Abstract
Phosphoinositide 3-kinase (PI3K) pathway exerts its effects through Akt, its downstream target molecule, and thereby regulates various cell functions including cell proliferation, cell transformation, apoptosis, tumor growth, and angiogenesis. Phosphatase and tensin homolog deleted on chromosome 10 (PTEN) has been implicated in regulating cell survival signaling through the PI3K/Akt pathway. However, the mechanism by PI3K/PTEN signaling regulates angiogenesis and tumor growth in vivo remains to be elucidated. Vascular endothelial growth factor (VEGF) plays a pivotal role in tumor angiogenesis. The effect of PTEN on VEGF-mediated signal in pancreatic cancer is unknown. This study aimed to determine the effect of PTEN on both the expression of VEGF and angiogenesis. Toward that end, we used the siRNA knockdown method to specifically define the role of PTEN in the expression of VEGF and angiogenesis. We found that siRNA-mediated inhibition of PTEN gene expression in pancreatic cancer cells increase their VEGF secretion, up-modulated the proliferation, and migration of co-cultured vascular endothelial cell and enhanced tubule formation by HUVEC. In addition, PTEN modulated VEGF-mediated signaling and affected tumor angiogenesis through PI3K/Akt/VEGF/eNOS pathway.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Phosphatase and tensin homolog deleted on chromosome 10 (PTEN), also known as MMAC1/TEP1, is localized to chromosome 10q23 that is often lost in late-stage human cancers, especially that of the prostate, brain, and endometrium [1, 2]. It is a dual specificity phosphatase that dephosphorylates phosphatidylinositol 3,4,5-trisphosphate to phosphatidylinositol-4,5-bisphosphate, and thus, is a potent antagonist of phosphoinositide-3-kinase (PI3K)/ATP-dependent tyrosine kinases (Akt) signaling [3]. It was initially discovered as a tumor suppressor with regulatory role in cell survival and proliferation, particularly in tumor-prone tissues, such as the breast and endometrium [4]. Description of germ line mutations and deletions of PTEN in two hereditary diseases with predisposition to cancer (Cowden Disease and the Bannayan-Riley-Ruvalcaba syndrome [5–7]) point to a role of PTEN as a tumor suppressor gene in the pathogenesis of both benign and malignant growth. PTEN has been implicated in regulating cell survival signaling through the PI3K/Akt pathway. PTEN blocks the action of PI3K by dephosphorylating the signal lipid phosphatidylinositol 3,4,5-triphosphate (PIP3). PIP3, produced by PI3K after activation by receptor tyrosine kinase, activated Ras, or G proteins, leads to the stimulation of several downstream targets, including the serine/threonine protein kinase Akt [8]. Thus, it is particularly important that PTEN is one of the most frequently mutated genes in a variety of cancers [8–10]. Although mutation of one PTEN allele and loss of the second is the most common mechanism underlying PTEN loss-of-function in human tumors, additional mechanisms have recently been discovered. Methylation of the PTEN promoter is a frequent occurrence in certain type of cancer, such as thyroid cancer [11], melanoma [12], lung cancer [13], and low grade/secondary glioblastoma mulitforme (GBM) tumors [14]. However, recent results have shown that Akt activity can influence methylation patterns [15]. While PTEN mutations rarely occur in the pancreatic cancer [16], the reduction of its expression provides an additional growth advantage to malignant pancreatic cells [7, 17, 18].
Vascular endothelial growth factor (VEGF) plays a key role in the endothelial cell differentiation and the sprouting of the new blood vessels from pre-existing ones (angiogenesis). Angiogentic responses play key roles in certain pathologies, including ischemic diseases, inflammation, and cancer. VEGF expression in endothelial cells is mediated by PI3K signaling [19]. Binding of VEGF to receptor-2 (VEGFR-2) lead to receptor phosphorylation and subsequent activation of PI3K, phospholipase C-γ1, Sre family tyrosine kinases, and other signaling pathway [20, 21]. One of major downstream targets of PI3K is a serine–threonine kinase Akt that regulates the critical roles of growth factors and oncogenes. Akt transmits angiogenic and oncogenic signals and can directly induce angiogenesis. Therefore, the PI3K/Akt signaling pathway is implicated in VEGF expression in ovarian cells and in the reduction of angiogenesis [15, 19]. The impact of PTEN on VEGF-mediated signaling in pancreatic cancer has not been reported.
While recent studies of PTEN have focused on its role in the tumor cell biology, a recent report demonstrated that the PTEN/PI3K pathway governs normal vascular development and tumor angiogenesis [22]. Several studies have strongly implied that PTEN was association with tumor-induced angiogenesis [2, 19, 23, 24]. Furthermore, nitric oxide (NO) produced by activated endothelial NO synthase (eNOS) play an important role in angiogenesis [25, 26]. Based on the roles of PI3K and downstream target Akt in signaling VEGF, we hypothesized that PTEN can regulate VEGF-mediated endothelial cellular responses and angiogenesis. In the present study, we used the siRNA knockdown of methodology to define the roles of PTEN in the expression of VEGF and in regulating angiogenesis. We found evidence that specific blockage of PTEN increased VEGF secretion by pancreatic cancer cells, and modulate the proliferation and migration of vascular endothelial cell by enhancing the angiogenesis. In addition, PTEN modulates VEGF-mediated signaling and angiogenesis effects through PI3K/Akt/VEGF/eNOS pathway.
