Abstract
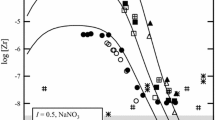
Stability constants of the form F β 1(M)=[MF2+][M3+]−1[F−]−1 (where [MF2+] represents the concentration of a yttrium or a rare earth element (YREE) complex, [M3+] is the free YREE ion concentration, and [F−] is the free fluoride ion concentration) were determined by direct potentiometry in NaNO3 and NaCl solutions. The patterns of log10 F β 1(M) in NaNO3 and NaCl solutions very closely resemble stability constant patterns obtained previously in NaClO4. For a given YREE, stability constants obtained in NaClO4 were similar to, but consistently larger than F β 1(M) values obtained in NaNO3 which, in turn, were larger than formation constants obtained in NaCl. Stability constants for formation of nitrate and chloride complexes ( \(_{\mathrm{NO}_{3}}\beta_{1}(\mathrm{M})=[\mathrm{MNO}_{3}^{2+}][\mathrm{M}^{3+}]^{-1}[\mathrm{NO}_{3}^{-}]^{-1}\) and Cl β 1(M)=[MCl2+][M3+]−1[Cl−]−1) derived from F β 1(M) data exhibited ionic strength dependencies generally similar to those of F β 1(M). However, in contrast to the somewhat complex pattern obtained for F β 1(M) across the fifteen member YREE series, no patterns were observed for nitrate and chloride complexation constants: neither \(_{\mathrm{NO}_{3}}\beta_{1}(\mathrm{M})\) nor Cl β 1(M) showed discernable variations across the suite of YREEs. Nitrate and chloride formation constants at 25 °C and zero ionic strength were estimated as log10 \(_{\mathrm{NO}_{3}}\beta_{1}^{\mathrm{o}}(\mathrm{M})=0.65\pm 0.06\) and log10 Cl β o1 (M)=0.71±0.05. Although these constants are identical within experimental uncertainty, the distinct ionic strength dependencies of \(_{\mathrm{NO}_{3}}\beta_{1}(\mathrm{M})\) and Cl β 1(M) produced larger differences in the two stability constants with increasing ionic strength whereby Cl β 1(M) was uniformly larger than \(_{\mathrm{NO}_{3}}\beta_{1}(\mathrm{M})\) .
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Byrne, R.H., Sholkovitz, E.R.: In: Gschneidner Jr., K.A., Eyring, L. (eds.) Handbook on the Physics and Chemistry of Rare Earths, vol. 23. Elsevier, Amsterdam (1996), Chap. 158
Quinn, K.A., Byrne, R.H., Schijf, J.: Sorption of yttrium and rare earth elements by amorphous ferric hydroxide: Influence of pH and ionic strength. Marine Chem. 99, 128–150 (2006)
Byrne, R.H., Kim, K.H.: Rare earth element scavenging in seawater. Geochim. Cosmochim. Acta 54, 2645–2656 (1990)
Lee, J.H., Byrne, R.H.: Complexation of trivalent rare earth elements (cerium, europium, gadolinium, terbium, and ytterbium) by carbonate ions. Geochim. Cosmochim. Acta 57, 295–302 (1993)
Luo, Y.R., Byrne, R.H.: The ionic strength dependence of rare earth and yttrium fluoride complexation at 25 °C. J. Solution Chem. 29, 1089–1099 (2000)
Luo, Y., Millero, F.J.: Effects of temperature and ionic strength on the stabilities of the first and second fluoride complexes of yttrium and the rare earth elements. Geochim. Cosmochim. Acta 68, 4301–4308 (2004)
Schijf, J., Byrne, R.H.: Determination of stability constants for the mono- and difluoro-complexes of Y and the REE, using a cation-exchange resin and ICP-MS. Polyhedron 18, 2839–2844 (1999)
Lee, J.H., Byrne, R.H.: Rare earth element complexation by fluoride ions in aqueous solution. J. Solution Chem. 22, 751–766 (1993)
Walker, J.B., Choppin, G.R.: Thermodynamic parameters of fluoride complexes of the lanthanides. Adv. Chem. Ser. 71, 127–140 (1967)
Avramenko, N.I., Andronov, E.A., Blokhin, V.V., Mironov, V.E.: Study of fluoride complexes of rare earth metals in aqueous salt solutions. Izv. Vyssh. Uchebn. Zaved. Khim. Techhnol. 26, 155–157 (1983) (in Russian)
Bilal, B.A., Herrmann, F., Fleischer, W.: Complex formation of trace elements in geochemical systems. 1. Potentiometric study of fluoro complexes of rare earth elements in fluorite bearing model systems. J. Inorg. Nucl. Chem. 41, 347–350 (1979)
Bilal, B.A., Becker, P.: Complex formation of trace elements in geochemical systems. 2. Stability of rare earths fluoro complexes in fluorite bearing model system at various ionic strengths. J. Inorg. Nucl. Chem. 41, 1607–1608 (1979)
Bilal, B.A., Kob, V.: Complex formation of trace elements in geochemical systems. 3. Studies on the distribution of fluoro complexes of rare earth elements in fluorite bearing model systems. J. Inorg. Nucl. Chem. 42, 629–630 (1980)
Bilal, B.A., Koss, V.: Complex formation of trace elements in geochemical systems. 4. Study on the distribution of sulfatocomplexes of rare earth elements in fluorite-bearing model system. J. Inorg. Nucl. Chem. 42, 1064–1065 (1980)
Becker, P., Bilal, B.P.: Lanthanide–fluoride ion association in aqueous sodium chloride solutions at 25 °C. J. Solution Chem. 14, 407–415 (1985)
Menon, M.P., James, J., Jackson, J.D.: Studies on the solubility and complexation of lanthanum and neodymium fluoride–water systems. Lanthan. Actinide Res. 2, 49–66 (1987)
Menon, M.P., James, J.: Solubilities, solubility products and solution chemistry of lanthanon trifluoride–water systems. J. Chem. Soc. Faraday Trans. 1 85, 2683–2694 (1989)
Luo, Y.R., Byrne, R.H.: Yttrium and rare earth element complexation by chloride ions at 25 °C. J. Solution Chem. 30, 837–845 (2001)
Smith, S.M., Martell, A.E.: NIST critically selected stability constants of metal complexes database, Version 8.0 (2004)
Mironov, V.E., Avramenko, N.I., Kopyrin, A.A., Blokhin, V.V., Eike, M.Yu., Isaev, I.D.: Thermodynamics of the formation of monochloride complexes of rare earth metals in aqueous solutions. Koord. Khim. 8, 636–638 (1982) (in Russian)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Luo, YR., Byrne, R.H. The Influence of Ionic Strength on Yttrium and Rare Earth Element Complexation by Fluoride Ions in NaClO4, NaNO3 and NaCl Solutions at 25 °C. J Solution Chem 36, 673–689 (2007). https://doi.org/10.1007/s10953-007-9141-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10953-007-9141-6