Abstract
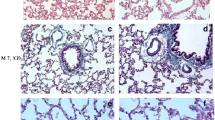
This study was designed to examine the effects of erdosteine on bleomycin (BLM)-induced lung fibrosis in rats. Thirty-three Sprague–Dawley rats were divided randomly into three groups, bleomycin alone (BLM), bleomycin + erdosteine (BLM + ERD), and saline alone (control). The BLM and BLM + ERD groups, were given 2.5 mg/kg BLM intratracheally. The first dose of oral erdosteine (10 mg/kg/day) in the BLM + ERD group was started 2 days before BLM administration and continued until animals were sacrificed. Animals were sacrificed 14 days after intratracheal instillation of BLM. The effect of erdosteine on pulmonary fibrosis was studied by analysis of bronchoalveolar lavage (BAL) fluid, histopathology, and biochemical measurements of lung tissue superoxide dismutase (SOD) and glutathione (GSH) as antioxidants, malondialdehyde (MDA) as an index for lipid peroxidation, and nitrite/nitrate levels. Bleomycin-induced lung fibrosis as determined by lung histology was prevented with erdosteine (grades of fibrosis were 4.9, 2.3, and 0.2 in BLM, BLM + ERD, and control groups, respectively). Erdosteine also prevented bleomycin-induced increase in MDA (MDA levels were 0.50 ± 0.15, 0.11 ± 0.02, and 0.087± 0.03 nmol/mg protein in BLM, BLM + ERD, and control groups, respectively) and nitrite/nitrate (nitrite/nitrate levels were 0.92 ± 0.06, 0.60 ± 0.09, and 0.56± 0.1 μmol/mg protein in BLM, BLM + ERD, and control groups respectively) levels. Bleomycin-induced decrease in GSH and SOD levels in the lung tissue also prevented by erdosteine [(GSH levels were 213.5 ± 12.4, 253.2± 25.2, and 287.9± 34.4 nmol/mg protein) (SOD levels were 1.42± 0.12, 1.75± 0.17, and 1.89± 0.09 U/mg protein) in BLM, BLM + ERD, and control groups respectively]. Erdosteine prevented bleomycin-induced increases in total cell number and neutrophil content of the BAL fluid. In conclusion, oral erdosteine is effective in prevention of BLM-induced lung fibrosis in rats possibly via the repression of neutrophil accumulation, inhibition of lipid peroxidation, and maintenance of antioxidant and free radical scavenger properties.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
McAnulty RJ, Laurent GJ: Pathogenesis of lung fibrosis and potential new therapeutic strategies. Exp Nephrol 3: 96–107, 1995
Weissler JC: Idiopathic pulmonary fibrosis: cellular and molecular pathogenesis. Am J Med Sci 297: 91–104, 1989
Kim JY, Lee KH, Lee BK, Ro JY: Peroxynitrite modulates release of inflammatory mediators from guinea pig lung mast cells activated by antigen–antibody reaction. Int Arch Allergy Immunol 137: 104–114, 2005
Bhatia M, Moochhala S: Role of inflammatory mediators in the pathophysiology of acute respiratory distress syndrome. J Pathol 202: 145–156, 2004
Fubini B, Hubbard A: Reactive oxygen species (ROS) and reactive nitrogen species (RNS) generation by silica in inflammation and fibrosis. Free Radic Biol Med 34: 1507–1516, 2003
Haddad IY, Panoskaltsis-Mortari A, Ingbar DH, YangS, Milla CE, Blazar BR: High levels of peroxynitrite are generated in the lungs of irradiated mice given cyclophosphamide and allogeneic T cells. A potential mechanism of injury after marrow transplantation. Am J Respir Cell Mol Biol 20: 1125–1135, 1999
Laurent GJ, McAnulty RJ: Protein metabolism during bleomycin-induced pulmonary fibrosis in rabbits. In vivo evidence for collagen accumulation because of increased synthesis and decreased degradation of the newly synthesized collagen. Am Rev Respir Dis 128: 82–88, 1983
Lazenby AJ, Crouch EC, McDonald JA, Kuhn C 3rd: Remodeling of the lung in bleomycin-induced pulmonary fibrosis in the rat. An immunohistochemical study of laminin, type IV collagen, and fibronectin. Am Rev Respir Dis 142: 206–214, 1990
