery and were maintained on normal drinking water for whole study period.
Group II (DOCA control): uninephrectomized rats were administered DOCA (20 mg/kg/s.c., twice weekly, 4 weeks) and on 5–6 weeks rats were maintained on normal drinking water without being treated with DOCA.
Group III (high dose ALK-treated hypertensive rats): rats were treated with high dose of ALK (50 mg/kg/i.p.; 9 days) [24], 4-week following DOCA administration.
Group IV [low dose (sub-effective dose) ALK-treated hypertensive rats]: rats were treated with low dose ALK (25 mg/kg/i.p.; 9 days), 4-week following DOCA administration.
Group V (high dose AVE 0991-treated hypertensive rats): rats were treated with high dose of AVE 0991 (576 μg/kg/i.p.; 9 days) [25], 4-week following DOCA administration.
Group VI (low dose AVE-0991-treated hypertensive rats): rats were treated with low dose of AVE 0991(288 μg/kg/i.p.; 9 days), 4-week following DOCA administration.
Group VII (low dose ALK + low dose AVE 0991-treated hypertensive rats): rats were treated with low dose ALK (25 mg/kg/i.p.; 9 days) 60 min before treatment with low dose AVE-0991 (288 μg/kg/i.p.; 9 days), 4-week following DOCA administration.
Drugs and chemicals
DOCA–salt was obtained from Sigma, St. Louis, MO, USA. AVE 0991 was obtained from Sanofi-Aventis, Germany as an ex-gratia. ALK was obtained from Novartis; USA as an ex-gratia. All other chemicals used were of analytical grade.
Statistical analysis
Values were expressed as mean ± SD. One-way ANOVA followed by Tukey’s multiple comparison test was used to determine the statistical significance (P < 0.05) between various groups.
Results
In group first, five animals were underwent sham surgery and were maintained on normal drinking water. There was no significant change observed in MABP during whole study period in these animals.
Effect of DOCA–salt on mean arterial blood pressure (MABP) in normal rats
A significant increase in MABP was noted after 1 week in DOCA-treated uninephrectomized rats, as compared with the base line value. A stable hypertension (201 ± 10 mmHg) developed after 4 week of DOCA administration. The MABP remained significantly high even in the DOCA free period from 5th to 6th weeks (Fig. 1).
Effect of low and high dose ALK on MABP in DOCA-treated rats
A significant increase in MABP (185.6 ± 11.4 mmHg) and (195.6 ± 13.9 mmHg) was noted in DOCA-treated rats in both groups (low dose ALK and high dose ALK-treated groups), respectively, as compared with their baseline values. Treatment with the low dose ALK significantly decreased MABP starting from day 4 and a maximum decrease in MABP (49 ± 2.6 mm/Hg) was on 9th day as compared with DOCA-treated rats, but did not reach the basal value even at the end of experiment. Treatment with the high dose of ALK significantly decreased MABP from day 3 onward and reached to the basal value on 7th day of treatment (Figs. 2, 3; Table 1).
Effect of low dose and high dose AVE 0991 on MABP in DOCA-treated rats
A significant increase in MABP (187.3 ± 15.0 mmHg) and (190.3 ± 13.5 mmHg) was noted in DOCA–salt-treated rats in both groups (low dose AVE0991 and high dose AVE0991-treated groups), respectively, as compared with the baseline value. Treatment with the low dose AVE0991 significantly decreased MABP starting from day 4 onward and a maximum decrease in MABP (45 ± 3.2 mm/Hg) was on 9th day as compared with DOCA–salt-treated rats, but did not reach the basal value even at the end of experiment. Whereas treatment with the high dose of AVE0991 significantly decreased MABP starting from day 3 onwards and reached to the basal value on 6th day of treatment (Figs. 4, 5; Table 1).
Effect of combination of low dose ALK and AVE 0991 on MABP in DOCA-treated rats
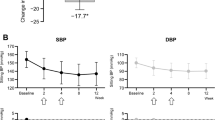
Treatment with combination of low dose ALK and AVE0991 significantly decrease MABP starting from day 2 and reached to the basal value on 8th day of treatment. The combination treatment significantly decreases MABP as compare with either drug alone in low doses (Fig. 6; Table 1).
Effect of combination of low dose of ALK and low dose of AVE 0991 on MABP in DOCA-treated rats: data is expressed as mean ± SD. a P < 0.05 versus basal; b P < 0.05 versus DOCA control; e P < 0.05 versus low dose ALK alone; f P < 0.05 versus low dose AVE0991 alone. B basal value, DC DOCA control (n = 6)
Discussion
Principle findings of the study are that, treatment with combination of low doses of ALK and AVE 0991 produced synergistic antihypertensive effect in Wistar rats.
