Abstract
Context
Tree canopy connectivity is important for supporting biodiversity. In urban landscapes, empirical examinations of habitat connectivity often overlook residential land, though yards and gardens often comprise a large portion of urban forests.
Objectives
We quantify structural composition (patches and paths), connectivity and fragmentation of an entire tree canopy network spanning 1220 Boston’s neighborhoods to assess the configuration of the urban forest potentially affecting tree-dependent wildlife species, such as some birds and arboreal mammals.
Methods
The urban landscape was classified by land use, and residential yards were further subdivided into front yards, backyards, and corner yards. Structural composition, connectivity and fragmentation of the tree canopy was assessed using morphological spatial pattern and network analysis. Canopy metrics were then related to the land use of 349,305 property parcels.
Results
Back yard tree canopy cover was 65.23%. The majority of canopy links were on residential land (60.95% total), and particularly in backyards. Back yards contained the highest number of canopy fragments (48.65% total). Fragmentation of the canopy network peaked at ~ 23% of total canopy cover. Canopy fragmentation, distance among patches and their shape complexity were lower in neighborhoods with more tree canopy.
Conclusions
The important role that yards have in sustaining canopy connectivity across urban landscapes poses challenges and opportunities. Urban land management and planning need to protect connectivity links within urban forests when located on private residential realm. A prioritization strategy aimed at expanding urban tree cover could focus on yards to ensure that urban landscape connectivity is maintained and increased.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The configuration of urban forests—the spatial arrangement of woody vegetation across urban landscapes—can greatly affect several organisms that are affected by the presence of trees and shrubs (Savard et al. 2000; Beninde et al. 2015). The relationship between urban forests and biodiversity depends upon species- and community-specific mechanisms that might relate to several factors linked to forest configuration: (i) the provision of resources within canopy patches (e.g., habitat, food, shelter, nesting, etc.) (Stagoll et al. 2012; Treby and Castley 2015), (ii) the interspecific interactions affected by urban forest structure (e.g., predation, competition) (Evans et al. 2009; Fontana et al. 2011), as well as (iii) the connectivity among canopy patches that can affect metapopulation dispersal (Villaseñor et al. 2014; Kolbe et al. 2016). Despite the importance of urban tree canopy, we have little information about large-scale patterns of forest cover, connectivity, and fragmentation across a metropolitan region (Ren et al. 2014).
Among the characteristics of urban forests, a connected tree canopy cover is an important factor affecting the distribution of urban organisms. Canopy cover predicts gene flow between white-footed mouse (Peromyscus leucopus) populations across New York City, USA (Munshi-South 2012). A connected canopy is necessary to keep viable populations of canopy-dependent organisms, such as threatened gliding mammals and bats in Australia cities (Goldingay et al. 2006; Hale et al. 2012). High canopy cover is associated with greater arthropod species richness and abundance in cities across Switzerland (Turrini and Knop 2015). Similarly, urban forest connectivity has been positively related to the species richness and abundance of avian, mammal and reptile assemblages across urban landscapes globally (Evans et al. 2009; Shanahan et al. 2011; Kang et al. 2015; Kolbe et al. 2016; Suarez-Rubio et al. 2018).
Oftentimes, even relatively low canopy cover and isolated tree patches can act as important corridors and stepping stones for organisms able to move through a densely built matrix (Threlfall et al. 2012). Large canopy patches generally offer higher nesting resources for vertebrates, though preferred nesting sites for some bird species are closer to forest edges (Kang et al. 2012). Likewise, the presence of edges along urban forests can increase avian movements and foraging activities of edge-tolerant species (Hodgson et al. 2007). Ultimately, the structural connectivity of urban forest networks can be an indicator of functional connectivity for some species (Hale et al. 2012).
Although much of the research on urban forests has focused on public urban green spaces (such as riparian corridors, parks, and reserves), private lands including residential yards and gardens, present an important resource for biodiversity across the urban matrix (Goddard et al. 2010; Braaker et al. 2014). Across Britain’s cities, 22 species of mammals were recorded in yards, with higher wildlife visitation rates in larger yards with greater vegetation structure and closer to natural habitats (Baker and Harris 2007). Species richness and abundance of moths across British gardens (195 species in total) was higher in yards with greater vegetation cover providing microhabitat (Bates et al. 2014). Similarly, diverse and dense yard vegetation with little lawn cover has been linked to higher bird diversity in residential landscapes (Belaire et al. 2014; Paker et al. 2014). Yards with tall trees have been found to host almost of half of the breeding couples of Cooper’s Hawks in Tucson, Arizona (Boal and Mannan 1998). Yards connected to larger landscape corridors and native urban forests are likely important to determine direction and extent of movement of both vertebrates and invertebrates (Vergnes et al. 2012, 2013). As such, the presence of yards with high vegetation cover can further mitigate the negative effects for wildlife due to habitat fragmentation in urban landscapes (Baker et al. 2003; Braaker et al. 2014).
These studies suggest the important role that yards, home gardens and the trees therein might have for urban habitat connectivity. However, empirical evidence comes from a limited number of neighborhoods and little information exists on how canopy connectivity varies across the entire urban landscape and different land uses (Casalegno et al. 2017; Ossola et al. 2018). Previous attempts at evaluating urban forest connectivity have mainly focused on the prioritization and protection of suburban forest patches, rather than trees in the urban matrix, despite the fact that yards and home gardens contain most of the urban vegetation and trees (Loram et al. 2007, 2008; Ossola et al. 2019).
