Abstract
In the present study various separation methods were investigated for the recovery of actinides from aqueous waste solutions. Extraction behavior and recovery of U(VI) from acidic waste solutions has been examined with Tri-iso-Amyl Phosphate (TiAP) impregnated on XAD-7. Recovery of Am from acidic waste solutions using octyl (phenyl)-N,N-diisobutylcarbamoylmethylphosphine oxide (CMPO) impregnated XAD-7 resin has been examined. Recovery of plutonium from acidic waste solution was carried out using a synergistic mixture of Di-2-ethylhexyl phosphoric acid (D2EHPA) and Tri-n-octyl phosphine oxide (TOPO).
Similar content being viewed by others

Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Radioactive waste solutions get generated during the various R&D activities in nuclear industry. For example acidic waste solutions containing U, Pu and Am get generated during the development of novel extractants for spent fuel reprocessing, acidic U-Zr solutions get generated during the dissolution of U-Zr and U-Pu-Zr metallic alloy fuels. Aqueous waste solutions containing uranium also get generated in the uranium mining industry. It is important to recover actinides from aqueous waste solutions due to its strategic significance, chemical and toxicity. Various separation methods involving suitable extractants or adsorbent materials can be employed for the recovery of actinides from aqueous waste solutions.
Extraction chromatography is preferred for the recovery of metal ions from dilute solutions. Extraction chromatography is the combination of solvent extraction and chromatographic processes, where in selectivity is achieved by solvent extraction process with the multistage character of chromatographic process and ease of operation of ion-exchange column chromatography [1, 2]. Actinides such as U, Pu, Th and Am from acidic waste solutions generated during the reprocessing of spent nuclear fuels is recovered by extraction chromatographic method [3]. Chromatographic technique is very useful for the recovery of actinides from high active waste solutions [4,5,6,7,8]. The selection of suitable solvent in extraction chromatographic system for the recovery of actinides is chosen depending on the solvents employed in solvent extraction process. Several neutral type extractants such as Tri-n-Butyl Phosphate (TBP), Diamylamyl Phosphonate(DAAP), Octyl (phenyl)-N,N-diisobutylcarbamoylmethylphosphine oxide (CMPO), N,N,N’,N’ -tetra-octyldiglycolamide (TODGA) etc, and acidic type extractant such as Di-2-ethylhexyl phosphoric acid (D2EHPA) impregnated on stationary inert support matrices were studied in literature for the recovery of actinides [9,10,11,12,13,14].
Acidic waste solutions containing Am in trace quantities is generated in the various stages of back end of nuclear fuel cycle [15, 16]. Recovery of Am from acidic waste solutions is the topic of interest for last many years and it is also important to reduce the release of Am to environment in the context of safety and radio toxicity. Many reagents such as carbamoylmethylphosphine oxides, malonamides, and diglycolamides are well studied for the extraction of trivalent actinides and lanthanides [17, 18]. Bidentate organophosphorous ligand such as CMPO has been well studied for the extraction of actinides and lanthanides by solvent extraction and is found to be the most preferred solvent [19,20,21].
Large volumes of analytical waste solutions are generated when the radionuclides such as uranium, plutonium and other minor actinides are analyzed in the process as well as in the waste streams. The waste solutions thus generated in each step need to be treated before final disposal considering its radio-toxicity to the environment. Analytical waste solution is usually generated in large volumes with relatively lower levels of radioactivity (µCi/L to mCi/L). The types of wastes generated are determined by the analytical processes used in the laboratory and the characteristics of the samples analyzed. Several processes such as chemical precipitation, solvent extraction, ion exchange etc. can be employed either as a single process or in combination for the recovery of radionuclides from various waste streams. Depending on the nature of the analytical waste, radionuclides and level of associated radioactivity, a suitable treatment scheme is chosen to concentrate bulk of the radioactivity to a small volume. Recovery of plutonium from general analytical waste appears challenging due to its complexity. Typical acidic analytical waste solution was generated in our laboratory during the analysis of uranium and plutonium in various processes. In addition to radionuclides (U, Pu and Am) other major impurities such as Fe, Cr, Ag, V, and Mo etc. are also present in liquid waste [22,23,24]. In literature, studies are reported on the recovery of uranium from phosphoric acid using D2EHPA and tri-n-octyl phosphine oxide (TOPO) mixture [25, 26].
