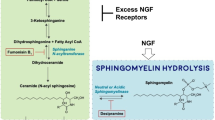
Abstract
The NGF secretion from cultured mouse astrocytes was enhanced by sublethal concentrations of phosphatidic acid (PA), ceramide, or sphingosine (Sph), and concentration dependently by lysophosphatidic acid (LPA), sphingosylphosphorylcholine (SPC), or sphingosine-1-phosphate (S1P), but was unaffected by any concentrations of phosphatidylcholine (PC), phosphatidylethanolamine (PE) or sphingomyelin (SM). The enhancement of NGF synthesis by Sph was completely inhibited by the addition of ceramide synthase inhibitor, fumonisin B1. LPA and S1P showed similar hyperbolic curves with maximum NGF secretion at concentrations of more than 50 μM, but they showed no proliferative effect on quiescent astrocytes. The mechanisms underlying the stimulation of NGF synthesis by 50 μM LPA and 50 μM S1P were further investigated by using various inhibitors. One of the protein kinase C (PKC) inhibitors, Gö6976, suppressed the LPA- and S1P-stimulated NGF synthesis by 70 and 80%, respectively. LPA and S1P were found to activate common multiple signaling pathways for NGF production, involving the activation of the protein kinase C (PKC), mitogen-activated protein (MAP) kinase, and phosphatidylinositol 3-kinase (PI-3K) pathways.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Neurotrophic factors (NTFs) are secreted proteins that regulate the survival, functional maintenance, and phenotype development of neural cells. NGF is a major NTF essential to preserve cholinergic neurons of the basal of Meynert in culture, and it has been considered as a potential therapeutic agent for the treatment of Alzheimer’s disease [1, 2]. Astrocytes support neural cells by secreting NTFs such as nerve growth factor (NGF), brain-derived neurotrophic factor (BDNF), and glial cell line-derived neurotrophic factor (GDNF). Therefore, inducers of NTF biosynthesis in astrocytes are expected to serve as effective therapeutic agents for neurodegenerative diseases, as well as for traumatic, ischemic, and inflammatory brain lesions.
Some naturally occurring lipids and their metabolites are known to act as bioactive lipids having various physiological functions. Increasing evidence indicates that bioactive lipids such as eicosanoids and lysophospholipids serve as intercellular and intracellular signaling molecules that participate in physiological and pathological functions in the brain. Previously, we found that prostaglandins and their metabolites powerfully induced NGF secretion from cultured mouse astrocytes [3]. NGF synthesis in astrocytes has already been shown to be induced by the addition of exogenous sphingomyelinase or C2-ceramide [4], or by that of lysophophatidic acid [5]. In the present study, we examined the effects of several glycerophospholipids and sphingolipids, and related compounds on the production of NGF in cultured mouse astrocytes, in order to explore the bioactive lipids having neuroprotective potential. Lysophosphatidic acid (LPA) and sphingosine-1-phosphate (S1P) were found to be the most effective for NGF synthesis in astrocytes. We further compared their intracellular signaling pathways by using various inhibitors and found that LPA and S1P activated common multiple signaling pathways for NGF production.
Materials and methods
Materials
Phosphatidylethanolamine (PE, dipalmitoyl), phosphatidic acid (PA, dipalmitoyl), lysophosphatidic acid (LPA, oleoyl), sphingomyelin (SM), sphingosine (Sph), and sphingosylphosphorylcholine (SPC) were purchased from Sigma-Aldrich (St. Louis, MO, USA). Phosphatidylcholine (PC) was from Avanti Polar Lipids (Alabaster, AL, USA). C2-, C6-, and C8-ceramides, and fumonisin B1 were from Cayman (Ann Arbor, MI, USA); and C2-dihydroceramide and sphingosine-1-phosphate (S1P) from Biomol (Plymouth Meeting, PA, USA). Pertussis toxin (PTX; an inhibitor of Gi/o) was obtained from List Biological Laboratories (Campbell, CA, USA). Wortmannin (an inhibitor of phosphatidylinositol 3-kinase), genistein (a tyrosine kinase inhibitor), H-89 (a PKA inhibitor), and indomethacin (an inhibitor of both COX1 and COX2) were purchased from Sigma-Aldrich. BIM, Gö6976, and Gö6983 (PKC inhibitors), PD98059 (a MAPK/ERK kinase inhibitor), SB203580 (a p38MAPK inhibitor), and KT-5720 (a PKA inhibitor) were from Calbiochem (San Diego, CA, USA); and NS-398 (a COX2 inhibitor) from Cayman. Dulbecco’s modified Eagle medium (DMEM) was obtained from Sigma-Aldrich; and fetal bovine serum (FBS) was from BioWhittaker (Walkersville, MD, USA). All other chemicals were obtained from either Nacalai Tesque (Kyoto, Japan) or Wako Pure Chemical Industries (Osaka, Japan).
