Abstract
Hygrothermal ageing of polyamide 6 (PA6) and polyamide 6 reinforced with 30 wt% of glass fibers (PA6GF30) was undertaken. Immersion was conducted in distilled water at 90 °C and 100% relative humidity (RH) for up to 80 days (1920 h). Results revealed a noteworthy decrease either in glass transition temperature Tg or in tensile properties, at early stage of ageing, for both studied materials. This decline was mainly caused by the plasticization effect of water and the weakness of the interfacial interactions leading as a consequence to a loss of adhesion between fiber and matrix. Afterwards, physical and mechanical properties decrease monotonically testifying the occurrence of exhaustive damages and chemical reaction phenomena. Such phenomena were yellowing and crazing formation which were observed for both materials after 1920 h of conditioning. The former is caused by the thermo- oxidation whereas the latter results from the release of internal stresses induced by water sorption. These chemical reactions were monitored by infrared spectroscopy. Thus, an increase of the free N-H stretch and the carbonyl groups (imides) was noted. Accordingly, it seems that long term immersion in distilled water at high temperature induces chemical reactions which indicate the severity of the damage.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The use of polyamide (PA) materials knows an astounding climb in many engineering components and structures. In particular PA-6 and PA6,6 are the most investigated polyamide materials. They show strong hydrogen bondings (H-bonding) which are typical in their chemical structure. Such characteristic ensures their particular mechanical performance and increases their widespread use in automotive industry. Hence, they are more often used for parts located in engine compartments and interior equipment of several car models [1,2,3]. However, these materials sustain several loads and different types of environmental conditions that attribute to their ageing in their particular environment. According to the severity of these conditions (high and/or low temperatures, water attack, UV exposure, saline atmosphere), different mechanisms may occur separately or simultaneously and decrease their performance and durability.
Because the prediction of the durability of polyamide materials is a very important issue for designers and users, numerous researches have been devoted to study their hygrothermal ageing [4,5,6,7,8,9]. According to the moisture-related works on these materials, PA 6 and PA 6–6 are sensitive to moisture. Thus, they absorb more than 8 wt% in 100% relative humidity (RH) at room temperature [10] due to the presence of polar functions and H-bonds [8, 11, 12]. Eventually, the sorption mechanism occurs preferentially into the amorphous regions of the polymer [7, 13] affecting as a consequence its physical, chemical and mechanical properties [14,15,16]. These effects arise through different mechanisms such as plasticization, crazing, hydrolysis, and swelling. Plasticization and swelling are the most important physical changes that may occur through the interaction of the water molecules with the hydrogen bonds [17]. These interactions increase the chain mobility inducing as a consequence a lowering of the glass transition temperature (Tg) [14] of about 65–93 °C [10].
Placed in humid condition, water can interpenetrate between -NH and C = O groups of the polymer and break the pre-existent bonds leading to a loss of its mechanical cohesion [6, 14, 15]. Thus, the tensile properties of polyamides have been further reduced when the samples are moisture conditioned as reported by Refs [14, 15, 17]. Thereby, the decrease of the stiffness in the presence of water is common to all polyamides to a certain degree. Nonetheless, for composite materials, the loss of mechanical properties is not only related to the plasticization effect of water but also to the degradation of the fiber-matrix interface. Thus, this region is extremely damaged after water absorption [6, 16, 18]. Afterwards, a severe degradation of the mechanical properties takes place. Thence, the water absorption lowers the interfacial stress transmissibility. It seems that the interface fiber/matrix was considered as a privileged site for the water molecules accumulation during the diffusion process [19, 20]. Therefore, its efficiency is critical for both short term properties and long term durability which limits its structural performance [21].
The macroscopic properties of most polyamides were affected by hygrothermal ageing after few months, or even weeks. No doubt, this tendency is considered as the most serious limitations of these materials. Numerous investigations are interested to study the effect of early stage of conditioning on mechanical behavior of such materials [4, 7,8,9,10,11,12,13,14,15,16,17,18], albeit poorly studied the long term exposure. Commonly, they have revealed that the degradation mechanism occurs at early stage of ageing leading to a loss of the mechanical characteristics. But what about the assessment of their performance after long term of conditioning? Otherwise, do their properties fall catastrophically or they keep a steady decrease? Are there other phenomena that can occur and affect the structural strength of polyamides such those found by Simar et al. [22] and Mezghani et al. [23] for other materials? To fulfill this requirement, this experimental investigation was undertaken. Whence, this paper aims to focus on the complete assessment of the performance of PA6 and PA6GF30 as well as the discernible damages that may appear under immersion in distilled water at 90 °C for long period. Initially, a comparison between the neat polymer’s behavior and its composite towards water during the ageing process will be established. Afterwards, optical and morphological observations were conducted to identify the surface damages of these materials under hygrothermal ageing. Then, physicochemical, mechanical and morphological properties were studied in order to ascertain the impact of the detected damages on the bulk properties of studied materials.
