Abstract
This paper addresses the issue of whether landscape structure affects A. terrestris population kinetics on a neighbourhood spatial scale, and if so, at what spatial scale is that effect at its maximum. We investigated how the growth of A. terrestris populations is influenced by the landscape context of parcels used for hay production in the French Jura Mountains. Five landscape metrics (relative area of grassland, mean patch area of grassland, patch density of grassland, woodland patch density in grassland, grassland–woodland edge density) were computed over an increasing radius around each parcel (max. 3 km). Redundancy analysis showed that the extent, rate and early onset of A. terrestris population growth were favoured in open grassland areas. Landscape effects on A. terrestris populations as determined by the five metrics are scale-dependent: mean patch area of grassland, patch density of grassland and woodland patch density in grassland had an impact on a grassland parcel within a neighbourhood radius of about 800 m, while relative area of grassland and grassland–woodland edge density had an impact within a neighbourhood radius of about 400 m. Those findings corroborate earlier hypotheses about a multifactorial regulation of A. terrestris populations and a spatial hierarchy of regulating factors. They have potential implications in terms of landscape management and small mammal pest control.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Post-war agricultural policies in temperate Europe led to more intensive farming practices with the less productive and less accessible fields being abandoned while hedgerows were uprooted creating more open landscapes. These changes have caused ecological problems such as environmental pollution and have modified animal and plant population dynamics (Alard and Poudevigne 1997; Roschewitz et al. 2005). The homogenization of landscape has improved conditions for both insect and small-mammal pests by increasing the proportion of source (optimal) habitats for certain species (Robinson and Sutherland 2002). For instance, grassland rodents have benefited from the increase in meadows and pastures in the mid-altitude mountains of Europe.
Rodents can cause major economic loss through crop damage (Teivainen 1979; Singleton et al. 2001) and may transmit diseases to humans (Gratz 1997). Changes in agricultural practices in mountain areas of France (Jura, Massif Central, Alps) since the 1970s have led farmers to specialize in milk production and to convert arable land into permanent grassland. This change in land use has created homogeneous grassland ecosystems with higher connectivity among grassland areas, fewer hedgerows and more open landscapes. This has led to outbreaks of grassland rodents, such as the fossorial water vole (Arvicola terrestris scherman Shaw) whose populations undergo travelling waves on a 6-year cycle with four successive phases: low density, population growth, high density and population decline (Giraudoux et al. 1997). High population densities of this species cause severe crop damage and substantial economic losses (Meylan 1981). They have also been linked to a higher prevalence of human alveolar echinococcosis, a lethal parasitical disease transmitted via a fox–small mammal cycle (Viel et al. 1999). Rodenticides have been used to control A. terrestris populations but they may result in secondary poisoning of non-target wild animals (Brakes and Smith 2005; Berny et al. 1997).
Previous studies have identified a connection between the risk of A. terrestris population outbreaks and the landscape features at different spatial scales. On the regional scale (area of about 2500 km2) larger variations in population densities occur where permanent grassland (optimal habitat for A. terrestris) makes up a large proportion of the agricultural landscape (>85%). This pattern has been termed a ‘landscape composition effect’ (Giraudoux et al. 1997; Fichet-Calvet et al. 2000). On the sectorial scale (area of about 25 km2), Duhamel et al. (2000) report that A. terrestris outbreaks generally occur in the more homogeneous areas of grassland (>51% of the landscape) rather than in areas with many hedgerow networks and woodland patches. This pattern has been termed a ‘landscape structure effect’. However, to our knowledge, no quantitative studies have determined either the effect of landscape on A. terrestris populations on a neighbourhood scale (area of about 0.01 km2) or the scale at which the landscape effect is maximum.
In this paper we will focus on the following questions: (1) can wood and grassland patches affect the population dynamics of Arvicola terrestris? (2) if so, what is the spatial scale at which this effect is maximum?
This study was carried out in Franche-Comté (France) where agricultural management is based on cattle breeding for milk and meat, with large permanent grassland and forest being the dominant landscape features of the area. We concentrated on the growth phase of an A. terrestris population cycle. Our findings are discussed in relation to grassland management practices and their implications for preventing rodent outbreaks.
