Abstract
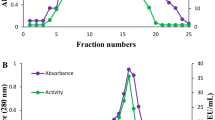
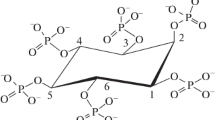
High-pressure liquid chromatography (HPLC) analysis established myo-inositol pentakisphosphate as the final product of phytate dephosphorylation by the phytate-degrading enzyme from Pantoea agglomerans. Neither product inhibition by phosphate nor inactivation of the Pantoea enzyme during the incubation period were responsible for the limited phytate hydrolysis as shown by addition of phytate-degrading enzyme and phytate, respectively, after the observed stop of enzymatic phytate degradation. In additon, the Pantoea enzyme did not possess activity toward the purified myo-inositol pentakisphosphate. Using a combination of High-Performance Ion Chromatography (HPIC) analysis and kinetic studies, the nature of the generated myo-inositol pentakisphosphate was established. The data demonstrate that the phytate-degrading enzyme from Pantoea agglomerans dephosphorylates myo-inositol hexakisphosphate in a stereospecific way to finally D-myo-inositol(1,2,4,5,6)pentakisphosphate.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- HPIC:
-
high performance ion chromotography
- HPLC:
-
high pressure liquid chromotography
- TBAH:
-
tetrabutyl ammonium hydroxide
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Greiner, R. Degradation of myo-inositol Hexakisphosphate by a Phytate-degrading Enzyme from Pantoea agglomerans. Protein J 23, 577–585 (2004). https://doi.org/10.1007/s10930-004-7884-0
Received:
Issue Date:
DOI: https://doi.org/10.1007/s10930-004-7884-0