Materials and methods
Cell lines and culture conditions
Bxpc-3, MIAPaCa-2, SW1990, and Capan-2 cells were obtained from the American Type Culture Collection (Rockville, MD). BxPC-3 cells were maintained in RPMI-1640 (Sigma Chemical Co., St. Louis, MO) supplemented with 10% fetal bovine serum (FBS). MIAPaCa-2, SW1990, and Capan-2 cells were cultured in Dullbecco modified Eagle medium (sigma Chemical Co.) with high glucose and 10% FBS. Human umbilical vein endothelial cell (HUVEC) was purchased from Kurabo Co. (Osaka, Japan). HUVECs were maintained in HuMedia-EG2 medium supplemented with 2% FBS, 5 ng/ml basic fibroblast growth factor, 10 μg/ml heparin, 10 ng/ml epidermal growth factor, and 1 μg/ml hydrocortisone according to the supplier’s instructions (Kurabo Co.). All cells were incubated at 37°C in a humidified atmosphere of 5% CO2 in air.
Reagents and antibodies
VEGF was provided by R&D System Inc. (Minneapolis, MN, USA). Tyrphostin SU 1498 (#T2710, a specific inhibitor of VEGFR2, Klt-1 (IC = 700 nM)) was purchased from LC Laboratories (Woburn, MA, USA). Akt inhibitor was ordered from Bio Vision (Mountain View, CA, USA). LY294002 (PI3-kinase inhibitor) was provided by Cell Signaling Technology (Beverly, MA, USA). The monoclonal antibodies included PTEN Rabbit mAb, Akt antibody, phosphor-Akt (ser473), PI3-kinase P85 antibody, phospho-PI3-kinase P85 (Tyr 458) antibody, eNOS antibody, and phospho-eNOS (Ser1177) antibody were purchased from Cell Signaling Technology. VEGF (VG-1): sc-53462 antibody was purchased from Santa Cruz Biotechnology, Inc.
Reverse transcription-polymerase chain reaction analysis of PTEN and VEGF mRNA expression
Total RNA was extracted from four pancreatic cancer cell lines using Isogen Kits (Nippon Gene Tokyo, Japan), and then quantities were determined spectrophotometrically. Total RNA aliquots (5 μg) were pretreated with Random Hexamers and dNTP Mix were incubated at 65°C for 5 min, chilled on ice, and then reverse-transcribed into cDNA using the SuperScript III RT System (Invitrogen, San Diego, CA). One microlitre of cDNA aliquots was used as the templates for PCR. The pairs of forward and reverse primer sets were designed using Primer 3 software. The primer sequences and PCR condition were described in Table 1. Amplification reactions were performed by a DNA Thermal Cycle (model TP300; Takara PCR Thermal Cycle MP). The amplified DNA fragments were displayed by electrophoresis on 1.5% agarose gels containing ethidium bromide.
Western blot analysis
Pancreatic cancer cells were treated with lysis buffer (20 mM Tris pH 7.5, 150 mM NaCl, 1 mM Na2EDTA, 1 mM EGTA, 1% Triton X-100, 2.5 mM sodium pyrophosphate, 1 mM β-glycerophosphate, 1 mM Na3VO4, 1 μg/ml leupeptin, and 1 mM PMSF). The protein concentration was measured with a BCA Protein Assay Kit (Pierce, Rockford, USA). The lysates (30 μg per lane) were separated using 10% SDS-polyacrylamide gel electrophoresis, transferred to polyvinylidene membrane (Immobilo PVDF; Nihon Millipore Ltd, Tokyo, Japan), following, blocked by blocking buffer for 1 h at room temperature. The blocking buffer consisted of 5% non-fat dry milk dissolved into Tris-buffered saline containing 0.1% Tween 20 (TBS-T). After three 5 min wishes, the membrane was immunoblotted with each primary antibody was diluted, 200- to 1,000-fold by primary antibody dilution buffer, and incubated overnight at 4°C. Then membrane was washed three times (5 min) with TBS-T, and subjected to HRP-conjugated secondary antibody for 1 h at room temperature. The protein-antibody complexes were visualized by ECL Western Blotting Detection Reagents (Amersham Biosciences, Buckinghamshire, UK). β-actin western blot was served as the control.