Experimental procedure
Materials
A commercial grade of polyamide 6 (PA6) ‘designed ALYAMID C2122’ and its composite reinforced with 30 wt% of glass fibers (PA6GF30) ‘commercially designed ALYAMID C2122 GF30’ supplied by POLYMERES SA, were used in the current study. These materials were injection molded in accordance with the ISO standard 527–2 type 1A.
Prior to testing, samples were dried at 70 °C for 24 h and immediately stored in vacuum desiccators.
Conditioning
Accelerated ageing was carried out in distilled water at a set temperature of 90 °C for 80 days in glass jar containers. The temperature was chosen in order to accelerate the diffusion process as well as to accentuate the materials degradation mechanism. Samples were fully immersed and periodically weighted using a digital balance of 0.001 g accuracy. Each material was taken off twice for testing; at equilibrium state and at the end of ageing. Thus, the former characterization was after 69 h and 87 h for PA6 and PA6GF30, respectively. Then, the second one was after 1920 h (80 days) for both materials.
The moisture uptake M (t) was measured through gravimetric method as following in Eq.1:
Where m0 and mt are respectively the dry weight and the wet weight of the sample after any specific time t.
In order to measure the water uptake only in the composite matrix (without taking into account the weight of the fiber), Carrascal et al. [8] and Ledieu [24] have suggested Eq. 2, where WGF is the fiber weight fraction.
Methods of characterization
Dynamic-mechanical analysis (DMA):
DMA measurements were performed on 8000 Perkin Elmer analyzer before and after ageing. Running a frequency scan of 1 Hz, samples were tested at a heating rate of 3 °C/min through a temperature range between −50 °C and 180 °C. The glass transition temperature (Tg) was settled by the damping peak (tanδ). Samples dimensions were 51x10x4 mm3.
Infrared (IR) spectroscopy analysis
Tests are realized with Agilent Cary 630 FTIR spectrophotometer at room temperature. The samples were scanned from 650 cm−1 to 4000 cm−1.
Tensile tests
An universal testing machine model WDW-E with a capacity of 10 kN was used to perform the tensile tests. Samples were tested according to ISO 527 procedure at a constant crosshead speed of 5 mm/min. Measurements were conducted at room temperature using a longitudinal extensometer with a gauge length of 50 mm.
Scanning electron microscopy (SEM)
After tensile tests, failure features of aged and unaged samples were examined using a scanning electron microscope (VOVA NANO SE 450/650) operating under an accelerating voltage of 10 and 20 kV. Before testing, samples were coated with a thin gold layer.
Results and discussion:
Water uptake
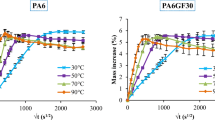
Fig. 1 shows the mass increase of PA6 and PA6GF30 conditioned in distilled water at 90 °C for 80 days versus the square root of time. The plotted curves exhibit the same trend involving two parts;
-
A noteworthy rapid initial water uptake which increases with the conditioning time. This linear part has been associated to the excess of water ingress within the amorphous regions of the polymer. The penetration of water molecule remains until an equilibrium saturation level M (∞) is reached.
-
The equilibrium rapidly disappears and a tendency of mass loss was noticed. This decrease may be ascribed to the polymer degradation and the occurrence of hydrolysis phenomenon [11, 25].
Moreover, it is interesting to note that the moisture content, as derived from the absorption curves (Fig. 1), differs according to the nature of tested material (PA6 or PA6GF30). Under the same ageing conditions, PA6 absorbs more water than PA6GF30. The former’s water uptake reached 8.55% while for the latter it attained only 5.26%. If we assume that only the polymer is the major contributor to the sorption mechanism it seems reasonable that PA6GF30 gets the lowest moisture uptake. Thus, the decrease of polymer contribution induces a decrease of the water uptake. This result is consistent with those reported in previous experimental investigations related to polyamide materials [6, 10, 11]. It has been established that the equilibrium moisture content of nylon 6,6 reached 8 ± 0.8%. Nevertheless, this content decreases with increasing fiber volume fraction [6, 26]. This fact is attributed to the decrease of the volume fraction of the hygroscopic matrix [6].
