Abstract
Sylvatic plague is a major factor influencing the dynamics of black-tailed prairie dog (Cynomys ludovicianus) colonies in the western Great Plains. We studied the nesting response of the mountain plover (Charadrius montanus), a grassland bird that nests on prairie dog colonies, to plague-driven dynamics of prairie dog colonies at three sites in the western Great Plains. First, we examined plover nest distribution on colonies that were previously affected by plague, but that had been recovering (expanding) for at least 6 years. Plovers consistently nested in both young (colonized in the past 1–2 years) and old (colonized for 6 or more years) portions of prairie dog colonies in proportion to their availability. Second, we examined changes in plover nest frequency at two sites following plague epizootics, and found that mountain plover nest numbers declined relatively rapidly (≤2 years) on plague-affected colonies. Taken together, our findings indicate that available plover nesting habitat associated with prairie dog colonies closely tracks the area actively occupied by prairie dogs each year. Given the presence of plague throughout most of the mountain plover’s breeding range in the western Great Plains, important factors affecting plover populations likely include landscape features that determine the scale of plague outbreaks, the distance that plovers move in response to changing breeding habitat conditions, and the availability and quality of alternate breeding habitat within the landscape.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The role that black-tailed prairie dogs (Cynomys ludovicianus) play in sustaining the biodiversity of the western Great Plains has been the subject of considerable controversy and research (Vermiere et al. 2004; Forrest 2005; Hoogland 2006). Prior to European settlement, black-tailed prairie dogs were a widespread and abundant component of the grazing regime in landscapes of central North America. Black-tailed prairie dogs (prairie dogs hereafter) maintained a broad geographic range during the late Pleistocene, and expanded into their current distribution around the onset of the Holocene (Goodwin 1995). Numerous plant and animal species of the western Great Plains exhibit adaptations to the resources and conditions provided by prairie dog colonies (Kotliar et al. 1999; Lomolino and Smith 2003; Smith and Lomolino 2004; Kotliar et al. 2006). In recent decades, declining populations have been documented for some species that rely to varying degrees on prairie dog colonies for habitat (Miller et al. 1996; Desmond et al. 2000; Knopf and Wunder 2006).
Plague caused by the bacterium Yersinia pestis, a disease introduced from Asia, is currently a major factor regulating the distribution and abundance of prairie dogs (Cully and Williams 2001; Collinge et al. 2005; Antolin et al. 2006). Prairie dogs have only been exposed to plague for approximately 60 years, and have not evolved resistance to this highly virulent pathogen (Cully et al. 2006). Epizootic plague outbreaks cause rapid and nearly complete mortality within a colony (Cully and Williams 2001; Pauli et al. 2006). In landscapes with a high degree of connectivity among colonies, epizootic plague can decimate colonies distributed over a large geographic area (e.g. >100,000 ha) in a single year (Collinge et al. 2005; Johnson 2005; Augustine et al. 2008). In contrast, in landscapes with greater inter-colony distances and higher densities of roads and water bodies, epizootic plague may only affect colonies distributed across smaller areas within a year (Stapp et al. 2004; Collinge et al. 2005). Epizootic plague outbreaks can induce dramatic fluctuations in the abundance of prairie dogs, and can reoccur within a locality at intervals of 6–15 years (Stapp et al. 2004; Hartley 2006; Cully et al. 2006; Augustine et al. 2008). If prairie dogs are indeed important to sustaining the biodiversity of shortgrass and mixed prairie landscapes, then we need to understand how species that use prairie dog colonies for habitat are influenced by the spatiotemporal dynamics of prairie dog colonies affected by plague.
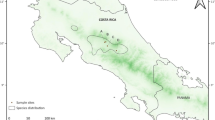
In grasslands of the Great Plains, breeding habitat for the mountain plover (Charadrius montanus) is associated with prairie dog colonies, heavy grazing by large ungulates, or recent fire (Dinsmore 2003; Knopf and Wunder 2006). In the shortgrass steppe, plovers can nest in grassland without prairie dogs (Knopf and Miller 1994), but occurred at 10 times greater density on prairie dog colonies compared to non-colonized rangeland in 2005 (Tipton 2007). In the more productive northern mixed prairie, plovers nest almost exclusively on prairie dog colonies (Olson and Edge 1985; Dinsmore et al. 2005). Plague occurs throughout most of the breeding range of the mountain plover (Fig. 1), such that plague effects on prairie dog colony dynamics, and the potential consequences for vegetative conditions, may have significant implications for mountain plovers.
