Abstract
Host prefeeding and acceptance behavior of herbivorous insects may strongly differ depending upon the plant species and previous experience. We examined prefeeding behaviors and acceptance of host and non-host plants by the beetle Phaedon cochleariae in populations with different rearing histories using a camera. Suitable host plants evoked short test biting followed by long feeding durations indicating that these first contacts provide sufficient information to stimulate feeding. In contrast, plants of intermediate quality caused prolonged test biting but feeding duration was rather low. When offered unsuitable plants, beetles showed only rare leaf contacts and test biting, demonstrating that test biting behavior is a useful measure of plant suitability. Rearing history had a minor influence on host plant exploitation behavior.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Host plant finding of phytophagous insects consists of sequential steps, such as habitat searching, host plant searching, selection and finally acceptance of the host plant (Schoonhoven et al. 2005). During this process, several behavioral steps occur before feeding begins. After arriving on the plant, the first contact with the host surface takes place via the tarsi and antennae of the searching insect. Cues on the plant surface such as cuticular hydrocarbons and primary or secondary metabolites offer the first information about plant quality (Adati and Matsuda 1993; Müller and Riederer 2005). If these cues are stimulatory, the chewing herbivore may start biting into the leaf and thereby come into contact with internal plant cell tissue (Eigenbrode and Espelie 1995). However, mechanical barriers such as the cuticular wax layer or trichomes may complicate access to leaf tissue for inspection of plant quality (Valverde et al. 2001; Reifenrath et al. 2005). Henderson et al. (2004) investigated the prefeeding behavior of the leaf beetle Phyllotreta crucifera Goeze (Coleoptera: Chrysomelidae) on host and non-host plant seedlings determining the role of this behavior in the onset of feeding. The prefeeding behavior of this beetle species could be divided into the sequence: acclimation, stimulation, initial feeding and continuous feeding, where the frequency and duration of these different steps was strongly dependent on the plant suitability (Henderson et al. 2004).
Prefeeding and acceptance of host plants may also strongly depend on former experience of the individual. In this sense, learning and physiological adjustments have been shown to induce preferences for host plant species that have been contacted first by larvae, for example, of the tobacco horn worm (Bernays and Wcislo 1994; del Campo et al. 2001), which could make the insect dependent on certain host allelochemicals, even leading to “addiction” (Renwick and Lopez 1999). In contrast, in other insect species host plant use was found to be stable despite experience over several generations, indicating that exploitation of host plant species may be genetically fixed (Via 1991).
Phaedon cochleariae F. (Coleoptera: Chrysomelidae), the mustard leaf beetle, is a pest insect on crops of Brassicaceae in several regions (Speyer et al. 1954; Finch and Kienegger 1997). Whereas the behavioral steps involved in the oviposition sequence of P. cochleariae have been investigated in detail by Müller and Rosenberger (2006), little is known about the prefeeding and acceptance behavioral sequence of adults. All members of the Brassicaceae contain non-volatile glucosinolates, which can act as feeding stimulants for P. cochleariae (Nielsen 1978; Reifenrath et al. 2005), but in some species can only be detected when the cuticle is slightly damaged (Reifenrath et al. 2005; Städler and Reifenrath 2009; Badenes-Pérez et al. 2011). Due to the activity of myrosinases, glucosinolates can be hydrolysed to volatile isothiocyanates (Halkier and Gershenzon 2006), which were shown to attract P. cochleariae (Kühnle and Müller 2011a). Various Brassicaceae, including host and non-host species, were attractive to P. cochleariae in an olfactometer (Kühnle and Müller 2009), probably due to the presence of isothiocyanates. With regard to the role of former experience, evidence exists that larval experience does not influence feeding preferences of adult P. cochleariae despite differences in performance on various host plant species (Kühnle and Müller 2011b).
In the present study, the prefeeding and acceptance behaviors of P. cochleariae were investigated in detail in individuals with different rearing histories to determine the role of prior experience in acceptance or rejection of potential hosts. Therefore, adult behavior was observed on host as well as non-host plant species within the families of Brassicaceae and Plantaginaceae, which strongly differ in leaf traits such as toughness, trichome density and secondary plant metabolites (Biere et al. 2004; Travers-Martin and Müller 2008). Tests were conducted with two beetle populations of different origin that were switched to a common host plant species for several generations and tested at the beginning and the end of rearing on this common host.
