Abstract
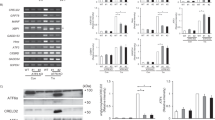
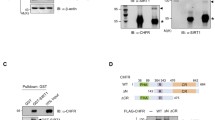
Sensitive to Apoptosis Gene (SAG), a RING component of SCF E3 ubiquitin ligase, was shown to be phosphorylated by protein kinase CK2 at the Thr10 residue. It is, however, unknown whether this phosphorylation is stress-responsive or whether the phosphorylation changes its E3 ubiquitin ligase activity. To address these, we made a specific antibody against the phosphor-SAGThr10. Transient transfection experiment showed that SAG was phosphorylated at Thr10 which can be significantly inhibited by TBB, a relatively specific inhibitor of protein kinase CK2. To determine whether this SAG phosphorylation is stress-responsive, we defined a chemical-hypoxia condition in which SAG and CK2 were both induced. Under this condition, we failed to detect SAG phosphorylation at Thr10, which was readily detected, however, in the presence of MG132, a proteasome inhibitor, suggesting that the phosphorylated SAG has undergone a rapid degradation. To further define this, we made two SAG mutants, SAG-T10A which abolishes the SAG phosphorylation and SAG-T10E, which mimics the constitutive SAG phosphorylation. The half-life study revealed that indeed, SAG-T10E has a much shorter protein half-life (2 h), as compared to wild-type SAG (10 h). Again, rapid degradation of SAG-T10E in cells can be blocked by MG132. Thus, it appears that CK2-induced SAG phosphorylation at Thr10 regulates its stability through a proteasome-dependent pathway. Immunocytochemistry study showed that SAG as well as its phosphorylation mutants, was mainly localized in nucleus and lightly in cytoplasm. Hypoxia condition did not change their sub-cellular localization. Finally, an in vitro ubiqutination assay showed that SAG mutation at Thr10 did not change its E3 ligase activity when complexed with cullin-1. These studies suggested that CK2 might regulate SAG-SCF E3 ligase activity through modulating SAG’s stability, rather than its enzymatic activity directly.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- CHX:
-
cycloheximide
- CK2:
-
protein kinase CK2
- ROC1:
-
Regulator of Cullin-1
- Rbx1:
-
RING box protein-1
- SAG:
-
Sensitive to Apoptosis Gene
- SCF:
-
Skp1, cullin, F-box protein complex
References
Deshaies RJ: SCF and Cullin/Ring H2-based ubiquitin ligases Annu Rev Cell Dev Biol 15: 435–467, 1999
Sun Y, Tan M, Duan H, Swaroop M: SAG/ROC/Rbx/Hrt, a zinc RING finger gene family: molecular cloning, biochemical properties, and biological functions Antioxid Redox Signal 3: 635–650, 2001
Sun Y: Induction of glutathione synthetase by 1,10-phenanthroline FEBS Lett 408: 16–20, 1997
Duan H, Wang Y, Aviram M, Swaroop M, Loo JA, Bian J, Tian Y, Mueller T, Bisgaier CL, Sun Y: SAG, a novel zinc RING finger protein that protects cells from apoptosis induced by redox agents Mol Cell Biol 19: 3145–3155, 1999
Swaroop M, Bian J, Aviram M, Duan H, Bisgaier CL, Loo JA, Sun Y: Expression, purification, and biochemical characterization of SAG, a RING finger redox sensitive protein Free Radicals Biol Med 27: 193–202, 1999
Kim SY, Bae YS, Park JW: Thio-linked peroxidase activity of human sensitive to apoptosis gene (SAG) protein Free Radic Res 36: 73–78, 2002
Kim SY, Lee JH, Yang ES, Kil IS, Bae YS: Human sensitive to apoptosis gene protein inhibits peroxynitrite-induced DNA damage Biochem Biophys Res Commun 301: 671–674, 2003
Sun Y: Alteration of SAG mRNA in human cancer cell lines: requirement for the RING finger domain for apoptosis protection Carcinogenesis 20: 1899–1903, 1999
Yang GY, Pang L, Ge HL, Tan M, Ye W, Liu XH, Huang FP, Wu DC, Che XM, Song Y, Wen R, Sun Y: Attenuation of ischemia-induced mouse brain injury by SAG, a redox- inducible antioxidant protein J Cereb Blood Flow Metab 21: 722–733, 2001
Chanalaris A, Sun Y, Latchman DS, Stephanou A: SAG attenuates apoptotic cell death caused by simulated ischaemia/reoxygenation in rat cardiomyocytes J Mol Cell Cardiol 35: 257–264, 2003
Duan H, Tsvetkov LM, Liu Y, Song Y, Swaroop M, Wen R, Kung HF, Zhang H, Sun Y: Promotion of S-phase entry and cell growth under serum starvation by SAG/ROC2/Rbx2/Hrt2, an E3 ubiquitin ligase component: association with inhibition of p27 accumulation Mol Carcinog 30: 37–46, 2001