Map showing the U.S. portion of the breeding distribution of mountain plovers (solid black outline, from Knopf and Wunder (2006)) and U.S. counties where evidence of plague has been documented in the western United States. The distribution of counties with plague is based on records from the Center for Disease Control and Prevention, Fort Collins, CO (dark grey shading) and observations of large-scale prairie dog mortality due to apparent plague in eastern Colorado (light grey shading; V. Dreitz, personal observation) and eastern Wyoming (light grey shading; Johnson 2005). The white background shows counties with no documented evidence of plague. Black dots show the Phillips County study area in north central Montana, the Pawnee study area in northern Colorado, and the Comanche study area in southeast Colorado
Increasing length of prairie dog occupancy at a site can lead to progressive reductions in tall-structure grasses, and increased abundance of forbs and subshrubs (Coppock et al. 1983; Archer et al. 1987). In shortgrass steppe, plague can alter prairie dog effects on the plant community by reducing differences in vegetative cover, biomass and species composition between colonized and uncolonized sites (Hartley 2006). Observations of plover nesting activity on a plague-affected prairie dog colony in the same area studied by Hartley (2006) documented a rapid decline in the number of plover nests 1–2 years after a plague outbreak, and only a small increase in the number of plover nests after the colony had returned to its pre-plague size (Fig. 2). This suggested that after a plague event, nesting habitat for plovers may not return as quickly as the prairie dogs. Observations in mixed prairie also suggested that soon after an epizootic plague outbreak, vegetative conditions on colonies may become unsuitable for mountain plovers (Dinsmore et al. 2005). Here, we examine responses of nesting mountain plovers to plague-driven dynamics of prairie dog colonies in the shortgrass steppe of Colorado and mixed prairie of Montana. Our first objective was to evaluate the time frame in which prairie dog colonies become suitable for plover nesting as colonies recover after epizootic plague outbreaks. Our second objective was to examine the response of plover nesting activity to recent epizootic plague outbreaks in shortgrass steppe and mixed prairie.
Study areas
Study areas and prairie dog colony surveys
Study areas consisted of (1) the Carrizo Unit of the Comanche National Grassland in Baca County, CO (Augustine et al. 2008), (2) the Pawnee National Grassland and adjacent Central Plains Experimental Range in Weld County, CO (Stapp et al. 2004), and (3) Bureau of Land Management and private lands in southern Phillips County, Montana (Dinsmore et al. 2005; Augustine et al. 2008; Table 1). On the Comanche study area, surveys of prairie dog colony boundary locations using handheld global position system (GPS) units were conducted in 1995, 1999, 2001, 2002, 2003 and 2004 (Augustine et al. 2008). Observations in 1994 and 1995 indicated that all colonies were affected by a plague epizootic that started in 1994 and continued after 1995. All colonies in the Comanche study area were expanding (i.e. boundaries of each colony encompassed a larger area in each successive year) during 1999–2004, and did not exhibit any signs of plague activity. Prairie dog colony boundaries were surveyed annually using GPS units during 1993–2006 on the Pawnee National Grassland and during 1999–2006 on the adjacent Central Plains Experimental Range. Epizootic plague affected a small proportion of colonies in this area in most years since 1981, with more extensive plague epizootics occurring in 1992 and 1999 (Stapp et al. 2004). Prairie dog colony boundaries in the Phillips County study area were surveyed using GPS units in 1993, 1995–1997, 1998, 2000, 2002 and 2004 (Augustine et al. 2008). During 1995–1997, a non-overlapping third of the colonies in the area were surveyed each year; results from these surveys were combined and are referred to hereafter as the 1996 survey. A plague epizootic was first recorded in Phillips County in 1992, when the area occupied by prairie dogs was reduced by approximately 50% (Collinge et al. 2005; M. R. Matchett, unpublished data), and declines continued through 1996. Most colonies in the study area were expanding during 1996–2002, and a portion of the colonies were affected by a plague epizootic during 2002–2004.