Material and Methods
Plant and Insect Rearing
The two host plant species cabbage (Brassica rapa L. ssp. chinensis) and white mustard (Sinapis alba L. cv. Silenda), as well as two non-host species, the Brassicaceae Turkish rocket (Bunias orientalis L.) and the Plantaginaceae ribwort plantain (Plantago lanceolata L.) were reared in a greenhouse at 20°C and under a 16:8 h L:D regime. Seeds were obtained from Kiepenkerl, Norken, Germany, except seeds of B. orientalis, which were obtained from the Botanical Garden Würzburg, Germany. Plants used for bioassays and feeding were 4–5 weeks old and non-flowering. Leaves of different age were randomly picked for bioassays, as it is known that leaf age influences feeding and performance of P. cochleariae (Reifenrath and Müller 2009).
Two beetle populations were used for bioassays. One beetle population of P. cochleariae had been reared in the laboratory on B. rapa for over forty generations after collection in the field near Berlin and Würzburg (Germany). From this population, about 100 larvae were transferred shortly after hatching to S. alba and continuously reared on this plant species for the subsequent eleven generations (Brassica population, ‘Br’). A second population was collected in May 2007 in the field on Rorippa sylvestris (L.) Bess. (Rorippa population, ‘Rs’) near Pevestorf, Germany, and also reared in the laboratory on S. alba for 19 generations. For rearing, insects were kept in plastic boxes (200 × 200 × 65 mm) covered with a gauze lid (500 μm mesh) in a controlled environment chamber (20°C, 70% R. H., and 16:8 h L:D). The bottom of each box was lined with filter paper. Stems with about 6–10 leaves of S. alba held in water-filled plastic tubes were replaced every other day.
Feeding Bioassays
To investigate prefeeding and acceptance behavior of P. cochleariae on different plant material in relation to rearing history, beetles of the first generation placed on S. alba and of the 11th and 19th generations of the ‘Br’ and ‘Rs’ populations, respectively, were used about 2 to 4 weeks after adult eclosion. Adults were starved for 1 to 2 h prior to bioassays to increase feeding motivation. One leaf disc (6 mm in diameter) of one of the different test plant species was placed in the center of a Petri dish (55 mm diameter) lined with white filter paper. The Petri dish was surrounded by a white paper wall (70 mm diameter, 70 mm height) to exclude visual influences of the environment. A beetle was placed at the edge of the Petri dish and filmed for 5 min with a camera (JVC Digital Camcorder GR-DV 4000, objective: F1.2 f = 3.8 mm to 38 mm, 10:1 motor zoom, 300x digital zoom, Friedberg, Germany) attached to a stereomicroscope. Beetles of the ‘Br’ and ‘Rs’ population were offered leaf discs of B. rapa, S. alba and B. orientalis. Additionally, beetles of the ‘Br’ population were offered P. lanceolata leaves to test behavioral responses to a non-host which does not belong to the Brassicaceae. After every test trial the leaf disc was exchanged to provide fresh plant material. At least ten beetles were tested per plant species and each beetle was observed only once. Only tests in which beetles contacted leaf discs at least once were incorporated in the statistical analyses. Therefore, actual sample size was lower in some cases than the number of tested beetles due to non-responders. Afterwards, the films were analyzed with the program The Observer 5.0 software (Noldus Information Technology bv, Wageningen, The Netherlands). The duration and frequency of the behavioral steps test biting, continuous feeding, and walking on leaf were recorded. From these values, total leaf contact duration and time without leaf contact were calculated. Test biting was defined as short, interrupted nibbling on the plant, whereas continuous feeding lasted at least 10 s and was not interrupted. Walking on leaf was defined as walking on the leaf surface (at least 2 s) without biting but including contact of antennae.