RNA interference (siRNA)-induced gene silencing
Pancreatic cancer cells were transfected with PTEN siRNA or control non-specific siRNA using Steaith™ siRNA Duplex Oligoribonucleotides (Invitrogen). Human pancreatic cancer cells were seeded at a density of 2 × 105 cells per 35-mm dish in medium with 10% FBS and without antibiotics, and allowed to adhere overnight before transfection. PTEN siRNA (200 pmol) or control siRNA were added to 500 μl of Opti-MEM® Ι Reduced Serum Medium, while 10 μl of Lipofectamine™ 2000 (Invitrogen) was added to 500 μl Opti-MEM® Ι Reduced Serum Medium. Each was mixed gently and incubated for 5 min, after which diluted siRNA and diluted Lipofectamine™ 2000 were then combined, gently mixed, and allowed to incubate for 20 min at room temperature. The siRNA plus Lipofectamine 2000 complex were added directly to the culturing cells. After 4 h incubation, complete medium with 10% FBS was added and cells were cultured for another 24–48 h.
VEGF protein enzyme-linked immunosorbent assay
Untreated, PTEN siRNA transfected, or control siRNA transfected human pancreatic cancer cells (BxPC-3, MIAPaCa-2, SW1990, and Capan-2) were plated in 35-mm dishes at a density of 2 × 105 containing media with 10% FBS and cultured overnight. The medium was exchanged, and cells were cultured for 48 h. Then, cells numbers were determined and culture media were collected and microfuged at 1,500 rmp for 5 min to remove any particles, and supernatants frozen at −80°C until used in an enzyme-linked immunosorbent assay (ELISA). The concentration of VEGF in the culture medium per 2 × 105 cells was measured using a VEGF ELISA kit (R & D System) according to the manufacturer’s instructions.
HUVEC proliferation assay
Cultured pancreatic cancer cell supernatants collected as follows for use in HUVEC proliferation assays. BxPC-3, MIAPaCa-2, SW1990, and Capan-2 were plated at a density of 2 × 105 cells per wells in 12-well trays containing medium with 10% FBS and cultured overnight. The medium was exchanged to one contained 2% FBS, and the cells were cultured for 24 h. The culturing media were collected, microfuged at 1,500 rpm for 5 min, and the supernatants frozen at −80°C until use in HUVEC proliferation assay. HUVEC was seeded in 96-well, flat-bottom plates at a density of 1 × 103 cells/100 μl containing HUVEC basal medium and allowed to adhere overnight. The culturing medium was changed and cells were then cultured with HUVEC basal medium only (untreated), or pancreatic cancer’s supernatant mixed with HUVEC basal medium (1:1) with or without 20 μM of SU 1498. The medium was exchanged every 24 h, and after incubation for 72 h, HUVEC proliferation was measured using the WST-1 assay.
Migration of HUVEC co-cultured with pancreatic cancer cells
The HUVEC migration assay was performed by BD BioCoat™ Matrigel™ Invasion Chamber (Becton Dickinson Biosciences, Bedford, MA) in vitro. In order to investigate the influence of PTEN siRNA transfected cells on HUVEC proliferation, we used a double chamber co-culture system. Pancreatic cancer cells (1 × 105) were plated into 24-well plates (low chambers) and incubated overnight. HUVEC (1 × 105 cells) were separately seeded in Matrigel pre-coated transwell chamber (upper chamber), which consist of polycarbonate membranes with 8 μm pores. The transwell chambers were then plated on the 24-well plates. The condition culture medium were mixed by DMEM or RPMI-1640 with 2% FBS and HUVEC basal culture medium only, or co-culture with untreated, PTEN siRNA transfected, and control siRNA transfected pancreatic cancer cells in low chambers with or without 20 μM of SU 1498. After 12 h incubation, the upper surfaces of the transwell chambers were wiped with cotton swab and invading cells were fixed and stained Giemsa solution. The invading cells were counted in five microscope fields (200×).
Angiogenesis assay
In order to investigate the influence of PTEN and PTEN siRNA transfected pancreatic cancer cells on tubule formation by HUVEC, HUVECs, and fibroblasts were co-culture in the basal medium using an angiogenesis Kit (Kurabo Co.) according to the manufacturer’s protocols. First, HUVECs and fibroblasts were co-cultured in 24-well plates with basal medium. The media were exchanged every two days, with co-incubation continuing for a total of 11 days. The co-culturing system was stained with anti-CD31 antibody. The area of angiogenesis was measured quantitatively over 10 different microscope fields for each well using an image analyzer (Kurabo Co.).
Angiogentic activity during co-cultivation with pancreatic cancer cells
We further investigate the influence of different pancreatic cancer cells (either untreated, or PTEN siRNA transfected, or control siRNA transfected) on tubule formation by HUVECs. Transfected or non-transfected pancreatic cancer cells (BxPC-3 or Capan-2) were co-incubated with HUVECs or fibroblasts using a double chamber method in 24-well plates. Pancreatic cancer cells (1 × 104 cells/well) were seeded in transwell chambers, consisting of polycarbonate membranes with 0.45 μm pores, and the cells allowed to adhere overnight. Then, the transwell chambers were placed in the HUVEC/fibroblast co-culture plate and the medium exchanged on day 6. Cells were incubated for 11 days, and HUVEC tubule formation was measured as above described.
