Abstract
Michigan has the 17th highest adult obesity rate in the United States. Among college-aged adults between 18 and 25 years old, the rate of obesity was 11.6%. Obesity is a key precedent for the development of metabolic syndrome. Accordingly, the purpose of this study was to examine the prevalence of metabolic syndrome and its individual components among a sample of students at Central Michigan University. A cross-sectional survey was conducted among 462 students, aged 18–25 years, in Spring 2015 and Fall/Spring 2016 semesters. Students were recruited throughout the campus via flyers, in-class, and Blackboard announcements. Biochemical, anthropometric, and blood pressure measurements were taken for all students. Prevalence of metabolic syndrome was estimated based on the National Cholesterol Education Program’s Adult Treatment Panel III guidelines. Multivariable analysis was used to assess the prevalence of metabolic risk components. To explore the association between metabolic risk factors and lifestyle behaviors, students filled out a validated online questionnaire related to their eating habits, physical activity, and sleep patterns. Metabolic syndrome was not prevalent in our sample. However, about one-third of the students had at least one metabolic abnormality, and 6.0% had two metabolic abnormalities. The most common metabolic abnormalities were low HDL-cholesterol levels (22.0%) and high waist circumference (12.6%), and elevated serum triglyceride (5.8%). Adjusting for other factors, excess adiposity and high visceral fat scores were associated with increased risk of metabolic risk factors, whereas healthy lifestyle practices such as daily breakfast consumption, eating three meals a day, being active, and not smoking were associated with lower risks for MetS. Given the adverse consequences of undiagnosed metabolic abnormalities, efforts to identify and manage MetS among asymptomatic college students, particularly women, is essential and warrants further research.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
According to the American College Health Association-National College Health Assessment (ACHA-NCHA), a nationwide survey conducted in Fall 2015 among 19,861 university students at 2- and 4-year institutions to assess health-related variables, four of ten of the surveyed students were either overweight or obese, 96.2% did not consume the daily recommended five or more servings of fruits and vegetables, and 79.1% did not participate in at least 30 min of moderate-intensity activity at least 5 days per week [1]. Obesity, unhealthy dietary practices, and physical inactivity are risk factors for the development of metabolic syndrome (MetS) [2–5], which is considered as a clinical marker to identify individuals at high risk for type 2 diabetes and cardiovascular disease (CVD) [6–11].
MetS prevalence has been documented among college students in the range of 0.6–13% [4, 12–17]. Findings from the Young Adult Health Risk Screening Initiative study, conducted among 2722 college students aged 18–24 years, showed that 3% of females and 10% of the male participants have MetS [14]. The authors also reported that 54% of the female participants and 77% of the males had at least one risk factor for MetS (low concentrations of HDL-C in women and elevated blood pressure in men) [14]. Similarly, Huang et al. noted that 26–40% of students at the University of Kansas had at least one metabolic risk factor (low concentrations of HDL-C) [4]. These findings are worrisome since individuals with MetS appear to have a two-fold increase in risk for CVD and a four-fold increase risk for type 2 diabetes [14]. Thus, efforts for early detection and intervention of MetS risk factors among college students is essential to prevent or reduce the development of MetS and, eventually, cardiovascular health problems later in life [18–21].
Michigan currently ranks 17th in adult obesity rate in the United States [22]. According to the Centers for Disease Control and Prevention (CDC), 30.7% of Michigan adults (31.1% female, 31.9% male) were obese in 2014 compared to 13.2% in 1990 [23], representing a 132.6% increase in the obesity rate. Among young adults between 18 and 25 years old, the rate of obesity was 11.6% [23]. Given the high rate of obesity in Michigan, and the long-term health consequences of metabolic abnormalities in young adults [24], the need for early screening and management of metabolic risk factors among college students has never been greater to prevent the development of MetS or halt the progression of metabolic risk factors into advanced disease later in life [4, 5, 24].
Universities are unique settings for raising awareness about obesity’s comorbidities and for identifying students who are at risk for developing MetS when appropriate intervention strategies to manage MetS risk factors can be implemented at an early age [24]. According to the Health Belief Model, risk perception is a primary motive to change behavior, and the greater the perceived threat, the more likely an individual will change his/her behavior [25, 26]. Students who are screened and thereby become aware of their MetS risk factors are more likely to perceive themselves as at risk and take appropriate behavioral lifestyle changes to manage their metabolic risk factors [27], whereas students who are unaware of their MetS risk factors (or not screened) may not perceive themselves as at risk for this condition and, consequently, may go undiagnosed until cardiovascular and diabetes complications occur [27]. Therefore, screening students for MetS risk factors is vital to developing any prevention strategy to reduce MetS among college students [28, 29]. Thus, the primary objective of this study was to estimate the prevalence of MetS among a sample of undergraduate students at Central Michigan University (CMU). The secondary objectives were to assess which of the MetS criteria are most prevalent among these students and to relate the criteria of MetS present in students to their body mass index (BMI)/adiposity, eating habits, sleep pattern, and physical activity.