Taooka Y, Maeda A, Hiyama K, Ishioka S, Yamakido M: Effects of neutrophil elastase inhibitor on bleomycin-induced pulmonary fibrosis in mice. Am J Respir Crit Care Med 156: 260–265, 1997
Nagai A, Aoshiba K, Ishihara Y, Inano H, Sakamoto K, Yamaguchi E, Kagawa J, Takizawa T: Administration of alpha 1-proteinase inhibitor ameliorates bleomycin-induced pulmonary fibrosis in hamsters. Am Rev Respir Dis 145: 651–656, 1992
Kida H, Yoshida M, Hoshino S, Inoue K, Yano Y, Yanagita M, Kumagai T, Osaki T, Tachibana I, Saeki Y, Kawase I: Protective effect of IL-6 on alveolar epithelial cell death induced by hydrogen peroxide. Am J Physiol Lung Cell Mol Physiol 288: 342–349, 2005
Ghazi-Khansari M, Nasiri G, Honarjoo M: Decreasing the oxidant stress from paraquat in isolated perfused rat lung using captopril and niacin. Arch Toxicol 2005
Jang YY, Song JH, Shin YK, Han ES, Lee CS: Depressant effects of ambroxol and erdosteine on cytokine synthesis, granule enzyme release, and free radical production in rat alveolar macrophages activated by lipopolysaccharide. Pharmacol Toxicol 92: 173–179, 2003
Dal Sasso M, Bovio C, Culici M, Fonti E, Braga PC: The SH-metabolite I of erdosteine, a mucolytic drug, enhances the inhibitory effect of salbutamol on the respiratory burst of neutrophils. Drugs Exp Clin Res 28: 147–154, 2002
Fadillioglu E, Erdogan H, Sogut S, Kuku I: Protective effects of erdosteine against doxorubicin-induced cardiomyopathy in rats. J Appl Toxicol 23: 71–74, 2003
Dal Sasso M, Culici M, Bianchi T, Fonti E, Braga PC: Inhibitory effects of metabolite I of erdosteine on the generation of nitric oxide and peroxynitrite chemiluminescence by human neutrophils. Pharmacology 71: 120–127, 2004
Hosoe H, Kaise T, Ohmori K: Effects on the reactive oxygen species of erdosteine and its metabolite in vitro. Arzneimittelforschung 52: 435–440, 2002
Ege E, Ilhan A, Gurel A, Akyol O, Ozen S: Erdosteine ameliorates neurological outcome and oxidative stress due to ischemia/reperfusion injury in rabbit spinal cord. Eur J Vasc Endovasc Surg 28: 379–386, 2004
Sogut S, Ozyurt H, Armutcu F, Kart L, Iraz M, Akyol O, Ozen S, Kaplan S, Temel I, Yildirim Z: Erdosteine prevents bleomycin-induced pulmonary fibrosis in rats. Eur J Pharmacol 494: 213–220, 2004
Dechant KL, Noble S: Erdosteine. Drug 52: 875–881, 1996
Ashcroft T, Simpson JM, Timbrell V: Simple method of estimating severity of pulmonary fibrosis on a numerical scale. J Clin Pathol 41: 467–470, 1988
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ: Protein measurement with the Folin phenol reagent. J Biol Chem 193: 265–275, 1951
Sun Y, Oberley LW, Li Y: A simple method for clinical assay of superoxide dismutase. Clin Chem 34: 497–500, 1988
Ellman GL: Tissue sulfhydryl groups. Arch Biochem Biophys 82: 70–77, 1959
Buege JA, Aust SD: Microsomal lipid peroxidation. Meth Enzymol 52: 302–310, 1978
Cortas NK, Wakid NW: Determination of inorganic nitrate in serum and urine by a kinetic cadmium-reduction method. Clin Chem 36: 1440–1443, 1990
Gross TJ, Hunninghake GW: Idiopathic pulmonary fibrosis. N Engl J Med 345: 517–525, 2001
Oury TD, Thakker K, Menache M, Chang LY, Crapo JD, Day BJ: Attenuation of bleomycin-induced pulmonary fibrosis by a catalytic antioxidant metalloporphyrin. Am J Respir Cell Mol Biol 25: 164–169, 2001
Otsuka M, Takahashi H, Shiratori M, Chiba H, Abe S: Reduction of bleomycin induced lung fibrosis by candesartan cilexetil, an angiotensin II type 1 receptor antagonist. Thorax 59: 31–38, 2004
Jules-Elysee K, White DA: Bleomycin-induced pulmonary toxicity. Clin Chest Med 11: 1–20, 1990
Dik WA, McAnulty RJ, Versnel MA, Naber BA, Zimmermann LJ, Laurent GJ, Mutsaers SE: Short course dexamethasone treatment following injury inhibits bleomycin induced fibrosis in rats. Thorax 58: 765–771, 2003