Mountain plover surveys
We surveyed mountain plovers on prairie dog colonies by systematically driving a vehicle slowly across each colony and periodically stopping to scan for plovers. We watched individual adult birds from a distance until they returned to a nest, and recorded the geographic coordinates of the nest. In the Comanche study area, we surveyed mountain plovers on 10 prairie dog colonies ≥2 times during May of 2005. A plague epizootic during 2006 affected all of this study area. In late April of 2007, we conducted a single systematic survey at four colonies that had the highest nest densities in 2005. In the Pawnee study area, we surveyed prairie dog colonies for mountain plovers during 2003–2007, as part of long-term studies on plover ecology (Wunder 2007; Dreitz and Knopf 2007). Colonies were surveyed ≥2 times during May–June of each year. In the Phillips County study area, we systematically searched active prairie dog colonies ≥3 times during May–July in 2003 and 2005.
Data analysis
We imported prairie dog colony boundary coordinates into a geographic information system (GIS). For the Comanche study area, we used the GIS to determine that all 10 colonies we surveyed for mountain plovers in 2005 had been expanding during 1999–2004. For the Pawnee and Phillips County study areas, we used the GIS to identify all colonies that were expanding for at least 6 years preceding a mountain plover survey.
For the analysis of plover nest distribution on expanding colonies, we calculated the area of each colony in each of three age classes: young, mid, and old. For all study areas, we define “young” portions of colonies as those areas colonied by prairie dogs only within the past 1–2 years prior to the mountain plover survey. Due to differences among sites in the timing of prairie dog colony surveys (every other year in Phillips County, no survey in 2000 for the Comanche study area), we define “mid”-aged portions of colonies as areas occupied by prairie dogs for the past 3–5 years at Comanche and Pawnee, and as areas occupied by prairie dogs for the past 3–6 years in Phillips County. Finally, we define “old” portions of colonies as areas occupied by prairie dogs for 6+ years at Comanche and Pawnee, and areas occupied by prairie dogs for 7+ years in Phillips County. The 1 year difference in colony age used to define age classes was necessary to ensure a balanced sample of age classes for each of the three sites. Because mountain plovers arrive on the breeding range in March–April of each year, which was at least several months prior to when prairie dog colonies were mapped, we used boundaries of prairie dog colonies mapped in the year prior to mountain plover nest surveys as the best measure of available plover habitat.
For each mountain plover nest, we used the GIS to determine the history of prairie dog occupancy at the nest location. We calculated the expected distribution of nests in the different occupancy classes for each individual prairie dog colony based on the total number of nests found on that colony and the area of each occupancy class on the colony. We then summed across colonies to determine the expected number of nests in each occupancy class, in order to account for variation among colonies in nest density. We used chi-squared tests (α = 0.05) to compare the observed versus expected distribution of nests in each study area.
For the Comanche study area, we examined the distribution of 24 mountain plover nests found in 2005 on 10 prairie dog colonies encompassing 827 ha. For the Pawnee study area, we identified two colonies in 2003, one colony in 2005, one colony in 2006 and two colonies in 2007 (six different colonies) that were surveyed for mountain plovers after 6 previous years of documented colony expansion. We examined the distribution of 20 mountain plover nests on these six colonies, which encompassed 632 ha. For Phillips County, we analyzed nest distributions in 2003 and 2005 separately because a sufficiently large number of nests was sampled in each year. In 2003, we studied 114 mountain plover nests located on 22 prairie dog colonies encompassing 1,114 ha. In 2005, we studied 76 nests located on 15 prairie dog colonies encompassing 867 ha. For Phillips County in 2005, a more detailed analysis of nest distribution relative to prairie dog occupancy history was possible due to both the large sample of nests and the longer available record of prairie dog colony boundaries. For this survey, we additionally calculated the area of each colony occupied for 1–2, 3–4, 5–6, 7–8 and 9+ years.
