Abstract
A preconcentration neutron activation analysis method has been developed for the determination of trace levels of Cu in natural waters including seawater with an absolute detection limit of 0.106 μg. The method involves the preconcentration of Cu onto Amberlite XAD-4 resin coated with 1-(2-thiazolylazo)-2-naphthol using reversed-phase extraction chromatography (RPEC). Copper is assayed through its neutron activation product 66Cu (half-life = 5.10 min) by direct irradiation of the resin bed. Factors affecting the uptake of Cu onto the ligand-impregnated resin have been investigated; these include flow rate, pH, column height, presence of other trace elements, major elements, and interfering matrix. The RPEC–NAA method has been validated by analyzing NIST and NRC water reference materials.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The chemical states as well as levels of certain toxic elements in potable and natural waters are of great concern. Some of them could be toxic even at low concentrations. Their measurements with high precision and accuracy require the development of reliable methods. In the case of seawater, the determination of trace elements by neutron activation analysis (NAA) is further complicated by the presence of major as well as interfering elements such as Na and Cl. As a result, pre-irradiation separation methods have become necessary for not only concentrating the elements of interest but also removing the interfering elements. Chromatographic techniques are generally preferred for the preconcentration of trace elements in aqueous samples because of their simplicity, efficiency, and ease of operation [1–3].
The RPEC techniques make use of a chelating agent bound to an inert support. The chelating agent may be either immobilized via a chemical bond to the resin or impregnated onto the porous surface of the support [4–7]. Of these two, ligand-impregnation appears to be more attractive. The synthesis of ligand-immobilized resins is generally more difficult and is limited in the variety of ligands which can be used [8]. Conversely, ligand-impregnated resins offer a greater choice of ligands for various applications, and the synthesis of such resins is typically rather facile. Pyrocatechol violet [9], EDTA [10], tributylphosphate [11], 8-hydroxyquinoline [6, 7], 1-nitroso-2-naphthol [12], thionalide [13], dithizone [5], 2-mercaptobenzo-thiazole [14], and 1-(2-pyridylazo)-2-naphthol [15] have been adsorbed or immobilized onto a variety of supports for use in RPEC.
Copper is the element of interest in the present study. Over the years we have developed several PNAA methods for its determination in water samples [16–21]. The chelating agent 1-(2-Thiazolylazo)-2-naphthol, abbreviated as TAN, has stability constants of the order of 108–1010 for metals such as Ag, Cd, Co, Cu, and Zn [22]. In the work reported here, we have employed Amberlite XAD-4 resin impregnated with TAN for the determination of Cu in water samples using RPEC–NAA.
Experimental
Reagents and apparatus
All chemicals we used in this work were of analytical grade or better quality. We purchased Amberlite XAD-4 and XAD-7 resins, 2-(N-morpholino)-ethanesulfonic acid (MES), piperazine-N,N′-bis(2-ethanesulfonic acid) (PIPES), tris(hydroxymethyl)-aminomethane (TRIZMA base) and N-(2-hydroxyethyl)-piperazine-N′-(2-ethanesulfonic acid) (HEPES) from the Sigma Chemical Co. We also purchased ultrapure grade nitric acid, acetic acid, and ammonia from Seastar Chemicals, plasma emission grade elemental standard solutions and copper wires from SPEX, and TAN from TCI American.
We prepared deionized distilled water (DDW) by passing quartz-distilled water through a mixed-bed ion-exchange column.
We prepared a stock solution of Cu standard in 0.2 M ultrapure nitric acid by adding the required amount of the elemental standard solution and diluting it to 100 mL. For most experiments, we used 200-μL of this solution containing 15 μg of Cu.
We cleaned Amberlite XAD-4 resin by successive washings for 1 h each on a wrist-action shaker with 2 M HNO3, DDW, 1 M aqueous NH3 solution, DDW, 3 M HNO3, and finally rinsed thoroughly with DDW. We washed the product with acetone, dried at 100 °C overnight, and stored. We determined the elemental impurities of the cleaned resin by INAA and found it to be free of the trace elements of interest. We first cleaned the Amberlite XAD-7 resin by a similar method and then stored in DDW.
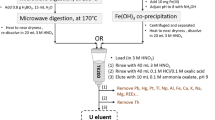
The column apparatus we employed in this study is shown in Fig. 1. We assembled it using a 10-cm long Bio-Rad Econo-Column (Stock No. 737–1011) with a 7-mm internal diameter. We fitted the bottom of the column with a fluoropolymer frit and the top with a Luer lock connector. We controlled the flow rate using a Teflon stopcock. The 100-mL Nalgene polyethylene separatory funnel on the top served as a reservoir for feeding the sample solutions to the column. We connected the separatory funnel to the head of the column with Tygon tubing. We washed all components of the column with soap and tap water, rinsed with dilute nitric acid and DDW, and air-dried prior to use.