In the present study, Tri-iso-Amyl Phosphate (TiAP) an alternate solvent to TBP in solvent extraction process has been examined to understand the extraction behavior of U(VI) by TiAP solvent impregnated on XAD-7. Separation of U from Zr was studied with extraction chromatographic resins, TiAP and TBP solvents impregnated on XAD-7. Recovery of Am from acidic aqueous waste solution demonstrated using 50% CMPO/XAD-7. Elution of Am from loaded column was performed by citric acid. In the present study a method has been developed for the recovery of Pu from analytical acidic waste solution by employing a synergistic mixture of D2EHPA and TOPO in xylene and the results are discussed. The chemical structures of the molecules employed in the present study are shown in Scheme 1.
Experimental
Chemicals
Solvent extraction grade TBP of purity (> 99%) provided by M/s Fluka was used. TiAP (> 98%) was received from Heavy Water Board, Tuticorin, India and further purified by washing with NaOH and water. Details of synthesis of CMPO are reported elsewhere [27, 28]. All the chemicals used were of AR or GR grade. 241Am in the form of americium nitrate was used for the measurement of distribution coefficeints. XAD-7 resin, mesh size 20–60 (M/s. Supelco1, USA) were washed with distilled water, acetone and methanol to remove preservatives and residual monomers and dried under vacuum. The main characteristics of the XAD-7 resin are presented elsewhere [28]. Nuclear grade zirconium metal (Nuclear Fuel Complex, Hyderabad) was used to prepare zirconium nitrate solutions and the details of zirconium metal dissolution are reported elsewhere [29]. It is reported that zirconium exists as Zr(IV) under these conditions of metal dissolution. Nuclear grade uranyl nitrate hexahydrate [Nuclear fuel complex, Hyderabad] was used as received.
Preparation of extraction chromatographic resins
Extractants such as TBP, TiAP and CMPO loaded on XAD-7 for the extraction chromatography experiments was prepared by the following procedure. Required amounts of XAD-7 and extractants were shaken in 25 mL of methanol: water (1: 1) mixture for 24 h using mechanical shaker. Subsequently, methanol/water was removed by evaporation at ~ 80oC under vacuum using a rotary evaporator. The weight % of extractant loaded onto a resin was calculated from the difference in the weight of resin before and after equilibration; about 50 wt% of extractants in XAD resins were prepared using this method.
Preparation of Stock solutions
The stock solutions of U(VI) containing 10 mg/mL and 100 mg/mL in 4 M nitric acid medium were prepared by mixing the acidic waste solutions of U(VI) which are generated from various solvent extraction experiments. The stock solution of U(VI)(~ 10 mg/mL) and Zr(IV)(~ 1 mg/mL) in 4 M nitric acid was prepared from uranyl nitrate and zirconium nitrate solutions.
Extraction studies of U(VI) with 50%TiAP/XAD-7
Extraction behavior of U(VI) by 50% TiAP/XAD-7 has been examined as a function of equilibrium aqueous phase U(VI) concentration from 3 to 5 M HNO3 at 25oC. Various concentrations of uranium solutions in 3 and 5 M HNO3 (3 mL) were equilibrated with 0.2 g of resin in a set of equilibration tubes for 30 min. After equilibration, contents were allowed to settle and the amount of U(VI) present in the supernatant solution was analyzed. The amount of uranium loaded (qe) onto the resin were calculated using the following equations.
Here, Co and Ce denote the initial and equilibrium concentrations (mg mL− 1) of U(VI), V is the volume of solution (L) added, and m is the weight of the dry chromatographic resin (g).
Determination of breakthrough capacities for the sorption of U(VI) by columns packed with 50% TiAP/XAD-7 resin
Slurry packing procedure was adopted for the column preparation. A glass column with ~ 10 mm inside diameter and ~ 270 mm length was used for packing. About 8 g of 50% TiAP/XAD-7 resin was mixed with distilled water and the column was filled with the slurry. The bed volume was found to be 15.35 mL and the bed density (i.e., the weight of resin per unit bed volume), was found to be 0.52 g/mL. After fixing the upper end by cotton, column pre-conditioning was completed by eluting with several bed volumes of 4 M HNO3. Fractions of 2 mL, 5 mL and 10 mL were collected in standard flasks with flow rates of 0.5 mL min− 1.
Column experiments for the separation of U(VI) and Zr(IV) from U-Zr feed solution with 50% TiAP/XAD-7 and 50% TBP/XAD-7 resins
Extraction chromatographic resins such as 50% TiAP/XAD-7 (~ 8 g) and 50% TBP/XAD-7 (~ 6.8 g) were mixed with distilled water and the column was filled with the slurry. After fixing the upper end by cotton, column preconditioning was completed by eluting with several bed volumes of 4 M HNO3. A feed solution containing U(VI) ~ 10 mg/mL and Zr(IV) ~ 1 mg/mL in 4 M HNO3 was passed through the column until 100% breakthrough was achieved. Fractions of 2 mL, 5 mL and 10 mL were collected in standard flasks with a flow rate of 0.5 mL/min. U and Zr were analyzed in effluent fractions.