In this study, we use morphological spatial pattern analysis and network analysis to describe the urban forest across the entire greater Boston metropolitan area. We quantify the structural connectivity of a canopy network and the arrangement of canopy patches and paths. We further assess the number, morphology, and distribution of canopy fragments, as well as the distribution of canopy areas that are most important within the canopy network. Because residential yards and gardens contain most vegetation and trees (Kenney et al. 2011; Daniel et al. 2016), we hypothesize that yards and gardens will be the most important land use for connectivity of the urban forest. In particular, we address the following research questions:
How does the composition of a canopy network (i.e., canopy patches and paths) vary across an urban landscape?
How do canopy fragmentation and connectivity (i.e., number and spatial relationship of canopy core patches) vary in an urban landscape in relation to land use?
We further expand on discussing the implications that urban forest connectivity has for urban landscape management and urban planning.
Methods
Study area
We investigated the city of Boston and 41 adjacent municipalities in this study (see Supplementary Fig. 1). The study area covers a total of 1220 block groups. These are statistical units defined by the US Census Bureau for collecting detailed population and socio-economic data at a local level; they usually contain between 600 and 3000 residents (US Census Bureau 2015). Census block groups are called “neighborhoods” from here forward for simplicity. About 1.5 million people live within the study area. Since its establishment in the year 1630, Boston has greatly expanded from its center to include a large suburban area characterized by leafy neighborhoods and large public green spaces and parks. About 44% of the study area is private residential land (Ossola et al. 2019), twice as large as the area occupied by public open spaces and public infrastructure (i.e., roads, parking areas, right of ways, etc.) (Table 1).
The relatively high tree canopy cover compared to other US cities (Ossola et al. 2019) is facilitated by Boston’s humid continental climate (mean annual temperature = 9.6 °C; mean annual precipitation = 1233 mm) (PRISM Climate Group 2015). However, there is substantial variation in the spatial distribution of canopy. Neighborhoods closer to the city center have canopy cover as low as 2% (of land area), and peripheral neighborhoods have as high as 84% canopy cover. This has stimulated the City of Boston and other underserved municipalities to increase canopy cover across the urban landscape by devising ambitious urban forest and greening strategies (Danford et al. 2014; Foo et al. 2018).
Geospatial data and yard identification
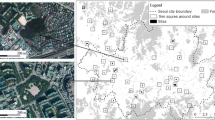
Residential parcel polygons, cadastral data, building footprints, road centerlines, and land use maps were obtained from local open data repositories (City of Boston 2017; Commonwealth of Massachusetts 2017). We classified land into six land use categories: residential (housing), mixed use (housing and commercial), infrastructure (transportation, communications, and utilities), open space (e.g., parks and preserves), commercial (shopping), and industrial (manufacturing) (Table 1). This is a standard urban land use classification scheme used in most municipalities in the US. A tree canopy cover map (1.5 m resolution) derived from spectral (i.e., visible and near infrared) and structural (i.e., LiDAR) characteristics of Boston’s urban tree canopy was obtained from Ossola et al. (2019) (Fig. 1a and Supplementary Fig. 1).
a Urban canopy cover across Boston, MA derived from Ossola et al. (2019) was used to b identify individual canopy fragments and c measure the structure and composition of the canopy network (morphological spatial pattern analysis). The area in the map is located in the suburb of Waverley, latitude: 42° 23′ 14.63″ N, longitude: 71° 10′ 55.78″ W
Front yards, backyards, and corner yards within all residential parcels across the greater Boston were geolocated and classified in ArcGIS Desktop 10.5 (ESRI, Redlands, CA), and results are detailed elsewhere (Ossola et al. 2019). Briefly, house centroids were identified in each parcel to calculate an offset segment from the closest street centerline. Corner parcels and yards, which are located within 15 m from street intersections (Ossola et al. 2019), lack a clear front/back dichotomy and thus were considered in their entirety. For the remaining residential parcels, front and backyards were distinguished by splitting each parcel polygon with a split line perpendicular to the offset segment and extending to each parcel’s lateral borders, while subtracting the area occupied by all buildings (Supplementary Fig. 2). Residential parcels containing multiple units, flats, deckers, apartment blocks, condominiums and mixed-use parcels were excluded from yard classification, as in these residential parcels it is not possible to accurately discriminate between yard types (e.g., front yard and backyard). The accuracy of yard location and classification across Boston’s neighborhoods exceeded 98% (Ossola et al. 2019).
Landscape analyses: composition of the tree canopy network
Landscape analyses of tree canopy cover were performed in GuidosToolbox (version 2.7, revision 2), which is a holistic platform for the analysis of composition, connectivity and fragmentation of habitats that is independent from the idiosyncratic responses of different faunal species (Vogt and Riitters 2017). A Morphological Spatial Pattern Analysis (MSPA) was used to calculate structure and composition of the tree cover network across the entire greater Boston area (Fig. 1c).