Furthermore, it can be seen also that water ingress within PA6 samples is faster than into PA6GF30 samples. Thus, the equilibrium of composite material is reached after a while (87 h) compared to the polymer (69 h). Therefore the glass fibers hinder and decelerate the sorption mechanism.
Using Eq. 2, it is possible to examine the matrix mass change during immersion time as shown in Fig. 1. It is obviously seen that at short conditioning time (< 3 h of conditioning) the level of water uptake between the matrix and the polymer are very close. After that, the gap between the curves is slightly increased until 80 days. All over the ageing process the PA6 absorbs more water compared to the matrix. Accordingly, the presence of the glass fibers reduces the ability of the polyamide matrix to absorb the same water content that was absorbed by the polymer in the same environment [27].
According to Fig.1, the water diffusion initially follows a Fickian process. Therefore, the diffusion coefficient ‘D’ was computed using the slope of the plotting (Mt/M∞) versus √t,.
Where e and Ms. denote the thickness of tested sample and the equilibrium saturation weight, respectively.
Results reveal that the lowest value was measured for PA6GF30 samples (Table 1). The obtained values of diffusivity correlate well with those reported by Mohd Ishak and Berry [5, 6] and Vlasveld et al. [14].
Surface damages
The first visual aspect change after hygrothermal ageing was the color change (yellowing) of PA6 (Fig. 2a) and PA6GF30 (Fig. 2b). Both materials reached a maximum level of absorbed water after 69 h and 87 h, respectively (Fig. 2a, b). However, the samples color remains as received before conditioning. After 80 days of ageing (1920 h), yellowness becomes more pronounced for both materials. This phenomenon has been already reported in studies dealing with thermo-oxidative degradation and photo-degradation [26, 28,29,30,31]. According to literature data the yellowing of polyamide materials is mainly caused by the formation of pyrrole derivates [26, 28] or conjugated enal or enone groups as reported in Ref [31]. Due to the degradation process, tested samples move from uncolored ones to yellow and finally turn to black as reported by Rudzinski et al. [28]. According to them, the thermal degradation of the polyamide 66 starts at the sample surface and progresses continuously into the core of the sample. Among thermo-oxidative ageing, sequences of conjugated double bonds can be formed due to the thermal degradation steps. It has been suggested that these bonds gives rise to the chromophore which are responsible for the observed yellowing of PA’s during oxidation [28, 30].
Another finding was observed in water-exposed samples after 1920 h which is the surface crazing (Fig. 3). Crazing occurs homogeneously throughout the material for both cases. This phenomenon occurs either due to the internal stresses induced by the water absorption and/or due to thermal effect [13, 26]. Thus, in related experiments, we tried to separate the combined effect of water and temperature. In this context a thermal ageing upon 90 °C over 1920 h were performed. Here, samples showed only yellowing (almost no crazing) (Fig. 4). Accordingly, water absorption is the main responsible of craze initiation due to internal stresses development. In this context, Pillay et al. [13] noted the presence of cracks on the carbon/nylon 6 composite. These cracks were expanded on the surface after exposure to water at 100 °C for more than 100 h. Likewise, Chevali et al. [26] noticed the presence of microcraking at the surface of NY66 40 LFT exposed to boiling water for the same period. According to them, the development of these cracks may be caused by either the residual stresses from the processing of the composite or other processing anomalies. Moreover, Ledieu [24] showed the formation of a crack network on the PA66GF30 and PA66 aged in water at 130 °C for 700 h.
Does these crazing, which are obviously seen at the surface, extend to the sample’s core? Optical observations were conducted on polished cross sections (Fig. 5). The measurement performed on aged samples at different temperature revealed that, the craze formation is deeper for unreinforced polymer than for reinforced one (Fig. 6) since the former absorbs more water than the latter.
Dynamic-mechanical analysis (DMA)
In order to evaluate the consequences of full immersion in distilled water for 80 days on the glass transition temperature Tg, DMA measurements were conducted. According to Fig.7a, b, at dry state, the Tg of PA6 and PA6GF30 are 52.5 °C and 50.8 °C, respectively. After ageing the glass transition temperature decreases monotonically with ageing time. At early stage of conditioning, damping peak of both materials shifted progressively towards lower temperatures. Thereby, it shifts of approximately 43.1 °C and 42.9 °C for PA6 and PA6GF30, respectively (Fig.7a, b). This reduction can be ascribed to the plasticization effect of water. Thus, under hygrothermal ageing, water molecules are mainly absorbed by the amorphous phase where they interfere with the hydrogen bonds and break the secondary bonds between the polar groups. This induces an increase in their chain mobility and decreases consequently the Tg. This effect on PA6 and other polyamides is widely reported in literature [14, 15]. Afterwards, Tg reaches −2.4 °C for the neat polymer whereas it attains −0.1 °C for the PA6GF30 after 1920 h of exposure. This continuous decrease of Tg with ageing time may assume the occurrence of chemical reaction in tested materials as reported by Simar et al. [22].
