Abstract
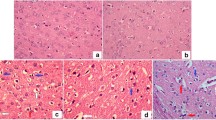
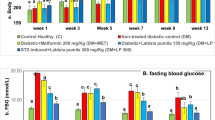
Vanadium has been reported to have broad pharmacological activity both in vitro and in vivo. Vanadium compound, sodium orthovanadate, Na3VO4, is well known for its hypoglycaemic effects. However, Na3VO4 exerts these effects at relatively high doses (0.6 mg/ml) and exhibit several toxic effects. In the present study lower doses of Na3VO4 (0.2 mg/ml) are combined with Trigonella foenum graecum seed powder (TSP), another hypoglycaemic agent, to reduce its toxicity without compromising its antidiabetic potential. The efficacy of the lower doses of Na3VO4 has been investigated in restoring the altered glucose metabolism and histological structure in the sciatic nerves in 21 and 60 days alloxan diabetic rats. A portion of the glucose was found to be channelled from the normal glycolytic route to polyol pathway, evident by the reduced hexokinase activity and increased polyol pathway enzymes aldose reductase and sorbitol dehydrogenase activity causing accumulation of sorbitol and fructose in diabetic conditions. Ultrastructural observation of the sciatic nerve showed extensive demylination and axonal loss after eight weeks of diabetes induction. Blood glucose levels increased in diabetic rats were normalized with the lower dose of vanadium and Trigonella treatment. The treatment of the diabetic rats with vanadium and Trigonella prevented the activation of the polyol pathway and sugar accumulations. The sciatic nerves were also protected against the structural abnormalities found in diabetes with Trigonella foenum graecum as well as Na3VO4. Results suggest that lower doses of Na3VO4 may be used in combination with TSP as an efficient antidiabetic agent to effectively control the long-term complications of diabetes in tissues like peripheral nerve.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Colciago A, Negri-Cesi P, Celotti F: Pathogenesis of diabetic neuropathy – Do hyperglycemia and aldose reductase inhibitors affect neuroactive steroid formation in the sciatic nerves? Exp Clin Endocrinol Diabetes 110: 22–26, 2002
Thurston JH, McDougal DBJ, Hauhart RE, Schulz DW: Effects of acute, subacute, and chronic diabetes on carbohydrate and energy metabolism in rat sciatic nerve. Relation to mechanisms of peripheral neuropathy. Diabetes 44: 190–195, 1995
Brownlee M: Biochemistry and molecular cell biology of diabetic complications. Nature 414: 813–820, 2001
Baynes JW: Role of oxidative stress in development of complications of diabetes. Diabetes 40: 405–412, 1991
Hunt JV, Dean RT, Wolff SP: Hydroxyl radical production and autoxidative glycosylation: Glucose autoxidation as the cause of protein damage in the experimental glycation model of diabetes mellitus and ageing. Biochem J 256: 205–212, 1988
Dvornik D: Hyperglycemia in the pathogenesis of diabetic complications. In: D. Porte (ed.) Aldose Reductase Inhibition. An Approach to the Prevention of Diabetic Complications. Biomedical Information Corp., New York, 1987, pp 69–151
Stevens MJ, Feldman EL, Thomas TP, Greene DA: The pathogenesis of diabetic neuropathy. In: Veves A and Conn PMC (eds) Clinical Management of Diabetic Neuropathy. Humana Press, Totowa, NJ, 1997, pp 13–47
Gabbay KH: The sorbitol pathway and the complication of diabetes. N Engl J Med 288: 831–837, 1973
Cameron NE, Cotter MA, Maxfield EK: Anti-oxidant treatment prevents the development of peripheral nerve dysfunction in streptozotocin-diabetic rats. Diabetologia 36: 299–304, 1993
Hotta N, Kakuta H, Fukasawa H, Koh N, Sakakibara F, Komori H, Sakamoto N: Effect of nicertrol on Streptozotocin-induced diabetic neuropathy in rats. Diabetes 41: 587–591, 1992
Shindo H, Tawata M, Aida K, Onaya T: The role of cyclic adenosine 3′,5′-monophosphate and polyol metabolism in diabetic neuropathy. J Clin Endocrinol Metab 74: 393–398, 1992
Nickander KK, Schmelzer JD, Rohwer DA, Low PA: Effect of alphatocopherol deficiency on indices of oxidative stress in normal and diabetic peripheral nerve. J Neurol Sci 126: 6–14, 1994