MSPA takes a canopy cover map and a user-defined edge width to derive canopy patch and path elements. For a given binary map (i.e., tree canopy and non-tree canopy) and a particular edge width, the results for each MSPA are always the same because each pixel is classified based on precise and known properties (Table 2). The algorithm underlying the MSPA classified each canopy pixel in one of seven canopy elements either representing canopy patches (i.e., canopy cores, outer and inner edges, and canopy islets), or canopy paths (i.e., canopy bridges, branches and loops) (see Table 2 for definitions). These canopy elements have different ecological attributes and importance to the structural connectivity of a network (Table 2) (Soille and Vogt 2009; Vogt et al. 2009; Saura et al. 2011). As such, these canopy elements have been previously used to investigate the spatial structure of native forests (Vogt et al. 2007; Riitters et al. 2016), as well as to simulate how different urban forms can maintain habitat connectivity (Tannier et al. 2012).
As most microclimatic and biophysical parameters of urban forests (i.e., temperature, moisture, shade, radiation) are mostly affected by edge effects between 0 and 5 m from a forest edge (Li et al. 2018), the MSPA was set up to include a standard edge width of 3 m (or 2 pixels). The selection of this width was based on microclimatic and biophysical parameters, rather than species-specific thresholds, and is thus independent from the idiosyncratic responses of different organisms and species to forest edges (Ries et al. 2004). This edge width threshold also insured that relatively small isolated trees and newly planted trees were not considered as canopy cores. MSPA was further set up by selecting the following parameters: (1) border and corner pixel connectivity (8-pixel connectivity), (2) enabled transition pixels (value = 1), and (3) enabled internal background (intex = 1), which are default values for MSPA in GuidosToolbox.
Landscape analyses: canopy fragmentation and connectivity
Following the MSPA, we used the GuidosToolbox to locate individual canopy fragments across the landscape, defined as areas of contiguous canopy cores (Fig. 1b). This was done with the “NW Components” (network components) tool in GuidosToolbox, using the same parameters we used for the MSPA. Average distance of fragments was calculated in ArcGIS Desktop 10.5 as the minimum distance between the edges of two canopy fragments. The outer shape of canopy fragments was expressed as Fractal Dimension (FD = 2 × ln(fragment perimeter/4)/ln(fragment area)) (Jiao and Liu 2012) and calculated in ArcGIS Desktop 10.5.
The output from the morphological spatial pattern analysis was used in GuidosToolbox to analyze landscape network connectivity, following the methods of Saura et al. (2011). Of the seven canopy cover classes from MSPA described above, only canopy cores and bridges contribute to landscape connectivity and are used in the network analysis. Cores are analogous to network nodes, and bridges provide links between the nodes. In the first step of the network analysis, landscape-level connectivity was measured as the probability of connectivity [PC; Saura and Pascual-Hortal (2007)]. In our network, movement through all links was considered feasible, regardless of the distance between connected nodes [the PC-infinite scenario in Saura et al. (2011)]. This method likely overestimates landscape-level connectivity, and underestimates the importance of individual nodes and links, but is less computationally demanding than other methods (Saura et al. 2011). The importance of each node and link in the network was determined by individually removing each node and link and calculating the resulting change in landscape-level connectivity. We focused on the connector fraction of PC (dPCconnector), which quantifies the degree to which a particular element connects other forest areas in the landscape, independent of the area of that element (Saura and Rubio 2010).
Statistical analyses
In order to evaluate how canopy fragmentation and connectivity vary in an urban landscape in relation to land use, all metrics related to canopy elements, canopy fragments and the node/link structure within the canopy connectivity network were firstly summarized within each land use and yard type, and then within each of the 1220 neighborhoods investigated. The relationships between canopy elements and land use types, yard types, and canopy cover within neighborhoods were assessed by fitting loess models to account for the non-linear patterns of several of these relationships. Average values are reported with their respective standard errors.
Results
Composition of the tree canopy network
Residential land and open spaces had proportionally higher canopy cover (on a per area basis) compared to all other land uses (Table 1). In particular, front yards, backyards, and corner yards collectively occupied 27.36% of land and contained 37.03% of total canopy cover. Backyards contained 21% of the study area’s total tree canopy cover, or more than twice as much cover as front yards (8.28% cover) on a per area basis. Canopy cover was greater in backyards (65.23%) when compared to corner (57.66%) and front yards (47.40%) (Supplementary Table 1). Average canopy cover across all yard types (56.77%) was comparable to that of open spaces (59.40%), but yards covered substantially more land (Table 1).
MSPA revealed that canopy cores and their outer edges represented more than 80% of total canopy cover across Boston’s landscape (Table 3). Canopy cores covered about a quarter of the total land area (Supplementary Tables 2, 3) and were most common in residential land (27.46% of all core habitat). Overall, yards contained a similar amount of canopy cores as open spaces (21.24 and 21.50%, respectively), but contained about three times as many outer edges (8.52 and 2.61%, respectively). All canopy path elements (i.e., canopy bridges, branches and loops) were more abundant in residential land and yards compared to all the other land uses (Table 3).