Moreover, it can be noticed that the relaxation α is divided in two relaxations (Fig. 7a, b). According to Bergeret et al. [32], these relaxations were attributed to water diffusion respectively within intra-spherulitic (strong linked water molecules) and inter-spherulitic (weak linked water molecules) phases.
Similarly, the decrease of storage moduli for both materials in the 0–50 °C temperature range with time testifies the occurrence of plasticization phenomenon (Fig.7a’b’). This phenomenon is related to the material water uptake as previously mentioned. Simultaneously, an increase in the moduli in the sub-ambient temperature range is obviously noted in Fig.7a’b’. This increase has been attributed to the ability of water to form bonds between chain segments at low temperatures which are sufficiently stable to produce an increase in modulus [33].
Infrared spectroscopy analysis
Infrared spectra of the PA6 and PA6GF30 samples before and after immersion in distilled water at 90 °C for different times are presented in Fig.8. Table 2 summarizes the assignment of IR bands for both materials before ageing. According to Fig.8a, a’, the absorbance intensity related to most bands evolved progressively after ageing. At early stage of immersion, contrarily to PA6GF30, a clear increase of the absorbance intensity of characteristic bands was observed for the PA6 (Fig.8a). However, at long term exposure it rises similarly for both materials. Furthermore, it can be noted that there are peaks which shifted either to lower frequencies (such as amide I) or to higher frequencies (as the bonded N-H stretch and the overtone of amide II). Simultaneously, detectable peaks were obviously noted after 1920 h of conditioning. For instance, the band associated to the water-absorption increase when the time of ageing increases overlapping the N-H stretching bands as reported by Lim et al. [4] (Fig.8b, b’). Thus, a significant increase in the absorbance of the free N-H stretch, as pointed by an arrow (≈3490 cm−1), can be observed. Hence, the intensity of this band rises with exposure time to reach the highest value for samples aged for 80 days. Indeed, the increase in the broad absorption bands in the range 1760–1700 cm−1 resulted from the carbonyl groups accumulated in the polymer chains due to the oxidation process (Fig.8c, c’) [34,35,36,37,38]. Two peaks are detectable after long term conditioning which are located at 1735 cm−1 and 1716/1718 cm−1 (Fig.8c, c’). The former has been related to the formation of imide groups [9, 36, 37]. The latter has been attributed to the formation of diametric acids [36, 37] or ketones [35]. The appearance of these bands suggests that the degradation of studied materials occurs due to immersion in water. Thus, the detectable bands appear only as a weak shoulder at early stage of ageing (69 h and 87 h). However, after long term ageing (1920 h), the absorbance of the carbon groups increases as shown in Fig. 8c, c’ indicating the occurrence of material degradation. [9, 36]. The overall changes of the IR spectrum obviously seen after long term conditioning were largely related to the degradation of both materials. This degradation was previously pointed out by surface damages phenomena: yellowing and crazing network.
Mechanical tests
Monotonic tensile tests were conducted on PA6 and PA6GF30 samples before and after ageing. Fig. 9 shows the superposition of the stress–strain curves for PA6 and PA6GF30 at different distilled water conditioning times. In order to follow the assessement of mechanical performance of studied materials versus the ageing time two sections will be presented below.
At early stage of ageing
According to Fig. 9, it is obvious that the water absorption has a crucial impact on the response stress-strain of both materials. Thus, after a few hours of immersion in distilled water (69 h), a noteworthy decrease in the Young’s modulus and the tensile strength of the PA6 was noticed (Fig.10a). In fact, the reduction rate in both characteristics was recorded approximately 80% and 50%, respectively. This decrease is the result of the water ingress into the polymer. Thus, water molecules are small and polar which rises the probability to interfere with the hydrogen bonds between the polymer chains. Thereby, these bonds are partially destroyed by water absorption and a loss of the stiffness and the strength are noted as reported by [11, 12, 14, 16, 41]. The loss is not only noted for the neat polymer but also for the composite one (Fig. 10b). Thus, a similar decrease in tensile strength was measured. Such reduction can be an indication of a weak adhesion between the fibers and the matrix polymer.