Data analysis
Statistical comparisons were performed using Student’s t-test for paired observations or one-way ANOVA with a post hoc test (Dunnett multiple comparison) for multiple group comparisons. All data are presented as mean ± standard deviation (SD). P < 0.05 was considered statistically significant. Mean values and SD were calculated for experiments carried out in triplicate.
Results
Expression of PTEN and VEGF in pancreatic cancer cell lines
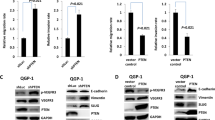
PTEN and VEGF mRNA levels were determined in all pancreatic cancer cell lines (Fig. 1a). Immunoblotting analysis revealed that the four pancreatic cancer cell lines are also expressed PTEN and VEGF protein (Fig. 1b). Knockdown of PTEN expression by siRNA pretreatment was confirmed by immunoblotting. Transfection of PTEN siRNA led to a near total loss of PTEN expression. An anti-β-actin antibody served as control (Fig. 1c).
Expression of PTEN and VEGF by pancreatic cancer cell lines. a PTEN and VEGF mRNA expression in pancreatic cancer cells were detected by RT-PCR. PCR amplification was carried out using primers pairs designed from PTEN and VEGF cDNA sequences. PCR products were subjected to 1.5% agarose gel electrophoresis and stained with ethidium bromide. β-actin served as the loading control. b Expression of PTEN and VEGF protein was determined in whole-cell lysates by Western Blotting analysis. Thirty micrograms of total cell lysates were subjected to 10% SDS-PAGE and transferred to a polyvinylidene difluoride membrane. The membrane was labeled with antibodies against PTEN and VEGF. β-actin was the loading control. c Knockdown of PTEN by siRNA was confirmed by Western blot. siRNA duplex oligoribonucleotides were transfected into cells for 48 h. Expression was assessed as above
Effect of VEGF secretion by PTEN siRNA transfected cells
Four human pancreatic cancer cell lines were examined under three different conditions: untreated, PTEN siRNA transfected, and control siRNA transfected. All the lines secreted VEGF protein under the three conditions as determined by the ELISA assay. VEGF secretion levels were highest in BxPC-3 cell line (2025.908 ± 82.740), and lowest in Capan-2 (428.565 ± 5.960). Transfection with PTEN siRNA significantly enhanced the cell’s secretion of VEGF. Specifically, the fold-increases were 1.8-, 1.7-, 2.3-, and 2.2-times in BxPC-3, MIAPaCa-2, SW1990, and Capan-2, respectively, compared to untreated cells (Fig. 2).
Effect of PTEN on VEGF secretion levels. Levels of Secreted VEGF levels in the culture media of pancreatic cancer cells were measured by ELISA. Left black column, center striped column, and the right striped column show untreated cells, PTEN siRNA transfected cells, and control siRNA transfected cells, respectively. Values are expressed as mean ± SD. Multiple comparisons were performed by one-way ANOVA with Dunnett test, * P < 0.01 compared with untreated cell
Effect of pancreatic cancer cell supernatants on HUVEC proliferation
We investigated the effect of culture supernatant from PTEN siRNA transfected cells on HUVEC proliferation. Briefly, we found that HUVEC proliferation was significantly enhanced by culture supernatants from four different PTEN siRNA transfected pancreatic cancer cell lines (* P < 0.01, Fig. 3a) compared with untreated cells and control siRNA transfected cells. Furthermore, we also investigated whether angiogenic factor other than VEGF can be increased by PTEN knockdown. VEGFR2 inhibitor SU 1498 was used to block the effect of VEGF on HUVEC proliferation from PTEN knockdown. We found that the enhancement HUVEC proliferation by PTEN knockdown was most blocked by SU 1498 (* P < 0.01, Fig. 3b). These results revealed that PTEN has a critical role in regulation of HUVEC proliferation. This function is mainly effect VEGF secretion level in pancreatic cancer.
Effect of knockdown of PTEN on HUVEC and treatment of SU 1498 on HUVEC proliferation. HUVEC were plated in 96-well plates at a density of 1 × 103 cells/100 μl with HUVEC basal medium only (untreated), or in conditioned culture media from BxPC-3, MIAPaCa-2, SW1990, or Capan-2 mixed 1:1 with HUVEC basal medium. a Each cell line in contained three sources of culture media: untreated cells, PTEN siRNA, or control siRNA transfected medium. b The condition medium containing 20 μM of SU1498 from PTEN siRNA transfected BxPC-3 cells. After 72 h of incubation, HUVEC proliferation was determined by WST-1 assay. Columns mean absorbance reading; bar, SD. Multiple comparison were performed by one-way ANOVA followed by Dunnett test. (3a: * P < 0.01, versus untreated; ** P < 0.05 versus MIAPaCa-2 and Capan-2 untreated or control siRNA condition medium. (3b: P < 0.01 versus PTEN siRNA)
Effect of PTEN and pancreatic cancer cells on the migration of HUVEC
Untreated pancreatic cancer cells or those transfected with PTEN siRNA or control siRNA were co-cultured with vascular endothelial cells for 12 h. The cells which migrated through the membrane were then stained. The migrating capability of HUVEC was significantly enhanced by PTEN siRNA transfected pancreatic cancer compared with control siRNA transfected cells or untreated cells in co-cultured system (* P < 0.01, Fig. 4a). Thus, the data demonstrated that PTEN depressed HUVEC migration. Knockdown of PTEN enhances HUVEC migration in human pancreatic cancer cell. On the other hand, the promoted HUVEC migration by PTEN knockdown was significantly block by presence of SU 1498 compared with PTEN siRNA transfected cells (* P < 0.01, Fig. 4b).