Huang Y, Duan H, Sun Y: Elevated expression of SAG/ROC2/Rbx2/Hrt2 in human colon carcinomas: SAG does not induce neoplastic transformation, but its antisense transfection inhibits tumor cell growth Mol Carcinog 30: 62–70, 2001
Swaroop M, Wang Y, Miller P, Duan H, Jatkoe T, Madore S, Sun Y: Yeast homolog of human SAG/ROC2/Rbx2/Hrt2 is essential for cell growth, but not for germination: Chip profiling implicates its role in cell cycle regulation Oncogene 19: 2855–2866, 2000
Sasaki H, Yukiue H, Kobayashi Y, Moriyama S, Nakashima Y, Kaji M, Fukai I, Kiriyama M, Yamakawa Y, Fujii Y: Expression of the sensitive to apoptosis gene, SAG, as a prognostic marker in nonsmall cell lung cancer Int J Cancer 95: 375–377, 2001
Pan ZQ, Kentsis A, Dias DC, Yamoah K, Wu K: Nedd8 on cullin: building an expressway to protein destruction Oncogene 23: 1985–1997, 2004
Pinna LA: A historical view of protein kinase CK2 Cell Mol Biol Res 40: 383–390, 1994
Meggio F, Pinna LA: One-thousand-and-one substrates of protein kinase CK2? Faseb J 17: 349–368, 2003
Ahmed K, Gerber DA, Cochet C: Joining the cell survival squad: an emerging role for protein kinase CK2 Trends Cell Biol 12: 226–230, 2002
Litchfield DW: Protein kinase CK2: structure, regulation and role in cellular decisions of life and death. Biochem J 369: 1–15, 2003
Guo C, Yu S, Davis AT, Wang H, Green JE, Ahmed K: A potential role of nuclear matrix-associated protein kinase CK2 in protection against drug-induced apoptosis in cancer cells J Biol Chem 276: 5992–5999, 2001
Lebrin F, Chambaz EM, Bianchini L: A role for protein kinase CK2 in cell proliferation: evidence using a kinase-inactive mutant of CK2 catalytic subunit alpha Oncogene 20: 2010–2022, 2001
Wang H, Davis A, Yu S, Ahmed K: Response of cancer cells to molecular interruption of the CK2 signal Mol Cell Biochem 227: 167–174, 2001
Ruzzene M, Penzo D, Pinna LA: Protein kinase CK2 inhibitor 4,5,6,7-tetrabromobenzotriazole (TBB) induces apoptosis and caspase-dependent degradation of haematopoietic lineage cell-specific protein 1 (HS1) in Jurkat cells Biochem J 364: 41–47, 2002
Ravi R, Bedi A: Sensitization of tumor cells to Apo2 ligand/TRAIL-induced apoptosis by inhibition of casein kinase II Cancer Res 62: 4180–4185, 2002
Yenice S, Davis AT, Goueli SA, Akdas A, Limas C, Ahmed K: Nuclear casein kinase 2 (CK-2) activity in human normal, benign hyperplastic, and cancerous prostate Prostate 24: 11–16, 1994
Daya-Makin M, Sanghera JS, Mogentale TL, Lipp M, Parchomchuk J, Hogg JC, Pelech SL: Activation of a tumor-associated protein kinase (p40TAK) and casein kinase 2 in human squamous cell carcinomas and adenocarcinomas of the lung Cancer Res 54: 2262–2268, 1994
Faust RA, Niehans G, Gapany M, Hoistad D, Knapp D, Cherwitz D, Davis A, Adams GL, Ahmed K: Subcellular immunolocalization of protein kinase CK2 in normal and carcinoma cells Int J Biochem Cell Biol 31: 941–949, 1999
Stalter G, Siemer S, Becht E, Ziegler M, Remberger K, Issinger OG: Asymmetric expression of protein kinase CK2 subunits in human kidney tumors Biochem Biophys Res Commun 202: 141–147, 1994
Romieu-Mourez R, Landesman-Bollag E, Seldin DC, Traish AM, Mercurio F, Sonenshein GE: Roles of IKK kinases and protein kinase CK2 in activation of nuclear factor-kappaB in breast cancer Cancer Res 61: 3810–3818, 2001
Landesman-Bollag E, Romieu-Mourez R, Song DH, Sonenshein GE, Cardiff RD, Seldin DC: Protein kinase CK2 in mammary gland tumorigenesis Oncogene 20: 3247–3257, 2001
Sarno S, Moro S, Meggio F, Zagotto G, Dal Ben D, Ghisellini P, Battistutta R, Zanotti G, Pinna LA: Toward the rational design of protein kinase casein kinase-2 inhibitors Pharmacol Ther 93: 159–168, 2002
Ahmad KA, Wang G, Slaton J, Unger G, Ahmed K: Targeting CK2 for cancer therapy Anticancer Drugs 16: 1037–1043, 2005
Son MY, Park JW, Kim YS, Kang SW, Marshak DR, Park W, Bae YS: Protein kinase CKII interacts with and phosphorylates the SAG protein containing ring-H2 finger motif Biochem Biophys Res Commun 263: 743–748, 1999