As the greatest water uptake was found for the neat polymer, the PA6GF30 exhibits the lowest reduction in the Young’s modulus (≈ 67%). Thus, it was commonly assumed that sorption mechanism of water molecules is impeded by the presence of glass fibers. Water molecules diffuse into the material through voids and cohesion defects that may appear at the interfaces due to the industrial process. Consequently, a dramatic decrease in both strength and stiffness are noticed as reported by Refs [6, 7, 13, 16].
Concerning the elongation at break two trends are observed; i) The first was a slight decrease (≈20%) for the PA6 while ii) the second was a rise noticed for the PA6GF30 (from 3.4% to 5.1%). Moisture merely acts as a plasticizer in the polymer and subsequently leads to an increase of the elongation at break. It appears that the absorbed moisture, at early stage of immersion, has significantly changed the mode of fracture of the composite from being more or less brittle to a ductile fracture [16, 17]. Nevertheless, the reduction noted in neat polymer at equilibrium state may be due to the initiation of polymer damage. Thus, the entanglement network is partially affected at early stage of immersion. This network allows the unwinding and drawing of chain segments located in the amorphous phase and connecting crystalline lamellae [10]. Therefore, the plasticizer effect is no longer physical in nature and the hydrolysis of the polymer may probably occur [41, 42].
Consequently, it has been well established that hygrothermal ageing leads to a significant decrease of mechanical properties of materials since the first hours of conditioning.
After long term of ageing
After 1920 h of conditioning, the decline remains steady and no longer detectable especially for the elastic modulus for both materials (Fig. 10). On the other hand, the tensile strength still decreases slowly to reach 55% and 65% for PA6 and PA6GF30, respectively. Meanwhile, the elongation at break is markedly decreased after 80 days of ageing. It reaches approximately 93% for PA6. The plastic plateau (Figs. 9a and 10a) disappears and samples failure occurs without neither necking nor stretching. After ageing, there is no entanglement network and the Van der Waals interactions are the only intermolecular forces present in the polymer. Therefore, the fracture properties of semi-crystalline polymers fall catastrophically when the entanglement network is deeply damaged [10]. Furthermore, a dramatic decrease in the elongation at break of the PA6GF30 was observed (from 5.1% to 1.7%) (Figs. 9b, and 10b). This indicates that either i) the chemical scission of the polymer chains is accelerated or ii) the interface was deeply attacked and destroyed by rising the immersion time. Accordingly, it is believed that the decrease in mechanical properties at long term ageing is mainly caused by hydrolysis process as reported by [11, 24].
Accordingly, the long term ageing has deeply affected the mechanical properties of PA6 and PA6GF30.
The interface role of PA6GF30 composite
It is well known that composite materials are characterized by the capacity to transfer loads from the matrix to the fiber across the interface. Thus, the enhancement of mechanical properties was attributed to the level of interfacial adhesion which can be measured by the interfacial shear strength (IFSS). Such parameter is usually considered as a good indicator for the interfacial adhesion for fiber reinforced polymer (FRP) composites [43].
In order to confirm that the interfacial region is preferentially damaged during hygrothermal ageing, a comparison between the IFSS values obtained after immersion and those after thermal ageing at 90 °C at the same durations remains important. The calculation of the interfacial shear stress was conducted using the mathematical model Kelly-Tyson that has been fully described elsewhere [43]. Results illustrate the detrimental water effect on the interface (Fig.11a). This effect rises proportionally to the ageing time. The first decrease was noted after 87 h of immersion (from τ0 h = 67 MPa to τ87 h = 48 MPa). After 1920 h (80 days) of immersion the IFSS decreases linearly to reach 32 MPa. However, after thermal ageing (Fig.11b), a slight decrease in the IFSS was noted after 80 days (from τ0 h = 67 MPa to τ1920 h = 56 MPa). Consequently, the interfacial interactions between the fiber and the matrix are extremely weakened after immersion compared to exposure to high temperature. Certainly, the interfacial region is preferentially attacked by water leading to a significant decrease of the IFSS [43]. Previous studies have shown that this region is the most damaged after immersion [13, 16, 17, 43]. It has been established that water is the major contributor to the loss of adhesion between the fibers and the matrix [44, 45] which has a decisive effect on the mechanical behavior of composite materials [46].