Statistical Analyses
For the general description of the behavioral sequence (Fig. 1), behavioral steps on each plant species were quantified by multiplying the relative proportion of beetles showing a given behavior with the mean duration (in s) of this behavior. Therefore, all beetles tested on one plant species were considered, regardless of their experience. The relative importance of each step is visualized by the thickness of the arrows (Fig. 1).
Steps of the prefeeding and acceptance behavior of Phaedon cochleariae adults when presented with (a) a suitable host plant species (i.e. Brassica rapa or Sinapis alba), (b) an unsuitable non-host plant species (i.e. Bunias orientalis), and (c) an unacceptable plant species (i.e. Plantago lanceolata) (c). Thickness of arrows indicates relative importance of the following behavioral step, quantified by multiplying the relative proportion of beetles showing a given behavior with the mean duration (in s) of this behavior. These values of behaviors are given along the arrows. The percentages in the upper part of the figure refer to the proportion of beetles that contacted the leaf at least once. Beetle experience was not considered in this analysis (all beetles tested on one plant species were pooled)
The different behavioral steps of acceptance behavior of the populations (‘Br’ and ‘Rs’ population) and generations (first and final generation reared on S. alba) in relation to test plant species and experience were analyzed by generalized additive models with a Gaussian distribution and identity link function (durations of leaf contact and feeding) and a Poisson distribution with a log link function (test biting frequency). The smooth function was represented using cubic regression splines with three knots. In this kind of regression spline a three-order polynomial is used. The dependent variable and its first and second derivates are continuous at all the knots (Guo and White 2005). Test plant species, generation and population were included as explanatory variables in the model. Additionally, two interactions were included, test plant species*generation and generation*population. Depending on the behavior under investigation, the model validation had to be adjusted resulting in slightly different models. The most suitable model was determined on the lowest generalized cross-validation value. Data for feeding and total leaf contact duration were transformed [(log(data2+1))6 for feeding and log(data) for contact duration] to achieve highest approximation to normal distribution of residuals. Statistical analyses were conducted with the program R 2.11.0 using the package mgcv (Wood 2003, 2011).
Results
General Behavioral Sequence
After placing an adult in a Petri dish, first contact with a potential feeding source, consisting of a very short touch of the leaf surface, occurred by the antennae or tarsi. Contact by tarsi increased when the beetle walked on the leaf disc facilitating recognition of potential chemotactic cues before test biting occurred. In general, the following behavioral sequence was observed: short first contact with the leaf by the insect’s antennae or tarsi, increased contact duration by walking on the leaf and test biting followed by either continuous feeding or leaving the leaf disc (Fig. 1). Test biting started mainly on the leaf edges but also occurred on the central parts of the leaf discs.
Behaviors Related to Food Suitability
Depending on leaf quality, tarsal contact duration and test biting were shorter or longer. A certain behavioral step could either stop and the prefeeding sequence could start again or the beetle could leave the leaf disc. When a highly suitable plant species such as B. rapa or S. alba was offered, tarsal contact was soon followed by a short test biting and subsequent long feeding (scenario A in Fig. 1). In the case of the non-host Brassicaceae species B. orientalis (scenario B in Fig. 1), test biting occurred more often but with short durations (Tables 1, 2), whereas subsequent feeding was only shown by 37% of all tested beetles and occurred only briefly (26 s ± 7.4 s, mean±SE). On the unacceptable Plantaginaceae species P. lanceolata (scenario C in Fig. 1), tarsal contact and test biting were rare and no feeding behavior occurred, indicating that the lack of attractants or stimulants and/or presence of repellents or deterrents were detected rapidly. Leaving of discs of this plant species was regularly recorded.
Overall, the test plant species had a highly significant effect on the different behavioral traits test biting frequency, feeding duration and total contact duration with the leaf [Table 1, s (Test plant species)]. The test biting frequencies were on average higher on the non-host B. orientalis, whereas feeding durations as well as total leaf contact duration were lowest on this plant species (Table 2).
