Summary
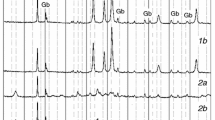
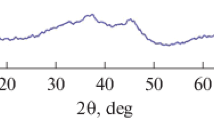
Two different processes of the thermal decomposition of synthetic bayerite, i.e., the non-isothermal decomposition of mechanically ground sample in flowing N2 and the controlled rate thermal decomposition of crystalline bayerite under vacuum, were investigated comparatively. In comparison with the conventional non-isothermal decomposition of crystalline bayerite in flowing N2, the reaction temperature of the thermal decomposition was lowered by the individual effects of mechanical grinding of the sample and the reaction rate control. These decomposition processes indicated similar behavior characterized by the restricted changes of the specific surface area during the course of decomposition reaction and the formation of an amorphous alumina as the decomposition product. Different thermal behaviors were observed for those amorphous Al2O3 produced by the respective decomposition processes.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Koga, N. A comparative study of the effects of decomposition rate control and mechanical grinding on the thermal decomposition of aluminum hydroxide. J Therm Anal Calorim 81, 595–601 (2005). https://doi.org/10.1007/s10973-005-0830-3
Issue Date:
DOI: https://doi.org/10.1007/s10973-005-0830-3