Consequently, the interfacial bonds are weakened with water uptake. This weakness causes the reduction of the mechanical properties of the composite at early stage of ageing. However, this effect remains till 80 days of immersion.
Scanning electron microscopy analysis
SEM observations of the fracture surface of PA6 and PA6GF30 samples before and after ageing in distilled water are shown in Fig. 12. Before conditioning, the failure feature of the unaged PA6 samples exhibits a rough fracture surface with some fibrils (Figs. 12a). These fibrils are indicative of a ductile fracture mechanism [42]. At early stage of ageing a considerable stretching of the polymer fibrils still observed (Figs. 12b). After 80 days of conditioning in distilled water, the feature shows a dramatic change in the fracture topography. The fibrils are absent as shown in Fig. 12c. It seems that ageing under high temperature for long period may hydrolyze the polymer. Whence, a loss of the material integrity with a noteworthy embrittlement will be noted [47].
Meanwhile, as it was expected, the morphology of the moisture absorbed composite samples is different to that of unaged ones particularly fibers adhesion. Before ageing fibers surfaces are well coated with the matrix (Fig. 12a’). This micrograph shows that there is a good adhesion between the fiber and the matrix. Nevertheless, this adhesion is progressively weakened due to hygrothermal ageing (Fig. 12b’, c’). Most of glass fibers are smooth and not coated with the polymer matrix either after 87 h or 1920 h of ageing. This denotes that, after few hours after immersion, the interfacial region has primordially attacked. This attack reduces its effectiveness leading to a drop down of the mechanical properties as previously presented. In this context, Mohd Ishak et al. [48] found that the fibers pull out and uncoated fibers are the predominant results of hygrothermal ageing of PBT composite. This confirms again that the major contributors of the stiffness loss are the interface degradation and the plasticization of the matrix [13, 15, 16]. Moreover, Athijayamani et al. [44] and Ray [45] showed that the cleaner fibers and the interfacial debonding are prevalent in fractured surface of aged composites. Initially the water acts as a plasticizer. Afterwards, the effect becomes chemical in nature.
Consequently, these SEM observations are in agreement with results previously founded.
Conclusions
This paper pointed out the effect of long term ageing on PA6 and PA6GF30 properties. Findings reveal that, the sorption mechanism is extremely affected by the presence of glass fibers. Thus, under the same ageing conditions, PA6 absorbs more water than PA6GF30. Moreover, yellowing and crazing formation were noted after 80 days (1920 h) of immersion for both materials due to thermal-oxidation and the release of internal stresses, respectively. The overall phenomena were monitored by IR spectroscopy. Thus, the appearance of new bands was clearly observed (free N-H stretching and carbonyl groups) testifying the occurrence of chemical reactions.
Furthermore, mechanical properties are deeply altered by hygrothermal ageing due to the plasticizer effect of water as well as the loss of adhesion between fiber and matrix.
References
Thomason JL (2008) The influence of fibre length, diameter and concentration on the strength and strain to failure of glass fibre-reinforced polyamide 6,6. Compos Part A 39:1618–1624
Bernasconi A, Davoli P, Basile A, Filippi A (2007) Effect of fibre orientation on the fatigue behaviour of a short glass fibre reinforced polyamide-6. Int J Fatigue 29:199–208
Teixeira D, Giovanela M, Gonella LB, Crespo JS (2013) Influence of flow restriction on the microstructure and mechanical properties of long glass fiber-reinforced polyamide 6.6 composites for automotive applications. Mater Des 47:287–294
Lim LT, Britt IJ, Tung MA (1999) Sorption and transport of water vapor in nylon 6,6 film. J Appl Polym Sci 7:197–206
Mohd Ishak ZA, Berry JP (1994) Effect of moisture absorption on the dynamic mechanical properties of short carbon fiber reinforced nylon 6,6. Polym Compos 15:223–230