Canopy path elements were generally lower when canopy cover was high (Fig. 2a, c) except for canopy loops that remained relatively stable along the canopy cover gradient (Fig. 2b). Canopy cores had a positive and linear relationship with canopy cover (Fig. 2e), whereas inner edges substantially increased above ~ 50% canopy cover (Fig. 2f). The distribution of canopy elements had similarly relationships in relation to yard cover (see Supplementary Fig. 4).
Relationships between percent canopy cover within neighborhoods and the canopy network in its canopy paths [canopy bridges (a); loops (b); branches (c) and islets (d)], canopy patches [canopy cores (e); inner edges (f); and outer edges (g)] calculated from the morphological spatial pattern analysis and annual median income (h) among the 1220 census block groups (i.e., neighborhoods) investigated across the greater Boston’s area. Lines represent loess models and bands the 95% confidence level interval for model predictions
Canopy fragmentation and connectivity
The network components tool in GuidosToolbox identified 87,209 distinct fragments (contiguous canopy core areas) in the urban canopy, 48.65% of which were at least partially contained in residential yards (Fig. 3). The number of canopy fragments peaked at ~ 23% of the total canopy cover (Fig. 4a), whereas fragment fractal dimension (FD), the shape complexity of a fragment, peaked around 40% canopy cover (Fig. 4c). Average minimum distance between fragments was highest in neighborhoods with low canopy cover (< 20%) to then level off at values below 10 m (Fig. 4b).
Number of canopy fragments for each land use type (a). Cumulative number of fragments in the figure is greater than the total number of fragments in the study area (n = 87,209) as fragments might cover two or more land use types. The area of connectivity links in the greater Boston area for each land use category (colored bars) and cumulative importance (black dots) for each land use (b)
Relationship between canopy cover and: a number of canopy fragments per unit of land area, b average fragment minimum distance from nearest fragment edge, c fractal dimension of fragments (i.e., outer shape complexity), d area of connectivity links per unit of land area, e percent mean link importance and f percent yard cover within a neighborhood. Points represent average values for the 1220 census block groups (i.e., neighborhoods) investigated across the greater Boston area. Lines represent loess models and bands are the 95% confidence level interval for model predictions. Fragments refer to areas of contiguous canopy cores, fractal dimension of fragments is a function of perimeter and area
The connectivity analysis highlighted that links in Boston’s canopy connectivity network were largely contained in residential land and yards (6.07 km2 out of 9.96 km2 total link area) (Fig. 3b). In terms of relative importance for the canopy network (defined by the change in overall landscape connectivity when the link was removed), links in residential land contributed to 60.24% of the total network importance, followed by canopy connectivity links along infrastructure (which includes street trees, 19.09%) and open spaces (16.05%). The extent of network links was lower in neighborhoods with lower canopy cover (Fig. 4d). The average link importance within the canopy network was greater when total canopy cover was higher than 60% (Fig. 4e).
Discussion
Composition of the tree canopy network
The structural composition of the tree canopy network varied greatly based on land use across the greater Boston area. As hypothesized, yards were the most important land use because they contained the majority of the tree canopy elements (i.e., patches and paths), and patches of primary importance (i.e., canopy cores and bridges). Among yards, backyards contained most of the canopy elements. In Boston, backyards have higher canopy cover and taller trees as compared to front yards (Ossola et al. 2019), because of their larger size that allows more living spaces for trees and vegetation (Smith et al. 2005).
Open spaces contained a substantial amount of canopy elements, but these were less numerous across neighborhoods and more spatially concentrated in Boston’s landscape compared to residential yards. This is important in relation to previous empirical evidence as it highlights the role yards might have in providing primary habitat patches (Baker and Harris 2007; Bates et al. 2014; Paker et al. 2014) and important vegetated paths (i.e., corridors) across the urban landscape (Vergnes et al. 2012, 2013).
Overall, as total canopy cover increases in a neighborhood, more of that canopy becomes core habitat. Thus, neighborhoods with more tree canopy are most likely to contain canopy patches that can serve as the primary habitat template for tree-dependent organisms. When canopy cover is higher, outer edges comprise a smaller portion of the total canopy within a patch, while inner edges (i.e., perforations) are more numerous. Inner edges, however, were 4–5 times less common than outer edges. In this way, increments in canopy cover can determine an overall decrease of the urban forest edge, thus potentially affecting edge-sensitive species as well as the ecological and behavioral processes occurring at this landscape interface (Kang et al. 2012).
In neighborhoods with low canopy cover, approximately 5–10% of the canopy comprises bridges. These important path types connect two distinct canopy cores, and are important for movement of obligate canopy users, which are more likely to be impacted by canopy fragmentation than other organisms (Goldingay et al. 2006). However, canopy bridges are often narrow strips that might be poorly used by some organisms, as in the case of some bird species sensitive to anthropogenic disturbance (Oprea et al. 2009). Thus, canopy bridges might offer higher chances for dispersal for some species more sensitive to anthropogenic disturbance if they are located in relatively less disturbed and trafficked areas, such as backyards, rather than along streets. Canopy branches and islets, in turn, might be still functionally important for some highly vagile or flying species by serving as stepping stones or temporary shelter during their movements across the built-up matrix (Threlfall et al. 2012).
