Abstract
The effects of habitat gaps on breeding success and parental daily energy expenditure (DEE) were investigated in great tits (Parus major) and blue tits (Cyanistes caeruleus) in urban parkland (Cardiff, UK) compared with birds in deciduous woodland (eastern England, UK). Tree canopy height, the percentage of gap in the canopy and the percentage of oak (in the wood only) within a 30 m radius of nest boxes were obtained from airborne remote-sensed data. Breeding success was monitored and parental DEE (great tits: both habitats; blue tits: park only) was measured using doubly labelled water in birds feeding young. In the park, mean (±SD) tree height (7.5 ± 4.7 m) was less than in the wood (10.6 ± 4.5 m), but the incidence of gaps (32.7 ± 22.6%) was greater (9.2 ± 14.7%). Great tits and blue tits both reared fewer young in the park and chick body mass was also reduced in park-reared great tits. Park great tits had a higher DEE (86.3 ± 12.3 kJ day−1) than those in the wood (78.0 ± 11.7 kJ day−1) and, because of smaller brood sizes, worked about 64% harder for each chick reared. Tits in the park with more than about 35% gap around their boxes had higher DEEs than the average for the habitat. In the wood, great tits with less oak around their boxes worked harder than average. Thus structural gaps, and functional gaps generated by variation in the quality of foraging habitat, increased the costs of rearing young.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Habitat fragmentation and loss are two of the major causes of current worldwide declines in biodiversity (Ehrlich and Wilson 1991; Heywood 1995). The extent of fragmentation in the UK (Fuller et al. 1994) and elsewhere means that substantial proportions of many species populations now live in such habitat (Vane-Wright et al. 1991). For birds and other wildlife, small patches, whether rural or urban, may constitute sub-optimal habitat. For example, small patches may lack food resources and be more exposed to poor weather conditions and certain predators (Andrén 1992; Burke and Nol 1998; McCollin 1998). In urban parkland, structural patchiness can be exacerbated by functional patchiness due to high proportions of exotic plant species which may support relatively few invertebrates, reducing the foraging opportunities for birds (Mills et al. 1989; Reichard et al. 2001; Stauss et al. 2005). The wider diversity of plant species may also generate temporal patchiness when differing phenologies create mismatches in timing between food supply and demand (Dias and Blondel 1996; Schoech and Bowman 2001; Thomas et al. 2001). Thus birds living in patchy habitat may have to travel more widely in search of food, increasing their workload (Eybert et al. 1995; Hinsley 2000). Continuous woodland has fewer physical gaps, but food resources vary between both tree species and individual trees of the same species resulting in functional patchiness and a patchy use of territory by foraging birds (Naef-Daenzer 2000; Stauss et al. 2005; Tremblay et al. 2005).
In this paper, we investigate the effects of structural and functional habitat gaps (quantified using airborne remote sensing) on parental energy expenditure (measured using doubly labelled water) and breeding success in great tits (Parus major) and blue tits (Cyanistes caeruleus) feeding young in urban parkland and in continuous woodland. Structural gaps are defined as physical spaces in the tree canopy; functional gaps are defined as arising from differences in the quality of different plant species as foraging habitat (Kennedy and Southwood 1984; Peck 1989; Lambrechts et al. 2004; Alexander et al. 2006). For altricial nestlings, all food must be carried to the nest and thus breeding success may be affected by the amount of time and energy the adults expend in crossing gaps. Similarly, the presence of gaps may increase travel times and distances if adults take longer routes around gaps to reduce potential exposure to aerial predators (Hinsley et al. 1995; Desrochers and Hannon 1997). Although principally forest birds, in the UK, great tits and blue tits are common breeders in secondary habitats such as small woods, farmland hedgerow-tree networks and suburban-urban gardens and parks. All these secondary habitats are characteristically patchy and reproductive success in both species is known to be reduced compared with continuous woodland (e.g. Schmidt and Steinbach 1983; Cowie and Hinsley 1987; Riddington and Gosler 1995; Hinsley et al. 1999). Both species also feed their young principally on tree-dwelling lepidopteran larvae (Perrins 1979; Perrins 1991) and the availability of caterpillars influences many aspects of tit breeding ecology including chick growth, survival and recruitment (Haywood and Perrins 1992; Keller and van Noordwijk 1994; Tremblay et al. 2003). The abundance and distribution of the food supply available to adults when feeding young can also affect adult body condition and survival by increasing the time and energy demands of foraging (Tinbergen and Dietz 1994; Moreno et al. 1995; Merilä and Wiggins 1997; Sanz et al. 1998; Thomas et al. 2001). If structural and functional gaps influence foraging habitat quality, then we hypothesize that parental energy expenditure should correlate with the availability and quality of trees around the nest site. Thus birds in patchy habitat may suffer the double penalty of having to work harder for a reduced reproductive success (Daan et al. 1996).