Behaviors Related to Rearing History
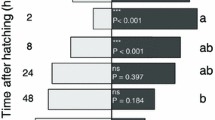
Apart from an effect of test plant species, a minor effect of generation was also indicated by the model for test biting frequency (Table 1), with test biting frequency generally increasing slightly from the first to the last generation (Table 2). Furthermore, generation affected feeding duration and total leaf contact duration in slightly different directions, depending on test plant species (Tables 1, 2). Additionally, feeding duration and total leaf contact duration differed between the two populations and the significant interaction effect generation*population showed that the first and the last generation behaved differently depending on the population. The first generation of the ‘Br’ population fed and staid (total leaf contact duration) especially longer on leaves of B. rapa than the last, whereas the ‘Rs’ population increased on average feeding and total contact duration on S. alba from the first to the last generation (Table 2).
Discussion
In this study two main points were investigated: first the prefeeding and acceptance behavior was characterized in P. cochleariae on plant species of differing suitability and second whether the behavioral sequence changes over time in relation to rearing history and beetles’ origin were investigated. The observed behavioral pattern clearly shows that the sequence of prefeeding and acceptance behavior followed a certain regularity; a first contact of the leaf by antennae and tarsi was followed by test biting, which resulted either in continuous feeding or in leaving of the plant (Fig. 1). Similar observations had been made in another leaf beetle species, Phyllotreta crucifera, in which the main steps, stimulation and initial feeding, were characterized by antennal waving and tarsal tapping, biting and feeding (Henderson et al. 2004).
In general, olfactory and chemotactic sensilla are located on the antennae, tarsi and mouthparts of herbivorous insects and are important for food selection (Jourdan et al. 1995; Mitchell et al. 1999; Schoonhoven et al. 2005; Siddiqi et al. 2006; Glendinning et al. 2007). Antennal contact can allow detection of phytochemicals without biting (Isidoro et al. 1998). The lack of contact or only rare contacts with test leaves may be also due to the detection of potential repellents reducing attraction of a plant species (Hori 1998; Egigu et al. 2011). The unacceptable plant P. lanceolata seemed not to be repellent but was rather neutral for P. cochleariae when tested in an olfactometer (Kühnle and Müller 2009).
After beetles had reached a leaf and touched it with tarsi and mouthparts, test biting occurred that was characterized by short nibbling on the leaf. Test biting was also observed in other species and enables the chewing herbivores to receive cues from the plant tissue (Mitchell and Low 1994; Henderson et al. 2004). Glendinning et al. (2007) showed that differences of plant quality or suitability were measurable in Manduca sexta caterpillars by their biting frequencies and depended on the stimulation of several taste sensilla on the mouthparts. In P. cochleariae, suitable plants evoked low frequencies of test biting followed by long feeding durations indicating that in this species gustatory sensilla of the mouth parts are similarly involved in testing of the plant quality. Highly frequent test biting was found on the non-host B. orientalis and may be evidence for the presence of both feeding stimulants and deterrents. Stimulants may cause herbivores to start nibbling but deterrents may hinder continuous feeding. Indeed, certain extracts of B. orientalis had a stimulatory effect on P. cochleariae probably due to the presence of glucosinolates, whereas another fraction contained deterrent compounds, likely flavonoids (Kühnle and Müller 2009). In contrast, extracts of the Plantaginaceae P. lanceolata decreased feeding when applied to an acceptable plant species (Kühnle and Müller 2009). Also in the present study, the non-host plant P. lanceolata evoked the lowest durations of test biting by P. cochleariae without any subsequent feeding. Iridoid glycosides, characteristic compounds of Plantaginaceae, are well-known deterrents against herbivores (Puttick and Bowers 1988; Biere et al. 2004) and were also shown to deter P. cochleariae (Kühnle and Müller 2009). Similarly, total duration of leaf contact mirrored the degree of acceptance of the tested plant species. In addition to time spent feeding, leaf contact duration also consists of walking over the test leaf, which may increase information about the environment by tarsal contact. Such undirected searching behavior was also observed in other arthropod taxa indicating that this behavior is an important part of orientation (Bell 1990; Bengtsson et al. 2004).