The effect of PTEN and pancreatic cancer cells and treatment of SU 1498 on HUVEC migration. HUVEC were co-cultured with four different cell lines which were either untreated, PTEN siRNA transfected, or control siRNA transfected (a), or presence of SU 1498 in co-cultured system with PTEN siRNA transfected BxPC-3 cells (b). After 12 h incubation, HUVEC migration was assessed by Matrigel assay. Column show relative number of migrating cells (%). Bars, SD. Multiple comparisons were performed by one-way ANOVA followed by Dunnett test. (* P < 0.01, versus untreated (a); * P < 0.01 versus PTEN siRNA group; A, control; B, co-cultured with BxPC-3; C, co-cultured with PTEN siRNA; D, co-cultured with PTEN siRNA +SU 1498)
Effect of PTEN by pancreatic cancer cells on angiogenesis
In order to further pursue the role of PTEN in angiogenesis, and focusing on the interaction between tumor cell and stromal cell by characterizing the angiogentic activity in co-cultured system consisting of vascular endothelial cells, fibroblasts, and pancreatic cancer cells. Thus, we examined the influence of PTEN on the formation of tube-like structures by HUVEC. We cultured two cell lines using the double chamber methods to determine the effect of PTEN knockdown on tube formation. Angiogenesis was significantly enhanced by co-culture with PTEN siRNA transfected cells compared to control (HUVECs and fibroblasts only), untreated cancer cells, or control siRNA transfected cells (* P < 0.01, Fig. 5a).These results showed that there is a close relationship between angiogenic formation and PTEN. That is, when PTEN expression was blocked in human pancreatic cancer cell lines in vitro, angiogentic processes in co-cultured cells were enhanced.
Knockdown of PTEN cell effect angiogenesis. a Angiogenesis assay by cultivation of HUVECs/fibroblasts with pancreatic cancer cells using the double chamber method. After incubation for 11 days, the tube formation was stained with anti-CD31 antibody and the angiogenic area was scaled using an image analyzer. Column mean pixels of HUVEC tube formation area; bars, SD. Multiple comparisons were performed by one-way ANOVA followed by the Dunnett test. * P < 0.01 versus untreated or control siRNA transfected cells. A, control; B1, Co-cultured with BxPC-3; B2, Co-cultured with PTEN siRNA transfected BxPC-3; B3, Co-cultured with control siRNA transfected BxPC-3; C1, Co-cultured with Capan-2; C2, Co-cultured with PTEN transfected Capan-2; C3, Co-cultured with control siRNA transfected Capan-2. Magnification 100×. Black column: untreated cancer cells; right striped column: PTEN siRNA transfected cancer cells; left heavy striped column: control siRNA transfected cancer cells. b The tube formation area was measured quantitatively over 10 different fields for each condition using image analyzer. Values are expressed as mean ± SD. Black column: untreated cancer cells; right striped column: PTEN siRNA transfected cancer cells; left heavy striped column: control siRNA transfected cancer cells
The PI3K–Akt–VEGF–eNOS signaling pathway was activated after blockage of PTEN in pancreatic cancer cells
After transfection with PTEN siRNA, we examined the activation of the PI3K/Akt signaling and downstream targets. Increased PI3K–Akt phosphorylation was seen in PTEN siRNA transfected cells (Fig. 6a, b), but not in untreated or in control siRNA transfected cells. Akt phosphorylation was inhibited by LY294002 (PI3-kinase inhibitor) in PTEN siRNA transfected cancer cell (Fig. 6c). We also investigated the interaction between VEGF expression and PTEN suppression. The data showed that inhibition of PTEN expression resulted in enhanced expression of VEGF, but this function was clearly decreased by LY294002 and Akt inhibitor in PTEN siRNA transfected cells. eNOS activity of pancreatic cancer cell was remarkably enhanced by VEGF stimulation in a time- and dose-dependent manner (Fig. 6d). Thus, in PTEN siRNA transfected pancreatic cancer cells, the PI3K–Akt signaling pathway was activated, subsequently enhancing VEGF expression and increasing eNOS activation.
