Abstract
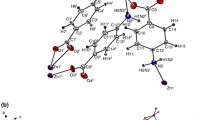
A new complex, diaquadi(1,2,4-triazol-5-one)zinc(II) ion nitrate formulated as {[Zn(TO)2(H2O)2](NO3)2}n (1) (1,2,4-triazole-5-one, abbreviated as: TO) was synthesized and characterized by elemental analysis, X-ray single crystal diffraction, infrared spectrum (IR), differential scanning calorimetry (DSC), thermogravimetric analysis and differential thermogravimetric analysis (TG-DTG). The X-ray structure analysis reveals that the complex is orthorhombic with space group Pbca and unit-cell parameters a=6.9504(2) Å; b=10.6473(3) Å; c=17.8555(5) Å. Based on the result of thermal analysis, the thermal decomposition process of the compound was derived. From measurement of the enthalpy of solution in water in 298.15 K, the standard molar enthalpy of solution of lignand TO and the complex were determined as 15.43±0.18 and 52.64±0.42 kJ mol−1, respectively. In addition, the standard molar enthalpy of formation of TO(aq) was calculated as −126.97±0.72 kJ mol−1.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
M. A. Hiskey, D. E. Chavez and D. Naud, U.S. Patent 6,458,227, 2002, October 1.
M. A. Hiskey, D. E. Chavez and D. Naud, U.S. Patent 6,342,589, 2002, January 29.
K.-Y. Lee and M. M. Stinecipher, U.S. Patent 5,256,792, 1993, October 26.
H. Xu, Y. Gao, B. Twamley and J. M. Shreeve, Chem. Mater., 17 (2005) 191.
K.-. Lee and M. M. Stinecipher, U.S. Patent 5,256,792, 1993, October 26.
G. W. Drake, USP 6,509,473, 2003, January 21.
G. Singh and S. Prem Felix, Combust. Flame, 135 (2003) 145.
P. B. Kulkarni, G. N. Purandare, J. K. Nair, M. B. Talawar, T. Mukundan and S. N. Asthana, J. Hazard. Mater. A, 119 (2005) 53.
P. B. Kulkarni, T. S. Reddy, J. K. Nair, A. N. Nazare, M. B. Talawar, T. Mukundan and S. N. Asthana, J. Hazard. Mater. A, 123 (2005) 54.
J. P. Agrawal, S. M. Walley and J. E. Field, Combust. Flame, 112 (1998) 62.
J. R. Song, R. Z. Hu, B. Kang and F. P. Li, Thermochim. Acta, 331 (1999) 49.
G. X. Ma, T. L. Zhang, J. G. Zhang and K. B. Yu, Anorg. Allg. Chem., 630 (2004) 423.
J.G. Zhang, T. L. Zhang and Z. Lu, Acta Chim. Sinica, 57 (1999) 1233.
J. G. Zhang, T. L. Zhang, L. Yang and L. Q. Mao, Chin. J. Inorg. Chem., 18 (2002) 284.
M. M. Boudakian and D. A. Fidler, U.S. Patent 4,927,940, 1990, May 22.
J. R. Li, B. R. Chen and Y. X. Ou, Ener. Mater., 6 (1998) 107 (in Chinese).
G. M. Sheldrick, SHELX-97, Program for Crystal Structure Refinement, University of Göttingen, Germany 1997.
M. Ji, M. Y. Liu, S. L. Gao and Q. Z. Shi, Instrum. Sci. Technol., 29 (2001) 53.
V. K. Marthada, J. Res. NBS Standards, 85 (1980) 467.
J. W. Rogers Jr., H. C. Peebles, R. R. Rye, J. E. Houston and J. S. Binkley, J. Chem. Phys., 80 (1984) 4513.
R. P. Singh, R. D. Verma, D. T. Meshri and J. M. Shreeve, Angew. Chem. Int. Ed., 45 (2006) 3584.
L. Zhou, Base of Explosion Chemistry, Beijing Institute of Technology Press, (2005), p. 248.
A. Finch, P. J. Gardner, A. J. Head and H. S. Majdi, J. Chem. Thermodyn., 23 (1991) 1169.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, N., Chen, S.P. & Gao, S.L. Crystal structure and thermal analysis of diaquadi (1,2,4-triazol-5-one)zinc(II) ion nitrate. J Therm Anal Calorim 89, 583–588 (2007). https://doi.org/10.1007/s10973-006-7600-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-006-7600-8