Mohd Ishak ZA, Berry JP (1994) Hygrothermal aging studies of short carbon fiber reinforced nylon 6.6. J Appl Polym Sci 51:2145–2155
Bergeret A, Pires I, Foulc MP, Abadie B, Ferry L, Crespy A (2001) The hygrothermal behaviour of glass-fibre-reinforced thermoplastic composites: a prediction of the composite lifetime. Polym Test 20:753–763
Carrascal I, Casado JA, Polanco JA, Gutiérrez-Solana F (2005) Absorption and diffusion of humidity in fiber glass- reinforced polyamide. Polym Compos 26:580–586. doi:10.1002/pc.20134
Gonçalves ES, Poulsen L, Ogilby PR (2007) Mechanism of the temperature-dependent degradation of polyamide 66 films exposed to water. Polym Degrad Stab 92:1977–1985
El-Mazry C, Correc O, Colin X (2012) A new kinetic model for predicting polyamide 6-6 hydrolysis and its mechanical embrittlement. Polym Degrad Stab 97:1049–1059
Regrain C (2009) comportement, endommagement et fissuration par fluage du polyamide 6 etude experimentale et modelisation: Ph.D.Thesis, Mines Paris Tech
Miri V, Persyn O, Lefebvre J-M, Seguela R (2009) Effect of water absorption on the plastic deformation behavior of nylon 6. Eur Polym J 45:757–762
Pillay S, Vaidya UK, Janowski GM (2009) Effects of moisture and UV exposure on liquid molded carbon fabric reinforced nylon 6 composite laminates. Compos Sci Technol 69:839–846
Vlasveld DPN, Groenewold J, Bersee HEN, Picken SJ (2005) Moisture absorption in polyamide-6 silicate nanocomposites and its influence on the mechanical properties. Polymer 46:12567–12576
Rajeesh KR, Gnanamoorthy R, Velmurugan R (2010) Effect of humidity on the indentation hardness and flexural fatigue behavior of polyamide 6 nanocomposite. Material Science and Engineering A 527:2826–2830
Haddar N, Ksouri I, Kallel T, Mnif N (2014) Effect of hygrothermal ageing on the monotonic and cyclic loading of glass fiber reinforced polyamide. Polym Compos 35:501–508
Ferreño D, Carrascal I, Ruiz E, Casado JA (2011) Characterisation by means of a finite element model of the influence of moisture content on the mechanical and fracture properties of the polyamide 6 reinforced with short glass fibre. Polymer Testing 30:420–428
Bernasconi A, Davoli P, Rossin D, Armanni C (2007) Effect of reprocessing on the fatigue strength of a fiberglass reinforced polyamide. Composites Part: A 38:710–718
Foulc MP, Bergeret A, Ferry L, Ienny P, Crespy A (2005) Study of hygrothermal ageing of glass fibre reinforced PET composites. Polym Degrad Stab 89:461–470
Mercier J, Bunsell A, Castaing P, Renard J (2008) Characterisation and modelling of aging of composites. Compos Part A 39:428–438
Scida D, Assarar M, Poilâne C, Ayad R (2013) Influence of hygrothermal ageing on the damage mechanisms of flax-fibre reinforced epoxy composite. Compos Part B 48:51–58
Simar A, Gigliotti M, Grandidier JC, Ammar-Khodja I (2014) Evidence of thermo-oxidation phenomena occurring during hygrothermal aging of thermosetting resins for RTM composite applications. Compos Part A 66:175–182
Mezghani K (2012) Long term environmental effects on physical properties of vinylester composite pipes. Polym Test 31:76–82
B. Ledieu (2010) vieillissement en milieu eau/glycol du polyamide 66 renforcé fibres de verres courtes pour l’application boîte à eau de radiateur de refroidissement moteur: Ph.D.Thesis, École Nationale Supérieure d'Arts et Métiers
Chaupart N, Serpe G, Verdu J (1998) Molecular weight distribution and mass changes during polyamide hydrolysis. Polymer 39:1375–1380
Chevali VS, Dean DR, Janowski GM (2010) Effect of environmental weathering on flexural creep behavior of long fiber-reinforced thermoplastic composites. Polym Degrad Stab 95:2628–2640
Thomason JL (2007) Structure-property relationships in glass-reinforced polyamide, part 3: effects of hydrolysis ageing on the dimensional stability and performance of short glass-fiber-reinforced polyamide 66. Polym Compos 28:344–354
Rudzinski S, Häussler L, Harnisch C, Mäder E, Heinrich G (2011) Glass fibre reinforced polyamide composites: Thermal behaviour of sizings. Composites: Part A 42:157–164
El-Mazry C, Ben Hassine M, Correc O, Colin X (2013) Thermal oxidation kinetics of additive free polyamide 6–6. Polymer Degradation and Stability 98:22–36