Canopy fragmentation and connectivity
Despite containing a high proportion of canopy, yards and residential areas also contained a higher number of canopy fragments (i.e., contiguous core patches) compared to other urban land uses. The number of canopy fragments, however, was the greatest in neighborhoods with ~ 25–30% canopy cover and then decreased in neighborhoods with greater tree canopy. Similarly, these fragments had a more complex and less regular morphology in neighborhoods with intermediate levels of canopy cover. This suggests that increasing canopy cover in cities above a certain threshold might help to (i) regularize the shape of canopy core patches while decreasing the canopy perimeter-to-area ratio, (ii) decrease the average distance among canopy cores, and (iii) coalesce nearby canopy cores to create larger contiguous canopy patches, as previously observed in simulated landscapes (Hargis et al. 1998).
Overall, increasing canopy cover in cities could increase the extent of core habitat far away from canopy edges for some species, thus facilitating some edge-sensitive species less able to cope with ecotone change and disturbance (Villaseñor et al. 2014). On the other hand, some urban-adapted species that live and move between large canopy cores and the urban matrix (e.g., for roosting and foraging, respectively), could be negatively impacted by a lower presence of arboreal edges (Carter et al. 2019). Increasing canopy cover would increase the overall connectivity of primary habitat for some canopy-dependent and forest-obligate species (Evans et al. 2017), as well as decrease the distance needed to be covered by some vagile species among primary habitat patches (Threlfall et al. 2012). This however may have the unintended consequence of altering the habitat of other species that might prefer more open landscapes with little or no canopy cover (e.g., grassland or open-habitat specialists). Most of the links in the canopy network were contained in Boston’s yards and residential areas, despite the high number of canopy fragments in these areas. This might represent a critical spatial mismatch between tree corridors, as an important resource to allow dispersal of some less vagile species, and the ability of some species to use canopy links in more disturbed areas (Hale et al. 2012).
It is important to note that our analysis is based on some important assumptions about connectivity and species movement. Firstly, habitat connectivity, as we define it here, requires canopy cores to be directly connected by canopy habitat that acts as a bridge between two or more canopy cores. In other words, we assume that arboreal species do not leave the tree canopy to cross gaps between core habitat. While this assumption might hold true for some less vagile species, some other species might be able to efficiently move across an urban landscape by using anthropogenic structures (e.g., powerlines, fences, etc.) or non-vegetated patches (e.g., road crossing during night time). Secondly, the edge width used in our models was selected based on changes to the biophysical and microclimatic conditions of urban forest edges (Li et al. 2018), rather than the ecological requirements of particular species. Although it is outside the scope of this paper, the modelling framework proposed in this study could be easily re-parameterized to better include species-specific knowledge the of habitat and movement requirements of targeted wildlife species.
Implications for urban landscape management and planning
Accounting for risks
Urban landscapes are far from being homogeneous in terms of tenure and management of the urban forest (Kenney et al. 2011; Ossola et al. 2018). The fact that the structure and connectivity of urban canopy networks is primarily defined by residential yards and home gardens might pose a series of threats for urban biodiversity.
First, residents in a neighborhood can have high variability in managing their yards (Cook et al. 2012; Locke et al. 2018). Oftentimes, even neighbors in nearby properties manage their private green spaces in completely different ways based on their perception of yard aesthetic, values and norms (Larsen and Harlan 2006; Ossola et al. 2019). Further, health and performance of urban forests is critically affected by land tenure, with trees on public and private land being maintained in different ways (Kenney et al. 2011). This can affect not only the structure and species composition of residential forests, but also the amount and frequency of anthropogenic disturbance (e.g., frequency and intensity of landscaping, gardening and maintenance activities), which might be particularly important for some sensitive species attempting to use residential trees (Boal and Mannan 1998).
Second, urban residential forests are highly dynamic and tree removal in residential yards represents a widespread event across urban landscapes in various cities (Ossola and Hopton 2018; Roman et al. 2018). This can exacerbate the negative effects of canopy loss due to urban infill and densification (Lin and Fuller 2013; Daniel et al. 2016). In this way, canopy elements within the urban forest might be impacted in abrupt and unanticipated ways, affecting landscape connectivity and habitat fragmentation. In fact, the loss of the canopy cover from private yards, which is critical to sustain a connected canopy network, could cause a great disruption in the whole connectivity of the urban forest.
Third, yards are urban habitats not encompassed by traditional conservation planning (e.g., protected areas, reserves, etc.), despite their potential role for urban biodiversity conservation (Goddard et al. 2010; Pirnat and Hladnik 2016). Often, tree removal in yards and private land is poorly regulated and/or enforced (Coughlin et al. 1988; Hill et al. 2010). This threat is likely compounded by the fact that conservation awareness is extremely variable among private landowners (Kittredge et al. 2015), often determining other undesirable or unintended changes to urban forests (e.g., spread of invasive species).
Prioritizing for landscape connectivity
At the city scale, future urban planning and greening interventions aimed at increasing connectivity and urban biodiversity should better value yards, home gardens and their trees. This is not trivial as most of municipal canopy cover targets focus on establishing and maintaining the “public” urban forest (i.e., streetscapes, public rights of ways, nature strips, etc.), whereas little attention is currently being paid to the “private” residential forest (Kenney et al. 2011). Occasionally, the urban forest in the public realm might have little value in sustaining habitat connectivity even for some vagile organisms (Oprea et al. 2009).
