Abstract
The ability of the monoclonal antibody TU-20 and its scFv fragment to bind specifically to the C-end of the class III β-tubulin makes these substances useful as potential diagnostics for neurodegenerative diseases—especially peripheral neuropathies. TU-20 and its scFv were labeled with 125I and 123I by chloramine-T (with radiochemical yield 75 and 50%, respectively). Radiochemical purity and stability was revealed by gel filtration (decrease to 80 and 50% in 2 months, respectively). Immunoreactivity of the labeled TU-20 was determined by ELISA—the range of the preserved immunoreactivity varies from 60 to 95% in accordance to the used radiolabeling process. RIA and affinity coupling analytic methods were specifically designed with focusing on specifics of the antibody and its fragment. The results of RIA differ in depandance on the type of the reaction vessel (glass or polystyrene) and the affinity coupling results depend on the experimental arrangement—in the batch or on the column. Fragmentation of the labeled antibody and its fragment was estimated by bis–tris gel electrophoresis followed by silver staining and autoradiography (over 95% of radioactivity bound in the substances). The antibody binding in tissue slices was studied in vitro by immunohistochemistry. The Purkinje cells were observed conjugated with the radiolabeled substances, either TU-20 or its ScFv fragment in the area of the cerebellum. In vivo biodistribution of 125I-TU-20, 125I-scFv TU-20, 123I-scFv TU-20 and Na125I was proceeded in normal mice (wild type C57B/6/J). Both biomolecules labeled by 123I were also proved in an imaging biodistribution study with use of the SPECT camera. Finally, a transgene population G93A1 Gur was used for comparative study to show the different behaviour of the substances in a normal mouse and in the modified organism with amyotrophic lateral sclerosis. The most part of differences is observed in the area of the muscles, rostal and caudal spinal cord. In summary, the monoclonal antibody TU-20 and its scFv were successfully radioiodinated and afterwards analysed by several quality control methods and biodistribution studies which confirmed their preserved or expected immunoanalytical characteristics in normal and genetically modified organism.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The principal goal of the investigation monoclonal antibodies and their fragments was to examine the possibility of developing of an imaging radiotracer that would be specific for cytoskeleta of destructed dendrites and neuronal bodies. One of the suitable fitting marker, characteristical for neuronal tissue, performs anti III β-tubulin (βTcIII) antibody—TU-20 with molecular weight 150 kDa and its scFv fragment with molecular weight 27.7 kDa [1]. The scFv fragment of TU-20 was synthesized for its higher mobility through tissue and vascular barriers. Biochemical characteristics (especially immunoafinity) of the specific binding substance—anti III β-tubulin scFv fragment—is preserved, and, moreover, the biological availability is much better than in case of the whole antibody.
To examine this hypothesis, it is necessary to radiolabel both substances with 125I and 123I. The next step is chemical analysis and, furthermore, biochemical properties are extensively investigated. The quality control, performed by gel filtration, electrophoresis, ELISA testing determines adequate properties of the radiolabeled substances for further studies. Affinity coupling and RIA analytic methods occur under development with focusing on specifics of the antibody and its fragment behavior [2]. In vitro experiment shows an extent of the preserved binding specificity of the species by incubation of the both radiolabeled substances with mice brain slices followed by an autoradiography. The in vivo biodistribution confirmes the elimination of the radiolabeled TU-20 and scFv from mice. The bi-exponential model for two-phase clearance to determine short phase half-life t1/2α and long phase half-life t1/2β values is used. For comparative study, a transgene population G93A1 Gur was chosen to show different behavior of the substances in normal mouse and in modified organism with amyotrophic lateral sclerosis (ALS).
The main objective of this work is to develop a method for direct imaging of the structural degradation of peripheral neurones by various types of neuropathies.
Methods and materials
The monoclonal antibody TU-20 and its scFv was purchased from Exbio, CZ. The antibody recognizes the peptide sequence ESESQGPK. ScFv TU-20 is a recombinant protein expressed in E. coli [1].
125I (T1/2 = 59.4 h) radioiodination of TU-20 and scFv TU-20 was performed via chloramine-T with or without stopping reaction with thiosulfate agent in the phosphate buffer saline (PBS) pH 7,4. The ratio of an amount of TU-20 to radioactivity was 1 μg to 5.5–7.0 MBq of 125I. The ratio of an amount of the fragment to radioactivity was 1 μg to 1.5–2.0 MBq of 125I.
123I (T1/2 = 13.3 h) radioiodination of the fragment scFv TU-20 was performed via chloramine-T with stopping reaction with thiosulfate in PBS. The ratio of an amount of the fragment to radioactivity was 1 μg to 3–5 MBq 123I.
In both cases, at the end of labeling, the reaction mixture was loaded on the top of a BSA-blocked polyacrylamide desalting column with an exclusion limit 6 kDa. Fractions were eluted with 0.1% BSA in PBS and measured for radioactivity.