Methods
Study sites
Parcels were selected in 21 communes (French municipality) of the Doubs and the Jura departments (French administrative divisions), 47.11°N, 6.24°E. The term ‘parcel’ refers to an area of several hectares belonging to a single holding and farmed as a single land-use unit. Farmland in the area is almost exclusively permanent grassland. The Fédération Régionale de Défense contre les Organismes Nuisibles (FREDON, a farmers’ organization for crop protection) surveys the entire area of each commune annually and monitors the travelling wave dynamics of A. terrestris populations for the purpose of outbreak control. Each commune is given an annual score: 0, no colony observed; 1, some isolated colonies; 2, colonies present in many pastures and meadows; 3, numerous colonies and serious damage to grassland. This monitoring provided the temporal framework for the multiannual variations in A. terrestris populations. It meant the population cycle phase could be identified annually for each parcel of each commune. An increasing density phase was studied from 2001 to 2004 in communes which were selected with low-density vole populations (score 0–1) recorded for two years after a high-density phase (score 3), i.e. communes at the onset of a cycle in 2001. Twenty-five parcels (size: 0.5–15 ha) representative of the agricultural management of Franche-Comté where A. terrestris was presumably absent or present at very low densities in spring 2001 were selected in typical landscapes (Delattre et al. 1996). The parcels were also selected as distant as possible from each other (range of the minimum distance between two parcels: 0.8–3.2 km; median: 2.8 km) to limit the risks of spatial autocorrelation (see also the data analysis section below).The geographical coordinates of each parcel were recorded using a GPS (Garmin® PN 12 MAP, PhaseTrac™ receiver, USA).
Survey of A. terrestris populations
A. terrestris abundance in each parcel was estimated by an index method described by Giraudoux et al. (1995). Such index methods, calibrated against density estimates based on trapping, are usually employed for large-scale studies and/or long-term monitoring (Hansson 1979; Delattre et al. 1999; Fichet-Calvet et al. 1999; Giraudoux et al. 1997; Quéré et al. 2000). The relative abundance of A. terrestris was determined along a diagonal line across each parcel. The diagonal was subdivided into 10 m intervals and the strip of land 5 m wide and centred on the transect line was observed (Fig. 1). The presence or absence of A. terrestris was recorded for each interval. The surface indices used were earth mounds and occurrence of A.terrestris holes. The relative abundance of A. terrestris was calculated as the ratio of the number of positive intervals to the total number of intervals (Duhamel et al. 2000; Quéré et al. 2000; Raoul et al. 2001). A. terrestris abundance was estimated for each parcel and for each year from 2001 to 2004 in early spring, when vegetation was at its shortest. Values from 2001 to the year of maximum abundance inclusive were considered in the first stage of the analysis in order to study the growth phase of the A. terrestris cycle. Three variables, Maximum Abundance, Parcel Rise Time, and Commune Offset, were investigated. Maximum Abundance was the maximum density value attained for each parcel; Parcel Rise Time was the time (months) taken to reach the maximum density value from 2001 in each parcel; Commune Offset was the time-lag between the onset of the population increase in each parcel and the onset in the corresponding commune as a whole. The onset of the population growth phase in each commune corresponded to the increase of the initial FREDON spring score.
Method of estimating A. terrestris relative abundance at the parcel scale (adapted from Giraudoux et al. 1995)
Characterization of landscape context
The search for landscape effect on population dynamics of A. terrestris involves the characterization of the landscape structures by means of landscape metrics. The first step in computation of these metrics is to define a land cover map, which can be derived from several data sources. We opted for remotely sensed data at 15 m spatial resolution which allows land cover to be mapped over a very large area (see below). One of the supposed limits of this choice is that narrow hedgerow networks (<15 m) may not be detected in their details. Actually, trees and large bushes alter the reflectance of a 15 m pixel at a larger range than their simple projection on the ground. This means that although some details that could have been provided by costly and time-consuming analysis of aerial photography may have escaped notice, most wooded features of about 10 m resolution can be captured using automated classification of the satellite data used (see below). Automated analysis over large areas with low-cost data is a definite advantage for further development targeted at landscape management and small mammal control, thus this option was used for the present study.