M.N. Grigg (2006) Thermo-oxydative Degradation of Polyamide 6: Ph.D.Thesis, School of Physical and Chemical Sciences Queensland University of Technology
Li R, Hu X (1998) Study on discoloration mechanism of polyamide 6 during thermo-oxidative degradation. Polym Degrad Stab 62:523–528
Bergeret A, Ferry L, Ienny P (2009) Influence of the fibre/matrix interface on ageing mechanisms of glass fibre reinforced thermoplastic composites (PA-6,6, PET, PBT) in a hygrothermal environment. Polym Degrad Stab 94:1315–1324
Thomason JL, Ali JZ, Anderson J (2010) The thermo-mechanical performance of glass-fibre reinforced polyamide 66 during glycol–water hydrolysis conditioning. Compos Part A 41:820–826
Wu Q, Liu X, Berglund LA (2002) FT-IR spectroscopic study of hydrogen bonding in PA6/clay nanocomposites. Polymer 43:2445–2449
Dong W, Gijsman P (2010) Influence of temperature on the thermo-oxidative degradation of polyamide 6 films. Polym Degrad Stab 95:1054–1062
Cerruti P, Lavorgnac M, Carfagna C, Nicolais L (2005) Comparison of photo-oxidative degradation of polyamide 6,6 films stabilized with HALS and CuCl2+KI mixtures. Polymer 46:4571–4583
Cerruti P, Carfagna C (2010) Thermal-oxidative degradation of polyamide 6,6 containing metal salts. Polymer Degradation and Stability 95:2405–2412
Diogo OO, Richaud E, Verdu J, Fernagut F, Guilment J, Fayolle B (2015) Molecular and macromolecular structure changes in polyamide 11 during thermal oxidation e kinetic modeling. Polym Degrad Stab 120:76–87
Jha A, Bhowmick AK (1998) Thermal degradation and ageing behaviour of novel thermoplastic elastomeric nylon-6/acrylate rubber reactive blends. Polym Degrad Stab 62:575–586
Su KH, Lin JH, Lin CC (2007) Influence of reprocessing on the mechanical properties and structure of polyamide 6. J Mater Process Technol 192–193:532–538
Taktak R, Guermazi N, Derbeli J, Haddar N (2015) Effect of hygrothermal aging on the mechanical properties and ductile fracture of polyamide 6: experimental and numerical approaches. Eng Fract Mech 148:122–133
Phua YJ, Chow WS, Mohd Ishak ZA (2011) The hydrolytic effect of moisture and hygrothermal aging on poly(butylene succinate)/organo-montmorillonite nanocomposites. Polym Degrad Stab 96:1194–1203
Ksouri I, Guermazi N, Haddar N, Ferid Ayedi H (2016) Effects of processing steps and hygrothermal ageing on mechanical performance of PA6GF30 composite: interfacial shear strength. Polym Compos. doi: 10.1002/pc.23961
Athijayamani A, Thiruchitrambalam M, Natarajan U, Pazhanivel B (2009) Effect of moisture absorption on the mechanical properties of randomly oriented natural fibers/polyester hybrid. Mater Sci Eng A 517:344–353
Ray BC (2006) Temperature effect during humid ageing on interfaces of glass and carbon fibers reinforced epoxy composites. Journal of Colloid Interface Science 298:111–117
Bergeret A, Ferry L, Ienny P (2009) Influence of the fibre/matrix interface on ageing mechanisms of glass fibre reinforced thermoplastic composites (PA-6,6, PET, PBT) in a hygrothermal environment. Polym Degrad Stab 94:1315–1324
Q.N. THI THUY (2013) Identification des propriétés morphologiques et hygrothermiques hétérogènes de nouveaux composites hautes performances soumis à des cycles de vieillissement thermo-hygro-mécaniques: Ph.D.Thesis, École Nationale Supérieure Saint-Étienne
Mohd Ishak ZA, Ishiaku US, Karger-Kocsis J (2000) Hygrothermal aging and fracture behavior of short-glass-fiber- reinforced rubber-toughened poly(butylene terephthalate) composites. Compos Sci Technol 60:803–815
Acknowledgements
The authors appreciate the helpful discussions and suggestions provided by Dr. Noamen Guermazi. Authors would like also to thank ICA team (EMAC-France) especially Pr. Thierry Cutard, E. Ramirez, S. Tovar,V. Vidal, and K. Choquet, for their help provided for this research project. This work was supported by the Ministry of Higher Education and Scientific Research- Tunisia.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ksouri, I., De Almeida, O. & Haddar, N. Long term ageing of polyamide 6 and polyamide 6 reinforced with 30% of glass fibers: physicochemical, mechanical and morphological characterization. J Polym Res 24, 133 (2017). https://doi.org/10.1007/s10965-017-1292-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10965-017-1292-6