Astrocyte culture
Cortical astrocytes were prepared from 2-day-old mice (ICR) as described previously [6]. The astrocytes were seeded in medium in 24-well plates, 48-well plates, or 6-cm dishes at 105 cells/cm2 and then cultured to confluence as described previously [3]. Confluent cells were then exposed to FBS-free DMEM containing 0.5% endotoxin-free bovine serum albumin (BSA, Nacalai Tesque) for 10 days. Afterward, the cells were incubated with different concentrations of various compounds for 24 h. PE, PA, PC, SM, Sph, SPC, and C2-, C6-, and C8-ceramides were dissolved in DMSO and diluted with the medium to give a final DMSO concentration of less than 1%. Finally, the culture medium and cells were separately collected and used for measurement of the NGF content.
The astrocytes seeded in 96-well plates were used for proliferation assays and the following NGF quantification. Proliferation assays were performed by using CellTiter 96 Aqueous One Solution Reagent (Promega, Madison, WI, USA).
Enzyme immunoassay (EIA) of NGF
The NGF content in the culture medium was measured by means of the sensitive two-site enzyme linked immunosorbent assay (ELISA) system described previously [3]. Briefly, polystyrene microtiter plates (Corning, Corning, NY, USA) were coated with anti-β-NGF monoclonal antibodies (Promega). Following treatment with 10% Block Ace (Dainippon Pharmaceutical, Osaka, Japan), aliquots of the samples were added to the plates. After overnight incubation at 4°C, the plates were incubated with anti-β-NGF monoclonal antibodies that had been biotinylated with 5-(N-succimidyl-oxycarbonyl)pentyl-d-biotinamide (Dojindo Laboratories, Kumamoto, Japan) [7]. After having been washed, the plates were incubated with streptavidin-linked horseradish peroxidase (HRP; Amersham Biosciences, Piscataway, NJ, USA); and then the enzyme reaction was carried out with a TMB Microwell Peroxidase Substrate Kit (Kirkegaard and Perry, Gaithersburg, MD, USA). The reaction product was quantitated at 450 nm with a microplate reader (Tecan Spectra 3).
Reverse transcription-polymerase chain reaction (RT-PCR)
The RT-PCR analysis was performed as described previously [3]. In brief, total RNA was extracted from astrocytes with an RN easy RNA Extraction Kit (Qiagen, Valencia, CA, USA) and then treated with DNase I (Invitrogen, San Diego, CA, USA). Reverse transcription (RT) was carried out with a Transcription Kit (Invitrogen). Aliquots of the cDNA were each amplified in 25 μl of a PCR cocktail containing each specific primer and KOD Dash DNA polymerase (Toyobo, Osaka, Japan). Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) transcripts were used as positive controls. The sets of primers for NGF and G3PDH were identical to those described previously [3]. Based on preliminary experiments that had been conducted to establish the optimal conditions for the amplification by each primer pair, the target cDNAs of NGF and GAPDH cDNA were amplified for 32 and 27 cycles, respectively. The expected product sizes were 937 bp for NGF and 983 bp for GAPDH. The PCR products were electrophoresed on an agarose gel containing ethidium bromide, and then photographed under UV light.
Statistical analysis
Data are presented as means ± S.D. Statistical significance was determined by means of one-way analysis of variance, followed by a Tukey–Kramer test with statistical analysis software (InStat, GraphPad Software).
Results
Effects of glycerolipids and sphingolipids on NGF secretion from cultured astrocytes
As shown in Fig. 1, we first studied the effects of glycerophospholipids, such as phosphatidylcholine (PC), phosphatidylethanolamine (PE), phosphatidic acid (PA), and lysophosphatidic acid (LPA), on the NGF secretion from cultured mouse astrocytes by means of a highly sensitive two-site ELISA. PC or PE had no significant effect on the NGF secretion, whereas PA or LPA stimulated it strongly. The NGF secretion was stimulated to a level of 9-fold over the control by 24 h treatment with 5 μM PA, above which concentration cell death ensued. LPA showed a hyperbolic dose-response curve with a maximum NGF level of about 12-fold over the control at concentrations of more than 50 μM without any astrocytic death (Fig. 1B).