Mounting evidence, on the other hand, suggests that the residential forest needs a specific urban form and enough physical space to thrive (Smith et al. 2005; Bigsby et al. 2014; Ossola et al. 2019). Thus, urban planning interventions aimed at improving urban sustainability and livability (e.g., densification, retrofitting, renewal, etc.) can have the undesired consequence of decreasing cover and structure of the residential forest, while reducing the connectivity of the canopy network.
Thus, the challenge ahead relies on increasing canopy cover in order to: (1) meet ambitious municipal canopy cover targets, (2) increase canopy patch size across urban landscapes, and (3) maintain, and possibly increase, connectivity of the urban forest. This can be achieved through a prioritization strategy that aims at minimizing and/or connecting canopy elements with lower landscape value (i.e., islets), while maximizing cover of important canopy elements in an urban forest (e.g., cores, bridges). Other urban forestry and planning objectives may require tradeoffs for connectivity (Locke et al. 2010). Tree planting efforts could be targeted at areas most likely to (re)establish the connectivity of the urban tree canopy network while acknowledging that resources other than those depending on the canopy structure, such as food provision, nesting or shelter, are often specific to the identity of tree species (Colding 2007). In this way, new set of policy interventions, governance tools and financial incentives will have to be devised, in order to ensure that critical canopy areas and tree elements are protected or at least replaced over time, particularly within private landscapes, yards and home gardens. Finally, as landscape connectivity is a relative concept that depends upon characteristics of the organism investigated, such as its ecology, life history, tolerance and adaptability to urban environments, it is important to consider how changes in urban forest connectivity might impact other species, communities and functional guilds.
References
Baker PJ, Harris S (2007) Urban mammals: what does the future hold? An analysis of the factors affecting patterns of use of residential gardens in Great Britain. Mamm Rev 37:297–315
Baker PJ, Ansell RJ, Dodds PAA, Webber CE, Harris S (2003) Factors affecting the distribution of small mammals in an urban area. Mamm Rev 33:95–100
Bates AJ, Sadler J, Grundy D, Lowe N, Davis G, Baker D, Bridge M, Freestone R, Gardner D, Gibson C, Hemming R, Howarth S, Orridge S, Shaw M, Tams T, Young H (2014) Garden and landscape-scale correlates of moths of differing conservation status: significant effects of urbanization and habitat diversity. PLoS ONE 9(1):e86925. https://doi.org/10.1371/journal.pone.0086925
Belaire JA, Whelan CJ, Minor ES (2014) Having our yards and sharing them too: the collective effects of yards on native bird species in an urban landscape. Ecol Appl 24:2132–2143
Beninde J, Veith M, Hochkirch A (2015) Biodiversity in cities needs space: a meta-analysis of factors determining intra-urban biodiversity variation. Ecol Lett 18:581–592
Bigsby KM, McHale MR, Hess GR (2014) Urban morphology drives the homogenization of tree cover in Baltimore, MD, and Raleigh, NC. Ecosystems 17:212–227
Boal CW, Mannan RW (1998) Nest-site selection by Cooper’s hawks in an urban environment. J Wildl Manag 62:864–871
Braaker S, Moretti M, Boesch R, Ghazoul J, Obrist MK, Bontadina F (2014) Assessing habitat connectivity for ground-dwelling animals in an urban environment. Ecol Appl 24:1583–1595
Carter N, Cooke R, White JG, Whisson DA, Isaac B, Bradsworth N (2019) Joining the dots: how does an apex predator move through an urbanizing landscape? Glob Ecol Conserv 17:e00532
Casalegno S, Anderson K, Cox DTC, Hancock S, Gaston KJ (2017) Ecological connectivity in the three-dimensional urban green volume using waveform airborne lidar. Sci Rep. https://doi.org/10.1038/srep45571
City of Boston (2017) Analyze Boston portal. https://data.boston.gov/. Accessed 1 Sept 2017
Colding J (2007) ‘Ecological land-use complementation’ for building resilience in urban ecosystems. Landsc Urban Plan 81(1):46–55
Commonwealth of Massachusetts (2017) MASSgis. https://docs.digital.mass.gov/massgis. Accessed 1 Sept 2017
Cook EM, Hall SJ, Larson KL (2012) Residential landscapes as social-ecological systems: a synthesis of multi-scalar interactions between people and their home environment. Urban Ecosyst 15:19–52
Coughlin RE, Mendes DC, Strong AL (1988) Local programs in the United States for preventing the destruction of trees on private land. Landsc Urban Plan 15:165–171
Danford RS, Cheng C, Strohbach MW, Ryan R, Nicolson C, Warren PS (2014) What does it take to achieve equitable urban tree canopy distribution? A Boston case study. Cities Environ 7: Article 2
Daniel C, Morrison TH, Phinn S (2016) The governance of private residential land in cities and spatial effects on tree cover. Environ Sci Policy 62:79–89