Methods
Study sites
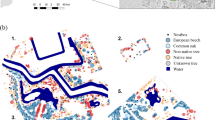
The two study sites were Bute Park (51°29′ N, 3°11′ W) in Cardiff, south Wales, UK, and Monks Wood National Nature Reserve (NNR) (52°24′ N, 0°14′ W) in Cambridgeshire, eastern England, UK. Bute Park comprises about 53 ha and is located in the centre of the city of Cardiff, lying more or less north-south along the east bank of the River Taff (Fig. 1). The southern end includes Cardiff Castle, formal gardens, mown grass and an arboretum while the northern end has sports pitches and some more extensive areas of woodland. Tree species diversity is high with many exotics often planted in groups of closely related species and varieties. The most abundant native species are common lime (Tilia europaea) and sycamore (Acer pseudoplatanus), plus smaller amounts of common ash (Fraxinus excelsior) and occasional English oaks (Quercus robur). The most frequently occurring species overall, including exotics, are limes (Tilia spp.), maples (Acer spp.), oaks (Quercus spp.) and pines (Pinus spp.). Conifers constitute about 15% of all formally planted trees. The park is heavily used by the public for recreation, sports, concerts and other events.
LiDAR first return Digital Surface Model of Bute Park, Cardiff (area shown is 1.4 km × 1.8 km) and Monks Wood, Cambridgeshire (area shown is 1.8 km × 1.7 km). The boundaries of the study areas are shown by dashed lines. Lighter shades of grey denote higher elevations, e.g. the northerly slope of Monks Wood is shown by the transition from light to darker shading from the bottom to the top of the image. Map of mainland UK shows site locations
Monks Wood comprises 157 ha of mixed deciduous woodland (Gardiner and Sparks 2005) (Fig. 1). It occupies a shallow north facing slope of maximum angle 14.5° and elevational range 6–46 m. The dominant tree species, in order of abundance, are common ash, English oak and field maple (Acer campestre) and are well mixed throughout the wood. Other tree species include small-leaved elm (Ulmus minor), silver birch (Betula pendula) and aspen (Populus tremula), and the main shrub species are hawthorn (Crataegus spp.), blackthorn (Prunus spinosa) and common hazel (Corylus avellana). These are all native species. The field layer is dominated by grasses and sedge (Carex pendula). There is a network of paths and rides, some wide enough to create gaps in the canopy, two open fields (4.3 ha and 1.7 ha), a number of smaller glades and several ponds. On part of its southern boundary, the wood is adjoined across a minor road by a mature 37 ha conifer plantation.
Site data
In Bute Park, a total of 26 sites throughout the centre and north of the park were provided with wooden nest boxes, with a hole-diameter of 32 mm, allowing access by both great tits and blue tits. Each year, 19 boxes were available during the breeding season (typically late March to the end of June), the additional seven sites arising through replacement of vandalized/stolen boxes. Boxes were located randomly with a spacing of about 40–100 m. The position of each nest box was recorded by differential GPS Real Time Kinematic survey (Topcon Hiper+ GPS receiver and Legacy-E Base Station) during winter, leaf-off conditions. Breeding success and energy expenditure of great tits was measured in Bute Park in 2003–2005, and in blue tits in 2004–2005. The extension of the project into 2005 in the park (but not in the wood, see below) was to compensate for a low sample size (one bird) in the park in 2003. In the park, in 2003–2005, the numbers of boxes in which great tits reached the chick-rearing stage were two, eight and five respectively, and energy expenditure was measured successfully at one, six and five of these. In 2004–2005, the numbers of boxes in which blue tits reached the chick-rearing stage were nine and six respectively, and energy expenditure was measured at six and four of these.
In Monks Wood, 36 nest boxes similar to those used in the park, and similarly located, were available each year. The position of each box was recorded during winter, leaf-off conditions using an electronic total station (Pentax R-125N), surveying from an established Ordnance Survey benchmark. Breeding success and energy expenditure of great tits was measured in Monks Wood in 2003 and 2004, and breeding success of blue tits in 2004 and 2005. Energy expenditure was only measured in great tits in the wood because too few boxes were used by blue tits (great tits, being larger, could out-compete blue tits for boxes). The numbers of boxes in which great tits reached the chick-rearing stage were 23 and 25 in 2003 and 2004 respectively and energy expenditure was measured successfully at 15 and nine of these. The numbers of boxes in which blue tits reached the chick-rearing stage in Monks Wood were five and two in 2004 and 2005 respectively. To obtain an adequate sample for blue tit breeding success in woodland, results from five boxes in another, similar, wood in Cambridgeshire were also used (Brampton Wood, 132 ha, mixed ash, oak, field maple, 9 km south of Monks Wood). No other data for Brampton Wood were used here. Ambient temperature was recorded in both Monks Wood and Bute Park using automatic loggers (Micro-T-log temperature datalogger, F.W. Parrett Ltd.) recording every 4 h.
Canopy height and habitat patchiness were measured using airborne Light Detection And Ranging (LiDAR). Airborne scanning LiDAR is a remote sensing technique which can provide finely resolved data describing vegetation structure (Lim et al. 2003; Næsett 2004) of particular value for ecological applications (Lefsky et al. 2002; Hill et al. 2004; Bradbury et al. 2005; Broughton et al. 2006; Hinsley et al. 2006). It uses a laser range finder to measure the elevation of points in a swath beneath the flight-path of an aircraft. Short duration pulses of near infrared laser light are fired at the ground and the return signals backscattered from the ground itself and/or surface features such as trees and buildings are recorded (Wehr and Lohr 1999). The timing of the returns, combined with measurement of the aircraft’s orientation and position, allow the 3D position of the ranged points to be calculated and geo-referenced (Ackermann 1999). Digital models of the surface of the ground and of vegetation canopy height can then be derived from these measurements (Hill et al. 2002). Further details of the analysis of the LiDAR data are given in the supplementary material.