Effects of glycerophospholipids on NGF secretion from mouse astrocytes. Cells were treated for 24 h with (A) PE (◯), PC (●), LPA (□), or PA (■) at concentrations from 0 to 20 μM or with (B) LPA (□) at concentrations from 0 to 100 μM. NGF was quantified with a two-site ELISA. Values represent the means ± SD of 3 independent determinations
Next, we examined the effects of sphingolipids and their metabolites on NGF secretion from the astrocytes. As shown in Fig. 2, the synthetic cell-permeable short alkyl-chain ceramides (C2-, C6-, and C8-ceramides) stimulated NGF secretion from the astrocytes. The C2-ceramide induced an approximately 20-fold increase in the NGF content at a concentration of 125 μM, whereas the C6- or C8-ceramide caused only an approximately 3-fold increase in the NGF content at a concentration of 20 μM. Concentrations of C6- and C8-ceramides above 20 μM and ones of C2-ceramide above 125 μM caused cell death. C2-Dihydroceramide, a structural analog of C2-ceramide, showed no significant effects, indicating the critical importance of the double bond between the fourth and fifth carbon atoms of the ceramide molecule for its enhancing effect.
Effects of sphingolipids on the NGF secretion from mouse astrocytes. Cells were treated for 24 h with the indicated concentrations of (A) C2-dihydroceramide (◯), C2-ceramide (●), C6-ceramide (□) or C8-ceramide (■), or (B) S1P (◯), SM ((●), SPC (□), and Sph (■). NGF was quantified with a two-site ELISA. Values represent the means ± SD of 3 independent determinations
Sphingomyelin (SM) had no significant effect on the NGF secretion from mouse astrocytes; whereas sphingosine, sphingosine-1-phosphate (S1P), and sphingosylphosphorylcholine (SPC) stimulated NGF secretion, the stimulation by S1P being especially pronounced (Fig. 2B). The NGF secretion was also stimulated to 9-fold of the control level by 24 h treatment with 8 μM sphingosine (Sph), above which concentration cell death occurred. S1P, which can be derived from Sph by sphingosine kinase, showed a hyperbolic dose-response curve with a maximum NGF level of about 30-fold over the control at concentrations of more than 50 μM without any cell death. SPC, having a phosphocholine moiety bound to the OH group at the first position of the sphingosine molecule, also showed a hyperbolic dose-response curve with a maximum NGF level of approximately 12-fold over the control at concentrations of more than 80 μM.
There is a pathway through which Sph is converted to ceramide by ceramide synthase, and this enzyme is known to be specifically inhibited by fumonisin B1 [8]. To test the possibility that the stimulation of NGF synthesis by Sph was due to the ceramide produced from Sph by ceramide synthase, we examined the effect of fumonisin B1. As shown in Fig. 3, fumonisin B1 inhibited the Sph-stimulated NGF secretion in a concentration-dependent manner, but did not inhibit the S1P-stimulated NGF secretion. The viability of astrocytes was unaffected by the addition of fumonisin B1. Since, neither the cell proliferation nor the stimulation of NGF synthesis were observed by the addition of fumonisin B1 alone, it was thought that activation of MAPK as reported in Swiss 3T3 fibroblasts [9] did not occur in astrocytes. The NGF secretion stimulated by Sph was almost completely inhibited by 100 μM fumonisin B1, suggesting that Sph was readily converted to ceramide by ceramide synthase and the Sph-derived ceramide enhanced the NGF synthesis.
Effect of fumonisin B1 on Sph- or S1P-stimulated NGF secretion from mouse astrocytes. Cells were incubated with the indicated agents for 24 h. Fumonisin B1 was added 1 h prior to the addition of 6 μM Sph or 50 μM S1P. NGF was quantified with a two-site ELISA. Values represent the means ± SD of 3 independent determinations
Likewise, there is a possibility that the enhancement of NGF synthesis by S1P was due to ceramide derived from Sph that had been produced from S1P by S1P phosphatase [10]. As shown in Fig. 3, fumonisin B1 caused no significant inhibition of the S1P-stimulated NGF secretion, suggesting that the effect of S1P on the NGF secretion was not due to ceramide produced from S1P, but was a direct effect of S1P.
