Abstract
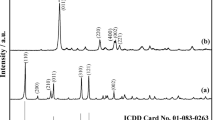
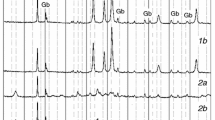
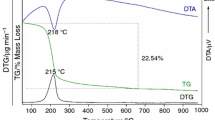
The thermal decomposition of aluminum phosphate monohydrate AlPO4·H2O-H4 was investigated in air using TG-DTG/DTA. The AlPO4·H2O-H4 decomposes in one step and final decomposition product (AlPO4) was studied by X-ray powder diffraction, FTIR and FT-Raman spectroscopy. The activation energies of dehydration reaction of AlPO4·H2O-H4 were calculated through the isoconversional methods of Ozawa and Kissinger-Akahira-Sunose (KAS), and the possible conversion functions have been estimated through the comparative methods. The activation energy calculated for the decomposition of AlPO4·H2O-H4 by different methods and techniques were found to be consistent. The kinetic model that better described the reaction of dehydration for AlPO4·H2O-H4 was the Fn model as simple n-order reaction and the corresponding function is f(α)=(1-α)2.75 and g(α)=-[1-(1-a)-1.75/(1.75)].
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
J. Hong, G. Guo and K. Zhang, J. Anal. Appl. Pyrolysis, 2 (2006) 111.
V. Logvinenko, J. Therm. Anal. Cal., 60 (2000) 9.
S. Vyazovkin, Anal. Chem., 74 (2002) 2749.
F. M. Bautista, J. M. Campelo, A. García, D. Luna, J. M. Marinas, R. A. Quirós and A. A. Romero, Appl. Catal. A., 243 (2003) 93, and references therein.
D. M. Poojary, K. J. Balkus, S. J. Riley, B. E. Gnade and A. Clearfield, Micropor. Mater., 2 (1994) 245 and references therein.
F. D’Yvoire, Bull. Soc. Chim. Fr., (1961) 1762.
K. Kunii, K. Narahara and S. Yamanaka, Macropor. Mesopor. Mater., 52 (2000) 159.
S. T. Wilsonm B. M. Lok, C. A. Messina, T. R. Cannan and E. M. Flanigen, J. Am. Chem. Soc., 104 (1982) 1146.
N. Venkatathri, Bull. Mater. Sci., 26 (2003) 279.
D. Stojakovic, N. Rajic, S. Sajic, N. Z. Logas and V. Kaucic, J. Therm. Anal. Cal., 87 (2007) 339.
T. Ozawa, Bull. Chem. Soc. Jpn., 38 (1965) 1881.
H. E. Kissinger, J. Anal. Chem., 29 (1957) 1702.
K. J. Balkus Jr., L. J. Sottile, S. J., Riley and B. E. Gnade, Thin Solid Films, 260 (1995) 4.
B. D. Cullity, Elements of X-ray Diffraction, 2nd Ed., Addison-Wesley Publishing, 1977.
G. K. Williamson and W. H. Hall, Acta Metall., (January) (1953) 22.
M. Zakeri, R. Yazdani-Rad, M. H. Enayati and M. R. Rahimipour, J. Alloys Compd., 403 (2005) 258.
P. Tarte, Spectrochim. Acta, 23A (1967) 2127.
R. L. Frost and M. L. Weier, J. Mol. Struct., 697 (2004) 207.
D. K. Breitinger, J. Mohr, D. Colognesi, S. F. Parker, H. Schukow and R. G. Schwab, J. Mol. Struct., 563-564 (2001) 377.
R. L. Frost and M. L. Weier, W. N. Martens, D. A. Henry and S. J. Mills, Spectrochim. Acta, 62A (2005) 181 and references therein.
T. V. Lyubomir, M. M. Nikolova and G. G. Gospodinov, J. Solid State Chem., 177 (2004) 2663.
S. Vyazovkin, Int. J. Chem. Kinet., 28 (1996) 95.
S. Vyazovkin, Thermochim. Acta, 355 (1996) 155.
K. Zhang, J. Hong, G. Cao, D. Zhan, Y. Tao and C. Cong, Thermochim. Acta, 437 (2005) 145.
A. W. Coats and J. P. Redfern, Nature, 20 (1964) 68.
B. N. Achar, Proc. Int. Clay Conf., 1 (1966) 67.
X. Gao and D. Dollimore, Thermochim. Acta, 215 (1993) 47.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Boonchom, B., Youngme, S., Srithanratana, T. et al. Synthesis of AlPO4 and kinetics of thermal decomposition of AlPO4·H2O-H4 precursor. J Therm Anal Calorim 91, 511–516 (2008). https://doi.org/10.1007/s10973-007-8420-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-007-8420-1