Land cover categories were defined from a set of multiband images and panchromatic bands (Landsat 7 ETM) acquired in September 1999. These data were geometrically corrected by using a polynomial model of spatial interpolation (Jensen 1996) to get a pixel matrix conforming to a Lambert conic projection. A multiband image (RMS error = 14.8 m) was merged with a panchromatic band (RMS error = 9.7 m) using color space transformation (Lillesand and Kiefer 2000) to obtain a multispectral image at 15 m-spatial resolution. Then, a maximum likelihood classifier was applied to obtain an image with four land cover classes: grassland, woodland, bare ground and water. The overall classification accuracy given by the global Kappa coefficient was of 0.95. The image was then used to compute landscape metrics for areas of varying size surrounding each parcel. Numerous metrics are commonly used in landscape ecology (McGarigal and Marks 1995; Gustafson 1998) but it is widely recognized that they provide redundant information and sometimes lead to ambiguous interpretations in terms of ecological processes (Tischendorf 2001; Li and Wu 2004). Starting from the ecological processes to be modelled through the landscape structure, we focused on metrics applied to the ‘grassland’ (the optimal habitat of A. terrestris) and ‘woodland’ (the habitat of the main predators of A. terrestris) categories. A neighbourhood analysis was performed using five landscape metrics computed for a series of circle buffers centred on each parcel. Measurements of landscape structures are sensitive to spatial scale (Withers and Meentemeyer 1999) defined by the grain and the extent of landscape data (Turner et al. 1989). The grain is defined here by the spatial resolution of the classified image that prevents us from considering fine elements of landscape but that is suitable for dealing with major landscape elements like wooded and grassland patches. One can hardly know a priori the neighbourhood size corresponding to the distance over which predators or resources impact vole populations. Consequently, landscape metrics were computed stepwise (75 m steps) by progressively increasing the circle buffer from a first circle of 225 m radius (including each parcel whatever its size), in order to quantify at each step the relationships between landscape metrics and vole population kinetics parameters. This method, recommended by Li and Wu (2004), was used in earlier works (Chust et al. 2000; Wu et al. 2000; Wu 2004). However, increasing the circle buffer may lead to overlap between computation areas, and so artificially increase spatial autocorrelation. To limit this problem while maintaining relevant ranges, the proportion of overlapping areas was estimated for each neighbourhood size. The maximum neighbourhood radius of 3 km was defined by limiting these overlapping areas to 30% of the total buffer. Model residuals were then checked for spatial autocorrelation (see below). The five landscape metrics were:
-
Relative area of grassland (percentage of area occupied by grassland), indicating the relative resource abundance for A. terrestris;
-
Mean patch area of grassland (a patch was defined as the set of adjacent pixels allocated to the same landscape class), representing the spatial continuity of habitat;
-
Patch density of grassland (number of patches per ha), an alternative characterization of the spatial pattern of habitat by measuring its fragmentation;
-
Woodland patch density in grassland (number of patches per ha), corresponding to the fragmentation of woodland relative to grassland areas, thought to represent the spatial ‘scattering’ of predator habitats in environments favoured by A. terrestris;
-
Grassland–woodland edge density, computed in metres per hectare and yielding a theoretical level of interaction between the two categories; this was taken as a measure of the exposure of A. terrestris to its predators.
Data analysis
Linear regressions between A. terrestris population kinetics variables and landscape metrics were computed. Residuals were examined for spatial autocorrelation. An empirical variogram was computed and compared to the envelope obtained from 99 random permutations of the residuals on their point location. No indication of spatial autocorrelation was found within a range of 40 km.
Spearman correlations between A. terrestris variables and landscape metrics were determined for each buffer and plotted against radius. Variables showing a correlation for which the probability of Ho (rs = 0) was less than 0.05 were selected for further analysis (Sokal and Rohlf 1995). For each metric, the choice of radius for further analysis was based on the largest statistically significant correlation between the A. terrestris variables and the corresponding metrics.
Redundancy analysis (RDA) is a direct extension of multiple regression to the modelling of multivariate response data (Legendre and Legendre 1998). It allows an assessment of how an array of response variables (in this case Maximum Abundance, Parcel Rise Time and Commune Offset) correlates with an array of explanatory variables (in this case landscape metrics). RDA was used to investigate the relationship between A. terrestris population kinetics variables (array of response variables) and landscape metrics (array of independent explanatory variables) at the onset of the A. terrestris cycle. The null hypothesis of independence between the two arrays was tested with a permutation test.
Statistical analyses were conducted with R 2.3.1 (R Development Core Team 2004), geoR (Ribeiro and Diggle 2001) and ADE4 (Thioulouse et al. 2004).