Preparation of ligand-impregnated XAD-4 resin
We prepared the TAN-impregnated XAD-4 (TAN-XAD-4) resin by equilibrating 10 g of precleaned resin with 500 mL of 0.05 M TAN solution in methanol on a wrist-action shaker for 2 h. After equilibration, we filtered the TAN-XAD-4 resin, washed it with DDW, and stored in DDW. We evaluated the loading of TAN onto the resin indirectly via its sulfur content by assaying the 3,102-keV photopeak of 37S using NAA. We determined the concentration of the chelating agent coated on the resin to be 0.38 ± 0.02 mmol per g of resin.
General preconcentration procedure
We slurry-packed the TAN-XAD-4 resin into the columns to a height of 5 cm. We used a glass rod to stir and eliminate air bubbles from the column. We then preconditioned the columns by passing 50 mL of buffer solution of appropriate pH (same as that for the sample) at a rate of 1 mL min−1. We fed the sample solution through the separatory funnel and passed through the column. We then washed the column apparatus with 3 × 10-mL portions of the buffer solution. We drained the column completely and dried it by passing pressurized air. We then transferred the resin beads to a polyethylene irradiation vial and shook it gently to ensure homogeneity and reproducible sample geometry for counting. We then heat-sealed the vial for irradiation.
Column capacity
To determine the capacity of the TAN-XAD-4 resin, we loaded the resin onto a column preconditioned to pH 6.00 with MES-aqueous NH3 buffer. We prepared a stock Cu solution by dissolving an appropriate amount of Cu wire in nitric acid and diluting it to 1 L with DDW. We determined the Cu concentration to be 0.322 ± 0.006 mg mL−1 by spectrophotometric titration with EDTA. We took a 50-mL portion of this Cu solution, adjusted its pH to 6.00 before passing it through the column at a rate of 1 mL min−1. We flushed the excess Cu(II) off the column with 4 × 10-mL portions of the pH 6.00 buffer. We analyzed the resin for Cu by NAA and the effluent for unretained Cu(II) by titration with 0.01 M EDTA using murexide indicator.
Comparator standards
We prepared a series of comparator standards by pipetting 200-μL aliquots of the stock solution onto 0.70-g portions of the precleaned resin placed in precleaned polyethylene vials. We thoroughly mixed the contents of the vials by shaking to ensure the same constant geometry as the samples.
Irradiations and counting
We used an inner pneumatic site of the Dalhousie University SLOWPOKE-2 reactor with a neutron flux of 5 × 1011 cm−2 s−1 for all irradiations. In order to determine the retention capacity of the TAN-XAD-4 resin for Cu(II), we irradiated the resin samples of identical geometry from replicate measurements for 30 s and counted for 10 min after a decay period of 10 min. For all other experiments, we used a scheme of 10-min irradiation, 1-min decay, and 10-min counting periods. We irradiated and counted the samples and comparator standards under identical conditions and geometries.
We employed a 60 cm3 APTEC Ge(Li) detector with a resolution of 1.9 keV at the 1,332-keV photopeak of 60Co, a peak-to-Compton ratio of 35:1, and an efficiency of 9.5 % in conjunction with an APTEC multichannel analyzer. We used the 1,039-keV photopeak of 66Cu (half-life = 5.10 min) to assay Cu content of the irradiated samples.
Results and discussion
Effect of pH
We investigated the effect of pH on the quantitative recovery of Cu using the TAN-XAD-4 resin. We spiked a 100-mL sample of 0.05 M buffer solution with a 200-μL aliquot of the standard Cu solution and re-adjusted the pH to the desired value. We then passed this solution through a column of TAN-XAD-4 resin preconditioned to the same pH. We investigated the recovery of Cu over the pH range 2.4–8.5. The results are shown in Fig. 2. It is evident that Cu was quantitatively recovered between pH 4.5 and 8.0. For all further experiments, we selected a pH of 6.00 as optimum while investigating other parameters.
Effect of column height
We varied the column height to determine the minimum amount of resin required to obtain a quantitative recovery of Cu. After the extraction and washing, we transferred the resin to a polyethylene vial. We added precleaned XAD-4 resin to the vial to normalize the sample height and shook the contents of the vial gently. The results of this study are shown in Fig. 3. We found that as little as 280 mg of resin (corresponding to 20 mm of column height) was enough to ensure quantitative recovery of Cu at levels typical of natural water samples. We then chose a column height of approximately 5 cm corresponding to a mass of about 0.70 g of dry resin for all other experiments.