To evaluate the influence of a recent epizootic plague outbreak in Phillips County, we also identified (1) all colonies that increased in size between 2002 and 2004, and for which at least one plover nest was documented in 2003 and/or 2005, and (2) all colonies that decreased by more than 50% between 2002 and 2004 due to apparent epizootic plague effects, and for which at least one plover nest was documented in 2003 and/or 2005. For this analysis, data were non-normally distributed so we used a Wilcoxon rank sum test (α = 0.05) to compare the number of nests on expanding and declining colonies.
Results
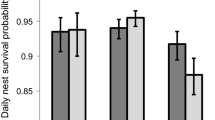
Nests were found in all prairie dog colony age classes in proportion to expected distribution based on the area of each age class available for the Comanche study area (χ2 = 0.08, df = 2, P = 0.96; Fig. 3a), the Pawnee study area (χ2 = 3.16, df = 2, P = 0.21; Fig. 3b), and the Phillips County study area in 2003 (χ2 = 4.17, df = 2, P = 0.12; Fig. 3c) and 2005 (χ2 = 0.07, df = 2, P = 0.97; Fig. 3d). A more detailed analysis of nest distribution on expanding colonies in Phillips County in 2005 showed the same trends as the grouped analysis, with nests distributed across all colony age classes, including areas that had been occupied by prairie dogs for 9 or more years (χ2 = 1.65, df = 4, P = 0.80).
Comparison of the number of mountain plover nests observed versus expected in areas of prairie dog colonies with different lengths of prairie dog occupancy on the Comanche National Grassland, Colorado in 2005 (a), the Pawnee National Grassland, Colorado, during 2003–2007 (b) and in Phillips County, Montana in 2003 (c) and 2005 (d). Age classes refer to the length of time that prairie dogs had occupied different areas of the colonies prior to the nest survey (see Methods). In each case, mountain plover nests were distributed in proportion to the availability of the three age classes within colonies (χ2 ≤ 4.17, df = 2, P ≥ 0.12 for all comparisons)
In the Comanche study area, we resurveyed four colonies in 2007 where nesting plovers were documented in 2005, and where prairie dogs were decimated by plague in 2006. In 2007, no live prairie dogs were observed at three of the four colonies, and only ∼1 ha of active prairie dogs was observed at the fourth colony. In 2005, we documented a total of 13 plover nests on these four colonies. In 2007, we observed only one plover at one colony. The bird did not exhibit behaviors indicative of nesting adults, and no plovers were observed during subsequent visits to this site.
In the Phillips County study area, we identified 21 colonies that were expanding between the 2002 and 2004 colony surveys, and on which plover nests were found in 2003 or 2005. We also identified nine colonies that contracted substantially (>50%; mean = 80%) between 2002 and 2004 due to apparent plague effects (Table 2). None of these colonies were extirpated completely between 2002 and 2004, but mean colony size declined from 102.2 ha in 2002 to 15.7 ha in 2004 (Table 2). The net change in number of nests per colony during 2003–2005 for declining colonies (mean = −2.44 nests colony−1; median = −2.0 nests colony−1) was significantly lower than the net change on expanding colonies (mean = −0.24 nests colony−1; median = +1.0 nest colony−1; Wilcoxon rank sum test, two-sided P = 0.027; Table 2).
Discussion
In mixed prairie, the abundance of forbs and subshrubs increases on older prairie dog colonies (Coppock et al. 1983; Archer et al. 1987) and preferential nesting by mountain plovers has been observed in portions of prairie dog colonies with increased cover of these plant functional groups (Olson and Edge 1985). In shortgrass steppe, older colonies contain more bare soil than young colonies or uncolonized grassland (Hartley 2006) and mountain plovers often nest in portions of the landscape with high cover of bare soil (Knopf and Miller 1994; Shackford et al. 1999; Knopf and Rupert 1999). As a result, we originally hypothesized that mountain plovers would be confined to nesting in older areas of the colonies. In contrast, we found that across widely distributed sites in the western Great Plains, mountain plovers nested in both the youngest (colonized in the past 1–2 years) and the oldest (colonized more than 6–9 years) portions of prairie dog colonies in nearly direct proportion to the availability of these age classes.