Results
Kinetic patterns of A. terrestris populations
Changes in the abundance of A. terrestris were monitored in 25 parcels for 36 months from spring 2001 to spring 2004 (Fig. 2).
Changes in A. terrestris abundance over time (2001–2004) recorded on the 25 study parcels. For each parcel, the analysis of A. terrestris population kinetics takes into account the data from low abundance (2001) to maximum abundance (•). The arrow (↔) represents the time-lag between the onset of the A. terrestris growth phase at the parcel scale compared with the commune scale. The onset of population growth in each commune was based on FREDON spring scores not shown here. For clarity, the time taken to reach maximum abundance is not shown
Correlations between population kinetics of A. terrestris and landscape metrics
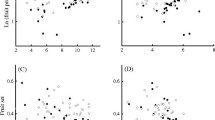
Commune Offset and Parcel Rise Time were negatively correlated with the relative area of grassland, and this effect was maximum for radii of 525 m and 375 m, respectively. Maximum Abundance did not appear to be related to a radius and to the relative area of grassland (Fig. 3a). The neighbourhood radius of 375 m was chosen for further analysis.
Spearman correlations between A. terrestris population kinetics variables and landscape metrics in relation to spatial scale. Landscape metrics were computed in neighbourhood radii progressively increasing from 0 to 3 km (75 m steps): (a) Relative area of grassland; (b) Mean patch area of grassland; (c) Patch density of grassland; (d) Woodland patch density in grassland; (e) Grassland–woodland edge density
Maximum Abundance was positively correlated with the mean patch area of grassland, and this effect was maximum for a radius of 825 m. Parcel Rise Time was negatively correlated with the mean patch area of grassland, for a radius of 350 m only. Commune Offset was negatively correlated with the mean patch area of grassland for almost all the neighbourhood radii (Fig. 3b). The neighbourhood radius of 825 m was chosen for further analysis.
Maximum Abundance was negatively correlated with the patch density of grassland, and this effect was maximum for a radius of 825 m. Commune Offset and Parcel Rise Time were not correlated with this metric (Fig. 3c).
Maximum Abundance was negatively correlated with the woodland patch density in grassland, and this effect was maximum for a radius of 750 m. Commune Offset and Parcel Rise Time did not appear to be related to a radius and to the woodland patch density in grassland (Fig. 3d).
Maximum Abundance was negatively correlated with the grassland–woodland edge density, and this effect was maximum for a radius of 450 m. Commune Offset and Parcel Rise Time were not correlated with this metric (Fig. 3e).
Redundancy analysis and landscape metrics
The relationship between the ‘landscape metrics’ array and the ‘A. terrestris population kinetic variables’ array was found to be globally significant (P < 0.01 after 999 permutations) with three explanatory variables: the relative area of grassland, the mean patch area of grassland and the grassland–woodland edge density. The corresponding scatter diagram shows that the landscape metric variables explained 18.3% and 7.9% of the variance of the canonical model on the first two axes respectively (total 26.9%) (Fig. 4). Maximum Abundances were clearly greater in parcels with larger mean patch areas of grassland. Furthermore, the parcels where abundance of A. terrestris populations peaked earlier were those with larger relative areas of grassland. Finally, the parcels for which the increase of A. terrestris populations began later in a commune were those with smaller relative areas of grassland and with larger grassland–woodland edge densities.
Redundancy analysis (RDA) biplot of axes 1 and 2 of RDA for the three variables describing A. terrestris population growth kinetics (Maximum Abundance, Parcel Rise Time and Commune Offset), constrained by three landscape variables (italicized labels). RAG 375: Relative area of grassland for a neighbourhood radius of 375 m; MPG 825: Mean patch area of grassland for a neighbourhood radius of 825 m; GWD 450: Grassland–woodland edge density for a neighbourhood radius of 450 m. Explained variation: axis 1: 18.3%, axis 2: 7.9%. The Monte Carlo permutation test was significant for the overall regression model (P < 0.01). Dots are sample parcels. Distances of parcels in the biplot are approximations of their Euclidean distances. Projections of parcels at right angles on the response variables (Maximum Abundance, Parcel Rise Time and Commune Offset) approximate the value of the parcels along Maximum Abundance, Parcel Rise Time and Commune Offset arrows. The angles between response and explanatory variables in the biplot reflect their correlations, i.e. the smaller the angle, the closer the correlation
Discussion
Kinetic patterns of A. terrestris populations
In the study area, the A. terrestris population cycle generally lasts six years. It includes four successive phases: low density, growth, high density and decline (Giraudoux et al. 1997). Our analysis of A. terrestris population kinetics took into account the relative abundances of A. terrestris (from low density to the maximum value) for each of the 25 parcels. We are therefore confident that our study period coincided with the growth phase of an A. terrestris population cycle. A. terrestris relative abundances were recorded each year in early spring from 2001 to 2004. Winter weather conditions led to a decrease in reproduction and survival. Short grass cover in early spring allowed a reliable estimation of A. terrestris abundance indices.