Chronic administration of DOCA–salt produces a progressive increase in systolic blood pressure and consequently, produced cardiac hypertrophy, and interstitial and perivascular fibrosis in uninephrectomized rats [26, 27]. In this study too, DOCA–salt-treatment resulted in significant increases in MABP in Wistar rats, which remained unchanged in the 2 week DOCA free period. It has been reported that, DOCA–salt HT is associated with up-regulation of the renal and cardiac RAAS [28]. Further, Tank et al. [29] found a significant increase in glomerular and proximal tubule renin mRNA in the remaining kidney after uninephrectomy. The intrarenally formed Ang II stimulates sodium and bicarbonate reabsorption possibly via activation of apical Na+/H+ exchange, basolateral Na+–HCO3− cotransport, and basolateral Na+/K+–ATPase and via insertion of H+–ATPase into the apical membrane and thereby regulates MABP in DOCA–salt-treated rats [30–35].
ALK is a new class of orally active, potent, low molecular weight, nonpeptide renin inhibitor [18, 19]. Renin inhibitors block the catalyzed hydrolytic cleavage of angiotensinogen by competitively binding to the active site and subsites of renin thus the generation of Ang I is inhibited and consequently less Ang II is available to maintain its biological effects. Further, ALK-reduced urinary aldosterone excretion and plasma aldosterone concentration by blocking plasma renin activity, reduces BP. Natriuresis was enhanced by ALK, whereas potassium excretion was not influenced. Reduction in aldosterone secretion can be regarded as a favorable neurohumoral effect of ALK, in light of the postulated detrimental pathophysiological role of aldosterone and the beneficial clinical effects of aldosterone antagonists in heart failure [36, 37]. Therefore, renin inhibitors have been suggested to provide more complete blockade of RAAS than ACEIs or ARBs [38, 39]. But it is likely that excessive inhibition of RAAS by renin inhibitor may also result in decreased formation of angiotensin (1–7) and may produced down-regulation of Mas-receptor axis. Our study, for the first time, shows that combination of low doses of ALK with AVE 0991, a Mas-receptor agonist, produced synergistic antihypertensive effect in DOCA–salt-treated rats.
ACE2–Ang-(1–7)–Mas-receptor axis has been documented to play a role as an ACE–Ang II–AT1-receptor counter-regulatory axis [12]. It has been well-documented that beneficial effects of ACE-2/Ang(1–7)/Mas-receptor axis are abolished in hypertensive’s, as ACE expression gets up-regulated and ACE-2 expression gets down-regulated leading to imbalance in two major axis of RAAS and consequent development of HT takes place [40]. Further, administration of Ang(1–7), a Mas-receptor agonist prevents development of severe HT and end-organ damage in spontaneously hypertensive rats (SHR) [25]. Furthermore, administration of Ang(1–7) has been shown to decrease the cardiac fibrotic changes associated with HT [38]. In this study, AVE0991, a Mas-receptor agonist produced antihypertensive effect for the first time in DOCA-treated Wistar rats in a dose-dependent manner.
After a single administration of ALK, peak plasma concentrations were reached in 1–2 h [38]. That may be sufficient for down regulation of angiotensin (1–7)/Mas-receptor counter regulating axis. Therefore, in this study, AVE 0991 was administrated 1 h after the ALK treatment in hypertensive rats.
Difference in the efficacy of angiotensin 1–7, a Mas-receptor agonist, in males and females rats have been reported earlier [41]. Therefore, we used animals of either sex in our study. But we did not find any sex-based differences in efficacy of AVE0991 in our study. However, our sample size (n = 5–6) is too small to contradict the results obtained by Sullivan et al. [41].
The result obtained in this study confirms the hypothesis that suppression of renin alone may result in down-regulation of ACE-2/Mas-receptor counter-regulatory axis of RAAS. Treatment with combination of low doses of ALK and AVE0991 for the first time, showed synergistic blood pressure lowering effect in DOCA–salt-treated rats. Therefore, combination of renin inhibitor with Mas-receptor agonist may prove beneficial for the treatment of hypertensive patients, than the effect of these drugs when used singly. Further research in this area is warranted.