Experiments with Am(III) solution using 50% CMPO/XAD-7 resin
Initially, distribution ratios (Kd) for the extraction of Am(III) were measured using 50% CMPO/XAD-7 as a function of nitric acid concentration(0.1–10 M) at 25oC by equilibrating 241Am tracer solutions in nitric acid (2 mL) with 100 mg of resin in a set of equilibration tubes. Supernatant solution was analyzed for 241Am by γ-counting after centrifugation. The distribution ratios were calculated from the following equation.
Where, C0 and C are the initial and final 241Am concentration in the aqueous phase before and after equilibration, respectively. V is the aqueous phase volume (in mL) and M is the weight of the dry chromatographic resin, 50% CMPO/XAD-7 (in gram).
After batch extraction experiments, extraction chromatographic resin i.e. 50% CMPO/XAD-7 was packed in a column by slurry procedure. A feed solution containing Am(III) about 0.3 mg/mL in 3 M HNO3 was passed through the column in three batches with different breakthroughs. After loading Am(III) in the column, elution was carried out with 0.01 M HNO3 in the first batch and in the next two batches elution was carried out with 0.1 M citric acid (pH = 3.4).
Solvent extraction experiments for recovery of Pu(IV)
The acidic waste solution generated during the analysis of U and Pu by potentiometry was analyzed for acidity and metal ions. The acidity of the waste solution was found to be 1.15 M. The acidic waste solution contain UO22+, Pu4+, and Am3+ and other impurities such as Fe3+, Cr3+, Ag+, Mo(VI), and VO2+. The organic extractant containing a synergistic mixture of 0.88 M D2EHPA and 0.22 M TOPO in xylene was prepared and used as such. The acidic aqueous waste was contacted with equal volume (5 mL) of 0.88 M D2EHPA and 0.22 M TOPO in xylene. The two phases were mixed in a flat-bottom equilibration tube and mixing was performed using a PTFE coated magnetic stirring bar with magnetic stirrer at 303 K for 30 min. After the equilibration, the phases were allowed to settle by gravity. Suitable aliquots of both the aqueous and organic phases were pipetted for the estimation of metal ion concentration. After the extraction, the organic phase was removed and aqueous phase was freshly contacted with 0.88 M D2EHPA and 0.22 M TOPO in xylene. This procedure was repeated for 3 times for complete removal of Pu from acidic waste solution. All the experiments were carried out in duplicate. The distribution ratio (D) (ratio of the concentration of metal ion in the organic phase to the concentration of metal ion in the aqueous phase at equilibrium) and the percentage extraction ((D/D + 1)*100) of each metal ion was measured.
Analytical methods
The concentration of nitric acid, zirconium, uranium and 241Am was determined by suitable analytical methods. Uranium in macro level (without any other metal ion) was determined by complexometric titration using Pyridine-2,6-dicarboxylic acid (PDCA) as the titrant and Arsenazo I as the indicator at pH 3.7 with a precision of ± 1% Relative Standard Deviation (RSD) [30]. A solution of 0.03 M PDCA was used for the analysis of uranium solutions. Uranium in macro levels (in presence of other metal ions) was determined by potentiometry using Davies & Gray method [23]. In this method uranium can be analyzed with a precision of ± 0.3% RSD. Uranium in micro quantities (in presence of other metal ions) was determined using spectrophotometry; uranium was analyzed using 2-(5-Bromo-2-pyridylazo)-5-(diethyl amino) phenol (Br-PADAP) as the chromogenic agent by measuring the absorbance of metal complex at 577 ± 1 nm [31]; in the spectrophotometric analysis, the lowest measurable concentration of uranium was found to be ~ 1 ppm, with a precision of ± 2% RSD. Zr in micro level was analyzed spectrophotometrically using xylenol orange as the chromogenic agent with a precision of ± 3% RSD [32]. The absorbance of Zr(IV)-Xylenol orange complex in the range of 0.5–3.5 µg/mL was measured at 535 nm. 241Am was determined by gamma counting using a NaI(Tl) detector based counting system at 60 kev with a precision of ± 5% RSD. Nitric acid concentration in the aqueous samples was determined by acid-base titration after complexing the metal ions with potassium oxalate-sodium fluoride solution and the solution was titrated with standard NaOH solution to pH 7. In this method, nitric acid can be analyzed to a precision of ± 1% [33]. Impurities (Fe, V, Mo, Ag, Cr) present in the acidic waste solution was analysed by Inductively Coupled Plasma-Optical Emission Spectrometry (ICP-OES) using sequential spectrometer of ULTIMA-C spectroanalyser system (John Yvon, France) with a precision of ± 5% RSD. Pu was analyzed by liquid scintillation counter using dioxane based solvents with a precision of ± 5% RSD.