Next to plant tissue chemistry, mechanical leaf traits like hairiness and plant cuticle thickness also may have influenced the prefeeding and acceptance behavior. Tough leaves and high trichome density often decrease insect performance and preference resulting in plant resistance against many herbivores (Valverde et al. 2001; Traw and Dawson 2002; Hui and Jin 2004; Medeiros and Moreira 2005). Likewise, leaf toughness had a negative influence on P. cochleariae larvae (Tanton 1962) and adults preferred leaves with removed epicuticular waxes over leaves with an intact cuticle (Reifenrath et al. 2005). Indeed, B. orientalis leaves, which were an unattractive feeding source for P. cochleariae, possess a higher trichome density than various other Brassicaceae species including S. alba and are also less accepted by other herbivorous species that are specialized on Brassicaceae (Travers-Martin and Müller 2008; personal observation).
In the present study, the influence of test plant species on prefeeding and acceptance behavioral steps was highly significant and obvious. However, the origin and the rearing history of the beetles also affected their behavior to different degrees. Test biting seemed to be a highly important trait for evaluation of plant quality and thus only marginally influenced by rearing history. Feeding duration and total leaf contact duration were influenced slightly by the duration of experience with S. alba over the experimental time (generation factor). They decreased in the eleventh generation when B. rapa was offered to the ‘Br’ population indicating that the lack of experience of B. rapa over time in this population lowered acceptance of this plant species (Table 2). The slight increase in feeding and total leaf contact duration on S. alba in the ‘Rs’ population from the first to the last generation indicates that this plant species became more acceptable over time (Table 2). Additionally, the origin of the beetles had an effect on feeding duration and total leaf contact. The beetles of the ‘Rs’ population spent less time feeding leaves of different species than the ‘Br’ population in the beginning of the experiment (first generation on S. alba). These differences may be due to the fact that P. cochleariae of the ‘Br’ population had been kept for several years in the lab and were thus highly used to the conditions, whereas the ‘Rs’ population was taken from the field. Differences in behavior due to the origin of the population have been also noted in other herbivore species (Bravo and Zucoloto 1998; Fox et al. 2010). For several insect species it was shown that changed or different rearing conditions and experience of a certain environment resulted in changed preferences towards the new environment (Tucić et al. 1995; Solarz and Newman 2001; Messina 2004). Mechanisms of adjustments with regard to acceptance behavior could be based on high phenotypic plasticity and/or genotypic variability indicating possible evolutionary changes in host plant exploitation (Via 1990; Agrawal 2001).
This study clearly shows that both the given plant tissue quality (mechanical and chemical traits) and plant suitability as well as the experience over generations and origin of insects have an impact on the actual prefeeding and acceptance behavior of the leaf beetle P. cochleariae. Differences in single behavioral traits of the behavioral sequence such as test biting allow deducing on overall plant acceptance of specific plant species.
References
Adati T, Matsuda K (1993) Feeding stimulants for various leaf beetles (Coleoptera: Chrysomelidae) in the leaf surface wax of their host plants. Appl Entomol Zool 28:319–324
Agrawal AA (2001) Ecology—Phenotypic plasticity in the interactions and evolution of species. Science 294:321–326
Badenes-Pérez FR, Reichelt M, Gershenzon J, Heckel DG (2011) Phylloplane location of glucosinolates in Barbarea spp. (Brassicaceae) and misleading assessment of host suitability by a specialist herbivore. New Phytol 189:549–556