References
Cheung BM, Ong KL, Man YB, Lam KS, Lau CP (2006) Prevalence, awareness, treatment, and control of hypertension: United States National Health and Nutrition Examination Survey 2001–2002. J Clin Hypertens 2:93–98
Pierdomenico SD, Di Nicola M, Esposito AL et al (2009) Prognostic value of different indices of blood pressure variability in hypertensive patients. Am J Hypertens 22:842–847
Schmieder RE, Hilgers KF, Schlaich MP et al (2007) Renin–angiotensin system and cardiovascular risk. Lancet 369:1208–1219
Yusuf S, Sleight P, Pogue J et al (2000) Effects of an angiotensin-converting-enzyme inhibitor, ramipril, on cardiovascular events in high-risk patients. The Heart Outcomes Prevention Evaluation Study Investigators. N Engl J Med 342:145–153
Israili ZH (2000) Clinical pharmacokinetics of angiotensin II (AT1) receptor blockers in hypertension. J Hum Hypertens 14:S73–S86
Chappell MC, Iyer SN, Diz DI, Ferrario CM (1998) Antihypertensive effects of angiotensin-(1–7). Braz J Med Biol Res 31:1205–1212
Roks AJM, van Geel PP, Pinto YM et al (1999) Angiotensin-(1–7) is a modulator of the human rennin–angiotensin system. Hypertension 34:296–301
Collister JP, Hendel MD (2003) The role of Ang-(1–7) in mediating the chronic hypotensive effects of losartan in normal rats. J Renin Angiotensin Aldosterone Syst 4:176–179
Donoghue M, Hsieh F, Baronas E et al (2000) A novel angiotensin-converting enzyme-related carboxypeptidase (ACE2) converts angiotensin I to angiotensin 1–9. Circ Res 87:E1–E9
Tipnis SR, Hooper NM, Hyde R, Karran E, Christie G, Turner AJ (2000) A human homolog of angiotensin-converting enzyme. Cloning and functional expression as a captopril-insensitive carboxypeptidase. J Biol Chem 275:33238–33243
Freeman EJ, Chisolm GM, Ferrario CM, Tallant EA (1996) Angiotensin-(1–7) inhibits vascular smooth muscle cells growth. Hypertension 28:104–108
Ferrario CM, Chappell MC, Tallant EA, Brosnihan KB, Diz DI (1997) Counter-regulatory actions of angiotensin-(1–7). Hypertension 30:535–541
Singh K, Singh T, Sharma PL (2011) Beneficial effect of angiotensin (1–7) in diabetic rats with cardiomyopathy. Ther Adv Cardiovasc Dis 5:159–167
Singh T, Singh K, Sharma PL (2010) Ameliorative potential of angiotensin (1–7)/Mas receptor axis in streptozotocin-induced diabetic nephropathy in the rat. Meth Find Exp Clin Pharmacol 32:19–25
Singh K, Sharma K, Singh M, Sharma PL (2012) Possible mechanism of cardio-reno protective effects of AVE0991, a nonpeptide Mas-receptor agonist in diabetic rats. J Renin Angiotensin Aldosterione Syst 13:334–340
Wiemer G, Dobrucki LW, Louka FR, Malinski T, Heitsch H (2002) AVE 0991, a nonpeptide mimic of the effects of angiotensin-(1–7) on the endothelium. Hypertension 40:847–852
Lemos VS, Silva DM, Walther T, Alenina N, Bader M, Santos RA (2005) The endothelium-dependent vasodilator effect of the nonpeptide Ang(1–7) mimic AVE 0991 is abolished in the aorta of Mas-knockout mice. J Cardiovasc Pharmacol 46:274–279
Sealey JE, Laragh JH (2007) Aliskiren, the first renin inhibitor for treating hypertension: reactive renin secretion may limit its effectiveness. Am J Hypertens 20:587–597
Jensen C, Herold P, Brunner HR (2008) Aliskiren: the first renin inhibitor for clinical treatment. Nat Rev Drug Discov 7:310–399
Pilz B, Shagdarsuren E, Wellner M et al (2005) Aliskiren, a human renin inhibitor, ameliorates cardiac and renal damage in double-transgenic rats. Hypertension 46:569–576
Takator LK, Ogura T, Ota Z (1991) Computerized approach using autoradiography to quantify atrial natriuretic peptide receptors in the DOCA–salt hypertensive rat kidney. Regul Pept 35:115–125
Montezano ACI, Callera GE, Mota AL et al (2005) Endothelin-1 contributes to the sexual differences in renal damage in DOCA–salt rats. Peptides 26:1454–1462
Cordellini S (1999) Endothelial dysfunction in DOCA–salt hypertension possible involvement of prostaglandin endoperoxides. Gen Pharmacol 32:315–320
Lastra G, Habibi J, Whaley-Connell AT et al (2009) Direct renin inhibition improves systemic insulin resistance and skeletal muscle glucose transport in a transgenic rodent model of tissue rennin overexpression. Endocrinology 150:2561–2568