Results and discussion
Recovery of U(VI) from acidic waste solutions
Extraction studies with U(VI) using 50% TiAP/XAD-7
Large quantities of acidic waste solutions containing uranium get generated during the development of novel extractants for spent fuel reprocessing [34, 35]. It is important to recover U(VI) from these waste solutions. In this context, novel extractant TiAP has been examined using extraction chromatography for the recovery of U(VI) from acidic waste solutions. Initially the sorption capacities for U(VI) by 50% TiAP/XAD-7 were computed followed by breakthrough curves were evaluated. The theoretical capacity for the extraction of U(VI) by 50% TBP/XAD-4 is calculated using the following equation; it was found to be about 223 mg of U(VI) per gram of 50% TBP/XAD-4 resin [36].
The maximum capacity depends on several factors such as concentration of metal ion, nitric acid in the aqueous phase, distribution equilibrium of metal ions between the aqueous phase and the resin phase etc. Suresh et al. have studied the extraction of U(VI) and Am(III) from 3 M HNO3 by XAD-4 and XAD-7 resins loaded with CMPO [28]. Their results indicate that XAD-7 is found to be a better support for CMPO as compared to XAD-4, both with regard to kinetics, as well as the equilibrium distribution ratio (Kd) for the extraction of U(VI) and Am(III) from 3 M HNO3. Therefore XAD-7 was chosen over XAD-4 for our studies. The extraction behavior of U(VI) with TiAP was found to be similar to that of TBP as shown in Eq. 2 [35]. The maximum capacity for the extraction of U(VI) by 50% TiAP/XAD-7 is calculated by using the following equation; it was found to be about 193 mg of U(VI) per gram of 50% TiAP/XAD-7 resin.
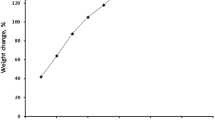
The sorption capacity of resin for U(VI) by 50% TiAP/XAD-7 was found to be higher at 5 M HNO3 compared to that of 3 M HNO3 (Fig. 1). The sorption capacity of resin increases with increase of equilibrium aqueous phase U(VI) concentration and the maximum sorption capacity was found to be about 170 and 181 mg of U(VI) per gram of 50% TiAP/XAD-7 resin at 3 and 5 M HNO3, respectively (Fig. 1).
Determination of Column Capacity of U(VI) by 50% TiAP/XAD-7
Breakthrough curves for U(VI) (Fig. 2) were obtained for the determination of sorption capacities of column packed with 50% TiAP/XAD-7 resin using uranyl nitrate solution with different concentrations such as 10 and 100 g/L in 4 M HNO3. Breakthrough capacities (milligrams of metal extracted per gram) of column packed with 50% TiAP/XAD-7 were determined when the concentration of U(VI) in effluent reached 50%, and 100% of the feed concentration (Table 1). Breakthrough capacities in general, increase with increasing the metal ion concentration in the feed solution. The maximum capacity is found to be about 134 and 331 mg/g resin for 50% TiAP/XAD-7 with U(VI) feed concentration of 10 g/L and 100 g/L, respectively in 4 M HNO3. The relatively high breakthrough capacity for the sorption of U(VI) by 50% TiAP/XAD-7 resin highlights the feasibility for the recovery of uranium from waste solutions generated during the nuclear fuel cycle operations. By using the Eq. (2), the maximum theoretical loadings of U(VI) is found to be 193 mg per gram of resin respectively. However, breakthrough capacities for uranium on 50% TiAP/XAD-7 column are higher than theoretical capacity indicating the possibility of formation of lower solvates (Table 1).
In order to examine the reusability of coated support, U(VI) loaded in column was washed with 4 M HNO3 and then, eluted with 0.01 M HNO3. A fresh lot of uranium (10 g/L) was loaded into the column and maximum breakthrough capacity was found to be ~ 133 mg U(VI)/g resin, indicating that U(VI) loading into the column is similar to the first cycle(~ 134 mg U(VI)/g resin). This highlights the stability of extraction chromatographic resin in nitric acid medium. Yamaura et al. studied the stability of 46% TBP/XAD-7 in nitric acid medium in the second cycle [37]. It was observed in their studies that uranium retention and elution efficiencies were found to be same even after 10 cycles, presenting a good stability in nitric acid medium. They have also calculated 10% breakthrough capacity of U(VI) at 4 M HNO3 for 46% TBP/XAD-7 with U(VI) feed concentration of 0.6 g/L and it was found to be about 21.2 mg U/g resin. Kimura studied the effect of flow rate and U(VI) concentration on maximum capacity of 50% TBP/XAD-4 [36]. He observed that maximum capacity increases with increasing uranium concentration. For example the maximum capacity is found to be about 153 mg U(VI)/g resin with a U(VI) feed concentration of 0.1 M and it was about 168 mg/g resin for 0.2 M solution of U(VI) in 3 M HNO3.
