Abstract
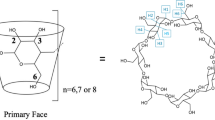
Density and heat capacity of the water+cyclodextrin (CD), water+nicotinic acid (NA) and water+CD+NA mixtures were determined at 298.15 K. CDs with different cavity size and alkylation were selected. From the experimental data the apparent molar properties were calculated. Assuming the formation of inclusion complexes of 1:1 stoichiometry, these properties were modeled and provided the stability constants of CD/NA inclusion complexes and the corresponding property change. The binding of NA with the smallest sized α-cyclodextrin (α-CD) generates more stable complexes accompanied by the lower volume and the heat capacity changes. These results are in agreement with earlier proposed binding mode according to which deep insertion of NA into α-CD takes place and it is governed by the hydrophobic-hydrophilic forces. The volume and the heat capacity changes caused by the interactions of CDs with NA were interpreted in terms of cosphere overlap model and the release of water molecules from the CD cavity due to the NA incorporation.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
T. Loftsson and N. E. Brewster, J. Pharm. Sci., 85 (1996) 1017.
R. A. Hedges, Chem. Rev., 98 (1998) 2035.
T. Kimura, M. Fujisawa, Y. Nakano, T. Kamiyama, T. Otsu, M. Maeda and S. Takagi J. Therm. Anal. Cal., 90 (2007) 581.
X. Lu and Y. J. Chen, Chromatogr. A, 955 (2002) 133.
Cs. Novák, Z. Éhen, M. Fodor, L. Jicsinszky and J. Orgoványi, J. Therm. Anal. Cal., 84 (2006) 693.
C. Baudin, C. Pean, B. Perly and P. Goselin, Int. J. Environ. Anal. Chem., 77 (2000) 233.
R. Kumar, J. S. Dahiya, D. Singh and P. Nigam, Bioresour. Technol., 28 (2001) 209.
R. Koukiekolo, V. Desseaux, Y. Moreau, M. G. Marchis and M. Santimone, Eur. J. Biol. Chem., 268 (2001) 841.
M. L. Bender and M. Komiyama, Cyclodextrin Chemistry, Springer Verlag, Berlin 1978.
J. Szejtli, Chem. Rev., 98 (1998) 1743.
E. M. Martin Del Valle, Process Biochem., 39 (2004) 1033.
L. Liu and Q.-X. Guo, J. Inclus. Phen. Macrocycl. Chem., 42 (2002) 1.
H. Aki, Y. Nakashima, Y. Kawasaki and T. Niiya, J. Therm. Anal. Cal., 85 (2006) 685.
K. Maturi and K. R. Reddy, Chemosphere, 63 (2006) 1022.
R. De Lisi, G. Lazzara, S. Milioto and N. Muratore, Chemosphere, 69 (2007) 1703.
I. V. Terekhova, N. A. Obukhova, R. S. Kumeev and G. A. Alper, Russ. J. Phys. Chem., 79 (2005) 2215.
I. V. Terekhova and N. A. Obukhova, J. Solution Chem., 36 (2007) 1167.
I. V. Terekhova and G. K. E. Scriba, J. Pharm. Biomed. Anal., 45 (2007) 688.
W. Zielenkiewicz, I. V. Terekhova, A. Marcinowicz, M. Koźbial and J. Poznanski, J. Therm. Anal. Cal., (2007) in press, DOI: 10.1007/s 10973-006-7974-7.
L. D. Wilson and R. E. Verrall, J. Phys. Chem. B, 104 (2000) 1880.
L. D. Wilson and R. E. Verrall, J. Phys. Chem. B, 101 (1997) 9270.
K. Spildo and H. Høiland, J. Solution Chem., 31 (2002) 149.
N. S. Isaacs and D. J. Young, Tetrahedron Lett., 40 (1999) 3953.
R. De Lisi, G. Lazzara, S. Milioto and N. Muratore, J. Phys. Chem. B, 107 (2003) 13150.
R. De Lisi, S. Milioto, A. De Giacomo and A. Inglese, Langmuir, 15 (1999) 5014.
R. De Lisi, S. Milioto, A. Pellerito and A. Inglese, Langmuir, 14 (1998) 6045.
R. De Lisi, G. Lazzara, S. Milioto and N. Muratore, Phys. Chem. Chem. Phys., 5 (2003) 5084.
F. J. Millero, Chem. Rev., 71 (1971) 147.
H. Høiland, Thermodynamic Data for Biochemistry and Biotechnology, H.-J. Hinz, Ed., Springer Verlag, New York 1986, p. 17.
J. E. Desnoyers, G. Perron and A. H. Roux, Surfactant Solutions. New Methods of Investigation, R. Zana, Ed., Marcel Dekker, New York 1987, pp. 2–51.
J. Lesley and J. Bullock, Chem. Soc. Faraday Trans., 78 (1982) 1177.
R. F. Evans, E. F. G. Herington and W. Kynaston, Trans. Faraday Soc., 49 (1953) 1284.
J. L. Fortier, P. A. Leduc and J. E. Desnoyers, J. Solution Chem., 3 (1974) 323.
T. F. Young and M. B. Smith, J. Phys. Chem., 58 (1954) 716.
H. L. Friedman and C. V. Krishnan, Water, A Comprehensive Treatise, Vol. 3, Ch. 1, F. Franks, Ed., Plenum Press, New York 1973.
P. Gianni and L. Lepori, J. Solution Chem., 29 (2000) 405.
K. A. Connors and J. M. Lipari, J. Pharm. Sci., 65 (1976) 379.
S. Simova and H.-J. Schneider, J. Chem. Soc. Perkin Trans., 2 (2000) 1717.
T. Stalin and N. Rajendiran, Chem. Phys., 322 (2006) 311.
P. K. Banipal, T. S. Banipal, J. C. Ahluwalia and B. S. Lark, J. Chem. Thermodyn., 34 (2002) 1825.
J. P. Morel, C. Lhermet and N. Morel-Desrosiers, Can. J. Chem., 64 (1986) 996.
R. Jasra and J. C. Ahluwalia, J. Solution Chem., 11 (1982) 325.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Terekhova, I.V., De Lisi, R., Lazzara, G. et al. Volume and heat capacity studies to evidence interactions between cyclodextrins and nicotinic acid in water. J Therm Anal Calorim 92, 285–290 (2008). https://doi.org/10.1007/s10973-007-8842-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-007-8842-9