LiDAR data for Bute Park were acquired on June 14th 2004 using an ALTM 3033 scanner, and for Monks Wood on June 10th 2000 using an Optech ALTM 1210 scanner. The ALTM is a small footprint (20–25 cm on the ground for these data sets), discrete return system supplying the first and last significant return per laser pulse. The ALTM 3033 had a 33 kHz repetition rate, and data were acquired with a scan angle of ±20° and a post spacing of one hit per 1.66 m2. The ALTM 1210 had a 10 kHz laser pulse repetition rate, and data were acquired with a scan angle of ±10° and a post spacing of one hit per 4.83 m2.
For Monks Wood, a tree species map was also available, produced from supervised classification of time series Airborne Thematic Mapper (ATM) multi-spectral data from 2003 (George 2005). Different types of vegetation cover, and at a finer scale, different tree species, have characteristic reflectance spectra due to differential reflection of solar radiation (Treitz and Howarth 2000; Carleer and Wolff 2004). Such differences between species can also be increased by differing leaf phenologies, e.g. rates of development and senescence (Wolter et al. 1995; Mickelson et al. 1998). Thus, using ATM data from five images of the wood, the six dominant tree species (see above) in the top canopy were mapped with an assessed accuracy of ca. 89%. The tree species map had a minimum height threshold of 8 m, which masked out areas of shrubs and young trees.
Bird breeding performance
All boxes were visited approximately weekly from the end of March until July. The following parameters were recorded (i) first egg date, (ii) clutch size, (iii) hatching date, (iv) number of young alive in the nest at 11 days of age where day of hatching = 0, (v) mean chick weight (g) at 11 days, excluding runts (runts were defined as chicks too small to be ringed at age 11 days and were rare), (vi) total live biomass (g) of young in the nest at 11 days (including runts), (vii) number of young fledged, and (viii) overall success calculated as the percentage of eggs producing fledged young. Chicks were weighed to 0.1 g using a spring balance, and were also ringed with a uniquely numbered ring of the British ringing scheme. After the young had fledged, the nest was removed from the box and searched for dead chicks and unhatched eggs.
Bird energy expenditure
Energy expenditure of birds feeding young was measured using doubly labelled water (DLW). This technique uses the differential turnover of oxygen-18 (18O) (excreted from the body in water and carbon dioxide) and deuterium (2H) (excreted in water) to measure carbon dioxide production which can then be converted to energy expenditure (Speakman 1997). The technique has been used on a wide range of animals, including humans, and provides the best means of measuring energy expenditure in free-living animals (Speakman 1998). Adults feeding ca. 11 day-old young were trapped at the nestbox, injected intraperitoneally with approximately 0.1 ml of DLW and a baseline blood sample collected after allowing 0.5 h for equilibration with the body water. Birds were then released to continue feeding the young and were retrapped about 24 h later to collect a final sample. To reduce disturbance at the nest, only one member of each pair was trapped. This was usually the female, but a few male blue tits were also trapped due to the greater difficulty of distinguishing the sexes. Further details of the DLW methodology and analysis are given in the supplementary material.
Of a total of 51 measurement attempts, 45 were successful, five failed (four great tits were not retrapped, one final sample dried out, all Monks Wood birds) and one park blue tit deserted and her brood of four died. The broods of all the other 50 birds fledged. All trapping, sampling and storage procedures were carried out under licence (see acknowledgements) and all operators were experienced and licenced bird ringers.
Bird-habitat analysis
Data on mean tree canopy height and the percentage of gap (defined as canopy <1 m tall) in the canopy were extracted from the digital canopy height models of Monks Wood and Bute Park for 30 m radius circles centred on the nest box locations. In addition, for 22 of the 36 Monks Wood nest boxes, the percentage of oak tree canopy was also extracted per 30 m radius circle. This information was extracted from the tree species map and was expressed as a percent of tree canopy rather than as a percent of the whole 30 m circle. However, given the low incidence of gaps in the wood, these two measures were similar for most boxes. Oak canopy could not be obtained for the remaining 14 Monks Wood boxes because the tree map did not include some woodland which lay outside the boundary of the NNR. A 30 m radius sample plot was used because this distance was representative of the typical foraging distances of tits (see discussion) (Stauss et al. 2005; Tremblay et al. 2005). Foraging distances may be greater (e.g. 40–50 m) in lower quality habitat, but the amounts of gap for radii of 30 m and 50 m were highly correlated (Bute Park: r = 0.957, P < 0.001, n = 26; Monks Wood: r = 0.960, P < 0.001, n = 36), and using 30 m for both study sites minimised assumptions concerning likely quality.
Breeding performance and parental DEE were summarised for each species and compared between the park and wood using two sample t tests. The effects of habitat structure (expressed as mean canopy height and percentage gap within 30 m in both the park and the wood, and as the percentage of oak canopy within 30 m in the wood) on parental DEE were examined using linear or quadratic regression analysis as appropriate to obtain best fit. Parental DEE was expressed as kJ day−1, and also as the percentage deviation from the mean DEE for each site calculated as (DEE–mean for the year)/mean for the year. All analyses were done using Minitab Release 12.
