Abstract
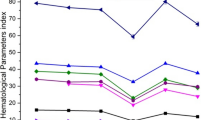
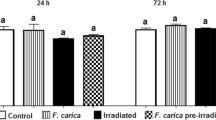
The present study reports the radioprotective properties of a hydro-alcoholic rhizome extract of Rhodiola imbricata (code named REC-7004), a plant native to the high-altitude Himalayas. The radioprotective effect, along with its relevant superoxide ion scavenging, metal chelation, antioxidant, anti-lipid peroxidation and anti-hemolytic activities was evaluated under both in vitro and in vivo conditions. Chemical analysis showed the presence of high content of polyphenolics (0.971 ± 0.01 mg% of quercetin). Absorption spectra analysis revealed constituents that absorb in the range of 220–290 nm, while high-performance liquid chromatography (HPLC) analysis confirmed the presence of four major peaks with retention times of 4.780, 5.767, 6.397 and 7.577 min. REC-7004 was found to lower lipid oxidation significantly (p < 0.05) at concentrations viz., 8 and 80 μg/ml respectively as compared to reduced glutathione, although the optimally protective dose was 80 μg/ml, which showed 59.5% inhibition of induction of linoleic acid degradation within first 24 h. The metal chelation activity of REC-7004 was found to increase concomitantly from 1 to 50 μg/ml. REC-7004 (10–50 μg/ml) exhibited significant metal chelation activity (p < 0.05), as compared to control, and maximum percentage inhibition (30%) of formation of iron-2,2′-bi-pyridyl complex was observed at 50 μg/ml, which correlated well with quercetin (34.9%), taken as standard. The reducing power of REC-7004 increased in a dose-dependent manner. The absorption unit value of REC-7004 was significantly lower (0.0183± 0.0033) as compared to butylated hydroxy toluene, a standard antioxidant (0.230± 0.091), confirming its high reducing ability. Superoxide ion scavenging ability of REC-7004 exhibited a dose-dependent increase (1–100 μg/ml) and was significantly higher (p < 0.05) than that of quercetin at lower concentrations (1–10 μg/ml), while at 100 μg/ml, both quercetin and REC-7004 scavenged over 90% superoxide anions. MTT assay in U87 cell line revealed an increase in percent survival of cells at doses between 25 and 125 μg/ml in case of drug + radiation group. In vivo evaluation of radio-protective efficacy in mice revealed that intraperitoneal administration of REC-7004 (maximally effective dose: 400 mg/kg b.w.) 30 min prior to lethal (10 Gy) total-body γ-irradiation rendered 83.3% survival. The ability of REC-7004 to inhibit lipid peroxidation induced by iron/ascorbate, radiation (250 Gy) and their combination [i.e., iron/ascorbate and radiation (250 Gy)], was also investigated and was found to decrease in a dose-dependent manner (0.05–2 mg/ml). The maximum percent inhibition of formation of MDA-TBA complex at 2 mg/ml in case of iron/ascorbate, radiation (250 Gy) and both i.e., iron/ascorbate with radiation (250 Gy) was 53.78, 63.07, and 51.76% respectively and were found to be comparable to that of quercetin. REC-7004 (1 μg/ml) also exhibited significant anti-hemolytic capacity by preventing radiation-induced membrane degeneration of human erythrocytes. In conclusion, Rhodiola renders in vitro and in vivo radioprotection via multifarious mechanisms that act in a synergistic manner.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Sweeney TR: A Survey of Compounds from the Anti-radiation Drug Development Program of the US Army Medical Research and Development Command. Government Printing Office, Washington, DC, 1979, pp 308–318
Arora R, Gupta D, Chawla R, Sagar R, Sharma AK, Prasad J, Singh S, Kumar R, Sharma RK: Radioprotection by plant products: Present status and future prospects. Phytother Res (in press), 2005
