Abstract
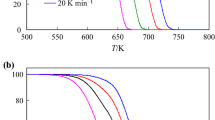
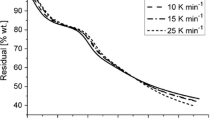
The non-isothermal decomposition process of the powder sample of palladium acetylacetonate [Pd(acac)2] was investigated by thermogravimetric (TG) and the X-ray diffraction (XRD) techniques. Model-free isoconversional method of Tang, applied to the investigated decomposition process, yield practically constant apparent activation energy in the range of 0.05≤α≤0.95. It was established, that the Coats-Redfern (CR) method gives several statistically equivalent reaction models, but only for the phase-boundary reaction models (R2 and R3), the calculated value of the apparent activation energy (E) is nearest to the values of E obtained by the Tang’s and Kissinger’s methods.
The apparent activation energy value obtained by the IKP method (132.4 kJ mol−1) displays a good agreement with the value of E obtained using the model-free analysis (130.3 kJ mol−1). The artificial isokinetic relationship (aIKR) was used for the numerical reconstruction of the experimental integral model function, g(α). It was established that the numerically reconstructed experimental function follows R3 reaction model in the range of α, taken from model-free analysis. Generally, decomposition process of Pd(acac)2 starts with initial nucleation which was characterized by rapid onset of an acceleratory reaction without presence of induction period.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
J. Halpern, Precious Met., 19 (1995) 411.
S. S. Stahl, Science, 309 (2005) 1824.
R. F. Heck, Palladium Reagents in Organic Synthesis, Academic Press, New York 1985.
R. Singh, M. S. Viciu, N. Kramareva, O. Navarro and S. P. Nolan, Org. Lett., 7 (2005) 1829.
J. Terao, A. Oda and N. Kambe, Org. Lett., 6 (2004) 3341.
B. M. Trost and J. Xu, J. Am. Chem. Soc., 127 (2005) 17180.
R. T. Jacobsen, Chem. Eng. Prog., 101 (2005) 20.
C. G. Anderson and S. M. Nordwick, Precious Met., 19 (1995) 123.
M. Aghamohammadi and N. Alizadeh, Anal. Chim. Acta, 480 (2003) 299.
J. Wang and K. Varughese, Anal. Chim. Acta, 199 (1987) 185.
G. Raber, K. Kalcher, C. G. Neuhold, C. Talaber and G. Kolbl, Electroanalysis, 7 (1995) 138.
S. Poston and A. Reisman, J. Electr. Mater., 18 (1989) 553.
S. Vyazovkin and C. A. Wight, Thermochim. Acta, 340–341 (1999) 53.
J. H. Flynn, Thermochim. Acta, 300 (1997) 83.
A. K. Galwey and M. E. Brown, Thermal Decomposition of Ionic Solids, Elsevier, Amsterdam 1999.
S. Vyazovkin and C. A. Wight, Annu. Rev. Phys. Chem., 48 (1997) 125.
W. J. Tang, Y. W. Liu, H. Zhang and C. X. Wang, Thermochim. Acta, 408 (2003) 39.
H. E. Kissinger, Anal. Chem., 29 (1957) 1702.
P. Budrugeac and E. Segal, J. Therm. Anal. Cal., 88 (2007) 703.
S. Vyazovkin and C. A. Wight, J. Phys. Chem. A, 101 (1997) 5653.
A. Khawam and D. R. Flanagan, J. Phys. Chem. B, 110 (2006) 17315.
A. Khawam and D.R. Flanagan, J. Pharm. Sci., 95 (2006) 472.
A. W. Coats and J. P. Redfern, Nature, 201 (1964) 68.
A. K. Galwey, Thermochim. Acta, 399 (2003) 1.
P. Budrugeac and E. Segal, Int. J. Chem. Kinet., 30 (1998) 673.
P. Budrugeac, C. Popescu and E. Segal, J. Therm. Anal. Cal., 64 (2001) 821.
P. Budrugeac, J. Therm. Anal. Cal., 89 (2007) 143.
S. Vyazovkin, Int. J. Chem. Kinet., 28 (1996) 95.
D. Dollimore, T. A. Evans, Y. F. Lee and F. W. Wilburn, Thermochim. Acta, 188 (1991) 77.
D. Dollimore, T. A. Evans, Y. F. Lee, G. P. Pee and F. W. Wilburn, Thermochim. Acta, 196 (1992) 255.
Y. F. Lee and D. Dollimore, Thermochim. Acta, 323 (1998) 75.
C. Popescu, E. Segal and C. Oprea, J. Therm. Anal. Cal., 38 (1992) 929.
N. L. Johnson and F. C. Leone, Statistics and Experimental Design in Engineering and the Physical Sciences, Wiley and Sons, New York 1977, Vol. I.
D. L. Massart, B. G. M. Vandeginste, L. M. C. Buydens, S. de Jong, P. J. Lewi and J. Smeyers-Verbke, Handbook of Chemometrics and Qualimetrics, Part A, Elsevier, Amsterdam 1997.
S. Vyazovkin and W. Linert, Chem. Phys., 193 (1995) 109.
S. Vyazovkin and A. I. Lesnikovich, Thermochim. Acta, 128 (1988) 297.
P. P. Semyannikov, V. M. Grankin, I. K. Igumenov and A. F. Bykov, J. Phys., 4 (1995) 205.
A. G. Nasibulin, P. P. Ahonen, O. Richard, E. I. Kauppinen and I. S. Altman, J. Nanopart. Res., 3 (2001) 385.
A. G. Nasibulin, I. S. Altman and E. I. Kauppinen, Chem. Phys. Lett., 367 (2003) 771.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Janković, B., Mentus, S. Model-fitting and model-free analysis of thermal decomposition of palladium acetylacetonate [Pd(acac)2]. J Therm Anal Calorim 94, 395–403 (2008). https://doi.org/10.1007/s10973-008-9118-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-008-9118-8