Separation of U(VI) and Zr(IV) from U-Zr feed solution in nitric acid medium
As an interim process aqueous based PUREX process has been examined in our laboratory for the reprocessing of metallic alloy fuels such as U-Zr and U-Pu-Zr. Several U-Zr feed solutions were generated during dissolution of metallic fuels [32, 38]. It is important to separate and recover U(VI) from U-Zr feed solutions. Therefore in the present study TiAP based extraction chromatographic resin has been examined for recovery of U(VI) from U-Zr feed solutions. Separation of U(VI) from U-Zr feed solutions was carried out by extraction chromatographic technique using 50% TiAP/XAD-7 and results were compared with 50% TBP/XAD-7. Breakthrough curves were obtained for U and Zr for 50% TiAP/XAD-7 and 50% TBP/XAD-7 at a flow rate of 0.5 mL/min(Fig. 3). The maximum capacities for U by TiAP and TBP based resins are found to be ~ 135 and 142 mg U/g resin, respectively. The maximum capacity of U in the case of U-Zr solution by 50%TiAP/XAD-7 is similar (~ 134 mg U/g resin) to the obtained in the case of pure uranyl nitrate solution (Table 2) signifying that loading of Zr into the resin is negligible. The maximum capacities for Zr by TiAP and TBP based resins were found to be very less ~ 2.39 and 3.04 mg Zr/g resin, respectively. The concentration of Zr in the effluent reaches its initial concentration within ~ 30 mL after passing through the column; however to achieve initial concentration of U (10 g/L), ~ 180 mL of U-Zr feed solution was passed through the column. These studies establish that extraction chromatographic resins can be employed for the selective separation and recovery of U from U-Zr feed solutions.
Recovery of Am(III) from acidic waste solution using 50% CMPO/XAD-7 resin
Acidic aqueous waste (raffinate) solution was generated in our laboratory during continuous solvent extraction runs carried out in a counter-current mode for the extraction of U(VI) and Pu(IV) by TiAP using an ejector mixer-settler [34]. It contained Am (III) with a concentration of about 0.3 mg/mL in 3 M HNO3, over a volume of 2 L. The objective of the present investigation is to develop a procedure for the recovery of Am from acidic solution and concentrate in a small volume. The compound, CMPO was examined in the literature for the extraction of actinides and it was found to be very efficient for the extraction of actinides [18,19,20,21]. Therefore in our studies CMPO has been employed for the recovery of Am from acidic waste solution. Initially distribution ratios (Kd) were measured for Am using 50% CMPO/XAD-7 as a function of nitric acid concentration, Kd values for Am(III) increases up to 3 M HNO3 and decreases thereafter(Fig. 4). Therefore the acidity of waste solution generated from mixer-settler runs was adjusted to 3 M HNO3. The concentration of Am in the solution after acidity adjustment was found to be ~ 0.275 g/L. The waste solution (330 mL) was passed through the column only up to 1% breakthrough to minimize the loss of Am to the effluent solution. About 91 mg of Am was loaded into the column with 1% breakthrough. In order to recover Am from the loaded column, 0.01 M HNO3 was initially employed as an eluant (Kd of Am at 0.01 M HNO3 is 3.06). Am(III) eluted using 0.01 M HNO3 (Fig. 5) resulted in large volume (~ 150 mL) which lead to very dilute solution of Am.
The objective of the present study is to concentrate Am in small volume. In this context, citric acid (CA) was examined as an eluant for the recovery of Am from loaded column. Kd values were measured for Am by 50% CMPO/XAD-7 at 0.1 M CA(pH 2.63) and it was found to be 1.89; the pH of the 0.1 M CA solution was adjusted to 3.4 and Kd was found to be negligible (< 0.01) indicating that Am can be recovered from column using 0.1 M CA(pH 3.4). Therefore 0.1 M CA (pH 3.4) was employed as eluant. After the quantitative elution of Am from column in the 1st batch, it was preconditioned with 3 M HNO3 and Am was again loaded into the column up to 1.8% breakthrough. About 750 mL of Am feed solution was passed through the column and 206 mg of Am was loaded into the column (Table 3). The loaded Am was eluted with 0.1 M CA (pH 3.4) and it was eluted as a sharp band (Fig. 6) in a small volume (~ 60 mL). It is because of the formation of Am(III)-Citrate complex, a relatively sharp band was observed unlike a broader one in the case of 0.01 M HNO3. After elution of Am from the column in the second cycle, Am was loaded again in the third cycle up to 4.2% breakthrough (Fig. 7) and elution profile of Am in third run was shown in Fig. 6. Am eluted in all the runs was collected and solution was evaporated near dryness and CA was destroyed by repeated fuming with HNO3. These studies established that Am can be concentrated in a small volume using CMPO based extraction chromatographic resin with citric acid as eluant.