Choice of landscape metrics
We selected indicators for the landscape context of the sample agricultural parcels making up the habitat of A. terrestris populations. The studied metrics thus reflected the landscape composition (relative area of grassland) and the landscape structure (mean patch area of grassland, patch density of grassland, woodland patch density in grassland, grassland–woodland edge density); resource availability (relative area of grassland, mean patch area of grassland, patch density of grassland) and the risk of predation for A. terrestris (woodland patch density in grassland, grassland–woodland edge density).
Influence of landscape context on A. terrestris populations
Analysis of the relationships between A. terrestris population growth and landscape metrics showed that landscape composition and structure could markedly affect A. terrestris population kinetics on the scale of a parcel (size: 0.5–15 ha).
The maximum values of A. terrestris abundance were reached in grassland areas which were not greatly fragmented suggesting that the intensity of A. terrestris population growth is favoured by an open landscape. Blant et al. (2004) already observed that A. terrestris population cycles reach higher densities in lower parts of poorly wooded valleys of the Swiss Jura High Chain. Moreover, our study indicates a negative impact of woodland patch density in grassland and grassland–woodland edge density on vole population growth. Following Anderson and Erlinge (1977), Delattre et al. (1999) in studies on Microtus arvalis populations, consider that homogenous landscapes (large open meadows) are refuge habitats for specialist predators (stoat, weasel), which are known to destabilize prey populations increasing both the amplitude of fluctuations and the duration of the high density phase. Conversely, heterogeneous landscapes (hedgerow networks) are dominated by generalist predators (fox, bird of prey) and act as regulating factors dampening vole population kinetics and shortening the phase of peak numbers. Moreover, residual A. terrestris populations are found in forest enclaves at low density. Although specific studies of predators could not be conducted in our study, it is remarkable that the landscape variables correlating with small mammal population dynamic characteristics were the same as those mentioned by earlier authors considering the predation hypothesis.
Our study indicates that A. terrestris abundance peaked earlier in areas dominated by grassland. These findings are consistent with results obtained by Giraudoux et al. (1997) on a regional scale. On that scale, the diffusion speed of A. terrestris populations was shown to be controlled by the proportion of Permanent Grassland (PG) in the Agricultural Landscape (AL), i.e. the higher PG/AL, the more likely vole population outbreaks are. Furthermore, the rate of A. terrestris population growth also was slowed by a grassland–woodland mixed (heterogeneous) landscape.
We found that the growth of A. terrestris populations occurred earlier in areas dominated by grassland and that were little fragmented. These results are consistent with those of Duhamel et al. (2000) on a sectorial scale (several tens of km2). Those authors showed that communes of the Doubs department where A. terrestris outbreaks begin are dominated by open landscapes, while communes where A. terrestris outbreaks spread later typically have a greater percentage of wooded habitats.
Our study has also shown for the first time that landscape features do not impact populations on the same range (e.g. 750 m for woodland patch density in grassland and Maximum Abundance versus 375 m for relative area of grassland and Parcel Rise Time). Although the impact of landscape features may be scale-invariant (from regional to more local, open and homogeneous grassland favours vole population outbreaks) the finest grain for a landscape effect on vole populations effect is 0.3–1 km radius.