The immunoreactivity of the radiolabeled monoclonal antibody TU-20 was determined by an enzyme linked immunosorbent assay (ELISA) using the commercial set for detection of mouse anti-β III tubulin antibodies from VIDIA, CZ [2, 3].
Affinity coupling was develop by use the basic matrix activated Sepharose 4 Fast Flow by Pierce which was modified specific binding octapeptide. RIA analytic method was developed in two modifications of surface of the reactive vessel.
Stability of the radiolabeled TU-20 and its scFv TU-20 was investigated on 4–12% Bis–Tris gel electrophoresis. Protein bands were visualized by staining the gels with Silver Stain Plus. 125I-labeled scFv fragment was processed by autoradiography exposing plate BAS-SR 2025, and finally developed by BAS-1800II. Autoradiographs were evaluated by AIDA 2.0 software.
Preserved binding properties of the radiolabeled MAb or scFv for neuronal tissue were confirmed by the “method of double labeling”. It is based on the immunohistochemistry and autoradiography of the brain tissue slices. The 50 μm thick brain slices from “wild type” mouse (C57B/6/J) were incubated with the radiolabeled TU-20. The second incubation was performed with anti-mouse IgG polyclonal antibody conjugated with horseradish peroxidase (Sigma–Aldrich, USA). Afterwards, the immunohistochemistry was finalized by staining with 3,3′-diaminobenzidine that revealed the neuronal structure [4–6].
The in vivo biodistribution was carried out with the male normal mice—“wild type” C57B/6/J. Biodistribution studies were performed following an i.v. injection. The main focus is intended for scFv fragment due to its better mobility in organism [7].
125I-labeled scFv fragment, for comparison with the biodistribution of Na125I, was applied in amount of 50 kBq/50 μl. 123I-labeled scFv fragment was injected in amount of 200 kBq/50 μl. Mice were sacrificed at designated times points in groups by three animals. The kinetic time intervals were: 3, 6, 12, 24, 48, 72, and 144 h for 125I-labeled scFv TU-20 fragment and 0.5, 1, 2, 3, 6, and 12 h for 123I-labeled scFv fragment. Blood and major organs (included thyroid gland, kidneys, lung, heart, brain, spleen, muscle, fat, skin, gallbladder, testicles, stomach, liver, small intestine, and colon) were removed, weighed, and counted in a gamma scintillation counter to determine the %ID/g (percentage of injected dose per gram) for each radiolabeled substance. Blood clearance data for 125I-labeled scFv fragment were obtained by analyzing blood samples by using a bi-exponential model for two-phase clearance to determine short phase half-life t1/2α and long phase half-life t1/2β values [7, 8].
[123I]scFv TU-20 and [123I]TU-20 behavior in mice (“wild type” C57B/6/J) was observed by use of the SPECT camera. Kinetic intervals were 0.5, 1, 2, and 3 h by [123I]scFv TU-20 and 1, 2, 3, and 6 h by [123I]TU-20.
Transgene population G93A1 Gur was used for comparative study to show different behavior of the substances in normal mouse and in modified organism with ALS. Biodistribution kinetic intervals were 3 h (125I-scFv) and 6 h (125I-TU-20) [9, 10].
Results
TU-20 and its scFv were labeled with 125I and 123I by chloramine-T (with radiochemical yield 72 and 50%, respectively). Radiochemical purity and stability was revealed by gel filtration (decrease to 80 and 50% in 2 months, respectively).
Fragmentation of the labeled antibody and its fragment was estimated by bis–tris gel electrophoresis followed by silver staining and autoradiography (over 95% of radioactivity bound in the substances), see Fig. 1.
Affinity coupling and RIA adaptation for the specific conditions showed 10–30% preserved immunoreactivity of the labeled compounds. Otherwise, these methods carry out quite high discrepancy and it will be necessary to provide further optimising search.
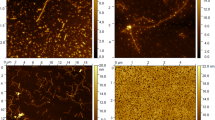
In vitro studies performed on mice brain slices confirmed several important assumptions. The antibody is preferentially bound in the layer of Purkinje cells in the cerebellum, see Fig. 2.
SPECT camera in vivo experiment deals with these results: activity bound in scFv is primarily distributed to the thyroid gland and digestive tract, then passes quickly through kidneys, see Fig. 3. Distribution images of the labeled TU-20 provides ambiguous because the substance is accumulated in the chest and ventral part and image resolution do not afford more detailed biodistribution identification. However, it is known from previous biodistribution preparative study that activity is distributed in lung, heart, liver, stomach and colon in first 6 h. The t1/2α values for 125I-labeled TU-20 was calculated from the previous study as 8.6 h and, respectively, in addition, the t1/2β value was calculated as 150.7 h [11].