LPA- and S1P-stimulated NGF synthesis in cultured astrocytes
The LPA and S1P are lipid mediators that evoke a variety of biological activities, and they are known to induce the proliferation of many cell types. NGF secretion from astrocytes was reported to be stimulated by cell proliferation [4]. We, therefore, studied the effect of LPA or S1P on astrocyte proliferation. As shown in Fig. 4, on the addition of 10% FCS, the proliferative activity of cultured mouse astrocytes increased to about 1.5-fold over the control, and the NGF secretion showed an approximately 20-fold increase. On the other hand, on the treatment of the astrocytes with LPA or S1P, the NGF secretion was enhanced, but the proliferative activity of the cells remained unchanged. These data indicate that LPA and S1P promoted the NGF secretion independently of cell proliferation.
Effects of LPA or S1P on the proliferative activity and NGF secretion. Cells were treated for 24 h with the indicated concentrations of LPA or S1P. NGF was quantified with a two-site ELISA (upper). The proliferation index was calculated from the ratio of the absorbance of the cells treated with the compounds to that of control cells (lower). Values represent the means ± SD of 3 independent determinations
Figure 5 shows the time course of the NGF secretion after treatment with 50 μM LPA or 50 μM S1P. The NGF content increased time-dependently and reached a maximum level at 24 h. We simultaneously investigated their effects on the NGF mRNA level by means of the RT-PCR method. The NGF mRNA level was increased from 3 h to 12 h after the treatment with LPA or S1P. Therefore, the increase in the NGF content caused by LPA or S1P was accompanied by increased NGF gene expression.
Time course of the secretion of NGF and the expression of NGF mRNA in mouse astrocytes after treatment with S1P or LPA. Cells were treated with 50 μM LPA (left) or 50 μM S1P (right) for the times indicated. NGF was quantified with a two-site ELISA (upper). Values represent the means ± SD of 3 independent determinations. Total RNA from the astrocytes treated with the compounds was analyzed by the RT-PCR method (lower)
The concentrations of LPA and S1P in serum have been reported to be 2–20 μM [11, 12] and 0.5–1.1 μM [13], respectively. But their local concentrations might exceed 50 μM at the site of inflammation. In order to clarify the mechanisms involved in the enhanced production of NGF caused by 50 μM LPA and 50 μM S1P, we investigated the effects of various inhibitors on the LPA- or S1P-stimulated NGF secretion (Fig. 6).
Effects of various inhibitors on the LPA- or S1P-induced NGF secretion. Cells were incubated with the inhibitors in the presence (closed column) or absence (open column) of 50 μM LPA (left) or 50 μM S1P (right). The inhibitors were used at the following concentrations: PTX, 100 ng/ml; BIM, 2 μM; Gö6976, 2 μM; Gö6983, 2 μM; wortmannin(1), 1 μM; wortmannin(10), 10 μM; PD98059(5), 5 μM; PD98059(20), 20 μM; SB203580(5), 5 μM; SB203580(20), 20 μM; NS-398, 20 μM; indomethacin, 20 μM; genistein, 1 μM; H-89, 10 μM; KT-5720(10), 10 nM; and KT-5720(100), 100 nM. The NGF content is expressed as the value (%) relative to that of the LPA- or S1P-induced NGF content in the absence of inhibitors. Values represent the means ± SD of 3 independent determinations. Asterisks indicate significant differences from the corresponding controls (*P < 0.01, **P < 0.001, by one-way analysis of variance)
The various physiological actions of both LPA and S1P are mediated through a G protein-coupled receptor on the surface of the cell membrane [14]. Therefore, we studied the effect of pertussis toxin (PTX), an inhibitor of Gi/o, on the LPA- or S1P-stimulated NGF secretion from cultured mouse astrocytes. As shown in Fig. 6, PTX suppressed the LPA- and S1P-stimulated NGF secretion by 52 and 25%, respectively. This result suggests that a part of the effect of LPA or S1P was due to the activation of PTX-sensitive G protein.