Izbicki G, Segel MJ, Christensen TG, Conner MW, Breuer R: Time course of bleomycin-induced lung fibrosis. Int J Exp Pathol 83: 111–119, 2002
Serrano-Mollar A, Closa D, Prats N, Blesa S, Martinez-Losa M, Cortijo J, Estrela JM, Morcillo EJ, Bulbena O: In vivo antioxidant treatment protects against bleomycin-induced lung damage in rats. Br J Pharmacol 138: 1037–1048, 2003
Mata M, Ruiz A, Cerda M, Martinez-Losa M, Cortijo J, Santangelo F, Serrano-Mollar A, Llombart-Bosch A, Morcillo EJ: Oral N-acetylcysteine reduces bleomycin-induced lung damage and mucin Muc5ac expression in rats. Eur Respir J 22: 900–905, 2003
Hagiwara SI, Ishii Y, Kitamura S: Aerosolized administration of N-acetylcysteine attenuates lung fibrosis induced by bleomycin in mice. Am J Respir Crit Care Med 162: 225–231, 2000
Thrall RS, Phan SH, McCormick JR, Ward PA: The development of bleomycin-induced pulmonary fibrosis in neutrophil-depleted and complement-depleted rats. Am J Pathol 105: 76–81, 1981
Gutteridge JM, Xiao Change F: Protection of iron-catalysed the radical damage to DNA and lipids by copper (II) bleomycin. Biochem Biophys Res Commun 99: 1354–1360, 1981
Yamazaki C, Hoshino J, Sekiguchi T, Hori Y, Miyauchi S, Mizuno S, Horie K: Production of superoxide and nitric oxide by alveolar macrophages in the bleomycin-induced interstitial pneumonia mice model. Jpn J Pharmacol 78: 69–73, 1998
Sleijfer S: Bleomycin-induced pneumonitis. Chest 120: 617–624, 2001
Hong JS, Ko HH, Han ES, Lee CS: Inhibition of bleomycin-induced cell death in rat alveolar macrophages and human lung epithelial cells by ambroxol. Biochem Pharmacol 66: 1297–1306, 2003
Tamagawa K, Taooka Y, Maeda A, Hiyama K, Ishioka S, Yamakido M: Inhibitory effects of a lecithinized superoxide dismutase on bleomycin-induced pulmonary fibrosis in mice. Am J Respir Crit Care Med 161: 1279–1284, 2000
Giri SN, Biring I, Nguyen T, Wang Q, Hyde DM: Abrogation of bleomycin-induced lung fibrosis by nitric oxide synthase inhibitor, aminoguanidine in mice. Nitric Oxide 7: 109–118, 2002
Yildirim Z, Turkoz Y, Kotuk M, Armutcu F, Gurel A, Iraz M, Ozen S, Aydogdu I, Akyol O: Effects of aminoguanidine and antioxidant erdosteine on bleomycin-induced lung fibrosis in rats. Nitric Oxide 11: 156–165, 2004
Kooy NW, Royall JA, Ye YZ, Kelly DR, Beckman JS: Evidence for in vivo peroxynitrite production in human acute lung injury. Am J Respir Crit Care Med 151: 1250–1254, 1995
El-Medany A, Hagar HH, Moursi M, At Muhammed R, El-Rakhawy FI, El-Medany G: Attenuation of bleomycin-induced lung fibrosis in rats by mesna. Eur J Pharmacol 509: 61–70, 2005
Anderson SE, Kallstrom L, Malm M, Miller-Learson A, Axellson B: Inhibition of nitric oxide synthase reduces sephadex-induced oedema formation in the rat lung. Inflamm Res 44: 418–422, 1995
Stuher DJ, Nathan CE: Nitric oxide : a macrophage product responsible for cytostasis and respiratory inhibition in tumor target cells. J Exp Med 1543–1555, 1989
Fadillioglu E, Yilmaz HR, Erdogan H, Sogut S: The activities of tissue xanthine oxidase and adenosine deaminase and the levels of hydroxyproline and nitric oxide in rat hearts subjected to doxorubicin: protective effect of erdosteine. Toxicology 191: 153–158, 2003
Yildirim Z, Sogut S, Odaci E, Iraz M, Ozyurt H, Kotuk M, Akyol O: Oral erdosteine administration attenuates cisplatin-induced renal tubular damage in rats. Pharmacol Res 47: 149–156, 2003
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Boyacı, H., Maral, H., Turan, G. et al. Effects of erdosteine on bleomycin-induced lung fibrosis in rats. Mol Cell Biochem 281, 129–137 (2006). https://doi.org/10.1007/s11010-006-0640-3
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11010-006-0640-3