Evans BS, Kilpatrick AM, Hurlbert AH, Marra PP (2017) Dispersal in the urban matrix: assessing the influence of landscape permeability on the settlement patterns of breeding songbirds. Front Ecol Evol 5:63
Evans KL, Newson SE, Gaston KJ (2009) Habitat influences on urban avian assemblages. Ibis 151:19–39
Fontana S, Sattler T, Bontadina F, Moretti M (2011) How to manage the urban green to improve bird biodiversity and community structure. Landsc Urban Plan 101:278–285
Foo K, McCarthy J, Bebbington A (2018) Activating landscape ecology: a governance framework for design-in-science. Landsc Ecol 33:675–689
Goddard MA, Dougill AJ, Benton TG (2010) Scaling up from gardens: biodiversity conservation in urban environments. Trends Ecol Evol 25:90–98
Goldingay RL, Sharpe DJ, Beyer GL, Dobson M (2006) Using ecological studies to understand the conservation needs of the squirrel glider in Brisbane’s urban forest-remnants. Aust Mamm 28:173–186
Hale JD, Fairbrass AJ, Matthews TJ, Sadler JP (2012) Habitat composition and connectivity predicts bat presence and activity at foraging sites in a large UK conurbation. PLoS ONE 7:e33300
Hargis CD, Bissonette JA, David JL (1998) The behavior of landscape metrics commonly used in the study of habitat fragmentation. Landsc Ecol 13:167–186
Hill E, Dorfman JH, Kramer E (2010) Evaluating the impact of government land use policies on tree canopy coverage. Land Use Policy 27:407–414
Hodgson P, French K, Major RE (2007) Avian movement across abrupt ecological edges: differential responses to housing density in an urban matrix. Landsc Urban Plan 79:266–272
Jiao L, Liu Y (2012) Analyzing the shape characteristics of land use classes in remote sensing imagery. ISPRS Ann Photogramm Remote Sens Spat Inf Sci I-7:135–140
Kang W, Lee D, Park C-R (2012) Nest distribution of magpies Pica pica sericea as related to habitat connectivity in an urban environment. Landsc Urban Plan 104:212–219
Kang W, Minor ES, Park CR, Lee D (2015) Effects of habitat structure, human disturbance, and habitat connectivity on urban forest bird communities. Urban Ecosyst 18:857–870
Kenney WA, Van Wassenaer PJE, Satel AL (2011) Criteria and indicators for strategic urban forest planning and management. Arboric Urban For 37:108–117
Kittredge DB, Short Gianotti AG, Hutyra LR, Foster DR, Getson JM (2015) Landowner conservation awareness across rural-to-urban gradients in Massachusetts. Biol Conserv 184:79–89
Kolbe JJ, VanMiddlesworth P, Battles AC, Stroud JT, Buffum B, Forman RTT, Losos JB (2016) Determinants of spread in an urban landscape by an introduced lizard. Landsc Ecol 31:1795–1813
Larsen L, Harlan SL (2006) Desert dreamscapes: residential landscape preference and behavior. Landsc Urban Plan 78:85–100
Li Y, Kang W, Han Y, Song Y (2018) Spatial and temporal patterns of microclimates at an urban forest edge and their management implications. Environ Monit Assess 190:93
Lin BB, Fuller RA (2013) FORUM: sharing or sparing? How should we grow the world’s cities? J App Ecol 50:1161–1168
Locke DH, Grove MJ, Lu JWT, Troy A, O'Neil-Dunne J, Beck BD (2010) Prioritizing preferable locations for increasing urban tree canopy in New York City. Cities Environ 3:1–18
Locke DH, Roy Chowdhury R, Grove JM, Martin DG, Goldman E, Rogan J, Groffman P (2018) Social norms, yard care, and the difference between front and back yard management: examining the landscape mullets concept on urban residential lands. Soc Nat Resour. https://doi.org/10.1080/08941920.2018.1481549
Loram A, Tratalos J, Warren PH, Gaston KJ (2007) Urban domestic gardens (X): the extent & structure of the resource in five major cities. Landsc Ecol 22:601–615
Loram A, Warren P, Gaston KJ (2008) Urban domestic gardens (XIV): the characteristics of gardens in five cities. Environ Manag 42:361–376
Munshi-South J (2012) Urban landscape genetics: canopy cover predicts gene flow between white-footed mouse (Peromyscus leucopus) populations in New York City. Mol Ecol 21:1360–1378
Oprea M, Mendes P, Vieira TB, Ditchfield AD (2009) Do wooded streets provide connectivity for bats in an urban landscape? Biodivers Conserv 18:2361–2371
Ossola A, Hopton ME (2018) Measuring urban tree loss dynamics across residential landscapes. Sci Total Environ 612:940–949
Ossola A, Locke DH, Lin BB, Minor E (2019) Greening in style: urban form, architecture and the structure of front and backyard vegetation. Landsc Urban Plan 185:141–157
Ossola A, Schifman L, Herrmann DL, Garmestani AS, Schwarz K, Hopton ME (2018) The provision of urban ecosystem services throughout the private-social-public domain: a conceptual framework. Cities Environ (CATE) 11:5
Paker Y, Yom-Tov Y, Alon-Mozes T, Barnea A (2014) The effect of plant richness and urban garden structure on bird species richness, diversity and community structure. Landsc Urban Plan 122:186–195