As also shown in Fig. 6, the protein kinase C (PKC) inhibitors (BIM, Gö6976, and Gö6983) decreased the NGF secretion by 26–80%. Therefore, PKC is involved in the mechanism underlying the LPA- and S1P-stimulated NGF secretion. The differences in the degree of inhibition among these inhibitors are thought to reflect their specificity as to inhibition of the PKC isozymes involved in NGF secretion. Since, the PKC inhibitors showed similar patterns of inhibition of the LPA- and S1P-stimulated NGF secretion, LPA and S1P were suggested to stimulate the NGF synthesis via a common signaling pathway. Gö6976 (an inhibitor of PKCα, PKCβ, and PKCμ) inhibited the NGF secretion much more strongly than BIM (an inhibitor of PKCα, PKCβ, PKCγ, PKCδ, and PKCε), and Gö6983 (an inhibitor of PKCα, PKCβ, PKCγ, PKCδ, and PKCζ), suggesting that PKCμ was involved in the pathway.
When PTX and Gö6976 were applied together, a synergistic inhibitory effect was observed. This finding suggests that PKC and Gi/o act in separate signaling pathways on the same inducer for stimulation of the NGF synthesis by LPA or S1P.
Wortmannin, a phosphatidylinositol 3-kinase (PI-3K) inhibitor, caused approximately 40% inhibition of both LPA- and S1P-stimulated NGF synthesis in a concentration-dependent manner (Fig. 6). These results indicate the involvement of the activation of PI-3K in the NGF synthesis. Inhibitors of the mitogen-activated protein (MAP) kinase pathway, PD98059 and SB203580, inhibited the LPA- and S1P-stimulated NGF synthesis in a concentration-dependent manner.
Cyclooxygenase (COX) inhibitors, i.e., NS-398 and indomethacin, had no significant effects on the LPA- or S1P-stimulated NGF synthesis. Furthermore, neither the tyrosine kinase inhibitor genistein nor the protein kinase A (PKA) inhibitors H-89 and KT-5720 had any significant effects except that H-89 slightly, but significantly, reduced the LPA-induced increase in the NGF content. These results suggest no significant involvement of COX, tyrosine kinase, or PKA in the stimulated NGF synthesis.
Discussion
The present study showed that the addition of exogenous PA, LPA, ceramide, S1P, Sph or SPC stimulated NGF synthesis and secretion in mouse astrocyte cultures. The stimulation by Sph was shown to be an indirect effect through the production of ceramide, whereas the stimulation by S1P was by a direct effect of S1P. SM did not have any effect on the NGF synthesis, suggesting that the addition of exogenous SM did not activate sphingomyelinase and thus did not enhance the intracellular ceramide level. Galve–Roperh et al. earlier reported an approximately 9-fold increase in the NGF level in cultured rat astrocytes at 24 h after the addition of 50 μM C2-ceramide [4]. The cell-permeable C2-ceramide was proposed to function as a second-messenger, modulating the activity of various protein kinases and thereby inducing NGF synthesis. In the same study, they showed through experiments involving PKC inhibitors or MAP kinase inhibitors that the ceramide-stimulated NGF synthesis was independent of the PKC pathway, but dependent on the MAP kinase pathway. In the present study, we also confirmed that an inhibitor of the MAP kinase pathway, PD98059, suppressed the C2-ceramide-stimulated NGF secretion by 82% (data not shown).
PA, LPA, S1P, and SPC belong to a class of bioactive phospholipids that evoke a wide range of biological effects in various cells. These effects are attributable to a family of cell-surface G-protein coupled receptors previously known as EDG receptors. The present study revealed that LPA and S1P, the best-studied lipid mediators, had similar dose-response and time-dependent effects in stimulating of NGF synthesis and secretion. Cultured mouse cortical astrocytes have been reported to express mRNA for multiple receptor subtypes of LPA (LPA1-3) and S1P (S1P1, 3, 4, and 5) receptors [15]. Since PTX suppressed the LPA- and S1P-stimulated NGF secretion by 52 and 25%, respectively (Fig. 6), the stimulation of NGF synthesis by these lipid mediators was shown to involve multiple receptor subtypes and thus multiple pathways as shown schematically in Fig. 7. Furthermore, the intracellular receptors for LPA and S1P other than EDG receptors might participate in the NGF synthesis and secretion. One such intracellular receptor, peroxisome proliferator-activated receptor γ (PPARγ), was identified as an intracellular receptor for LPA [16]. Although the intracellular receptor for S1P remains unknown, some reports have supported the concept of intracellular actions of S1P independent of S1P receptors [17]. The high concentrations (50 μM) of LPA and S1P used in the present study might indicate that LPA- and S1P-stimulated NGF secretion was mediated via these intracellular receptors. The Gi/o proteins were reported to contribute to the LPA- and S1P-mediated activation of MAPK pathways, since PTX suppressed the phosphorylation of ERK1/2 and SAPK/JNK [15]. NGF production by ceramide was reported to be mediated through activation of the MAP kinase cascade and to be independent of the PKC pathway [4]. In contrast to the case of ceramide, the NGF secretion caused by LPA and S1P was found to be highly dependent on the PKC pathway and only partially on the MAP kinase pathway (Fig. 6). The experiments involving astrocytes treated with various pharmacological inhibitors revealed very similar inhibitory profiles for the LPA- and S1P-stimulated NGF synthesis. Therefore, we consider LPA and S1P to have activated common multiple signaling pathways to produce NGF. When the inhibitory profiles were compared in detail, the LPA-stimulated NGF synthesis seemed more sensitive to the inhibitors of the Gi/o and MAP kinase pathways, and less sensitive to the PKC inhibitors, than the S1P-stimulated NGF synthesis. These differences might reflect differences between the multiple LPA and S1P receptors.