Our findings suggest that changes in vegetation and bare soil induced by multiple years of prairie dog presence are not required to create habitat in which plovers will nest. Rather, prairie dog presence and grazing activity, even in recently-colonized grassland, are sufficient to create nesting habitat. One possibility is that plovers utilize all portions of active colonies because vegetation remains cropped throughout the nesting and brood rearing season, thus maintaining conditions where plovers can effectively avoid predation. A second non-mutually exclusive possibility is that arthropod availability influences plover habitat use (Olson 1985; Schneider et al. 2006). For example, compared to grassland without prairie dogs, young grasshoppers can be more abundant on prairie dog colonies (Russell and Detling 2003). Densities of ground-dwelling arthropods are also enhanced in the immediate vicinity of prairie dog mounds (Davidson and Lightfoot 2007), and this microhabitat is distributed throughout active colonies.
Following plague-driven declines in prairie dog colonies, we found that mountain plover nesting activity declined relatively rapidly, within 1–2 years. This result is consistent with both the loss of the short-term influence of prairie dog grazing activity and the relatively rapid recovery of vegetative conditions that can occur on prairie dog colonies following plague (Hartley 2006). Taken together, our findings indicate that available plover nesting habitat associated with prairie dog colonies closely tracks the area actively occupied by prairie dogs each year. We note that this does not imply that all areas of active colonies are suitable habitat, as plovers also select relatively flat terrain for nesting (Graul 1975), and localized features of colonies such as topography may influence suitability for plover nesting. However, our results do indicate that plague-driven fluctuations in the extent of active prairie dog colonies within the landscape also directly reflect fluctuations in the extent of prairie dog habitat used by nesting plovers.
Mountain plovers have been reported to exhibit strong philopatry in their breeding territory (Graul 1973, P. D. B. Skrade, unpublished data), but less is known about the distance individual plovers may move to new breeding locations in response to changing habitat conditions. Plague epizootics often affect clusters of prairie dog colonies located near one another, but can vary considerably in size of the area they affect within a year. Some landscape features such as lakes, rivers and high-traffic roads may serve as barriers to plague transmission, and at increasing densities can reduce the spatial extent of plague outbreaks (Collinge et al. 2005). Colony size and connectivity also strongly influence the occurrence and transmission of epizootic plague within the landscape (Stapp et al. 2004; Collinge et al. 2005; Antolin et al. 2006). These landscape features affecting spatial patterns of plague epizootics could in turn influence the nesting activity of mountain plovers.
In northern mixed prairie, where mountain plovers nest almost exclusively on prairie dog colonies, the spatial extent of plague epizootics may influence the degree to which plovers can shift from plague-affected colonies to other nearby active prairie dog colonies. In Phillips County, colonies affected by plague during 2002–2004 were all less than 7 km from a colony not affected by plague, and interannual movements among colonies separated by up to 15 km have been documented for banded plovers in this study area (S. Dinsmore, unpublished data). For plague epizootics that affect larger areas, the degree to which plovers may respond by moving longer distances or by nesting off of prairie dog colonies is not clear. A negative effect of plague on plover nesting activity in mixed prairie is consistent with our observation that the large decline in nests on plague-affected colonies during 2003–2005 was not balanced by an increase in nests on expanding colonies within the Phillips County study area (Table 2).
In shortgrass steppe, mountain plovers can nest in several habitat types with high cover of bare soil where prairie dogs are not present. In landscapes where plague affects an extensive area within a given year (e.g. ∼200,000 ha of the Comanche study area during 2006), the sustainability of mountain plover populations may depend upon the availability and quality of such alternate habitats as recently burned grasslands (Svingen and Giesen 1999) or agricultural fields (Knopf and Rupert 1999; Dreitz and Knopf 2007). Because plague incidence increases following years with above-average precipitation (Stapp et al. 2004), which coincides with increased grass production and fuel loads, prescribed burning may be one means to provide alternate plover nesting habitat in years and portions of the landscape where plague outbreaks have recently occurred. Given the presence of plague throughout the mountain plover’s breeding range in the western Great Plains, important factors affecting plover populations may include landscape features determining the scale of plague outbreaks, the distance that plovers can move in response to changing habitat conditions, and the availability and quality of alternate breeding habitat within the landscape.