Recovery of Plutonium from Acidic Analytical Waste Solution
Venugopal et al. reported the recovery of plutonium from uranium analytical waste solution containing phosphate using 2-ethylhexyl 2-ethylhexyl phosphonic acid (PC88A). The plutonium extracted into the organic phase was stripped using 1 M oxalic acid containing 0.2 M ascorbic acid [39]. The extraction behavior of various metal ions such as V, Mo, Cr(III), Fe(III), Ag(I), etc. was examined using D2EHPA [40,41,42]. Recovery of uranium from phosphoric acid medium was demonstrated using D2EHPA and tri-n-octyl phosphine oxide (TOPO) mixture [25, 26]. Studies reported in the literature shows that D2EHPA more versatile molecule and it has been examined for the extraction of various metal ions. Therefore, in the present study, a synergistic mixture of D2EHPA and TOPO in xylene was employed for the recovery of Pu from acidic analytical waste generated during the analysis of U and Pu using Davies-Gray and Drummond-Grant methods respectively. The acidic analytical waste solution contain UO22+, Pu4+, and Am3+ and other impurities such as Fe3+, Cr3+, Ag+, Mo(VI), and VO2+. The typical concentrations of each metal ion presnt in the waste solution are given in the Table 4. The organic extractant containing a synergistic mixture of 0.88 M D2EHPA and 0.22 M TOPO in xylene was employed. The extraction of plutonium in the first contact is found to be about 91% and it was found to be about 98% in the second contact. Quantitative extraction (~ 99%) of plutonium was achieved within three contacts.
The extraction behavior of U, Am and other impurities were also examined in the first contact by 0.88 M D2EHPA and 0.22 M TOPO in xylene. The distribution ratios for Fe3+(9.6) and Mo(VI) (11.65)(Table 4) is found to be higher compared to other metal ions(UO22+, Am3+ Cr3+, Ag+, and VO2+) indicating that Fe(III) and Mo(VI) also extracted into the organic phase along with Pu(IV). Correspondingly the extraction(%) of Fe3+ and Mo(VI) is higher compared to other metal ions(Table 4). The extraction of Fe(III) and Mo(VI) into the organic phase reduces the purity of the Pu product. The extraction of other metal ions (Am, Ag, Cr, V) was found to be significantly low as their D values were found to be less than 0.2. The extraction of Fe3+ and Mo(IV) along with Pu is due to the presence of free extractant concentration(D2EHPA and TOPO). The concentration of Pu in the acidic waste solution is found to be less (178 ppm); therefore the loading of Pu into the organic phase is also less. It is reported in our earlier paper that distribution ratios of impurities decreases with increase of loading of U or Pu in the organic phase [43]. In the present study the distribution ratios of impurities is found to be high due to the loading of Pu into the organic phase is less; thereby the decontamination factors/purity of Pu against impurities decreases. The decontamination factors can be increased by reducing the free extractant concentration by employing the low concentration of extractant.
The synergistic mixture was reused six-times after stripping the plutonium with saturated oxalic acid solution and its performance was found to be good. Use of D2EHPA-TOPO in other proportions however was not very effective in extracting plutonium such as 3:1 D2EHPA/TOPO or 4:1 TOPO/D2EHPA. The potential application of this method includes direct discharge of the aqueous analytical waste by reducing the total activity less than acceptable limits of waste management facility (< 5500 Bq/mL).
Conclusions
In the present study various separation methods such as extraction chromatography, ion exchange, and solvent extraction were employed for the recovery of actinides from different aqueous streams. TiAP based extraction chromatographic resin supported on XAD-7 was examined to understand the extraction behavior of uranium. Breakthrough capacities for U(VI) by 50% TiAP/XAD-7 were obtained for different concentrations of uranium feed solutions. Separation of U(VI) from U-Zr feed solutions was demonstrated with TiAP and TBP based extraction chromatographic resins. Americium from acidic waste solution was recovered using CMPO based extraction chromatographic resin supported on a XAD-7 resin. The Am from the loaded column was eluted using 0.1 M citric acid. Solvent extraction method was demonstrated for the recovery of plutonium from analytical waste using a synergistic mixture of D2EHPA and TOPO. The present study demonstrates various separation methods for recovery of actinides from different aqueous streams.