Results
In the park (n = 26), the mean canopy height within 30 m of each nest box was less than in the wood (n = 36) (mean ± SD canopy height: park = 7.5 ± 4.7 m; wood = 10.6 ± 4.5 m; t 60 = −2.62, P = 0.011). In particular, there were many more gaps in the tree canopy in the park (% gap: park = 33 ± 23%; wood = 9 ± 15%; t 60 = 4.96, P < 0.001). In Monks Wood, the amount of gap was less than 10% for 26 of the 36 boxes (Fig. 2), and less than 1% for 21 of them. The occurrence of gaps in the wood was related to the presence of large rides and proximity to the edge of the wood.
Great tits in the park bred earlier than those in the wood, but all measures of breeding performance were significantly reduced (Table 1). Female DEE was greater in the park (and the difference close to significance at P = 0.058), but the smaller brood sizes and fledging success increased the females’ costs per chick by about 64% (Table 1). These differences between the park and the wood remained when comparing within the same year, i.e. 2004 (DEE: t 13 = −2.47, P = 0.028; DEE per chick: t 13 = −3.58, P = 0.003, Table 1). In blue tits, there was no difference between the park and the wood in the timing of breeding or clutch size, but fewer chicks were reared to fledging (Table 2). However, the quality of the young, measured as mean chick body mass (Lloyd 1987; Slagsvold et al. 1995), was comparable with that of woodland chicks. Earlier timing of breeding by urban great tits, but not blue tits, has been noted in previous work (Perrins 1979; Cowie and Hinsley 1987) and may be related to differential use of artificial food (Dhondt et al. 1984).
In the park (great tit, n = 12; blue tit, n = 10), we predicted that DEE would decrease with canopy height and increase with the amount of gap around the box, and while these trends were evident for both species, the relationships were not significant. However, when DEE was expressed as the percentage deviation from the mean DEE for the year, females working harder than average were found to have more patchy habitat around their nest boxes (great tits and blue tits combined, one outlier omitted, Fig. 3). Daily energy expenditure, relative to the mean, decreased with increasing canopy height and increased with increasing amounts of gap. The quadratic relationship (Fig. 3, % gap) showed that DEE, relative to the mean, was not linearly related to the amount of gap, but showed an increasing trend above about 35% gap.
Percentage deviation from mean daily energy expenditure (DEE) of great tits (closed circles) and blue tits (open circles) rearing young in Bute Park in relation to (a) mean canopy height (m) and (b) the amount of gap (%) measured within 30 m radius sample areas around the nest box. Regression equations are (i) canopy height: % deviation = 25.197–5.800 canopy height + 0.252 canopy height2, r 2 = 0.32, P = 0.031, n = 21; (ii) gap: % deviation = −1.868–0.506% gap + 0.013% gap2, r 2 = 0.42, P = 0.008, n = 21. Deviations were calculated separately for 2004 and 2005, and an overall mean used for 2003. Arrows indicate an outlier which was omitted from the calculations of the fitted lines, see discussion
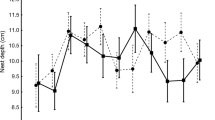
In the wood (great tit, n = 23), two females had relatively high DEEs in 2003 (as apparent in Fig. 4, see below). Without the results from these two birds, both DEE and the percentage deviation from the mean DEE for the year declined linearly with increasing canopy height (as found in the park), but the former relationship was weak (r 2 = 0.17, P = 0.061, n = 21) and the latter non-significant and they are not shown. Gaps were relatively rare in Monks Wood (Fig. 2), and there were no relationships with the amount of gap. However, both DEE and the percentage deviation declined as the percentage of oak canopy around the nest box increased (Fig. 4). Similar to the pattern of results for canopy gaps in the park, as the percentage of oak decreased below about 30%, birds worked increasingly harder. There was also an indication of a year effect. Both the females with the highest DEEs in 2003 had little oak within 30 m of their boxes, whereas the influence of oak appeared to be less in 2004 (Fig. 4).
Daily energy expenditure (DEE) (a) and percentage deviation from mean daily energy expenditure (b) of female great tits rearing young in Monks Wood in relation to the presence of oak canopy (as percentage of the canopy area of all trees ≥8 m in height) within a 30 m radius of the nest box. Regression equations are (i) DEE = 101.893–1.189% oak + 0.0113% oak2, r 2 = 0.56, P = 0.001, n = 19; (ii) % deviation = 30.267–1.145% oak + 0.0135% oak2, r 2 = 0.60, P = 0.001, n = 19. Closed triangles show results for 2003 and open triangles results for 2004
Mean and minimum ambient temperatures for the 24 h during which DEE was measured did not differ between Bute Park and Monks Wood (park: mean ± SD = 14.4 ± 1.7°C, minimum = 11.6 ± 1.7°C, n = 22; wood: mean = 15.1 ± 1.9°C, minimum = 11.7 ± 2.1°C, n = 23. Mean: t 43 = −1.26, P = 0.213; minimum: t 43 = −0.21, P = 0.837).