In the present study, PA and SPC also stimulated the NGF secretion in mouse astrocyte cultures. The PA-induced NGF production might have been mediated via the hypothetical PA receptor, since PA was found to produce NGF more effectively than LPA (Fig. 1), which is probably produced from PA by PA-specific phospholipase A2 [18]. SPC-induced NGF production seemed to be mediated via S1P receptors, because SPC showed a hyperbolic dose-response curve similar to that for S1P (Fig. 2B) and many of the SPC effects were reported to be mediated by low-affinity interactions with the S1P receptors [19].
NGF synthesis was reported to be closely associated with cell growth in cultured mouse astrocytes [20]. LPA and S1P have been shown to induce the proliferation of astrocytes in culture [5, 21–25]. LPA-induced NGF expression has already been studied in rat astrocytes, and it was reported to be accompanied by cellular proliferation [5]. S1P was shown to induce the expression of GDNF in rat cortical astrocytes, and this S1P-induced GDNF production was shown to be accompanied by cellular proliferation [25]. On the other hand, there have been some reports indicating that LPA is not a mitogen for astrocytes [26–28]. These discrepancies in the responses to LPA might be due to differences in the origin, species, developmental stage, and culture conditions of the astrocytes. In the present study, both LPA and S1P induced NGF production in mouse astrocytes in culture, but this production was not accompanied by cellular proliferation. Therefore, LPA- and S1P-induced NGF production proved to be independent of cell proliferation. Since the astrocytes used in the present study were quiescent cells that had been cultured for 10 days in FBS-free medium containing 0.5% BSA, the proliferative response of the cells might have been suppressed. Quiescent astrocytes might be less sensitive to the proliferative activity of S1P and LPA than growing astrocytes.
References
Weis C, Marksteiner J, Humpel C (2001) Nerve growth factor and glial cell line-derived neurotrophic factor restore the cholinergic neuronal phenotype in organotypic brain slices of the basal nucleus of Meynert. Neuroscience 102:129–138
Lad SP, Neet KE, Mufson EJ (2003) Nerve growth factor: structure, function and therapeutic implications for Alzheimer’s disease. Curr Drug Targets CNS Neurol Disord 2:315–334
Toyomoto M, Ohta M, Okumura K et al (2004) Prostaglandins are powerful inducers of NGF and BDNF production in mouse astrocyte cultures. FEBS Lett 562:211–215
Galve-Roperh I, Haro A, Diaz-Laviada I (1997) Induction of nerve growth factor synthesis by sphingomyelinase and ceramide in primary astrocyte cultures. Brain Res Mol Brain Res 52:90–97
Tabuchi S, Kume K, Aihara M et al (2000) Expression of lysophosphatidic acid receptor in rat astrocytes: mitogenic effect and expression of neurotrophic genes. Neurochem Res 25:573–582
Furukawa S, Furukawa Y, Satoyoshi E et al (1986) Synthesis and secretion of nerve growth factor by mouse astroglial cells in culture. Biochem Biophys Res Commun 136:57–63
Murase K, Takeuchi R, Furukawa S et al (1990) Highly sensitive enzyme immunoassay for beta-nerve growth factor (NGF): a tool for measurement of NGF level in rat serum. Biochem Int 22:807–813
Merrill AH Jr., van Echten G, Wang E et al (1993) Fumonisin B1 inhibits sphingosine (sphinganine) N-acyltransferase and de novo sphingolipid biosynthesis in cultured neurons in situ. J Biol Chem 268:27299–27306
Wattenberg EV, Badria FA, Shier WT (1996) Activation of mitogen-activated protein kinase by the carcinogenic mycotoxin fumonisin B1. Biochem Biophys Res Commun 227:622–627