References
Braun T, Ghersini G (1975) Extraction chromatography. Elsevier
Philip Horwitz E, Mcalister DR, Dietz ML (2006) Extraction chromatography versus solvent extraction: how similar are they? Sep Sci Technol 41:2163–2182
Horwitz EP, Chiarizia R, Dietz ML (1993) Separation and preconcentration of actinides from acidic media by extraction chromatography. Anal Chim Acta 281:361–372
Naik P, Dhami P, Misra S (2003) Use of organophosphorus extractants impregnated on silica gel for the extraction chromatographic separation of minor actinides from high level waste solutions. J Radioanal Nucl Chem 257:327–332
Watanabe S, Senzaki T, Shibata A (2019) Improvement in flow-sheet of extraction chromatography for trivalent minor actinides recovery. J Radioanal Nucl Chem 322:1273–1277
Modolo G, Asp H, Schreinemachers C (2007) Recovery of actinides and lanthanides from high-level liquid waste by extraction chromatography using TODGA + TBP impregnated resins. Radiochim Acta 95:391–397
Watanabe S, Goto I, Nomura K (2011) Extraction chromatography experiments on repeated operation using engineering scale column system. Energy Procedia 7:449–453
Watanabe S, Sano Y, Kofuji H (2018) Am, Cm recovery from genuine HLLW by extraction chromatography. J Radioanal Nucl Chem 316:1113–1117
Ramanujam A, Dhami P, Gopalakrishnan V (1993) Application of extraction chromatographic technique in the recovery of actinides from Purex waste solutions using CMPO. No. BARC–1993/E/024. Bhabha Atomic Research Centre
Mathur J, Murali M, Iyer R (1995) Extraction chromatographic separation of minor actinides from PUREX high-level wastes using CMPO. Nucl Technol 109:216–225
Sankhe RH, Sengupta A, Mirashi N (2014) Simultaneous recovery of plutonium and americium from assorted analytical waste solutions using extraction chromatography. J Radioanal Nucl Chem 302:617–622
Ansari SA, Mohapatra PK (2017) A review on solid phase extraction of actinides and lanthanides with amide based extractants. J Chromatogr A 1499:1–20
Horwitz E, Mcalister D, Bond A (2005) Novel extraction of chromatographic resins based on tetraalkyldiglycolamides: characterization and potential applications. Solvent Extr Ion Exch 23:319–344
Gujar R, Ansari S, Verboom W (2016) Multi-podant diglycolamides and room temperature ionic liquid impregnated resins: An excellent combination for extraction chromatography of actinides. J Chromatogr A 1448:58–66
Mathur J, Murali M, Nash K (2001) Actinide partitioning—a review. Solvent Extr Ion Exch 19:357–390
Michael K, Rizvi G, Mathur J (1997) Recovery of plutonium and americium from laboratory acidic waste solutions using tri-n-octylamine and octylphenyl-NN-diisobutylcarbamoylmethylphosphine oxide. Talanta 44:2095–2102
Ansari SA, Pathak P, Mohapatra PK (2011) Aqueous partitioning of minor actinides by different processes. Sep Purif Rev 40:43–76
Saipriya K, Kumaresan R, Nayak P (2016) Extraction behaviour of Am (III) and Eu (III) from nitric acid medium in CMPO-HDEHP impregnated resins. Radiochim Acta 104:67–75
Mathur J, Murali M, Natarajan P (1993) Partitioning of actinides from high-level waste streams of PUREX process using mixtures of CMPO and TBP in dodecane. Waste Manage 13:317–325
Ramanujam A, Dhami P, Gopalakrishnan V (1999) Partitioning of actinides from high level waste of PUREX origin using octylphenyl-N, N’-diisobutylcarbamoylmethyl phosphine oxide (CMPO)-based supported liquid membrane. Sep Sci Technol 34:1717–1728
Mincher BJ, Schmitt NC, Case ME (2011) A TRUEX-based separation of americium from the lanthanides. Solvent Extr Ion Exch 29:247–259
Xavier M, Nair P, Lohithakshan K (1991) Determination of uranium in the presence of iron and plutonium by Ti (III) reduction and biamperometric titration. J Radioanal Nucl Chem 148:251–256
Davies W, Gray U (1964) A rapid and specific titrimetric method for the precise determination of uranium using iron (II) sulphate as reductant. Talanta 11:1203–1211
Drummond J, Grant R (1966) Potentiometric determination of plutonium by argentic oxidation, ferrous reduction and dichromate titration. Talanta 13:477–488