Conclusions
A stream of converging data indicates that, in complex ecosystems, the population dynamics of small mammals is regulated by both top-down (predation, parasitism) and bottom-up (ressources) forces in a multivariate context (Lidicker 2000; Lindström et al. 2001; Korpimäki et al. 2004). The results of both monovariate and multivariate approaches showed that the overall configuration of several landscape variables best explains the demographic response of A. terrestris populations. This emphasizes the importance of taking into account multiple scales in a systems approach in studies implying community processes. This is particularly important in temperate ecosystems, which are more complex than the relatively simple arctic and boreal ones, and where population cycles may be caused by the combined action of a hierarchy of many regulating factors acting both spatially and temporally (Lidicker 1995, 2000; Hansson 2002; Hudson and Bjørnstad 2003). Although cross-scale studies have proved essential in understanding the population dynamics of most living species, few empirical studies have actually been conducted at multiple scales and over the long term addressing landscape issues and placing them in a spatial and temporal context. Among a chain of studies carried out across space–time scales on grassland small mammal population dynamics, the current study addresses the local effects of landscape in the inclusive context of temporally and spatially explicit events (the phase of a small mammal cycle and its geographical extent). Our results are consistent with those of our previous studies carried out at regional and sectorial scales (Delattre et al. 1992; Giraudoux et al. 1997; Fichet-Calvet et al. 2000; Delattre et al. 1996, 1999; Duhamel et al. 2000). Furthermore, this study is the first one to quantify the effect of landscape on A. terrestris population dynamics at such a local scale and the minimal area (areas of some tens of ha) at which such effects can be observed (Table 1). This corroborates the thesis that the population dynamics of Arvicola terrestris is the result of a spatial hierarchy of regulating factors expressed through various space–time scale (regional, sectorial and local) and landscape metrics. Our study has shown that locally, the extent, rate and early onset of the kinetics of A. terrestris population growth are favoured in open grassland areas. The moderate amount of variation (30%) explained by multivariate analysis here indicates that other unexplained factors play a role in the regulation of A. terrestris populations, corroborating Lidicker’s multifactorial hypothesis about small mammal population regulation (Lidicker 2000).
A. terrestris is an agricultural pest in temperate Europe. Chemical rodenticides currently used to prevent extensive crop damage are also hazards to non-target animals (Brakes and Smith 2005). Farmers must adopt a more diversified approach towards pest control to reduce dependence on chemicals and their impact on non target species. Possible options include landscape manipulation at various spatial extents including the very local scale of the agricultural parcel and its surroundings. This study shows that such landscape management should be designed considering a minimal area of 10–100 ha. This, with results of previous studies, paves the way to making multi-scale models to design alternative landscapes reducing the risk of A. terrestris population outbreaks. Practically, with regard to A. terrestris control, at local scale, our specific recommendations are (i) to reduce the size of open grassland areas (by preserving existing hedgerows and replanting where needed) (ii) to monitor as a priority parcels that may be considered at risk on the landscape criteria provided here (e.g open grassland areas, those which are distant from woodland); (iii) to target prevention and control operations (moderating fertilization; increasing vole and mole gallery disturbance by ploughing and grazing/cattle trampling; trapping and/or chemical control) in the parcels ‘at risk’ as early as possible in the cycle phase (specifically, during the low density phase). However, to be more efficient and considering ecological forces (predation, dispersal sink, etc.) that are implied in small mammal regulation, this approach at local scale should also be completed by supplementary actions directed at the inclusive contexts (e.g. landscape and practices on areas that are much larger than a farm holding). Actually, they must integrate the consequences of decisions taken by a broad array of land owners and stakeholders and this implies that a consistent policy should be designed explicitly considering the multi-scale and multiple usage of landscape in areas where controlling small mammal population outbreaks is an issue.