Johnson KR, Johnson KY, Becker KP et al (2003) Role of human sphingosine-1-phosphate phosphatase 1 in the regulation of intra- and extracellular sphingosine-1-phosphate levels and cell viability. J Biol Chem 278:34541–34547
Eichholtz T, Jalink K, Fahrenfort I et al (1993) The bioactive phospholipid lysophosphatidic acid is released from activated platelets. Biochem J 291:677–680
Tokumura A, Iimori M, Nishioka Y et al (1994) Lysophosphatidic acids induce proliferation of cultured vascular smooth muscle cells from rat aorta. Am J Physiol 267:C204–C210
Caligan TB, Peters K, Ou J et al (2000) A high-performance liquid chromatographic method to measure sphingosine 1-phosphate and related compounds from sphingosine kinase assays and other biological samples. Anal Biochem 281:36–44
Anliker B, Chun J (2004) Lysophospholipid G protein-coupled receptors. J Biol Chem 279:20555–20558
Sorensen SD, Nicole O, Peavy RD et al (2003) Common signaling pathways link activation of murine PAR-1, LPA, and S1P receptors to proliferation of astrocytes. Mol Pharmacol 64:1199–1209
McIntyre TM, Pontsler AV, Silva AR et al (2003) Identification of an intracellular receptor for lysophosphatidic acid (LPA): LPA is a transcellular PPARγ agonist. Proc Natl Acad Sci USA 100:131–136
Spiegel S, Milstien S (2003) Exogenous and intracellularly generated sphingosine 1-phosphate can regulate cellular processes by divergent pathways. Biochem Soc Trans 31:1216–1219
Thomson FJ, Clark MA (1995) Purification of a phosphatidic-acid-hydrolysing phospholipase A2 from rat brain. Biochem J 306:305–309
Meyer zu Heringdorf D, Himmel HM, Jakobs KH (2002) Sphingosylphosphorylcholine-biological functions and mechanisms of action. Biochim Biophys Acta 1582:178–189
Furukawa S, Furukawa Y, Satoyoshi E et al (1987) Synthesis/secretion of nerve growth factor is associated with cell growth in cultured mouse astroglial cells. Biochem Biophys Res Commun 142:395–402
Keller JN, Steiner MR, Holtsberg FW et al (1997) Lysophosphatidic acid-induced proliferation-related signals in astrocytes. J Neurochem 69:1073–1084
Ramakers GJ, Moolenaar WH (1998) Regulation of astrocyte morphology by RhoA and lysophosphatidic acid. Exp Cell Res 245:252–262
Pebay A, Toutant M, Premont J et al (2001) Sphingosine-1-phosphate induces proliferation of astrocytes: regulation by intracellular signalling cascades. Eur J Neurosci 13:2067–2076
Steiner MR, Urso JR, Klein J et al (2002) Multiple astrocyte responses to lysophosphatidic acids. Biochim Biophys Acta 1582:154–160
Yamagata K, Tagami M, Torii Y et al (2003) Sphingosine 1-phosphate induces the production of glial cell line-derived neurotrophic factor and cellular proliferation in astrocytes. Glia 41:199–206
Tigyi G, Dyer DL, Miledi R (1994) Lysophosphatidic acid possesses dual action in cell proliferation. Proc Natl Acad Sci U S A 91:1908–1912
Fuentes E, Nadal A, McNaughton PA (1999) Lysophospholipids trigger calcium signals but not DNA synthesis in cortical astrocytes. Glia 28:272–276
Pebay A, Torrens Y, Toutant M et al (1999) Pleiotropic effects of lysophosphatidic acid on striatal astrocytes. Glia 28:25–33
Acknowledgment
This work was supported by a Grant-in-Aid for High Technology Research from the Ministry of Education, Culture, Sports, Science, and Technology, Japan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Furukawa, A., Kita, K., Toyomoto, M. et al. Production of nerve growth factor enhanced in cultured mouse astrocytes by glycerophospholipids, sphingolipids, and their related compounds. Mol Cell Biochem 305, 27–34 (2007). https://doi.org/10.1007/s11010-007-9524-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-007-9524-4