Column capacity
We determined the capacity of this resin for Cu(II) to be 8.0 ± 0.2 mg Cu per gram of resin. Copper concentrations in natural water samples are typically of the order of μg L−1, so the saturation of this column by Cu present in natural waters should not be a problem.
Effect of other trace elements
We investigated the effect of other trace elements present in the sample on the recovery of Cu by preparing a multielement standard solution containing μg quantities of Co, Mn, Th, U, and V in addition to Cu. We observed that these trace elements did not hinder the quantitative recovery of Cu. Furthermore, the 1,039-keV photopeak used for assaying 66Cu was not affected by the presence of gamma-rays from the nuclides of the above elements.
Effect of selected major interferents
We prepared a solution containing Ca, Mg, Na, Br, and Cl such that a 10-mL spike of this solution in a 100-mL sample would simulate the elemental concentrations in seawater. We prepared the samples at pH 4.50, 6.00, and 8.00 to evaluate the effect of these interfering elements. We found that 0.4 M NaCl, 0.005 M Ca, 0.001 M Br, and 0.05 M Mg had no effect on the uptake and quantification of Cu. The results of four replicate runs at pH 6.00 are presented in Table 1.
The presence of Mg in the irradiation vials made it impossible to obtain reproducible results for the uptake of Mg on the TAN-XAD-4 column. However, neither the extraction of Cu nor the assay of the 1,039-keV photopeak of 66Cu was hindered by the presence of Mg and its 27Mg nuclide.
Effect of flow rate
We varied the flow rate over the range of 0.3–5.0 mL min−1. We concluded that flow rates as high as 5.0 mL min−1 had no adverse effect on the recovery of Cu at pH 6.00. The results of this study are given in Table 2. We checked the presence of TAN in the effluent spectrophotometrically to estimate the amount of TAN bleeding off the column. We observed that there was practically no bleeding of the chelating agent into the mobile phase even at the elevated flow rates.
Recommended preconcentration procedure
We slightly modified the preliminary method quoted earlier [23, 24] to develop the recommended procedure. We carried out a radiotracer experiment to evaluate the entire procedure. We used a 2-mL sample of the Cu solution in the column capacity study and irradiated it for 2 h to produce 66Cu and 64Cu. After a suitable time period to allow for the decay of 66Cu, we diluted the activated copper solution to 100 mL, producing a solution of 6.36 μg mL−1 Cu. We added a 1,000-μL aliquot of this solution to a 100-mL portion of DDW adjusted to pH 6.00 with MES-NH3 aqueous buffer. We then passed this solution down a column of TAN-XAD-4 using the optimized procedure. We assayed the concentration of Cu in these experiments using the 1,346-keV photopeak of 64Cu. The extraction of Cu onto the TAN-XAD-4 resin column was 101 ± 2 %. We also counted the effluents to check for the activity of 64Cu which might have passed through the column. Results from replicate measurements of the effluent showed no detectable activity of 64Cu, verifying that all of the Cu was extracted onto the resin.
Analysis of blanks
Each set of experimental runs was coupled with a blank run, in which we subjected an appropriate volume of the buffer solution to the same preconcentration procedure as the samples. We did not find any detectable amount of trace element contaminants in the blank.
Validation of the method
We evaluated the reproducibility of the method by analyzing six replicates of 100-mL size DDW samples spiked with 15 μg of Cu. We calculated the imprecision as the percent relative standard deviation (% RSD) of six replicates, and found them to be less than ±6 %.
The RPEC–NAA method was validated by analyzing four reference materials. We purchased the standard reference material (SRM) 1643a (Trace Elements in Water) from the US National Institute of Standards and Technology (NIST), and three natural water reference materials, namely SLEW-1 (estuarine water), SLRS-1 (riverine water), and NASS-2 (seawater) from the National Research Council (NRC) of Canada. We took two 1-L aliquots of NASS-2 and three 250-mL aliquots of the other reference materials, adjusted their pH values to 1.00 with nitric acid, digested them at 80 °C for 20 min, and passed them through columns of XAD-7 to remove any dissolved organic materials. We then readjusted the sample pH values to 6.00 with ultrapure ammonia solution in presence of MES buffer and passed them through a column of TAN-XAD-4. We calculated the recoveries of Cu and the results are given in Table 3. We found good agreements between the experimental and certified values.
We used the procedure described by Currie [25] for calculating the detection limit, LD = 2.71 + 4.65√μB where μB is the number of counts in the blank. We found the absolute detection limit for Cu as 0.106 μg which is considered very low.