Weiss JF, Landauer MR: Protection against ionizing radiation by antioxidant nutrients and phytochemicals. Toxicology 189: 1–20, 2003
Gupta D, Arora R, Garg AP, Goel HC: Radiation-induced damage to mitochondria and its modification by Podophyllum hexandrum Royale in HepG2 cells. Mol Cell Biochem 247: 25–40, 2003
Goel HC, Prasad J, Sharma AK, Singh B: Antitumor and radioprotective action of Podophyllum hexandrum. Ind J Exp Biol 36: 583–587, 1998
Goel HC, Prasad J, Singh S, Sagar RK, Prem Kumar I, Sinha AK: Radioprotection by a herbal preparation of Hippophae rhamnoides, RH-3, against whole body lethal irradiation in mice. Phytomedicine 9: 15–25, 2002
Brown RP, Gerbarg PL, Ramazanov Z: Rhodiola rosea: A phytomedicinal overview. Herbal Gram 56: 40–52, 2002
Ohwi J: Flora of Japan. Smithsonian Institution, Washington D.C. p 495, 1984
Duhan DM: The antimutagenic activity of biomass extracts from the cultured cells medicinal plants in the Ame’s test. Tsitol Genet 33: 19–25, 1999
Udintsev SN, Schakhov VP: Decrease of cyclophosphamide hematotoxicity by Rhodiola rosea root extract in mice with Ehrlich and Lewis transplantable tumours. Eur J Cancer 27: 1182, 1991
Baranov AI: Medicinal uses of Ginseng and related plants in the Soviet Union: Recent trends in the Soviet literature. J Ethnopharmacol 6: 339–353, 1982
Boon-Niermeijir EK, Vanden Berg A, Wikman G, Wiegant FA: Phytoadaptogens protect against environmental stress-induced death of embryos from the fresh water snail Lymnea stagnalis. Phytomedicine 7: 389–399, 2000
Saratikov AS, Krasnov EA: Adaptogenic properties of Rhodiola rosea. In: A.S. Saratikov, E.A. Krasnov (eds). Rhodiola rosea is a Valuable Medicinal Plant (golden root). Tomsk State University Press, Tomsk, Russia, 1987, pp 194–215
Rege NN, Thatte UM, Dahanukar SA: Adaptogenic properties of six rasayana herbs used in Ayurvedic medicines. Phytother Res 13: 275–291, 1999
Maslova LV, Kondratev BI, Maslov LN, Lishmanov IB: The cardioprotective and antiadrenergic activity of an extract of Rhodiola rosea in stress. Eksp Klin Farmakol 57: 61–63, 1994
Darbinyan V, Kteyan A, Panossian A: Rhodiola rosea in stress-induced fatigue-a double blind cross-over study of a standardized extract SHR-5 with a repeated low dose regimen on the mental performance of healthy physicians on night duty. Phytomedicine 7: 365–371, 2000
Lazarova MB, Petkov VD, Markovska VL: Effects of meclofenoxate and extract Rhodiola rosea L. on electroconvulsive shock-impaired learning and memory in rats. Methods Ind Exp Clin Pharmacol 8: 547–552, 1986
Salikhova RA, Aleksandrova IV, Mazwik VK, Mikhailov VF, Ushenkova LN, Poroshenko GG: Effect of Rhodiola rosea on the yield of mutation alterations and DNA repair in bone marrow cells. Patol Fiziol Ter 4: 22–24, 1997
Spasov AA, Wikman GK, Mandrikov VB, Mironova IA, Neumoin VV: A double-blind, placebo-control pilot study of the stimulating and adaptogenic effect of Rhodiola rosea SHR-5 extract on the fatigue of students caused by stress during an examination period with a repeated low dose regimen. Phytomedicine 7: 85–89, 2000
Narr H: Phytochemical and Pharmacological investigation of the adaptogens: Eleutherococcus senticosus, Ocimum sanctum, Codonopsis pilosula, Rhodiola crenulata. Doctoral Dissertation, Faculty of Chemistry and Pharmacy, Ludwig-Maxmillans University – Munchen, Munchen, Germany, 1993
Saratikov AS, Krasnov EA: Stimulative properties of Rhodiola rosea. In: Saratikov, Krasnov (eds). Rhodiola rosea is a valuable medicinal plant (golden root). Tomsk State University Press, Tomsk, Russia, 1987, pp.~69–90
Ohsugi M, Fan W, Hase K: Active oxygen scavenging activity of traditional nourishing tonic medicines and active constituents of Rhodiola sacra. J Ethnopharmacol 67: 111–119, 1999