Activation of PI3K/Akt/VEGF/eNOS signaling pathway by Knockdown of PTEN. a PTEN siRNA-induced phosphorylated PI3K in human pancreatic cancer cells. After transfected with PTEN siRNA for 48 h, harvested cells were lysed and 30 µg of lysate protein were used for immunoblotting with anti-phospho-PI3K antibody. Detection of total PI3K levels served as a loading control. b Effect of knockdown of PTEN on phosphorylated PI3K in pancreatic cancer cells. After transfected with PTEN siRNA and incubated for 24 h, then added 50 μM PI3-kinase inhibitor (LY294002), and culture continued for 24 h. Lysates were immunoblotting as above, but with anti-phospho-Akt antibody. Total Akt protein served as a loading control. c Effect of LY294002 and Akt inhibitors on expression of VEGF by PTEN siRNA transfected cells. BxPC-3 and Capan-2 were treated with PTEN siRNA and incubated for 24 h, at which point 50 μM LY294002, and 50 μM Akt inhibitor were added and cells incubated for another 24 h. Harvested cells were lysed and 40 µg were used for immunoblotting with anti-VEGF antibody. β-actin was served as a loading control. d Involvement of VEGF with the activation of eNOS in pancreatic cancer cells. VEGF and downstream eNOS activation were assessed as described in Materials and Methods. BxPC-3 and Capan-2 cells were incubated with 50 ng/ml VEGF for 0–1 5 min or with 0–100 ng/ml for 5 min, and cell lysates were assessed for phosphorylated and total eNOS by western blot
Discussion
Angiogenesis is a complex multistep process mediated by several endothelial receptor tyrosine kinase and their ligands. These receptor–ligand systems regulate diverse functions of endothelial cells, including extracellular matrix remodeling, endothelial cell growth, migration, capillary tube formation, and survival, all of which are necessary for proper vascular development. VEGF/VEGF receptor signaling is well known to regulate each of these process [20, 25, 26], and many of them have been linked to PI3K signaling [21, 27]. Thus, the phospholipid second messengers generated by PI3K provide a common mechanism for multiple steps during angiogenesis. In previous studies, we showed that IGF-1 induced the dephosphorylation of PTEN and up-regulated cellular invasiveness and proliferation through PI3K–PTEN–Akt–NF-κB signaling pathway. We also found a negative correlation between PTEN expression and liver metastasis in pancreatic cancer cells. Knockdown of PTEN enhanced the invasiveness and proliferation of pancreatic cancer cells [28]. Previous reports demonstrated that activation of the PI3K/Akt survival signal pathway and the endothelial specific eNOS/NO pathway are closely associated with vascular remodeling and angiogenesis [29–31]. VEGF increases endothelial cell survival and angiogenesis by promoting Akt-dependent eNOS phosphorylation and NO production, indicating that NO production can be regulated by phosphorylation-dependent activation of eNOS [32].
In the present study, we found that PTEN regulates angiogenesis and enhances the secreted VEGF by pancreatic cancer cells. Our ELISA result showed that VEGF secretion was regulated by PTEN. When PTEN was knocked down, VEGF secretion increased 1.5–2 times, and no significant inhibition of VEGF secretion by exogenous PTEN was observed (data not shown). Therefore, to further investigate the role of PTEN in the metastatic potential of pancreatic cancer cells, we examined the impact of PTEN on the interaction between tumor and tumor microenvironment, especially from the standpoint of tumor angiogenesis rather than direct influence on tumor itself. Our results indicated that HUVEC proliferation and migration were significantly enhanced by co-culture with PTEN siRNA transfected caner cells, but not by control siRNA and untreated cells. We also investigated capillary tube formation by vascular endothelial cell in vitro using the HUVEC/fibroblast system. Tube formation by HUVEC was significantly enhanced by co-culture with PTEN knockdown pancreatic cancer cells, and this function was most blocked by specific VEGFR2 inhibitor. These results suggested that PTEN play an important role in blockade of tumor angiogenesis. That is, knockdown of PTEN enhance stimulation of angiogenesis, by which mainly increased the secretion level of VEGF in pancreatic cancer through the active PI3K/Akt/VEGF/eNOS signaling pathway. To our knowledge, this is the first report that PTEN siRNA modulates tumor angiogenesis in pancreatic cancer cell in vitro.
The mechanism by which PTEN modulates tumor angiogenesis is not well understood. Recent in vitro studies have shown that loss of PTEN expression significantly up-regulated VEGF expression through modulation of HIF-1 alpha expression. Furthermore, VEGF-mediated pro-angiogenesis signaling via PI3K/Akt-dependent signaling pathway enhance the anti-apoptotic, proliferative, and chemotactic activity of endothelial cells as well as tumor formation [33–35]. PI3K signaling regulates many of the endothelial response required for angiogenesis. The activation of PI3K signaling pathway may be due to multiple mechanisms, including the amplification and mutations of PI3K, lost function of PTEN, and activation of RTKs. Akt is an essential downstream of PI3K in regulating tumor growth and angiogenesis [36]. Knockdown of PTEN could significantly enhance these responses. PI3K is activated downstream of other endothelial receptor tyrosine kinase that are required for vascular development, including Tie2 [37] and Tie1 [21], which play important roles in vascular maturation. Targeting PI3K or PTEN may have advantages over this approach, since many of the effects of PI3K are independent of Akt. Based on these data, we conclude that PTEN and VEGF should be considered targets in new therapeutic approach to inhibiting angiogenesis in pancreatic cancer.