Kim YS, Lee JY, Son MY, Park W, Bae YS: Phosphorylation of threonine-10 on CKBBP1/SAG/ROC2/Rbx2 by protein kinase CKII promotes the degradation of IκBα and p27kip1 J. Biol. Chem 278: 28462–28469, 2003
Tawfic S, Davis AT, Faust RA, Gapany M, Ahmed K: Association of protein kinase CK2 with nuclear matrix: influence of method of preparation of nuclear matrix J Cell Biochem 64: 499–504, 1997
Tan P, Fuchs SY, Chen A, Wu K, Gomez C, Ronai Z, Pan ZQ: Recruitment of a ROC1-CUL1 ubiquitin ligase by Skp1 and HOS to catalyze the ubiquitination of IkBa Mol Cell 3: 527–533, 1999
Zien P, Bretner M, Zastapilo K, Szyszka R, Shugar D: Selectivity of 4,5,6,7-tetrabromobenzimidazole as an ATP-competitive potent inhibitor of protein kinase CK2 from various sources Biochem Biophys Res Commun 306: 129–133, 2003
Unger GM, Davis AT, Slaton JW, Ahmed K: Protein kinase CK2 as regulator of cell survival: implications for cancer therapy Curr Cancer Drug Targets 4: 77–84, 2004
Wang G, Ahmad KA, Ahmed K: Role of protein kinase CK2 in the regulation of tumor necrosis factor-related apoptosis inducing ligand-induced apoptosis in prostate cancer cells Cancer Res 66: 2242–2249, 2006
Desagher S, Osen-Sand A, Montessuit S, Magnenat E, Vilbois F, Hochmann A, Journot L, Antonsson B, Martinou JC: Phosphorylation of bid by casein kinases I and II regulates its cleavage by caspase 8 Mol Cell 8: 601–611, 2001
Li PF, Li J, Muller EC, Otto A, Dietz R, von Harsdorf R: Phosphorylation by protein kinase CK2: a signaling switch for the caspase-inhibiting protein ARC Mol Cell 10: 247–258, 2002
Van Antwerp DJ, Martin SJ, Kafri T, Green DR, Verma IM: Suppression of TNF-alpha-induced apoptosis by NF-kappaB [see comments] Science 274: 787–789, 1996
Wang CY, Mayo MW, Baldwin AS Jr: TNF- and cancer therapy-induced apoptosis: potentiation by inhibition of NF-kappaB [see comments] Science 274: 784–787, 1996
Mayo MW, Wang CY, Cogswell PC, Rogers Graham KS, Lowe SW, Der CJ, Baldwin AS Jr: Requirement of NF-kappaB activation to suppress p53-independent apoptosis induced by oncogenic Ras Science 278: 1812–1815, 1997
Ahmed K, Davis AT, Wang H, Faust RA, Yu S, Tawfic S: Significance of protein kinase CK2 nuclear signaling in neoplasia J Cell Biochem Suppl Suppl 35: 130–135, 2000
Faust M, Montenarh M: Subcellular localization of protein kinase CK2. A key to its function? Cell Tissue Res 301: 329–340, 2000
Yu S, Wang H, Davis A, Ahmed K: Consequences of CK2 signaling to the nuclear matrix Mol Cell Biochem 227: 67–71, 2001
Wang H, Yu S, Davis AT, Ahmed K: Cell cycle dependent regulation of protein kinase CK2 signaling to the nuclear matrix J Cell Biochem 88: 812–822, 2003
Channavajhala P, Seldin DC: Functional interaction of protein kinase CK2 and c-Myc in lymphomagenesis Oncogene 21: 5280–5288, 2002
McElhinny JA, Trushin SA, Bren GD, Chester N, Paya CV: Casein kinase II phosphorylates I kappa B alpha at S-283, S-289, S-293, and T-291 and is required for its degradation Mol Cell Biol 16: 899–906, 1996
Tawfic S, Olson MO, Ahmed K: Role of protein phosphorylation in post-translational regulation of protein B23 during programmed cell death in the prostate gland J Biol Chem 270: 21009–21015, 1995
Torres J, Pulido R: The tumor suppressor PTEN is phosphorylated by the protein kinase CK2 at its C terminus. Implications for PTEN stability to proteasome-mediated degradation J Biol Chem 276: 993–998, 2001
Besson A, Gurian-West M, Chen X, Kelly-Spratt KS, Kemp CJ, Roberts JM: A pathway in quiescent cells that controls p27Kip1 stability, subcellular localization, and tumor suppression Genes Dev 20: 47–64, 2006
Malek NP, Sundberg H, McGrew S, Nakayama K, Kyriakides TR, Roberts JM: A mouse knock-in model exposes sequential proteolytic pathways that regulate p27Kip1 in G1 and S phase Nature 413: 323–327, 2001
Acknowledgments
This work was supported by NIH grants R01-CA111554 and R21-CA116982 (YS), and by V.A. Medical Research Fund and NIH grant CA-15062 (KA).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
He, H., Tan, M., Pamarthy, D. et al. CK2 phosphorylation of SAG at Thr10 regulates SAG stability, but not its E3 ligase activity . Mol Cell Biochem 295, 179–188 (2007). https://doi.org/10.1007/s11010-006-9287-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-006-9287-3