References
Antolin ML, Savage L, Eisen R (2006) Landscape features influence genetic structure of black-tailed prairie dogs (Cynomys ludovicianus). Landsc Ecol 21:867–875
Archer S, Garrett MG, Detling JK (1987) Rates of vegetation change associated with prairie dog (Cynomys ludovicianus) grazing in North American mixed-grass prairie. Vegetatio 72:159–166
Augustine DJ, Matchett MR, Toombs T, Cully JF, Johnson TL, Sidle JG (2008) Spatiotemporal dynamics of black-tailed prairie dog colonies affected by plague. Landsc Ecol 23:255–267
Collinge S, Johnson W, Ray C, Matchett R, Grensten J, Cully J, Gage K, Kosoy M, Loye J, Martin A (2005) Landscape structure and plague occurrence in black-tailed prairie dogs on grasslands of the western USA. Landsc Ecol 20:941–955
Coppock DL, Ellis JE, Detling JK, Dyer MI (1983) Plant–herbivore interactions in a North American mixed-grass prairie. I. Effects of black-tailed prairie dogs on intraseasonal aboveground plant biomass and nutrient dynamics. Oecologia 56:10–15
Cully JF, Williams ES (2001) Interspecific comparisons of sylvatic plague in prairie dogs. J Mammal 82:894–905
Cully JK, Biggins DE, Seery DB (2006) Conservation of prairie dogs in areas with plague. In: Hoogland JL (ed), Conservation of the black-tailed prairie dog. Island Press, Washington, pp 157–168
Davidson AD, Lightfoot DC (2007) Interactive effects of keystone rodents on the structure of desert grassland arthropod communities. Ecography 30:515–525
Desmond MJ, Savidge JA, Eskridge KM (2000) Correlations between burrowing owl and black-tailed prairie dog declines: a 7-year analysis. J Wildl Manage 64:1067–1075
Dinsmore SJ (2003) Mountain Plover (Charadrius montanus): a technical conservation assessment. [Online]. USDA Forest Service, Rocky Mountain Region. Available: http://www.fs.fed.us/r2/projects/scp/assessments/mountainplover.pdf
Dinsmore SJ, White GC, Knopf FL (2005) Mountain plover population responses to black-tailed prairie dogs in Montana. J Wildl Manage 69:1546–1553
Dreitz VJ, Knopf FL (2007) Mountain plovers and the politics of research on private lands. BioScience 57:681–687
Forrest S (2005) Getting the story right: a response to Vermeire and colleagues. BioScience 55:526–530
Goodwin HT (1995) Pliocene–pleistocene biogeographic history of prairie dogs, genus Cynomys (Sciuridae). J Mammal 76:100–122
Graul WD (1973) Breeding adaptations of the mountain plover (Charadrius montanus). PhD Dissertation, University of Minnesota, St. Paul, MN
Graul W (1975) Breeding biology of the mountain plover. Wilson Bull 87:6–31
Hartley LM (2006) Plague and the black-tailed prairie dog: an introduced disease mediates the effects of an herbivore on ecosystem structure and function. PhD Dissertation, Department of Biology, Colorado State University, Fort Collins, CO
Hoogland JL (ed) (2006) Conservation of the black-tailed prairie dog. Island press, Washington DC, 350 pp
Johnson TL (2005) Spatial dynamics of a bacterial pathogen: sylvatic plague in black-tailed prairie dogs. MS Thesis, Kansas State University, Manhattan, KS
Johnson-Nistler CM, Sowell BF, Sherwood HW, Wambolt CL (2004) Black-tailed prairie dog effects on Montana’s mixed-grass prairie. J Range Manage 57:641–648
Knopf FL, Miller BJ (1994) Charadrius montanus—montane, grassland, or bare-ground Plover? Auk 111:504–506
Knopf FL, Rupert JR (1999) Use of cultivated fields by breeding mountain plovers in Colorado. Stud Avian Biol 19:81–86
Knopf FL, Wunder MB (2006) Mountain Plover (Charadrius montanus). In: Poole A (ed) In the birds of North America online. Cornell Laboratory of Ornithology, Ithaca. From the birds of North America online database: http://bna.birds.cornell.edu/BNA/account/Mountain_Plover