References
Tao J, Xiong J, Li T, Yang Z et al (2006) Correlation between protein expression of PTEN in human pancreatic cancer and the proliferation, infiltration, metastasis and prognosis. J Huazhong Univ Sci Technol Med Sci 26:444–447. doi:10.1007/s11596-006-0417-7
Hsu SC, Volpert OV, Steck PA, Mikkelsen T et al (1996) Inhibition of angiogenesis in human glioblastomas by chromosome 10 induction of thrombospondin-1. Cancer Res 56:5684–5691
Stambolic V, Suzuki A (1998) Negative regulation of PKB/Akt-dependent cell survival by the tumor suppressor PTEN. Cell 95:29–39. doi:10.1016/S0092-8674(00)81780-8
Stambolic V, Tsao MS, Macpherson D, Suzuki A et al (2000) High incidence of breast and endometrial neoplasia resembling human Cowden syndrome in pten+/− mice. Cancer Res 60:3605–3611
Liaw D, Marsh DJ, Li J, Dahia PL et al (1997) Germline mutations of the PTEN gene in Cowden disease, an inherited breast and thyroid cancer syndrome. Nat Genet 16:64–67. doi:10.1038/ng0597-64
Marsh DJ, Dahia PL, Zheng Z, Liaw D et al (1997) Germline mutations in PTEN are present in Bannayan-Zonana syndrome. Nat Genet 16:333–334. doi:10.1038/ng0897-333
Chow JY, Quach KT, Cabrera BL, Cabral JA et al (2007) RAS/ERK modulates TGFbeta-regulated PTEN expression in human pancreatic adenocarcinoma cells. Carcinogenesis 28:2321–2327. doi:10.1093/carcin/bgm159
Cantley LC, Neel BG (1999) New insights into tumor suppression: PTEN suppresses tumor formation by restraining the phosphoinositide 3-kinase/AKT pathway. Proc Natl Acad Sci USA 96:4240–4245. doi:10.1073/pnas.96.8.4240
Dong JT, Sipe TW, Hyytinen ER, Li CL, Heise C et al (1998) PTEN/MMAC1 is infrequently mutated in pT2 and pT3 carcinomas of the prostate. Oncogene 17:1979–1982. doi:10.1038/sj.onc.1202119
Simpson L, Parsons R (2001) PTEN: life as a tumor suppressor. Exp Cell Res 264:29–41. doi:10.1006/excr.2000.5130
Alvarez-Nunez F, Bussaglia E, Mauricio D, Ybarra J et al (2006) PTEN promoter methylation in sporadic thyroid carcinomas. Thyroid 16:17–23. doi:10.1089/thy.2006.16.17
Mirmohammadsadegh A, Marini A, Nambiar S, Hassan M et al (2006) Epigenetic silencing of the PTEN gene in melanoma. Cancer Res 66:6546–6552. doi:10.1158/0008-5472.CAN-06-0384
Marsit CJ, Zheng S, Aldape K, Hinds PW et al (2005) PTEN expression in non-small-cell lung cancer: evaluating its relation to tumor characteristics, allelic loss, and epigenetic alteration. Hum Pathol 36:768–776. doi:10.1016/j.humpath.2005.05.006
Wiencke JK, Zheng S, Jelluma N, Tihan T et al (2007) Methylation of the PTEN promoter defines low-grade gliomas and secondary glioblastoma. Neuro-oncology 9:271–279. doi:10.1215/15228517-2007-003
Aoki M, Schetter C, Himly M, Batista O et al (2000) The catalytic subunit of phosphoinositide 3-kinase: requirements for oncogenicity. J Biol Chem 275:6267–6275. doi:10.1074/jbc.275.9.6267
Sakurada A, Suzuki A, Sato M, Yamakawa H et al (1997) Infrequent genetic alterations of the PTEN/MMAC1 gene in Japanese patients with primary cancers of the breast, lung, pancreas, kidney, and ovary. Jpn J Cancer Res 88:1025–1028
Escriva M, Peiro S, Herranz N, Villagrasa P, Dave N et al (2008) Repression of PTEN phosphatase by Snail1 transcriptional factor during gamma radiation-induced apoptosis. Mol Cell Biol 28:1528–1540. doi:10.1128/MCB.02061-07
Wang Q, Zhou Y, Wang X, Chung DH et al (2007) Regulation of PTEN expression in intestinal epithelial cells by c-Jun NH2-terminal kinase activation and nuclear factor-kappaB inhibition. Cancer Res 67:7773–7781. doi:10.1158/0008-5472.CAN-07-0187
Jiang BH, Zheng JZ, Aoki M et al (2000) Phosphatidylinositol 3-kinase signaling mediates angiogenesis and expression of vascular endothelial growth factor in endothelial cells. Proc Natl Acad Sci USA 97:1749–1753. doi:10.1073/pnas.040560897