References
Alard D, Poudevigne I (1997) Les facteurs de contrôle de la biodiversité dans un paysage rural: une approche agro-écologique. Ecologie 28:337–350
Anderson M, Erlinge S (1977) Influence of predation on rodent populations. Oikos 29:591–597
Berny PJ, Buronfosse T, Buronfosse F, Lamarque F, Lorgue G (1997) Field evidence of secondary poisoning of foxes (Vulpes vulpes) and buzzards (Buteo buteo) by bromadiolone, a 4-year survey. Chemosphere 35:1817–1829
Blant M, Beuret B, Ducommun A, Joseph E, Meyrat-Paratte MA, Poitry R, Lehmann A (2004) Le paysage de la Haute Chaîne Jurassienne Suisse influence-t-il les pullulations cycliques du campagnol terrestre Arvicola terrestris sherman (Shaw, 1801)? Bulletin de la Société Neuchâteloise des Sciences Naturelles 127:103–115
Brakes CR, Smith RH (2005) Exposure of non-target small mammals to rodenticides: short-term effects, recovery and implications for secondary poisoning. J Appl Ecol 42:118–128
Chust G, Lek S, Deharveng L, Ventura D, Ducrot D, Pretus J (2000) The effects of the landscape pattern on arthropod assemblages: an analysis of scale-dependence using satellite data. Belg J Entomol 2:99–110
Delattre P, Giraudoux P, Baudry J, Truchetet D, Musard P, Toussaint M, Stahl P, Poule ML, Artois M, Damange JP, Quere JP (1992) Land use patterns and types of common vole (Microtus arvalis) population kinetics. Agric Ecosyst Environ 39:153–169
Delattre P, Giraudoux P, Baudry J, Quere JP, Fichet E (1996) Effect of landscape structure on Common Vole (Microtus arvalis) distribution and abundance at several space scales. Landsc Ecol 11:279–288
Delattre P, De Sousa B, Fichet E, Quéré JP, Giraudoux P (1999) Vole outbreaks in a landscape context: evidence from a six year study of Microtus arvalis. Landsc Ecol 14:401–412
Delattre P, Clarac R, Melis JP, Pleydell DRJ, Giraudoux P (2006). How moles contribute to colonization success of water voles in grassland: implications for control. J Appl Ecol 43:353–359
Duhamel R, Quéré JP, Delattre P, Giraudoux P (2000) Landscape effects on the population dynamics of the fossorial form of the water vole (Arvicola terrestris sherman). Landsc Ecol 15:89–98
Fichet-Calvet E, Jomaa I, Giraudoux P, Ashford RW (1999) Estimation of fat sand rat Psammomys obesus abundance by using surface indices. Acta theriologica 44:353–352
Fichet-Calvet E, Pradier B, Quéré JP, Giraudoux P, Delattre P (2000) Landscape composition and vole outbreaks: evidence from an eight year study of Arvicola terrestris scherman. Ecography 23:659–668
Giraudoux P, Pradier B, Delattre P, Deblay S, Salvi D, Defaut R (1995) Estimation of water vole abundance by using surface indices. Acta theriologica 40:77–96
Giraudoux P, Delattre P, Habert M, Quere JP, Deblay S, Defaut R, Duhamel R, Moissenet MF, Salvi D, Truchetet D (1997) Population dynamics of fossorial water vole (Arvicola terrestris scherman): a land usage and landscape perpective. Agric Ecosyst Environ 66:47–60
Gratz NG (1997) The burden of rodent-borne diseases in Africa south of the Sahara. Belg J Zool 127:71–84
Gustafson EJ (1998) Quantifying landscape spatial pattern: what is the state of the art? Ecosystems 1:143–156
Hansson L (1979) Field signs as indicators of vole abundance. J Appl Ecol 16:339–347
Hansson L (2002) Cycles and travelling waves in rodent dynamics: a comparison. Acta Theriologica 47(Suppl. 1):9–22
Hudson PJ, Bjørnstad ON (2003) Vole stranglers and lemming cycles. Science 302:797–798
Jensen JR (1996) Introductory digital image processing, a remote sensing perspective. Prentice Hall, Upper Saddle River, USA
Korpimäki E, Brown PR, Jacob J, Pech R (2004) The puzzles of population cycles and outbreaks of small mammals solved? Bioscience 54:1071–1079
Legendre P, Legendre L (1998) Numerical ecology. Elsevier, Amsterdam
Li H, Wu J (2004) Use and misuse of landscape indices. Landsc Ecol 19:389–399
Lidicker WZJ (1995) The landscape concept: something old, something new. In: Lidicker WZJ (ed) Landscape approaches in mammalian ecology and conservation. University of Minnesota Press, pp 3–19
Lidicker WZJ (2000) A food web/landscape interaction model for microtine rodent density cycles. Oikos 91:435–445
Lillesand TM, Kiefer RW (2000) Remote sensing and image interpretation. Wiley, New York