Methods
Design and Sample
This study was a cross-sectional survey conducted in a convenience sample of 462 students. Of the 462 students who volunteered to participate, 364 students (78.3% females and 21.7% males) completed all the required study assessments, yielding a study completion rate of 78.8%. Students were recruited during spring 2015 and fall/spring 2016 semesters through invitations the researchers presented in Foods and Nutrition classes, via Blackboard announcements, through posters displayed in the Foods and Nutrition building, and by direct recruitment by a CMU Nutrition and Dietetics faculty member. The Foods and Nutrition classes were introductory nutrition classes offered to all undergraduate students as University Program (UP)/General Education courses. Eligibility criteria were the following: undergraduate student between the age of 18 and 25 years, not taking any medications, drugs, or experiencing any illnesses, not dieting, and not pregnant. Students voluntarily entered the study and were provided adequate information about the study’s protocol and methodology. Students agreeing to participate were asked to sign a consent form, in harmony with the Helsinki declaration, and then to come to a laboratory for anthropometric measurements and to receive a numerical code for completing a self-administered online questionnaire. Also, students were asked to report to a local hospital for clinical screening, which included measurement of blood pressure and determination of fasting blood lipid and glucose concentrations. Students were not offered any incentives for their participation. The study protocol was approved by the CMU Institutional Review Board (IRB) (IRB project ID: 678896-11).
Data Collection
Anthropometric Measurements
Data collection took place in two steps. First, students’ anthropometric measurements including height, weight, waist circumference, percentage body fat, and visceral fat score were measured by a CMU Nutrition and Dietetics professor and trained undergraduate senior students, using a standardized protocol in accordance with the CDC’s Anthropometry Procedures Manual [30]. Weight, percentage body fat, visceral fat score, and BMI were measured using a Tanita body composition analyzer (SC-331S) (Tanita, Arlington Heights, IL, U.S.). The student’s height, age, and gender were entered into the Tanita scale before testing. Then, wearing light clothing (a single layer of clothing), the student stepped into the Tanita scale footpads with bare feet (both feet touching the electrodes). Weight, percentage body fat, visceral fat, and BMI were recorded from the Tanita body composition analyzer readings. As fluctuations in body hydration status may affect body composition results, students were instructed to fast and to refrain from any heavy physical activity before Tanita scale measurements were taken in the morning (within 3 h after waking up). Also, students were asked to wipe off the bottom of their feet before stepping onto the measuring platform, since unclean foot pads may interfere with conductivity [27]. Height was measured to the nearest 0.1 cm using a height rod (Seca stadiometer model 217, Quick Medical, Issaquah, WA, U.S.). Students were asked to take off their shoes for height measurements. BMI was used to assess students’ weight status [31]. Weight status was stratified into four groups based on the guidelines published by the CDC for BMI classifications: underweight (BMI < 18.5 kg/m2), normal weight (18.5 ≤ BMI < 24.9 kg/m2), overweight (25 ≤ BMI < 29.9 kg/m2), and obese (BMI ≥ 30 kg/m2) [32]. The healthy range for body fat percentage was considered as 8–19% for males and 17–32% for females (Tanita body fat ranges for healthy adults). Visceral fat ratings from 1 to 12 were considered healthy while ratings from 13 to 59 indicated an excess level of visceral fat (Tanita visceral fat ranges for healthy adults). Waist circumference was measured using a non-stretchable tape (QM2000 Measure Mate, Quick Medical, Issaquah, WA, U.S.) in accordance with the CDC’s Anthropometry Procedures Manual [30].