Kotliar NB, Baker BW, Whicker AD, Plumb GE (1999) A critical review of assumptions about the prairie dog as a keystone species. Environ Manage 24:177–192
Kotliar NB, Miller BJ, Reading RP, Clark TW (2006) The prairie dog as a keystone species. In: Hoogland JL (ed) Conservation of the black-tailed prairie dog. Island Press, Washington DC, pp 53–64
Lomolino MV, Smith GA (2003) Terrestrial vertebrate communities at black-tailed prairie dog (Cynomys ludovicianus) towns. Biol Conserv 115:89–100
Miller B, Reading RP, Forrest S (1996) Prairie night: black-footed ferrets and the recovery of endangered species. Smithsonian Institution Press, Washington DC
Olson S (1985) Mountain Plover food items on and adjacent to a prairie dog town. Prairie Nat 17:83–90
Olson S, Edge D (1985) Nest site selection by Mountain plovers in north central Montana. J Range Manage 38:280–282
Pauli JN, Buskirk SW, Williams ES, Edwards WH (2006) A plague epizootic in the black-tailed prairie dog (Cynomys ludovicianus). J Wildl Dis 42:74–80
Russell RE, Detling JK (2003) Grasshoppers (Orthoptera: Acrididae) and black-tailed prairie dogs (Sciuridae: Cynomys ludovicianus (Ord)): associations between two rangeland herbivores. J Kans Entomol Soc 76:578–587
Schneider SC, Wunder MB, Knopf FL (2006) Relationship between shrubs and food in mountain plover habitat in Park County, Colorado. Southwest Nat 51:197–202
Shackford JS, Leslie DM, Harden WD (1999) Range-wide use of cultivated fields by Mountain plovers during the breeding season. J Field Ornithol 70:114–120
Smith G, Lomolino M (2004) Black-tailed prairie dogs and the structure of avian communities on the shortgrass plains. Oecologia 138:592–602
Stapp P, Antolin MF, Ball M (2004) Patterns of extinction in prairie dog metapopulations: plague outbreaks follow El Niño events. Front Ecol Environ 2:235–240
Svingen D, Giesen K (1999) Mountain plover (Charadrius montanus) response to prescribed burns on the Comanche National Grassland. J Colorado Field Ornithol 33:208–212
Tipton HC (2007) Occupancy, abundance and density of Colorado breeding grassland birds: estimation and habitat correlations. MS Thesis, Colorado State University, Fort Collins, CO
Vermeire LT, Heitschmidt RK, Johnson PS, Sowell BF (2004) The prairie dog story: do we have it right? BioScience 54:689–695
Winter SL, Cully JF Jr, Pontius JS (2002) Vegetation of prairie dog colonies and non-colonized shortgrass prairie. J Range Manage 55:502–508
Wunder MB (2007) Geographic structure and dynamics in mountain plover. PhD Dissertation, Graduate Degree Program in Ecology, Colorado State University, Fort Collins, CO
Acknowledgements
We thank M. Ball, J. F. Cully, J. J. Grensten, E. Humphrey, T. L. Johnson, and M. R. Matchett, for their contributions collecting and providing the prairie dog colony survey data. We thank M. D. Margulies, C. D. Hanson, C. T. Wilcox, R. Schmitz for assistance with mountain plover nest surveys, and J. Hoogland, M. R. Matchett, M. W. Miller and two anonymous reviewers for helpful reviews of the manuscript. Funding was provided by the USDA—Forest Service, USDA—Agricultural Research Service, U.S. Bureau of Land Management (Phillips Resource Area), Montana Fish, Wildlife, and Parks, Colorado Division of Wildlife, U.S. Geological Survey—Biological Resources Division, the Lois Webster Fund of the Audubon Society of Greater Denver, and U.S. Fish and Wildlife Service.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Augustine, D.J., Dinsmore, S.J., Wunder, M.B. et al. Response of mountain plovers to plague-driven dynamics of black-tailed prairie dog colonies. Landscape Ecol 23, 689–697 (2008). https://doi.org/10.1007/s10980-008-9230-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10980-008-9230-y