Discussion
In the park, the reproductive success of great tits was reduced compared with the wood, and yet female energy expenditure was higher, despite the smaller brood sizes. Thus great tits in the park worked harder for less return. We did not measure food abundance directly (the high tree species diversity in parkland makes frass trapping ineffective (Zandt 1994)), but the increase above average DEE in birds whose territories had more than about 35% gap indicated that the availability of trees per se had a direct effect on the costs of rearing young. Work on great tits in the Netherlands (Verhulst and Tinbergen 1997; Tinbergen and Verhulst 2000) has suggested that adults feeding young are constrained by time, rather than by intrinsic (e.g. phylogenic or physiological) limits to energy expenditure, due to a shortage of daylight for foraging. Our results for the park birds are consistent with this because, despite their higher work rate compared with Monks Wood birds, their DEE was similar or slightly less than values reported for great tits in various woodland sites in the Netherlands (Tinbergen and Dietz 1994; Tinbergen and Verhulst 2000) and were not excessive in terms of average (Daan et al. 1990; Bryant and Tatner 1991) or maximal suggested limits for sustainable energy expenditure (Lindström and Kvist 1995; Hammond and Diamond 1997). Tinbergen and Verhulst (2000) also suggested that ambient temperature might impose an extrinsic limit to energy expenditure (an “energetic ceiling”), but temperatures during the measurement of DEE did not differ between Bute Park and Monks Wood.
The results for the blue tit indicated by the arrows in Fig. 3 were omitted from the calculations of the fitted lines because the value of DEE for this bird was low compared to the rest of the data set. In a study of marsh tits (Poecile palustris), Nilsson (2002) found that six out of 12 females took 4–10 h to resume feeding their young after the initial procedures of the DLW method and that this reduced their DEE by an average of 46% compared with the others which resumed within 1 h. Although such adverse reactions are unusual (Speakman 1997), it is possible that the park blue tit was slow to resume normal feeding behaviour resulting in the low value. Results for great tits and blue tits in the park were combined by expressing DEE as the deviation from the mean because the breeding ecology of both species is well known to be particularly dependent on the spring abundance of tree-dwelling caterpillars (e.g. van Balen 1973; Perrins 1979; Perrins 1991). The use of alternative food supplies by either species would tend to obscure relationships with tree canopy height and gaps and the data for the two species showed no sign of segregation by species across the relationships (Fig. 3).
In Monks Wood, the relationships between both DEE and the percentage deviation from the mean for the year and the availability of oak around the nest box were analogous to that between the percentage deviation and amount of gap in Bute Park (Figs. 2 and 3). In the wood, birds with less oak around their boxes worked harder than average, while in the park, birds with fewer trees worked harder. In the park, English oak was rare within 30 m of any experimental box and thus the prime problem for the park birds appeared to concern quantity, rather than quality, of trees. Obviously, the non-oak species in Monks Wood do provide the birds with foraging opportunities, but the relationship with oak highlights its importance as a source of caterpillars as found in other studies (e.g. Perrins 1991; Fischbacher et al. 1998; Naef-Daenzer et al. 2004) and suggests that functional gap effects may commonly influence bird foraging behaviour. Tinbergen and Verhulst (2000) found an unusually high DEE for one female great tit which spent much of her time flying to a relatively distant oak with a high density of small caterpillars. Thus, the two females in Monks Wood with little oak canopy within 30 m and high DEEs in 2003 may have been travelling further than average to find oak trees. Other work has also shown that functional gap effects may operate at a landscape scale due to a heterogeneous distribution of forest patch sizes and tree species composition (e.g. Lambrechts et al. 2004).
The difference between the years in Fig. 4 suggested that the importance of oak to territory quality may differ between years (Lõhmus 2003; Hinsley et al. 2006). In blue tits breeding in suburban gardens, the amount of oak and rowan (Sorbus acuparia) within 25 m of the nest explained 21% of the variation in fledging success in one out of three years (Cowie and Hinsley 1987). In this 1 year, but not the other two, the time of peak chick demand (age 10–11 days, Perrins 1991) coincided with days of heavy rain.
Thomas et al. (2001) reported that a mismatch between the timing of breeding and peak caterpillar abundance could double the cost of rearing young in blue tits breeding in evergreen holm oak (Quercus ilex) forest in southern France (but also see Verhulst and Tinbergen 2001). For the most badly timed birds, DEE was in the region of 120–130 kJ day−1, which raises the question of why the Bute Park birds did not work harder, if such work rates are possible? If the evergreen oak habitat was more continuous than that of the park, then possibly the park tits were more constrained by the time taken to cross, or avoid, gaps than the additional energy expenditure. In a study of blue tits on Corsica (Tremblay et al. 2005), birds at a site with low caterpillar abundance, had average foraging distances from the nest of more than twice that of birds where caterpillar abundance was high (53 m versus 25 m). However, total flight distances per hour were similar because the birds in poor habitat made longer, but fewer, trips and fed their young a similar total biomass of caterpillars comprising fewer, larger prey. Tremblay et al. (2005) suggested that foraging costs, although not measured directly, would therefore be similar in both habitats because the costs of longer, but fewer, foraging trips would be offset by lower costs whilst searching more selectively within particular trees for large prey, i.e. the costs of searching would be less than the costs of flight (Goldstein 1990; Hinsley 2000). This was in contrast to the shorter, but more frequent, foraging trips and shorter search times of the birds in the good habitat. Similarly, starlings (Sturnus vulgaris) rearing experimentally enlarged broods, were able to maintain food delivery rates per chick without increasing DEE by adjustments in foraging and social behaviour and prey selection (Wright et al. 1998). Thus parkland birds might be able to minimise overall DEE by adopting a selective, large prey, strategy.