In vivo experiments were focused on investigation of the blood clearance and organ distribution of the radiolabeled TU-20 and scFv fragment in mice. Let’s show especially the results from scFv biodistribution study in preference, see Figs. 4 and 5. It was verified that the major part of activity, according to the amount of the labeled scFv fragment, was eliminated from blood during 2–3 h. Minor part of activity, according to the amount of the labeled scFv fragment (0.5–1.0%), was kept in the blood for some days. The value t1/2α for 125I-labeled scFv fragment was calculated as 2.3 h and the t1/2β was estimated as 62.4 h.
The half-life for overall elimination of Na125I from blood was 4.5 h. In comparison, we found that the 125I-labeled scFv fragment uptake in thyroid gland appeared much lower than for Na125I, as expected. The t1/2α value for 123I-labeled scFv fragment was calculated as 1.4 h, but the long phase elimination half-life t1/2β was not estimated due to short half-life of the isotope 123I.
The radiolabeled scFv fragment passed in general through the digestive tract (stomach and intestine) and finally was eliminated through kidneys in preference TU-20 and ScFv TU-20 showed suitable properties for further investigation in animals which are genetically modified mutants with the ALS. Comparing biodistribution experiments in modified organism confirmed expected behavior. The most significant biodistribution differences occured in the area of the limbs and caudal part of spinal cord and spine.
Conclusion
TU-20 and its scFv fragment were successfully labeled with radioiodine 123I and 125I, and, subsequently, the biochemical and analytical characteristics were investigated. Biological properties of the radiolabeled TU-20 and its scFv were evaluated in vivo by biodistribution studies. The expected behavior of biomolecules during their elimination was observed—much faster kinetics and better in the case of the scFv fragment, but, on the other hand better in vivo stability in the case of the whole antibody TU-20. Furthermore, the elimination parameters were calculated—the half-life of the both phases in the biexponential elimination model. 125I-labeling of the TU-20 and its scFv is very suitable for investigation of the radiolabeled antibody fragment behavior and properties due to the long 125I half-life. On the other hand, 123I-labeling of the scFv fragment TU-20 is intended for practical imaging at SPECT camera.
In summary, TU-20 shows better immunospecific behavior in organism together with slower kinetics, on the other hand, scFv TU-20 reveales worse immunospecific characteristics in combination with much faster kinetics.
References
Dráberová E, Lukáš Z, Ivanyi D, Viklický V, Dráber P (1998) Expression of class III β-tubulin in normal and neoplastic human tissues. Histochem Cell Biol 109:231–239
Fraker PJ, Speck JC (1978) Protein and cell membrane iodinations with a sparingly soluble 1,3,4,6-tetrachloro-3α, 6α-diphenylglycouril. Biochem Biophys Res Commun 80:849–857
Holliger P, Hudson PJ (2005) Engineered antibody fragments and the rise of single domains. Nat Biotechnol 23:1126–1136
Robles AM, Balter HS, Oliver P, Welling MM, Pauwels EK (2001) Improved radioiodination of biomolecules using exhaustive chloramine-T oxidation. Nucl Med Biol 28:999–1008
Chizzonite R, Truitt T, Podlaski FJ, Wolitzky AG, Quinn PM, Nunes P, Stern AS, Gately MK (1991) IL-12: monoclonal antibodies specific for the 40 kDa subunit block receptor binding and biologic activity on activated human lymphoblasts. J Immunol 147:1548–1556
Hamilton S, Odili J, Wilson GD, Kupsch JM (2002) Reducing renal accumulation of single-chain Fv against melanoma-associated proteoglycan by coadministration of l-lysine. Melanoma Res 12:373–379
Katsetos CD, Herman MM, Mörk SJ (2003) Class III β-tubulin in human development and cancer. Cell Motil Cytoskeleton 55:77–96
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Heiman-Patterson TD, Deitch JS, Blankenhorn EP et al (2005) Background and gender effects on survival in the TgN(SOD1–G93A)1Gur mouse model of ALS. J Neurol Sci 236(1–2):1–7
Naini A, Mehrazin M, Lu J, Gordon P, Mitsumoto H (2007) Identification of a novel D109Y mutation in Cu/Zn superoxide dismutase (sod1) gene associated with amyotrophic lateral sclerosis. J Neurol Sci 254(1–2):17–21
Svecova H et al (2008) Radioiodination of mouse anti-III β tubulin antibodies and their evaluation with respect to their use as diagnostic agents for peripherial neuropathies. Appl Rad Isot 66(3):310–316
Acknowledgments
This work was supported by the projects No. E!3177—DIAGIM (1P040E167) and E!2510—NEUROTUB (0E91) of EUREKA, and by the project No. IBS1048301 of Grant Agency CAS.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kleinova, V., Chaloupkova, H., Svecova, H. et al. Radioiodination and biodistribution of the monoclonal antibody TU-20 and its scFv fragment. J Radioanal Nucl Chem 286, 847–851 (2010). https://doi.org/10.1007/s10967-010-0856-2
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-010-0856-2