Lee MW, Lee YA, Park HM: Antioxidative phenolic compounds from the roots of Rhodiola sacchalinensis A. Bor Arch Phram Res 23: 455–458, 2000
Germano C, Ramazanov Z, Bernal SM: Arctic root (Rhodiola rosea): The powerful new Ginseng alternative. Kensington Publishing Corporation, New York, USA, 1999
Komar VB, Kit SM, Sischuk LV, Sischuk VM: Effect of Rhodiola rosea on the human mental activity. Pharmaceutic J 36: 62–64, 1981
Ganzera M, Yayla Y, Khan IA: Analysis of the marker compounds of Rhodiola rosea L. (golden root) by reversed phase high performance liquid chromatography. Chem Pharm Bull (Tokyo) 49: 465–467, 2001
Kurkin VA, Zapeesochnaya GG: Chemical composition and pharmacological characteristics of Rhodiola rosea (review). J Med Plants Russian Academy Science, Moscow 1231–1445, 1985
Yoshikawa M, Shimada H, Shimoda H, Matsuda H, Yamahara J, Murdeami N: Rhodiocyanosides A and B, new antiallergic cyanoglycosides from Chinese natural medicine ‘si lie hong jing tian’, the underground part of Rhodiola quadrifolia (Pall.) Fisch et Mey. Chem Pharm Bull 43: 1245–1247, 1995
Yoshikawa M, Shimada H, Horikawa S: Bioactive constituents of Chinese natural medicine.IV. Rhodiola Radix (2): On the histamine release inhibitors from the undergrowth part of Rhodiola sacra (Prain ex Hamet) S.H. Fu (Crassulaceae): Chemical structures of rhodiocyanoside D and sacranosides A and B. Chem Pharm Bull 45: 1498–1503, 1997
Zong Y, Lowell K, Ping JA, Che CT, Pezzuto JM, Fong HH: Phenolic constituents of Rhodiola coccinea, a Tibetan folk medicine. Planta Med 57: 589, 1991
Chaurasia OP, Singh B: Cold Desert Plants. Published by Defence Research and Development Organization, Vol 1 Leh Valley, pp. 146, Field Research Laboratory (FRL), DRDO, Leh, J&K, 1996
Singleton VL, Rossi JA, Jr: Colorimetry of total phenolics with phosphomolybdic acid-phosphotungstic acid reagents. Am J Enol Viticult 16: 144–158, 1965
Asamari AM, Addis PB, Epley RJ, Krick TP: Wild rice hull antioxidants. J Agric Food Chem 44: 126–130, 1996
Srour MA, Bitto YY, Juma M, Irhimeh MR: Exposure of human erythrocytes to oxygen radicals causes loss of deformability, increased osmotic fragility, lipid peroxidation and protein degradation. Clin Hemorheol Microcircul 23: 13–21, 2000
Harris GM, Livingstone SE: Bidendate Chelates. In: F.P. Dwyer, D.P. Mellor (Eds). Chelating Agents and Metal Chelates, Vol 1, Academic Press, New York, USA, 1964, p 95
Oyaizu M: Antioxidative activity of browning products of glucosamine fractionated by organic solvent and thin layer chromatography. Nippon ShoKuhin Kogyo Gakkaishi 35: 771–775, 1986
Kakkar P, Das B, Viswanathan PN: A modified spectrophometric assay of superoxide dismutase. Ind J Biophys Biochem 21: 110–133, 1984
Heckenkamp J, Leszezynski D, Schiereck J, Kung J, LaMuraglia FM: Different effects of photodynamic therapy and gamma-irradiation on vascular smooth muscle cells and matrix: implication for inhibiting restenosis. Arterioscler Thromb Vasc Biol 19: 2154–2161, 1999
Dacie JV, Lewis SM: Practical Hematology, 8th Edn. Churchill Livingstone, Edinburgh, 1995
Chung KT, Wong TY, Wei CI, Huang YW, Lin Y: Tannins and human health: A review. Crit Rev Food Sci Nutr 38: 421–464, 1998
Di Lusio NR: Antioxidants lipid peroxidation and chemical-induced injury. Fed Proc Fed Am Soc Exp Biol 32: 1875–1881, 1973
Gutteridge JMC: Age pigments and free radicals: Fluorescent lipid complexes formed by iron- and copper-containing proteins. Biochem Biophys Acta 834: 144, 1985
Rice Evans CA, Miller NJ, Pananga G: Structure-antioxidant activity relationship of flavonoids and phenolic acids. Free Radic Biol Med 20: 93–956, 1996
Halliwell B, Gutteridge JMC: Free radicals and metal ions in human diseases. Methods Enzymol 186, 1990