A study of foraging distances in blue tits in Germany (Stauss et al. 2005), again found shorter foraging distances in high quality habitat (deciduous woodland, 22 m) compared with low quality habitat (mixed woodland, 40 m). Prey size was not measured, but unlike the Corsican birds, feeding rates did not differ between the two habitats, suggesting that parental costs in the poor habitat should have been greater. In both Corsica and Germany, clutch and brood sizes were smaller in the poor habitats, whereas chick body mass was also lower in Corsica, but not in Germany. The lower body mass in poor habitat in Corsica was thought to be due to infestation of the young by blow-fly larvae (Protocalliphora spp.) because the total amount of prey delivered was the same as in the high quality habitat. In Bute Park, blue tits reared fewer young than in Monks Wood, but chick body mass did not differ. In contrast, great tits in the park reared both fewer and lighter young. Juvenile survival and recruitment into the breeding population is positively correlated with fledging mass (Tinbergen and Boerlijst 1990; Lindén et al. 1992); thus blue tits appeared to cope better in the park than great tits, as has been found for these two species in other secondary habitats such as suburban gardens (Cowie and Hinsley 1987) and small woods (Hinsley et al. 1999). Blue tits are smaller than great tits (ca. 10 g versus 18 g) and feed their young proportionately smaller prey items. Therefore, they may have an advantage over great tits in the application of a foraging strategy involving the selection of larger prey and fewer nest visits.
Habitat gaps, both structural and functional, can increase the costs of rearing young, and may also reduce breeding success. Given these effects on parental DEE of gaps in the park and of oak trees in the wood, it is clear that habitat quality for breeding tits, and other arboreal insectivores, could be improved. In parks and other secondary habitats, and especially where space to increase total habitat area is limited, this could be done by increasing the proportion of native tree species. In woodland, increasing the proportion of oak, and other tree species with rich invertebrate faunas, is one possibility, but would have to be balanced against additional requirements of both the birds and other taxa.
References
Ackermann F (1999) Airborne laser scanning – present status and future expectations. ISPRS J Photogramm Remote Sens 54:64–67
Alexander K, Butler J, Green T (2006) The value of different tree and shrub species to wildlife. Brit Wildl 18:18–28
Andrén H (1992) Corvid density and nest predation in relation to forest fragmentation: a landscape perspective. Ecology 73:794–804
Bradbury RB, Hill RA, Mason DC, Hinsley SA, Wilson JD, Balzter H, Anderson QA, Whittingham MJ, Davenport IJ, Bellamy PE (2005) Modelling relationships between birds and vegetation structure using airborne LiDAR data: a review with case studies from agricultural and woodland environments. Ibis 147:443–452
Broughton RK, Hinsley SA, Bellamy PE, Hill RA, Rothery P (2006) Marsh Tit territory structure in a British broadleaved woodland. Ibis 148:744–752
Bryant DM, Tatner P (1991) Intraspecific variation in avian energy expenditure: correlates and constraints. Ibis 133:236–245
Burke DM, Nol E (1998) Influence of food abundance, nest-site habitat, and forest fragmentation on breeding ovenbirds. Auk 115:96–104
Carleer A, Wolff E (2004) Exploitation of very high resolution satellite data for tree species identification. Photogramm Eng Remote Sens 70:135–140
Cowie RJ, Hinsley SA (1987) Breeding success of blue tits and great tits in suburban gardens. Ardea 75:81–90
Daan S, Masman D, Groenewold A (1990) Avian basal metabolic rates: their association with body composition and energy expenditure in nature. Am J Physiol 259:333–340
Daan S, Deerenberg C, Dijkstra C (1996) Increased daily work precipitates natural death in the kestrel. J Anim Ecol 65:539–544
Desrochers A, Hannon SJ (1997) Gap crossing decisions by forest songbirds during the post-fledging period. Conserv Biol 11:1204–1210
Dhondt AA, Eyckerman R, Moermans R, Hublé J (1984) Habitat and laying date of the Great Tit and Blue Tit (Parus major and Parus caeruleus). Ibis 126:388–397
Dias PC, Blondel J (1996) Breeding time, food supply and fitness components of Blue Tits Parus caeruleus in Mediterranean habitats. Ibis 138:644–649
Ehrlich PR, Wilson EO (1991) Biodiversity studies: science and policy. Science 253:758–762
Eybert MC, Constant P, Lefeuvre JC (1995) Effects of changes in agricultural landscape on a breeding population of linnets Acanthis cannabina L. living in adjacent heathland. Biol Conserv 74:195–202
Fischbacher M, Naef-Daenzer B, Naef-Daenzer L (1998) Estimating caterpillar density on trees by collection of frass droppings. Ardea 86:121–129