Tap water samples
We analyzed eight 1-L samples of Halifax city tap water by following the preconcentration procedure given above for the reference materials. Then we measured the Cu levels by NAA, as previously described. The results are presented in Table 4. The concentration of Cu in the first set of six tap water samples was 11.5 ± 0.6 μg mL−1. We also analyzed tap water samples from seven other sites around Halifax, with fewer replicates. The Cu concentrations ranged from 11.5 to 83 μg L−1. These values are well below the maximum acceptable concentration value of 1.0 mg L−1 for Cu in drinking water according to Health Canada [26].
Conclusions
We found the extraction of Cu from water samples by the RPEC–NAA method to be fast, simple, reliable, and cost-effective. Copper is quantitatively extracted onto the TAN-XAD-4 resin over a wide pH range, and can be assayed even in the presence of large excess of major elements such as Na, Ca, Mg, Br and Cl. We concluded that this procedure is amenable to neutron activation in that analysis can be performed directly on the solid resin rather than eluting the chelated elements prior to instrumental determination. This step circumvents problems, such as the danger of contamination and/or the possibility of irreproducible as well as incomplete recovery, commonly associated with an elution step. By concentrating a 1-L sample onto 2 cm3 of resin we achieved a concentration enrichment factor of 500.
References
Leyden DE, Wegschieder W (1983) Anal Chem 53:1
Van Grieken R (1982) Anal Chim Acta 143:3
Maccarthy P, Klusman RW, Cowling SW, Rice JA (1991) Anal Chem 63:301-R
Gennaro MC, Mentasti E, Sarzanini C (1986) Polyhedron 5:1013
Chikuma M, Aoki H (1993) J Anal At Spectrom 8:415
Abollino O, Mentasti E, Porta V, Sarzanini C (1990) Anal Chem 62:21
Sturgeon RE, Berman SS, Willie SN, Desaulniers JAH (1981) Anal Chem 53:2337
Isshiki K, Tsuji F, Kuwamoto T, Nakayama E (1987) Anal Chem 59:2491
Brajter K, Olbrych-Sleszynska E, Staskiewicz M (1988) Talanta 35:65
Guedes Da Mota MM, Jonker MA, Griepink B, Fres Z (1979) Anal Chem 296:345
Bark LS, Duncan G, Graham RJT (1967) Analyst 92:347
Ghosh JP, Das HA (1981) Talanta 28:274
Matsui M, Matsumoto K, Terada K (1987) Anal Chim Acta 193:381
Satake M, Ishida K, Puri BK, Usami S (1986) Anal Chem 58:2502
Braun T, Abbas MN (1980) Anal Chim Acta 119:113
Milley JE, Chatt A (1987) J Radioanal Nucl Chem 110:345
Rao RR, Goski DG, Chatt A (1992) J Radioanal Nucl Chem 161:89
Rao RR, Chatt A (1993) J Radioanal Nucl Chem 168:439
Beazley PI, Rao RR, Chatt A (1994) J Radioanal Nucl Chem 179:267
Perez-Gramatges A, Chatt A (2012) J Radioanal Nucl Chem. doi:10.1007/s10967-012-1708-z
Goodwin WE (1997) Ph.D. Thesis, Dalhousie University, Halifax, NS, Canada
Sillen LG (1971) Stability constants of metal-ion complexes. The Chemical Society, Burmington House
Goodwin WE, Rao RR, Chatt A (1994) International topical conference methods and applications of radioanalytical chemistry (MARC-III) Abstracts Booklet, Kona, HI, USA
Goodwin WE, Rao RR, Chatt A (1999) Trans Am Nucl Soc 80:59
Currie LA (1968) Anal Chem 40:586
Federal-Provincial Working Group on Drinking Water (1989) Guidelines for Canadian drinking water, 4th edn. Canadian Government Publishing Centre, Ottawa
Acknowledgments
The authors would like to thankfully acknowledge the cooperation of the Dalhousie University SLOWPOKE-2 Operations Group; financial assistance from the Natural Sciences and Engineering Research Council (NSERC) of Canada for Research Operating, Discovery, and Major Facilities Access grants; and the award of a Sumner Fellowship to WEG.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Goodwin, W.E., Rao, R.R. & Chatt, A. Reversed-phase extraction chromatography–neutron activation analysis (RPEC–NAA) for copper in natural waters using Amberlite XAD-4 resin coated with 1-(2-thiazolylazo)-2-naphthol. J Radioanal Nucl Chem 296, 489–494 (2013). https://doi.org/10.1007/s10967-012-2056-8
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-012-2056-8