Dyck PJ, Zimmerman BR, Vilen TH, Minnerath SR, Karnes JL, Yao, JK, Poduslo JF: Nerve glucose, fructose, sorbitol, myo-inositol, and fiber degeneration and regeneration in diabetic neuropathy. N Engl J Med 319(9): 542–548, 1988
Cameron NE, Leonard MB, Ross IS, Whiting PH: The effects of sorbinil on peripheral nerve conduction velocity, polyol concentrations and morphology in the streptozotocin-diabetic rat. Diabetologia 29: 168–174, 1986
Demir N, Akkoyunlu G, Yargicoglu P, Agar A, Tanriover G, Demir R: Fibre structure of optic nerve in cadmium-exposed diabetic rats: An ultrastructural study. Int J Neurosci 113: 323–337, 2003
Greene DA, Sima AA, Stevens MJ, Feldman EL, Lattimer SA: Complications: neuropathy, pathogenetic considerations. Diabetes Care 15: 1902–1925, 1992
Greene DA, Sima AF, Pfeifer MA, Albers JW: Diabetic neuropathy. Annu Rev Med 41: 303–317, 1990
Yagihashi S, Kudo K, Nishihira M: Peripheral nerve structures of experimental diabetes rats and the effect of insulin treatment. Tohoku J Exp Med 127: 35–44, 1979
Pierson CR, Zhang W, Murakawa Y, Sima AA: Insulin deficiency rather than hyperglycemia accounts for neurotrophic responses and nerve fiber regeneration in type 1 diabetic neuropathy. Neuropathol Exp Neurol 62: 260–271, 2003
Sumner CJ, Sheth S, Griffin JW, Cornblath DR, Polydefkis M: The spectrum of neuropathy in diabetes and impaired glucose tolerance. Neurology 60: 108–111, 2003
Cameron NE, Cotter MA: Potential therapeutic approaches to the treatment or prevention of diabetic neuropathy: Evidence from experimental studies. Diabet Med 10: 593–605, 1993
Brichard SM, Okitolonda W, Henquin, JC: Long term improvement of glucose homeostasis by vanadate treatment in diabetic rats. Endocrinology 123: 2048–2053, 1988
Cameron NE, Cotter MA, Basso M, Hohman TC: Comparison of the effects of inhibitors of aldose reductase and sorbitol dehydrogenase on neurovascular function, nerve conduction and tissue polyol pathway metabolites in Streptozotocin-diabetic rats. Diabetologia 40: 271–281, 1997
Raju J, Gupta D, Rao AR, Yadava PK, Baquer NZ: Trigonella foenum graecum (fenugreek) seed powder improves glucose homeostasis in alloxan diabetic rat tissues by reversing the altered glycolytic, gluconeogenic and lipogenic enzymes. Mol Cell Biochem 224: 45–51, 2001
Domingo JL: Vanadium and tungsten derivatives as antidiabetic agents: a review of their toxic effects. Biol Trace Elem Res 88: 97–112, 2002
Sochor M, Baquer NZ, McLean P: Glucose over- and underutilization in diabetes: Comparative studies on the change in activities of enzyme of glucose metabolism in rat kidney and liver. Mol Physiol 7: 51–68, 1985
Sochor M, Baquer NZ, McLean P: Effect of aldose reductase inhibiter sorbinil on metabolites in diabetic lens. Biochem Pharmacol 37: 3349–3356, 1988
Gumma K, McLean P: The kinetic quantitation of ATP. D-glucose-6-phosphotransferase. FEBBS Lett 27: 293–297, 1972
Kinoshita JH, Futterman S, Satoh K, Marola LO: Factors affecting the formation of sugar alcohols in ocular len. Biochem Biophys Acta 74: 340–350, 1963
Gerlach U, Hiby W: Assay of sorbitol. In: H.U. Bergmeyer (ed), Methods in Enzymatic Analysis. Academic Press, New York, 1974, pp 569–573
Baquer NZ, McLean P, Greenbaum AL: Enzymic differentiation in pathways of carbohydrate metabolism in developing brain. Biochem Biophys Res Commun 53: 1282–1288, 1973
Bergmeyer HU, Bernt E, Schmidt F, Stork F: Assay for hexoses. In: H.U. Bergmeyer (ed), Methods in Enzymatic Analysis, Vol 3, Academic Press, New York, 1974, pp 1196–1201
Bergmeyer HU, Bernt E: Assay for hexoses. In: H.U. Bergmeyer (ed), Methods in Enzymatic Analysis. Academic Press, New York, 1974, pp 1304–1307
Malone JI, Knox G, Benford S, Tedesco TA: Red cell sorbitol: An indicator of diabetic control. Diabetes 29: 861–864, 1980
Griffith OW: Determination of glutathione and glutathione disulfide using glutathione reductase and 2-vinylpyridine. Anal Biochem 107: 207–212, 1980
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ: Protein measurement with the folin phenol reagent. J Biol Chem 193: 265–275, 1951
Raheja BS, Iyer PD, Bhargava D, Krishnaswamy PR: Glycosylated haemoglobin: Its value in the management of diabetes mellitus. J Assoc Physicians India 29: 609–613, 1981