Fuller RM, Groom GB, Jones AR (1994) The Land Cover Map of Great Britain: an automated classification of Landsat Thematic Mapper data. Photogramm Eng Remote Sens 60:553–562
Gardiner C, Sparks T (eds) (2005) Ten years of change: woodland research at Monks Wood NNR, 1993–2003. In: Proceedings of the 50th anniversary symposium. English Nature Research Report 613. English Nature, Peterborough
George M (2005) Tree species classification from remote sensing data. Dissertation, University of Leicester
Goldstein DL (1990) Energetics of activity and free living in birds. In: Morrison ML, Ralph CJ, Verner J, Jehl JR (eds) Avian foraging: theory, methodology and applications. studies in avian biology no 13. Cooper Ornithological Society, Los Angeles, pp 423–426
Hammond KA, Diamond J (1997) Maximal sustained energy budgets in humans and animals. Nature 386:457–462
Haywood S, Perrins CM (1992) Is clutch size in birds affected by environmental conditions during growth? Proc R Soc Lond B 249:195–197
Heywood VH (eds) (1995) Global biodiversity assessment. United Nations Environment Programme. Cambridge University Press, Cambridge
Hill RA, Smith GM, Fuller RM, Veitch N (2002) Landscape modelling using integrated airborne multi-spectral and elevation data. Int J Remote Sens 23:2327–2334
Hill RA, Hinsley SA, Gaveau DLA, Bellamy PE (2004) Predicting habitat quality for Great Tits (Parus major) with airborne laser scanning data. Int J Remote Sens 25:4851–4855
Hinsley SA (2000) The costs of multiple patch use by birds. Landsc Ecol 15:765–775
Hinsley SA, Bellamy PE, Moss D (1995) Sparrowhawk Accipiter nisus predation and feeding site selection by tits. Ibis 137:418–420
Hinsley SA, Rothery P, Bellamy PE (1999) Influence of woodland area on breeding success in Great Tits Parus major and Blue Tits Parus caeruleus. J Avian Biol 30:271–281
Hinsley SA, Hill RA, Bellamy PE, Balzter H (2006) The application of LiDAR in woodland bird ecology: climate, canopy structure and habitat quality. Photogramm Eng Remote Sens 72:1399–1406
Keller LF, van Noordwijk AJ (1994) Effects of local environmental conditions on nestling growth in the great tit Parus major L. Ardea 82:349–362
Kennedy CEJ, Southwood TRE (1984) The number of species of insect associated with British trees: a re-analysis. J Anim Ecol 53:455–478
Lambrechts MM, Caro S, Charmantier A, Gross N, Galan M-J, Perret P, Cartan-Son M, Dias PC, Blondel J, Thomas DW (2004) Habitat quality as a predictor of spatial variation in blue tit reproductive performance: a multi-plot analysis in a heterogeneous landscape. Oecologia 141:555–561
Lefsky MA, Cohen WB, Parker GG, Harding DJ (2002) Lidar remote sensing for ecosystem studies. BioScience 52:19–30
Lim K, Treitz P, Wulder M, Stonge B, Flood M (2003) LiDAR remote sensing of forest structure. Prog Phys Geog 27:88–106
Lindén M, Gustafsson L, Pärt T (1992) Selection on fledging mass in the Collared Flycatcher and the Great Tit. Ecology 73:336–343
Lindström Å, Kvist A (1995) Maximum energy intake rate is proportional to basal metabolic rate in passerine birds. Proc R Soc Lond B 261:337–343
Lloyd DG (1987) Selection on offspring size at independence and other size-versus-number strategies. Am Nat 129:800–817
Lõhmus A (2003) Are certain habitats better every year? A review and case study on birds of prey. Ecography 26:545–552
McCollin D (1998) Forest edges and habitat selection in birds: a functional approach. Ecography 21:247–260
Merilä J, Wiggins DA (1997) Mass loss in breeding blue tits – the role of energetic stress. J Anim Ecol 66:452–460
Mickelson JG, Civco DL, Silander JA (1998) Delineating forest canopy species in the northeastern United States using multi-temporal TM imagery. Photogramm Eng Remote Sens 64:891–904
Mills GS, Dunning JB, Bates JM (1989) Effects of urbanization on breeding bird community structure in southwestern desert habitats. Condor 91:416–428
Moreno J, Cowie RJ, Sanz JJ, Williams RSR (1995) Differential response by males and females to brood manipulation in the Pied flycatcher: energy expenditure and nestling diet. J Anim Ecol 64:721–732
Næsett E. (2004). Practical large-scale forest stand inventory using small-footprint airborne scanning laser. Scand J Forest Res 19:164–179
Naef-Daenzer B (2000) Patch time allocation and patch sampling by foraging great and blue tits. Anim Behav 59:989–999
Naef-Daenzer L, Nager RG, Keller LF, Naef-Daenzer B (2004) Are hatching delays a cost or a benefit for Great Tit Parus major parents? Ardea 92:229–237
Nilsson J-Å (2002) Metabolic consequences of hard work. Proc R Soc Lond B 269:1735–1739
Peck KM (1989) Tree species preferences shown by foraging birds in forest plantations in northern England. Biol Conserv 48:41–57
Perrins CM (1979) British tits. Collins, London
Perrins CM (1991). Tits and their caterpillar food supply. Ibis 133(suppl 1):49–54
Reichard SH, Chalker-Scott L, Buchanan S (2001) Interactions among non-native plants and birds. In: Marzluff JM, Bowman R, Donnelly R (eds) Avian ecology and conservation in an urbanizing world. Kluwer Academic Publishers, pp 179–223
