Abstract
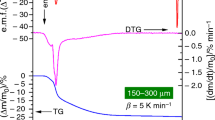
The paper presents a non-isothermal kinetic study of the decomposition of Zn acetate-based gel precursors for ZnO thin films, based on the thermogravimetric (TG) data. The evaluation of the dependence of the activation energy (E) on the mass loss (Δm) using the isoconversional methods (Friedman (FR), Flynn-Wall-Ozawa (FWO) and Kissinger-Akahira-Sunose (KAS)) has been presented in a previous paper. It was obtained that the sample dried at 125°C for 8 h exhibits the activation energy independent on the heating rate for the second decomposition step. In this paper the invariant kinetic parameter (IKP) method is used for evaluating the invariant activation parameters, which were used for numerically evaluation of the function of conversion. The value of the invariant activation energy is in a good agreement with those determined by isoconversional methods. In order to determine the kinetic model, IKP method was associated with the criterion of coincidence of the kinetic parameters for all heating rates. Finally, the following kinetic triplet was obtained: E=91.7 (±0.1) kJ mol−1, lnA(s−1)=16.174 (±0.020) and F1 kinetic model.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
V. Musat, P. Budrugeac, R. Monteiro, E. Fortunato and E. Segal, J. Therm. Anal. Cal., accepted for publication.
H. L. Friedman, J. Polym. Sci., Part C, 6 (1964) 183.
J. H. Flynn and L. A. Wall, J. Res. Natl. Bur. Standards, A. Phys. Chem., 70A (1966) 487.
T. Ozawa, Bull. Chem. Soc. Jpn., 38 (1965) 1881.
T. Akahira and T. Sunose, Res. Report Chiba Inst. Technol. (Sci. Technol.), 16 (1971) 22.
A. I. Lesnikovich and S. V. Levchik, J. Thermal Anal., 27 (1983) 83.
A. I. Lesnikovich and S. V. Levchik, J. Thermal Anal., 30 (1985) 667.
L. A. Perez Maqueda, J. M. Criado, F. G. Gotor and J. Malek, J. Phys. Chem., 106 (2002) 2862.
J. M. Criado and J. Morales, Thermochim. Acta, 19 (1977) 305.
Z. Adonyi and G. Korosi, Thermochim. Acta, 60 (1983) 23.
N. Koga and H. Tanaka, J. Thermal Anal., 37 (1991) 347.
J. Madarasz, G. Pokol and S. Gal, J. Thermal Anal., 42 (1995) 539.
S. Vyazovkin and W. Linert, Int. Rev. Phys. Chem., 14 (1995) 355.
N. Rose, M. Le Bras, S. Bourbigot and R. Delobel, Polym. Degrad. Stab., 45 (1994) 45.
S. Bourbigot, R. Delobel, M. Le Bras and D. Normand, J. Chim. Phys., 90 (1993) 1909.
L. R. Campisi, S. Bourbigot, M. Le Bras and R. Delobel, Thermochim. Acta, 275 (1996) 37.
B. Mortaigne, S. Bourbigot, M. Le Bras, G. Cordellier, A. Baundry and J. Dufay, Polym. Degrad. Stab., 64 (1999) 443.
X. Almeras, F. Dabrawski, M. Le Bras, R. Delobel, S. Bourbigot, G. Marosi and P. Anna, Polym. Degrad. Stab., 77 (2002) 315.
P. Budrugeac, Polym. Degrad. Stab., 71 (2001) 125.
P. Budrugeac and E. Segal, J. Therm. Anal. Cal., 82 (2005) 677.
P. Budrugeac, J. M. Criado, F. J. Gotor, J. Malek, L. A. Perez-Maqueda and E. Segal, Int. J. Chem. Kinet., 36 (2004) 309.
P. Budrugeac and E. Segal, Rev. Roum. Chim., 49 (2004) 193.
P. Budrugeac, E. Segal, L. A. Perez-Maqueda and J. M. Criado, Polym. Degrad. Stab., 84 (2004) 193.
A. W. Coats and J. P. Redfern, Nature, 201 (1964) 68.
P. Budrugeac, A. L. Petre and E. Segal, J. Thermal Anal., 47 (1996) 123.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Budrugeac, P., Muşat, V. & Segal, E. Non-isothermal kinetic study on the decomposition of Zn acetate-based sol-gel precursor. J Therm Anal Calorim 88, 699–702 (2007). https://doi.org/10.1007/s10973-006-8086-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-006-8086-0