Biochemical Tests and Blood Pressure Measurement
Students were provided with a free voucher for blood testing and were asked to report to a local hospital (McLaren-Central Michigan Hospital, Mount Pleasant, MI, U.S.) to have their blood pressure taken and to provide a venous blood sample (after an overnight fast of at least 10–12 h) to measure their blood lipids (total cholesterol, LDL-cholesterol, HDL-cholesterol, and serum triglyceride), and fasting blood glucose (FBS). The volume of the venous blood sample that was taken from students was about 8.5 ml. Prevalence of metabolic syndrome was estimated based on the National Cholesterol Education Program’s Adult Treatment Panel (NCEP-ATP) III revised guidelines [33]. MetS was identified in students when three or more of the following criteria were met: increased waist circumference (>102 cm for males and >88 cm for females), elevated blood pressure (systolic arterial BP ≥ 130 mmHg and/or diastolic arterial BP ≥ 85 mmHg), elevated serum triglyceride (≥150 mg/dl), reduced HDL cholesterol (HDL-C) (<40 mg/dl for males and <50 mg/dl for females), and elevated FBS (≥100 mg/dl) [33].
Online Questionnaire
Demographic and Student Characteristics
In the second step, students were asked to complete a self-administered online questionnaire consisting of questions related to age, gender, year-of-study at CMU, major of study, living condition (on/off campus), dietary habits, physical activity, sleep pattern, smoking status, family history, and the results of their anthropometric measurements, blood pressure, and biochemical tests. The demographic and student characteristics questions were adapted from Yahia et al. [34]. The dietary habits questions were adopted from a previous study by Turconi et al. [35] and were validated for use among university students in previous studies [34, 35].
Physical Activity
Physical activity was assessed using the International Physical Activity Questionnaire -short form (IPAQ-S) [36]. The IPAQ-S form consists of seven questions that assess physical activity at three levels (vigorous-intensity, moderate-intensity, and walking) undertaken by students for the last consecutive 7 days across four different physical activity domains (leisure time physical activity, domestic and gardening (yard) activities, work-related physical activity, and transport-related physical activity). Examples of vigorous-intensity activity include aerobics or fast bicycling, and moderate-intensity activities include bicycling at a regular pace or doubles tennis [36, 37]. The IPAQ obtains data about the frequency and the amount of time spent across each activity domain. Computation of the total score for the weekly physical activity (PA) was determined by summation of the duration (in minutes) and frequency (days) reported within each activity domain (i.e. vigorous-intensity, moderate-intensity and walking) multiplied by an appropriate Metabolic Equivalent of activity (MET) assigned to each category as follows: walking MET-minutes/week = 3.3 × walking minutes × walking days; moderate MET-minutes/week = 4.0 × moderate-intensity activity minutes × moderate days, and vigorous MET-minutes/week = 8.0 × vigorous-intensity activity minutes × vigorous-intensity days. The IPAQ assigns individual MET levels of 8.0, 4.0, and 3.3 to vigorous-intensity, moderate-intensity, and walking activities, respectively. The overall PA score expressed as MET-minutes/week, was determined using the following formula: total physical activity MET-minutes/week = sum of walking + moderate + vigorous MET-minutes/week scores [36, 37]. According to the IPAQ scoring protocol, students were categorized into three groups as follows: low (>600 MET-minutes/week), moderate (600–1500 MET-minutes/week), and high (1500 MET-minutes/week). A detailed description of the scoring methodology is available online at http://www.ipaq.ki.se [37] and discussed in a previous study [38].
Sleep Pattern
Subjects’ sleep patterns were assessed using the Pittsburgh Sleep Quality Index (PSQI) [39]. The PSQI is a standardized self-rated questionnaire consisting of 19 questions that assess a wide variety of factors related to sleep quality, including estimates of sleep duration and latency and the frequency and severity of specific sleep-related problems, over a 1-month time interval (for the last 30 days). According to the PSQI scoring protocol, the questions were grouped into seven component scores; each weighted equally on a scale of 0–3. The seven component scores were then summed to yield a global PSQI score, which ranged between 0 and 21; the higher the score, the worse the sleep quality [39]. Approval to use the questionnaire was obtained from the author prior to the onset of the study [39].
The online questionnaire was available online via SurveyMonkey Pro (SurveyMonkey.com, LLC, Palo Alto, CA) for about 10 weeks to accommodate students’ response times. Students were given instructions on how to fill out the questionnaire completely and were informed that they could withdraw from the study at any time. The online questionnaire was pilot-tested on a randomly selected group of 20 students before its administration.
Data Analysis
Statistical analyses were performed using the SAS (9.3, Cary, NC) software. Gender differences for study variables were assessed using Student’s independent t test, Wilcoxon Rank-Sum test, and Chi-squared test for independence. Fisher’s Exact test was used to assess gender differences in the prevalence of individual metabolic risk components and the Chi-squared test was used to assess gender differences in the total number of MetS components, and also the prevalence of at least one MetS component. Chi-squared test, Fisher Exact test and the Wilcoxon Rank-sum test were used to compare life style characteristics between students with at least one MetS component compared with students with no MetS components, stratified by gender. Results are expressed as means ± SD (standard deviation) for continuous variables and percent frequency for categorical variables. All reported p values were made on the basis of 2-sided tests and compared to significance level of 5%.