Riddington R, Gosler AG (1995) Differences in reproductive success and parental qualities between habitats in the Great Tit Parus major. Ibis 137:371–378
Sanz JJ, Tinbergen JM, Orell M, Rytkönen S (1998) Daily energy expenditure during brood rearing of great tits (Parus major) in Northern Finland. Ardea 86:101–107
Schmidt K-H, Steinbach J (1983) Niedriger Bruterfolg der Kohlmeise (Parus major) in städtischen Parks und Friedhöfen. J Ornithol 124:81–83
Schoech SJ, Bowman R (2001) Variation in the timing of breeding between suburban and wildland Florida scrub jays: do physiologic measures reflect different environments? In: Marzluff JM, Bowman R, Donnelly R, (eds) Avian ecology and conservation in an urbanizing world. Kluwer Academic Publishers, pp 289–306
Slagsvold T, Amundsen T, Dale S (1995) Costs and benefits of hatching asynchrony in blue tits Parus caeruleus. J Anim Ecol 64:563–578
Speakman JR (1997) Doubly labelled water: theory and practice. Chapman and Hall, London
Speakman JR (1998) The history and theory of the doubly labelled water technique. Am J Clin Nutr 68(suppl):S932–S938
Stauss MJ, Burkhardt JF, Tomiuk J (2005) Foraging flight distances as a measure of parental effort in blue tits Parus caeruleus differ with environmental conditions. J Avian Biol 36:47–56
Thomas DW, Blondel J, Perret P, Lambrechts MM, Speakman JR (2001) Energetic and fitness costs of mismatching resource supply and demand in seasonally breeding birds. Science 291:2598–2600
Tinbergen JM, Boerlijst MC (1990) Nestling weight and survival in individual great tits (Parus major). J Anim Ecol 59:1113–1127
Tinbergen JM, Dietz MW (1994) Parental energy expenditure during brood rearing in the Great Tit (Parus major) in relation to body mass, temperature, food availability and clutch size. Funct Ecol 8:563–572
Tinbergen JM, Verhulst S (2000) A fixed energetic ceiling to parental effort in the great tit? J Anim Ecol 69:323–334
Treitz P, Howarth P (2000) Integrating spectral, spatial and terrain variables for forest ecosystem classification. Photogramm Eng Remote Sens 66:305–317
Tremblay I, Thomas DW, Lambrechts MM, Blondel J, Perret P (2003) Variation in blue tit breeding performance across gradients in habitat richness. Ecology 84:3033–3043
Tremblay I, Thomas DW, Blondel J, Perret P, Lambrechts MM (2005) The effect of habitat quality on foraging patterns, provisioning rate and nestling growth in Corsican Blue Tits Parus caeruleus. Ibis 147:17–24
van Balen JH (1973) A comparative study of the breeding ecology of the Great Tit Parus major in different habitats. Ardea 61:1–93
Vane-Wright RI, Humphries CJ, Williams PH (1991) What to protect? Systematics and the agony of choice. Biol Conserv 55:235–254
Verhulst S, Tinbergen JM (1997) Clutch size and parental effort in the great tit Parus major. Ardea 85:111–126
Verhulst S, Tinbergen JM (2001) Variation in food supply, time of breeding, and energy expenditure in birds. Science 294 (Technical Comments) 471a. Full text available from http://www.Sciencemag.org/cgi/content/full/294/5542/471a
Wehr A, Lohr U (1999) Airborne laser scanning–an introduction and overview. ISPRS J Photogramm Remote Sens 54:68–82
Wolter PT, Mladenoff DJ, Host GE, Crow TR (1995) Improved forest classification in the northern lakes states using multi-temporal Landsat imagery. Photogramm Eng Remote Sens 61:1129–1143
Wright J, Both C, Cotton PA, Bryant D (1998) Quality vs. quantity: energetic and nutritional trade-offs in parental provisioning strategies. J Anim Ecol 67:620–634
Zandt HS (1994) A comparison of three sampling techniques to estimate the population size of caterpillars in trees. Oecologia 97:399–406
Acknowledgements
We would like to thank the Esmée Fairbairn Foundation for funding the DLW procedure. Thanks also to Natural England (especially Chris Gardiner), Cardiff City Council, (especially Chris Powell and Jonathan Green), and David Gaveau. The LiDAR data for Monks Wood were supplied by the Environment Agency, and the ATM data by the NERC Airborne Research and Survey Facility (ARSF). The LiDAR data for Bute Park were supplied by the NERC ARSF in conjunction with the Unit for Landscape Modelling at the University of Cambridge. Analysis of all remote sensed data was carried out whilst RAH was based at CEH Monks Wood. The DLW procedure, trapping and storage of samples were carried out under licences issued to SAH: Home Office Licence PPL 80/1756; Natural England: 20030897 & 20040579; Countryside Council for Wales: OTH: SB:03:2003, OTH:SB:02:2004 & OTH:SB:03:2005.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hinsley, S.A., Hill, R.A., Bellamy, P.E. et al. Effects of structural and functional habitat gaps on breeding woodland birds: working harder for less. Landscape Ecol 23, 615–626 (2008). https://doi.org/10.1007/s10980-008-9225-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10980-008-9225-8