Singh SK, Tripathi S, Singh D (2010) Studies on the separation and recovery of uranium from phosphoric acid medium using a synergistic mixture of (2-ethylhexyl) phosphonic acid mono 2-ethyl hexyl ester (PC-88A) and tri-n-octylphosphine oxide (TOPO). Sep Sci Technol 45:824–831
Botella T, Gasos P (1989) Recovery of uranium from phosphoric acid: An Overview. Recovery of uranium from phosphoric acid p17
Gatrone RC, Kaplan L, Philip Horwitz E (1987) The synthesis and purification of the carbamoylmethylphosphine oxides. Solvent Extr Ion Exch 5:1075–1116
Suresh A, Brahmmananda Rao C, Deivanayaki R (2003) Studies on the extraction behavior of octyl (phenyl)-N, N‐diisobutylcarbamoylmethylphosphine oxide in polymeric adsorbent resins. Solvent Extr Ion Exch 21:449–463
Rakesh KB, Suresh A, Sivaraman N (2016) Extraction and third phase formation behaviour of tri-iso-amyl phosphate and tri-n-butyl phosphate with Zr (IV) and Hf (IV): a comparative study. J Radioanal Nucl Chem 309:1037–1048
Celon E, Degetto S, Marangoni G (1979) Rapid-determination of milligram amounts of uranium in organic complexes with pyridine-2,6-dicarboxylic acid as titrant and arsenazo-I as indicator after oxygen-flask combustion. Talanta 26:160–162
Suresh A, Patre DK, Srinivasan TG (2002) A new procedure for the spectrophotometric determination of uranium (VI) in the presence of a large excess of thorium (IV). Spectrochim Acta A Mol Biomol Spectrosc 58:341
Sreenivasulu B, Suresh A, Sivaraman N (2017) Dissolution and characterisation studies on U–Zr and U–Pu–Zr alloys in nitric acid medium. J Radioanal Nucl Chem 311:789–800
Mayankutty P, Ravi S, Nadkarni M (1982) Determination of free acidity in uranyl nitrate solutions. J Radioanal Nucl Chem 68:145
Suresh A, Sreenivasulu B, Jayalakshmi S (2015) Mixer-settler runs for the evaluation of tri-iso-amyl phosphate (TiAP) as an alternate extractant to tri-n-butyl phosphate (TBP) for reprocessing applications. Radiochim Acta 103:101–108
Rakesh KB, Suresh A, Rao PV (2014) Extraction and stripping behaviour of tri-iso-amyl phosphate and tri-n-butyl phosphate in n-dodecane with U (VI) in nitric acid media. Radiochim Acta 102:619–628
Kimura T (1990) Extraction chromatography in the TBP-HNO3 system. J Radioanal Nucl Chem 141:307–316
Yamaura M, Matsuda H (1997) Actinides and fission products extraction behavior in TBP/XAD7 chromatographic column. J Radioanal Nucl Chem 224:83–87
Sreenivasulu B, Parida SK, Chandran K (2021) Oxidation and dissolution behavior of UZr alloys for aqueous reprocessing applications.Prog Nucl Energy104087
Venugopal Chetty K, Gamare JS, Godbole A (2006) Recovery of plutonium from uranium analytical waste solution containing phosphate using 2-ethylhexyl 2-ethylhexyl phosphonic acid (PC88A). J Radioanal Nucl Chem 269:15–20
Kamitani M, Shibata J, Sano M, EXTRACTION OF METAL IONS WITH DZEHPA FROM MIXED AQUEOUS-ORGANIC MEDIA (1988) Solvent Extr Ion Exch 6:605–619
Chen JH, Kao YY, Lin CH (2003) Selective Separation of Vanadium from Molybdenum Using D2EHPA-Immobilized Amberlite XAD‐4 Resin. Sep Sci Technol 38:3827–3852
Singh D, Yadav KK, Singh H (2013) Extraction and stripping behavior of iron (III) from phosphoric acid medium by D2EHPA alone and its mixtures with TBP/TOPO. Sep Sci Technol 48:1556–1564
Sreenivasulu B, Suresh A, Sivaraman N (2015) Solvent extraction studies with some fission product elements from nitric acid media employing tri-iso-amyl phosphate and tri-n-butyl phosphate as extractants. J Radioanal Nucl Chem 303:2165–2172
Acknowledgements
Authors are also thankful Shri. S. Subramaniam, former colleague, Materials Chemistry and Metal Fuel Cycle Group for his major contributions in experiments related to recovery of plutonium from acidic analytical waste Solution.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sreenivasulu, B., Brahmananda Rao, C.V., Suresh, A. et al. Recovery of actinides from acidic waste solutions generated in research facilities using adsorption and extraction techniques. J Radioanal Nucl Chem 331, 3623–3632 (2022). https://doi.org/10.1007/s10967-022-08451-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-022-08451-6