Graf E, Mahoney JR, Bryant RG, Eaton JW: Iron catalyzed hydroxyl radical formation: Strignent requirement for free iron coordination site. J Biol Chem 259: 3620–3624, 1984
Biaglow JE, Manevich Y, Uckun F, Held KD: Quantitation of hydroxyl radicals produced by radiation and copper linked oxidation of ascorbate by 2-deoxy-d-ribose method. Free Radic Biol Chem 22: 1129–1138, 1997
Minlotti G, Aust SD: The requirement of iron (III) in the initiation of lipid peroxidation by iron (II) and hydrogen peroxide. J Biol Chem 262: 1098, 1987
Davies MJ, Slater TF: Studies on metal ion and lipoxygenase-catalyzed breakdown of hydroperoxides using electron-spin-resonance spectroscopy. Biochem J 245: 167, 1987
Kumar PI, Goel HC: Iron chelation and related properties of Podophyllum hexandrum, a possible role in radioprotection. Ind J Exp Biol 38: 1003–1006, 2000
Uma Devi P, Ganasoundari A, Vrinda B, Srinivasan KK, Unnikrishnan MK: Radiation protection by Ocimum sanctum flavonoids orientin and vicenin -mechanisms of action. Radiat Res 154: 455–460, 2000
Goel HC, Prem Kumar I: Free radical scavenging potential of Tinospora cordifolia, a possible role in radioprotection. Ind J Exp Biol 40: 727–734, 2002
Yen GC, Duh PD: Scavenging effects of methanolic extracts of peanut hulls on free radical and active oxygen species. J Agric Food Chem 42: 629–632, 1995
Duh PD, Yen GC: Antioxidative activity of three herbal water extracts. Food Chem 42: 629–645, 1997
Song EK, Kim JH, Kim JS, Cho H, Nan JX, Sohn DH, Ko GI, Oh H, Kim HC: Hepatoprotective phenolic constituents of Rhodiola sachalinensis on tacrine-induced cytotoxity in Hep G2 cells. Phytother Res 17: 563–565, 2003
Xiong Q, Fan W, Tezuka Y, Adnyana I, Stampoulis P, Hattori M, Namba T, Kadota S: Hepatoprotective effect of Apocynum venetum and its active constituents. Planta Med 66: 127–133, 2000
Middleton EJR, Kandaswamy C, Theoharidies TC: The effects of plant flavonoids on mammalian cells: Implications for inflammation, heart disease, and cancer. Pharmacol Rev 52: 673–751, 2000
Diaz-Lanza AM, Abad-Martinez MJ, Matellano LF, Carretero CR, Castillo LV, Sen AM, Benito PB: Lignan and phenylpropanoid glycosides from Phillyrea latifolia and their in vitro antiinflammatory activity. Planta Med 67: 219–223, 2001
Devasagayan TPA, Boloor, Ramasarma T: Methods for estimating lipid peroxidation: An analysis of merits and demerits. Ind J Biochem Biophys 40: 300–308, 2003
Noda Y, Mc Geer PL, Mc Geer EG, Comporti M: Lipid peroxidation: Biopathological significance. Mol Aspects Med 14: 199–207, 1993
Rayleigh JA, Kremers W, Gaboury B: Dose-rate and oxygen effects in models of lipid membranes: Linoleic acid. Int J Radiat Biol Relat Stud Phys Chem Med 31: 203–213, 1977
Glaber G, Graefe EU, Struck F, Veit M, Gebhardt R: Comparison of antioxidative potential metabolites capacities and inhibitory effects of chlosterol biosynthesis of quercetin and rutin. Phytomedicine 9: 33–40, 2002
Koga TM, Moro K, Terao J: Protective effect of vitamin E analog phosphatidyl chromanol, against oxidative hemolysis of human erythrocytes. Lipid 33: 589–599, 1998
Naderi GA, Asgary S, Zadegan NS, Oroojy H, Afshin-Nia F: Antioxidant activity of three extracts of Morus nigra. Phytother Res 18: 365–369, 2004
Gupta D, Arora R, Garg AP, Bala M, Goel HC: Modification of radiation damage to mitochondrial system in vivo by Podophyllum hexandrum: Mechanistic aspects. Mol Cell Biochem 266: 65–77, 2004
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Arora, R., Chawla, R., Sagar, R. et al. Evaluation of radioprotective activities of Rhodiola imbricata Edgew – A high altitude plant. Mol Cell Biochem 273, 209–223 (2005). https://doi.org/10.1007/s11010-005-0822-4
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11010-005-0822-4