Bell WJ (1990) Searching behavior patterns in insects. Annu Rev Entomol 35:447–467
Bengtsson G, Nilsson E, Ryden T, Wiktorsson M (2004) Irregular walks and loops combines in small-scale movement of a soil insect: implications for dispersal biology. J Theor Biol 231:299–306
Bernays EA, Wcislo WT (1994) Sensory capabilities, information processing, and resource specialization. Q Rev Biol 69:187–204
Biere A, Marak HB, van Damme JMM (2004) Plant chemical defense against herbivores and pathogens: generalized defense or trade-offs? Oecologia 140:430–441
Bravo ISJ, Zucoloto FS (1998) Performance and feeding behavior of Ceratitis capitata: comparison of a wild population and a laboratory population. Entomol Exp Appl 87:67–72
del Campo ML, Miles CI, Schroeder FC, Müller C, Booker R, Renwick JA (2001) Host recognition by the tobacco hornworm is mediated by a host plant compound. Nature 411:186–189
Egigu MC, Ibrahim MA, Yahya A, Holopainen JK (2011) Cordeauxia edulis and Rhododendron tomentosum extracts disturb orientation and feeding behavior of Hylobius abietis and Phyllodecta laticollis. Entomol Exp Appl 138:162–174
Eigenbrode SD, Espelie KE (1995) Effects of plant epicuticular lipids on insect herbivores. Annu Rev Entomol 40:171–194
Finch S, Kienegger M (1997) A behavioural study to help clarify how undersowing with clover affects host-plant selection by pest insects of brassica crops. Entomol Exp Appl 84:165–172
Fox CW, Bush ML, Messina FJ (2010) Biotypes of the seed beetle Callosobruchus maculatus have differing effects on the germination and growth of their legume hosts. Agr Forest Entomol 12:353–362
Glendinning JI, Jerud A, Reinherz AT (2007) The hungry caterpillar: an analysis of how carbohydrates stimulate feeding in Manduca sexta. J Exp Biol 210:3054–3067
Guo Q, White RE (2005) Cubic spline regression for the open-circuit potential curves of a lithium-ion battery. J Electrochem Soc 152:A343–A350
Halkier BA, Gershenzon J (2006) Biology and biochemistry of glucosinolates. Annu RevPlant Biol 57:303–333
Henderson AE, Hallett RH, Soroka JJ (2004) Prefeeding behavior of the crucifer flea beetle, Phyllotreta cruciferae, on host and nonhost crucifers. J Insect Behav 17:17–39
Hori M (1998) Repellency of rosemary oil against Myzus persicae in a laboratory and in a screenhouse. J Chem Ecol 24:1425–1432
Hui X, Jin C (2004) Interspecific variation of plant traits associated with resistance to herbivory among four species of Ficus (Moraceae). Ann Bot-London 94:377–384
Isidoro N, Bartlet E, Ziesmann J, Williams IH (1998) Antennal contact chemosensilla in Psylliodes chrysocephala responding to cruciferous allelochemicals. Physiol Entomol 23:131–138
Jourdan H, Barbier R, Bernard J, Ferran A (1995) Antennal sensilla and sexual dimorphism of the adult ladybird beetle Semiadalia undecimnotata Schn. (Coleoptera, Coccinellidae). Int J Insect Morphol 24:307–322
Kühnle A, Müller C (2009) Differing acceptance of familiar and unfamiliar plant species by an oligophagous beetle. Entomol Exp Appl 131:189–199
Kühnle A, Müller C (2011a) Relevance of visual and olfactory cues for host location in the mustard leaf beetle Phaedon cochleariae. Physiol Entomol 36:68–76
Kühnle A, Müller C (2011b) Responses of an oligophagous beetle species to rearing for several generations on alternative host-plant species. Ecol Entomol 36:125–134
Medeiros L, Moreira GRP (2005) Larval feeding behavior of Gratiana spadicea (Klug) (Coleoptera: Chrysomelidae: Cassidinae) on its host plant, Solanum sisymbriifolium Lamarck (Solanaceae): interaction with trichomes. Coleopt Bull 59:339–350
Messina FJ (2004) How labile are the egg-laying preferences of seed beetles? Ecol Entomol 29:318–326
Mitchell BK, Low R (1994) The structure of feeding behavior in the Colorado potato beetle, Leptinotarsa decemlineata (Coleoptera, Chrysomelidae). J Insect Behav 7:707–729
Mitchell BK, Itagaki H, Rivet MP (1999) Peripheral and central structures involved in insect gustation. Microsc Res Tech 47:401–415