Benter IF, Yousif MHM, Anim JT, Cojocel C, Diz DI (2006) Angiotensin-(1–7) prevents development of severe hypertension and end-organ damage in spontaneously hypertensive rats treated with l-NAME. Am J Physiol Heart Circ Physiol 290:H684–H691
Young M, Head G, Funder J (1995) Determinants of cardiac fibrosis in experimental hypermineralocorticoid states. Am J Physiol Endocrinol Metab 269:E657–E662
Dobrzynski E, Wang C, Chao J, Chao L (2000) Adrenomedullin gene delivery attenuates hypertension, cardiac remodeling, and renal injury in deoxycorticosterone acetate–salt hypertensive rats. Hypertension 36:995–1001
Fowler JD, Krueth SB, Bernlohr DA, Katz SA (2009) Renin dynamics in adipose tissue: adipose tissue control of local renin concentrations. Am J Physiol Endocrinol Metab 296:E343–E350
Tank JE, Moe OW, Star RA, Henrich WL (1996) Differential regulation of rat glomerular and proximal tubular renin mRNA following uninephrectomy. Am J Physiol 270:F776–F783
Rosivall L, Navar LG (1983) Effects on renal hemodynamics of intra-arterial infusions of angiotensins I and II. Am J Physiol 245:F181–F187
Liu FY, Cogan MG (1988) Angiotensin II stimulation of hydrogen ion secretion in the rat early proximal tubule: modes of action, mechanism, and kinetics. J Clin Invest 82:601–607
Garvi JL (1991) Angiotensin stimulates bicarbonate transport and Na+/K+ ATPase in rat proximal straight tubules. J Am Soc Nephrol 1:1146–1152
Mitchell KD, Braam B, Navar LG (1992) Hypertensinogenic mechanisms mediated by renal actions of renin–angiotensin system. Hypertension 19:118–127
Eiam-Ong S, Hilden SA, Johns CA, Madias NE (1993) Stimulation of basolateral Na+–HCO3− cotransporter by angiotensin II in rabbit renal cortex. Am J Physiol 265:F195–F203
Wang T, Giebisch G (1996) Effects of angiotensin II on electrolyte transport in the early and late distal tubule in rat kidney. Am J Physiol 271:F143–F149
Stanton A, Jensen C, Nussberger J, O’Brien E (2003) Blood pressure lowering in essential hypertension with an oral renin inhibitor, aliskiren. Hypertension 42:1137–1143
McMurray JJC, Pitt B, Latine R, Aliskiren Observation of Heart Failure Treatment (ALOFT) Investigators et al (2008) Effect of the oral direct renin inhibitor aliskiren in patients with symptomatic heart failure. Circ Heart Fail 1:17–24
Wood JM, Schnell CR, Cumin F, Menard J, Webb RL (2005) Aliskiren, a novel, orally effective renin inhibitor, lowers blood pressure in marmosets and spontaneously hypertensive rats. J Hypertens 23:417–426
Azizi M, Webb R, Nussberger J, Hollenberg NK (2006) Renin inhibition with aliskiren: where are we now, and where are we going? J Hypertens 24:243–256
Koka V, Huang XR, Chung ACK, Wang W, Truong LD, Lan HY (2008) Angiotensin II up-regulates angiotensin I-converting enzyme (ACE), but down-regulates ACE2 via the AT1-ERK/p38 MAP kinase pathway. Am J Pathol 175:1174–1183
Sullivan JC, Bhatia K, Yamamoto T, Elmarakby AA (2010) Angiotensin (1–7) receptor antagonism equalizes angiotensin II-induced hypertension in male and female spontaneously hypertensive rats. Hypertension 56:658–666
Acknowledgments
This study was supported partially by a Research Grant obtained from All India Council for Technical Education, New Delhi (File no. 8023/BOR/RID/RPS-167/2007–2008). This paper is dedicated to the fond memories of Prof. Manjeet Singh who expired on 30-03-2009 while this study was in progress. We wish to express our gratitude to Sanofi-Aventis, Germany and Novartis; USA for providing AVE0991 and ALK as an ex-gratia, respectively. We also wish to our sincere thank to the management of ISF college of pharmacy for providing research facility.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Singh, Y., Singh, K. & Sharma, P.L. Effect of combination of renin inhibitor and Mas-receptor agonist in DOCA–salt-induced hypertension in rats. Mol Cell Biochem 373, 189–194 (2013). https://doi.org/10.1007/s11010-012-1489-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-012-1489-2