Pirnat J, Hladnik D (2016) Connectivity as a tool in the prioritization and protection of sub-urban forest patches in landscape conservation planning. Landsc Urban Plan 153:129–139
PRISM Climate Group (2015) 30-year Normals. http://www.prism.oregonstate.edu/. Accessed 26 Sept 2016
Ren Y, Deng L, Zuo S, Luo Y, Shao G, Wei X, Hua L, Yang Y (2014) Geographical modeling of spatial interaction between human activity and forest connectivity in an urban landscape of southeast China. Landsc Ecol 29(10):1741–1758
Ries L, Fletcher RJ, Battin J, Sisk TD (2004) Ecological responses to habitat edges: mechanisms, models, and variability explained. Annu Rev Ecol Evol Syst 35:491–522
Riitters K, Wickham J, Costanza JK, Vogt P (2016) A global evaluation of forest interior area dynamics using tree cover data from 2000 to 2012. Landsc Ecol 31:137–148
Roman LA, Pearsall H, Eisenman TS, Conway TM, Fahey R, Landry S, Vogt J, Van Doorn NS, Grove M, Locke DH, Bardekjian AC, Battles JJ, Cadenasso ML, ven den Bosch CK, Avolio M, Berland A, Jenerette D, Mincey SK, Pataki DE, Staudhammer CL (2018) Human and biophysical legacies shape contemporary urban forests: a literature synthesis. Urban For Urban Green 31:157–168
Saura S, Pascual-Hortal L (2007) A new habitat availability index to integrate connectivity in landscape conservation planning: comparison with existing indices and application to a case study. Landsc Urban Plan 83:91–103
Saura S, Rubio L (2010) A common currency for the different ways in which patches and links can contribute to habitat availability and connectivity in the landscape. Ecography 33:523–537
Saura S, Vogt P, Velázquez J, Hernando A, Tejera R (2011) Key structural forest connectors can be identified by combining landscape spatial pattern and network analyses. For Ecol Manag 262:150–160
Savard J-PL, Clergeau P, Mennechez G (2000) Biodiversity concepts and urban ecosystems. Landsc Urban Plan 48:131–142
Shanahan DF, Miller C, Possingham HP, Fuller RA (2011) The influence of patch area and connectivity on avian communities in urban revegetation. Biol Conserv 144:722–729
Smith RM, Gaston KJ, Warren PH, Thompson K (2005) Urban domestic gardens (V): relationships between landcover composition, housing and landscape. Landsc Ecol 20:235–253
Soille P, Vogt P (2009) Morphological segmentation of binary patterns. Pattern Recog Lett 30:456–459
Stagoll K, Lindenmayer DB, Knight E, Fischer J, Manning AD (2012) Large trees are keystone structures in urban parks. Conserv Lett 5:115–122
Suarez-Rubio M, Ille C, Bruckner A (2018) Insectivorous bats respond to vegetation complexity in urban green spaces. Ecol Evol 8:3240–3253
Tannier C, Foltête J-C, Girardet X (2012) Assessing the capacity of different urban forms to preserve the connectivity of ecological habitats. Landsc Urban Plan 105:128–139
Threlfall CG, Law B, Banks PB (2012) Sensitivity of insectivorous bats to urbanization: implications for suburban conservation planning. Biol Conserv 146:41–52
Treby DL, Castley JG (2015) Distribution and abundance of hollow-bearing trees in urban forest fragments. Urban For Urban Green 14:655–663
Turrini T, Knop E (2015) A landscape ecology approach identifies important drivers of urban biodiversity. Glob Change Biol 21:1652–1667
US Census Bureau (2015). Geographic areas reference manual (GARM). https://www2.census.gov/geo/pdfs/reference/GARM/Ch11GARM.pdf. Accessed 6 Sept 2019
Vergnes A, Kerbiriou C, Clergeau P (2013) Ecological corridors also operate in an urban matrix: a test case with garden shrews. Urban Ecosyst 16:511–525
Vergnes A, Viol IL, Clergeau P (2012) Green corridors in urban landscapes affect the arthropod communities of domestic gardens. Biol Conserv 145:171–178
Villaseñor NR, Driscoll DA, Escobar MAH, Gibbons P, Lindenmayer DB (2014) Urbanization impacts on mammals across urban-forest edges and a predictive model of edge effects. PLoS ONE 9:e97036
Vogt P, Riitters K (2017) GuidosToolbox: universal digital image object analysis. Eur J Remote Sens 50:352–361
Vogt P, Ferrari JR, Lookingbill TR, Gardner RH, Riitters KH, Ostapowicz K (2009) Mapping functional connectivity. Ecol Indic 9:64–71
Vogt P, Riitters KH, Estreguil C, Kozak J, Wade TG, Wickham JD (2007) Mapping spatial patterns with morphological image processing. Landsc Ecol 22:171–177
Acknowledgements
Authors kindly acknowledge MASS-GIS and the City of Boston for providing geospatial datasets. The National Socio-Environmental Synthesis Center (SESYNC) under funding received from the National Science Foundation DBI-1052875 supported this work. Findings and conclusions in this publication are those of the Authors and should not be construed to represent any official USDA or US Government determination or policy. The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ossola, A., Locke, D., Lin, B. et al. Yards increase forest connectivity in urban landscapes. Landscape Ecol 34, 2935–2948 (2019). https://doi.org/10.1007/s10980-019-00923-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10980-019-00923-7