Lindström J, Ranta E, Kokko H, Lundberg P, Kaitala V (2001) From arctic lemmings to adaptative dynamics: Charles Elton’s legacy in population ecology. Biol Rev Camb Philos Soc 76:129–158
Meylan A (1981) Bilan de quelques années de recherches fondamentales et appliquées sur le Campagnol terrestre, Arvicola terrestris scherman (Shaw). La Défense des Végétaux 208:143–154
McGarigal K, Marks B (1995) Fragstat: spatial pattern analysis program for quantifying landscape structure. Ge. Technical Report PNW-GTR-351, Portland, http://www.umass.edu/landeco/research/fragstats/fragstats.html
Morilhat C, Bernard N, Bournais C, Meyer C, Lamboley C, Giraudoux P (2007) Responses of Arvicola terrestris scherman populations to agricultural practices, and to Talpa europaea abundance in Eastern France. Agric Ecosyst Environ 122:392–398
Quéré JP, Raoul F, Giraudoux P, Delattre P (2000) An index method applicable at landscape scale to estimate relative population densities of the common vole (Microtus arvalis). Revue d’Écologie, Terre et Vie 55:25–32
Raoul F, Defaut R, Michelat D, Montadert M, Pépin D, Quéré JP, Tissot B, Delattre P, Giraudoux P (2001) Landscape effects on the populations dynamics of small mammal communities and prey-resource variations: a preliminary analysis. Revue d’Écologie, Terre et Vie 56:339–352
R Development Core Team (2004) R: A language and environment for statistical computing. R foundation for statistical computing, Vienna, Austria. ISBN 3–900051-07-0, URL http://www.R-project.org
Ribeiro PJ, Diggle PJ (2001) geoR: a package for geostatistical analysis, R. News 1(2):15–18
Robinson RA, Sutherland WJ (2002) Post-war changes in arable farming and biodiversity in Great Britain. J Appl Ecol 39:157–176
Roschewitz I, Thies C, Tscharntke T (2005) Are landscape complexity and farm specialisation related to land-use intensity of annual crop fields? Agric Ecosyst Environ 105:87–99
Singleton GR, Krebs CJ, Davies S, Chambers L, Brown PR (2001) Reproductive changes in fluctuating house mouse populations in southeastern Australia. Proc Roy Soc Lond 268:1741–1748
Sokal RR, Rohlf FJ (1995) Biometry: the principles and practice of statistics in biological research. Freeman WH & Co., New York, USA
Teivanen T (1979) Vole damage to forest seedlings in reforested areas and fields in Finland in the years 1973–76. Folia forestalia 387:1–23
Thioulouse J, Dufour AB, Chessel D (2004) Ade4: Analysis of environmental data: exploratory and euclidean methods in environmental sciences. R package version 1.3-3 http://pbil.univ-lyon1.fr/ADE-4
Tischendorf L (2001) Can landscape indices predict ecological processes consistently? Landsc Ecol 16:235–254
Turner MG, Dale VH, Gardner RH (1989) Predicting across scales: theory development and testing. Landsc Ecol 3:245–252
Viel JF, Giraudoux P, Bresson-Hadni S, Abrial V (1999) Water vole (Arvicola terrestris scherman) density as risk factor for human alveolar echinococcosis. Am J Trop Med Hyg 61(4):559–565
Withers MA, Meetenmeyer V (1999) Concepts of scale in landscape ecology. In: Klopatek JM, Gardner RH (eds) Landscape ecological analysis, issues and applications. Springer-Verlag, New York, pp 205–252
Wu J (2004) Effects of changing scale on landscape pattern analysis: scaling relations. Landsc Ecol 19:125–138
Wu J, Jelinski DE, Luck M, Tueller PT (2000) Multiscale analysis of landscape heterogeneity: scale variance and pattern metrics. Geogr Inf Sci 6:6–19
Acknowledgements
We thank the Service Régional de la Protection des Végétaux (SRPV) and the Fédération Régionale de Défense contre les Organismes Nuisibles (FREDON) of Franche-Comté for providing data and for helpful field information. We are also grateful to the farmers who allowed access to their parcels and provided information on their land-use practices. Financial support was provided by the Région of Franche-Comté (‘Plan d’Action Régional 2000–2006’). This study is part of a work undertaken with the support of pole 4 of the Maison des Sciences de l’Homme Claude Nicolas Ledoux (UMS 2913 CNRS).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Morilhat, C., Bernard, N., Foltête, JC. et al. Neighbourhood landscape effect on population kinetics of the fossorial water vole (Arvicola terrestris scherman). Landscape Ecol 23, 569–579 (2008). https://doi.org/10.1007/s10980-008-9216-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10980-008-9216-9