Müller C, Riederer M (2005) Review: plant surface properties in chemical ecology. J Chem Ecol 31:2621–2651
Müller C, Rosenberger C (2006) Different oviposition behaviour in Chrysomelid beetles: characterisation of the interface between oviposition secretion and the plant surface. Arthropod Struct Dev 35:197–205
Nielsen JK (1978) Host plant discrimination within Cruciferae: feeding responses of four leaf beetles (Coleoptera: Chrysomelidae) to glucosinolates, cucurbitacins and cardenolides. Entomol Exp Appl 24:41–54
Puttick GM, Bowers MD (1988) Effect of qualitative and quantitative variation in allelochemicals on a generalist insect: iridoid glycosides and the Southern armyworm. J Chem Ecol 14:335–351
Reifenrath K, Müller C (2009) Larval performance of the mustard leaf beetle (Phaedon cochleariae, Coleoptera, Chrysomelidae) on white mustard (Sinapis alba) and watercress (Nasturtium officinale) leaves in dependence of plant exposure to ultraviolet radiation. Environ Pollut 157:2053–2060
Reifenrath K, Riederer M, Müller C (2005) Leaf surface wax layers of Brassicaceae lack feeding stimulants for Phaedon cochleariae. Entomol Exp Appl 115:41–50
Renwick JAA, Lopez K (1999) Experience-based food consumption by larvae of Pieris rapae: addiction to glucosinolates? Entomol Exp Appl 91:51–58
Schoonhoven LM, van Loon JJA, Dicke M (2005) Insect-plant biology. Oxford University Press, Oxford
Siddiqi MJI, Rana SA, Ismail M, Suhail A (2006) Role of antennal sensilla of soil beetles as food selection in the fields of sugarcane. Pakistan Entomol 28:39–44
Solarz SL, Newman RM (2001) Variation in hostplant preference and performance by the milfoil weevil, Euhrychiopsis lecontei Dietz, exposed to native and exotic watermilfoils. Oecologia 126:66–75
Speyer W, Heikertinger F, von Winning E (1954) Chrysomelidae, Blattkäfer. In: Blunck H (ed) Handbuch der Pflanzenkrankheiten Vol. 5. Paul Parey, Berlin, pp 270–379
Städler E, Reifenrath K (2009) Glucosinolates on the leaf surface perceived by insect herbivores: review of ambiguous results and new investigations. Phytochem Rev 8:207–225
Tanton MT (1962) The effect of leaf “toughness” on the feeding of larvae of the mustard beetle Phaedon cochleariae Fab. Entomol Exp Appl 5:74–78
Travers-Martin N, Müller C (2008) Matching plant defense syndroms with performance and preference of a specialist herbivore. Funct Ecol 22:1033–1043
Traw MB, Dawson TE (2002) Reduced performance of two specialist herbivores (Lepidoptera: Pieridae, Coleoptera: Chrysomelidae) on new leaves of damaged black mustard plants. Environ Entomol 31:714–722
Tucić N, Milanović D, Mikuljanac S (1995) Laboratory evolution of host plant utilization in the bean weevil (Acanthoscelides obtectus). Genet Sel Evol 27:491–502
Valverde PL, Fornoni J, Nunez-Farfan J (2001) Defensive role of leaf trichomes in resistance to herbivorous insects in Datura stramonium. J Evol Biol 14:424–432
Via S (1990) Ecological genetics and host adaptation in herbivorous insects—the experimental study of evolution in natural and agricultural systems. Annu Rev Entomol 35:421–446
Via S (1991) Specialized host plant performance of pea aphid clones is not altered by experience. Ecology 72:1420–1427
Wood SN (2003) Thin-plate regression splines. J Roy Stat Soc B 65:95–114
Wood SN (2011) Fast stable restricted maximum likelihood and marginal likelihood estimation of semiparametric generalized linear models. J Roy Stat Soc B 73:3–36
Acknowledgements
The authors thank the gardeners of Bielefeld University and Helge Landskron for plant cultivation and insect breeding. We thank Ann-Kathrin Höwing and Stefanie Bukovec for help with the video analyses. This work was funded by the grant MU 1829/3-1 of the Deutsche Forschungsgemeinschaft.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kühnle, A., Müller, C. Prefeeding and Acceptance Behavior of an Oligophagous Beetle is Dependent on Plant Suitability and Rearing History. J Insect Behav 25, 155–165 (2012). https://doi.org/10.1007/s10905-011-9284-6
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10905-011-9284-6