Pearse AGE: Histochemistry: Theoretical and Applied. Vol. 1. J & A Churchill Ltd., 1968, p. 576
Sima AA, Bouchier M, Christensen H: Axonal atrophy in sensory nerves of diabetic BB-Wistar rat: A possible early correlate of human diabetic neuropathy. Ann Neurol 13: 264–272, 1983
Dolman CL: The morbid anatomy of diabetic neuropathy. Neurology 13: 135–142, 1963
Becker DJ, Ongemba LN, Henquin JC: Comparison of the effects of various vanadium salts on glucose homeostasis in streptozotocin-diabetic rats. Eur J Pharmacol 260: 169–175, 1994
Meyerovitch J FZ, Sack J, Shechter Y: Oral administration of Vanadate normalizes blood glucose levels in Streptozotocin-treated rats. J Biol Chem 262: 6658–6662, 1987
Heylinger CE, Tahiliani AG, McNeill JH: Effect of Vanadate on elevated blood glucose and depressed cardiac performance of diabetic rats. Science 227: 1474–1477, 1985
Vats V, Yadav SP, Grover JK: Effect of T. foenum graecum on glycogen content of tissues and the key enzymes of carbohydrate metabolism. J Ethnopharmacol 85: 237–242, 2003
Anonymous: The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. The Diabetes Control and Complications Trial Research Group. N Engl J Med 329: 977–986, 1993
Kazuhiro S, Soroku Y: Peripheral nerve pathology in rats with Streptozotocin-induced insulinoma. Acta Neuropathol 91: 616–623, 1996
Kamakura K, Ishiura S, Sugita H, Toyokura Y: Identification of Ca+2-activated neutral protease in the peripheral nerve and its effects on neurofilament degradation. J Neurochem 40: 908–913, 1983
Greene DA: Metabolic abnormalities in diabetic peripheral nerve: Relation to impaired function. Metabolism 32: 118–123, 1983
Wolff SP: The potential role of oxidative stress in diabetes and its complications: Scientific aspects. New York, Churchill Livingstone, 1987, pp 167–220
Kamiya H, Nakamura J, Hamada Y, Nakashima E, Naruse K, Kato K, Yasuda YNH: Polyol pathway and protein kinase C activity of rat Schwannoma cells. Diabetes Metab Res Rev 19: 131–139, 2003
Demir N, Akkoyunlu G, Agar A, Yargicoglu P, Tanriover G, Demir R: Effect of cadmium on sciatic nerve in diabetic rats: An ultrastructural study. Int J Neurosci 112: 779–796, 2002
Hammes HP: Pathophysiological mechanisms of diabetic angiopathy. J Diabetic Complications 17: 16–19, 2003
Sharma S, Pandey BN, Mishra KP, Sivakami S: Amadori product and age formation during nonenzymatic glycosylation of bovine serum albumin in vitro. J Biochem Mol Biol Biophys 6: 233–242, 2002
Brownlee M: The pathological implications of protein glycation. Clin Invest Med 18: 275–278, 1995
Schmidt RE, Dorsey DA, Beaudet LN, Parvin CA, Zhang W, Sima AA: Experimental rat models of types 1 and 2 diabetes differ in sympathetic neuroaxonal dystrophy. J Neuropathol Exp Neurol 63: 450–460, 2004
Yoshikawa M, Murakami T, Komatsu H, Murakami N, Yamahara J, Matsuda H: Medicinal foodstuffs. IV. Fenugreek seed. (1): Structures of trigoneosides Ia, Ib, IIa, IIb, IIIa, and IIIb, new furostanol saponins from the seeds of Indian Trigonella foenum-graecum. L Chem Pharm Bull (Tokyo) 45: 81–87, 1997
Sauvaire Y, Petit P, Broca C, Manteghetti M, Baissac Y, Fernandez-Alvarez J, Gross R, Roye M, Leconte A, Gomis R, Ribes G: 4-Hydroxyisoleucine: A novel amino acid potentiator of insulin secretion. Diabetes 47: 206–210, 1998
Broca C, Gross R, Petit P, Sauvaire Y, Manteghetti M, Tournier M, Masiello P, Gomis R, Ribes G: 4-Hydroxyisoleucine : Experimental evidence of its insulinotropic and antidiabetic properties. Am J Physiol 227: E617–E623, 1999
Devaskar SU, Giddings SJ, Rajakumar PA, Carnaghi LR, Menon RK, Zahn DS: Insulin gene expression and insulin synthesis in mammalian neuronal cells. J Biol Chem 269: 8445–8454, 1994
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Preet, A., Gupta, B.L., Siddiqui, M.R. et al. Restoration of ultrastructural and biochemical changes in alloxan-induced diabetic rat sciatic nerve on treatment with Na3VO4 and Trigonella—a promising antidiabetic agent. Mol Cell Biochem 278, 21–31 (2005). https://doi.org/10.1007/s11010-005-7815-1
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11010-005-7815-1