Ferrara N (2000) Vascular endothelial growth factor and the regulation of angiogenesis. Recent Prog Horm Res 55:15–35 (discussion 35–16)
Thakker GD, Hajjar DP, Muller WA et al (1999) The role of phosphatidylinositol 3-kinase in vascular endothelial growth factor signaling. J Biol Chem 274:10002–10007. doi:10.1074/jbc.274.15.10002
Hamada K, Sasaki T, Koni PA, Natsui M et al (2005) The PTEN/PI3K pathway governs normal vascular development and tumor angiogenesis. Genes Dev 19:2054–2065. doi:10.1101/gad.1308805
Giri D, Ittmann M (1999) Inactivation of the PTEN tumor suppressor gene is associated with increased angiogenesis in clinically localized prostate carcinoma. Hum Pathol 30:419–424. doi:10.1016/S0046-8177(99)90117-X
Wen S, Stolarov J, Myers MP, Su JD et al (2001) PTEN controls tumor-induced angiogenesis. Proc Natl Acad Sci USA 98:4622–4627. doi:10.1073/pnas.081063798
Tahir SA, Yang G, Goltsov AA et al (2008) Tumor cell-secreted caveolin-1 has proangiogenic activity in prostate cancer. Cancer Res 68:731–739. doi:10.1158/0008-5472.CAN-07-2668
Chung BH, Kim CK (2008) Icariin stimulates angiogenesis by activating the MEK/ERK-and PI3K/Akt/eNOS-dependent signal pathway in human endothelial cells. Biochem Biophys Res Commun 376:404–408. doi:10.1016/j.bbrc.2008.09.001
Ferrara N, Chen H, Davis-Smyth T et al (1998) Vascular endothelial growth factor is essential for corpus luteum angiogenesis. Nat Med 4:336–340. doi:10.1038/nm0398-336
Ma J, Sawai H, Matsuo Y, Ochi N et al (2008) IGF-1 mediates PTEN suppression and enhances cell invasion and proliferation via activation of IGF-1/PI3K/Akt signaling pathway in pancreatic cancer cells. J Surg Res. doi:10.1016/j.jss.2008.08.016
Dayanir V, Meyer RD, Lashkari K, Rahimi N (2001) Identification of tyrosine residues in vascular endothelial growth factor receptor-2/FLK-1 involved in activation of phosphatidylinositol 3-kinase and cell proliferation. J Biochem 276:17686–17692
Lee SJ, Kim NamkoongYM, Kim CK, Lee H et al (2006) Fractalkine stimulates angiogenesis by activating the Raf-/MMK/ERK-and PI3K/Akt/eNOS-dependent signal pathway. Am J Physiol Heart Circ Physiol 291:H2836–H2846. doi:10.1152/ajpheart.00113.2006
Papapetropoulos A, Rudic RD, Sessa WC (1999) Molecular control of nitric oxide synthases in the cardiovascular system. Cardiovasc Res 43:509–520. doi:10.1016/S0008-6363(99)00161-3
Dimmeler S, Dernbach E, Zeiher AM (2000) Phosphorylation of the endothelial nitric oxide synthase at ser-1177 is required for VEGF-induced endothelial cell migration. FEBS Lett 477:258–262. doi:10.1016/S0014-5793(00)01657-4
Gomez-Manzano C, Fueyo J, Jiang H, Glass TL et al (2003) Mechanisms underlying PTEN regulation of vascular endothelial growth factor and angiogenesis. Ann Neurol 53:109–117. doi:10.1002/ana.10396
Koul D, Shen R, Garyali A, Ke LD et al (2002) MMAC/PTEN tumor suppressor gene regulates vascular endothelial growth factor-mediated angiogenesis in prostate cancer. Int J Oncol 21:469–475
Huang J, Kontos CD (2002) PTEN modulates vascular endothelial growth factor-mediated signaling and angiogenic effects. J Biol Chem 277:10760–10766. doi:10.1074/jbc.M110219200
Jiang BH, Liu LZ (2008) PI3K/PTEN signaling in tumorigenesis and angiogenesis. Biochim Biophys Acta 1784:150–158
Kontos CD, Stauffer TP, Yang WP, York JD et al (1998) Tyrosine 1101 of Tie2 is the major site of association of p85 and is required for activation of phosphatidylinositol 3-kinase and Akt. Mol Cell Biol 18:4131–4140
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ma, J., Sawai, H., Ochi, N. et al. PTEN regulate angiogenesis through PI3K/Akt/VEGF signaling pathway in human pancreatic cancer cells. Mol Cell Biochem 331, 161–171 (2009). https://doi.org/10.1007/s11010-009-0154-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-009-0154-x