Results
Participants’ Characteristics
The demographic characteristics of the study sample are presented in Table 1. More than two-thirds of the study participants were female with a mean age of 20.52 ± 1.52 years. The mean height and weight of the female students were 164.4 ± 6.66 cm and 64.6 ± 11.23 kg, respectively, whereas the mean height of the male students was 178.31 ± 7.32 cm and the mean weight was 78.85 ± 13.73 kg (Table 2). Of the participating students, 87.4% were white, 3.1% were African-American, 2.8% were Asian, and 6.7% reported another ethnic origin, reflecting the composition of ethnic groups at CMU. A higher percentage of participants were in their 1st (23.1%) and 4th years (25.6%) of academic study. Eighty-seven percent of participants (88.6% female, 79.7% male) reported being non-smokers; 1.4% reported current cigarette usage; 3.6% were former smokers; and 8.3% were occasional smokers. The majority (83.5%) of both male (89%) and female students (82%) reported sleeping more than 7 h per night. The mean total PSQI for male students was 5.31 ± 2.09 and for females was 5.61 ± 2.66 (Table 1). Male students reported being more active than female students. Vigorous activity was significantly more common among male students than female students (P < 0.001) (Table 1).
Overall, except for sleep pattern, there were no significant differences between the characteristics of the 98 students (74 female and 24 male) who did not complete the entire study and those who completed all the study’s requirements, as presented in Table 3.
Anthropometric Parameters
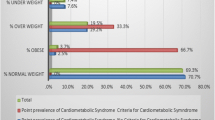
Results indicated that 63.9% of the female and 51.9% of the male students were within the healthy BMI range, and 30.5% of females and 46.8% of males were overweight or obese (Fig. 1). Obesity was more prevalent in females (7.7%) than in males (2.5%), yet more men (44.3%) than women (22.8%) were overweight. Overall mean values of BMI and percentage body fat were 24.0 ± 3.59 and 24.9 ± 8.66 kg/m2, respectively (Table 2). Male students had a significantly lower percentage of body fat but higher waist circumference (14.4 ± 6.49%, 85.3 ± 8.25 cm) than female students (27.9 ± 6.68%, 78.9 ± 11.5 cm) (P < 0.001) (Table 2). There were no significant differences between male and female students regarding visceral fat. However, women had a lower mean value of visceral body (2.1 ± 1.53) than men (2.4 ± 2.27) (Table 2).
Clinical and Biochemical Parameters
Blood pressure and biochemical measurements by gender are presented in Table 2. Blood pressure values for male and female students were within the normal ranges. However, women had significantly lower mean values of systolic blood pressure (113.7 ± 10.91 mmHg) and diastolic blood pressure (67.7 ± 7.72 mmHg) (P < 0.0001) than men (119.6 ± 11.00 mmHg, 71.4 ± 8.68 mmHg, respectively) (Table 2). Values of high-density lipoprotein cholesterol were normal for both genders, but values were significantly higher for female (60.2 ± 13.26 mg/dl) than for male students (50.2 ± 11.77 mg/dl) (P < 0.001) (Table 2). Also, female students had higher serum cholesterol levels but lower mean values of FBS (84.7 ± 5.81 mg/dl) than male students (86.7 ± 5.78 mg/dl) (Table 2).
Metabolic Syndrome Prevalence
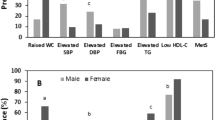
Based on the NCEP ATP definition of MetS, MetS was not prevalent in our sample (Fig. 2). However, 29.1% of the students had at least one MetS abnormality (30.2% of females and 25.3 of males), and 6.0% had two metabolic abnormalities (2.5% of males and 7.0% of females) (Fig. 2). The most common metabolic abnormalities were low HDL-cholesterol (defined as <50 mg/dl for women and <40 mg/dl for men) (22.0%) (19.0% in males and 22.8% in females) and high waist circumference (defined as >88 cm for women and >120 cm for men) (12.6%) (2.5% in males and 15.4% in females) (Fig. 3). The percentage of female students with elevated waist circumference was significantly greater than that of males (P = 0.001) (Fig. 3). Elevated blood pressure (defined as systolic arterial BP ≥ 130 mmHg and diastolic arterial BP ≥ 85 mmHg) was the least common MetS abnormality (0.5%) found among students (1.3% in males and 0.4% in females) (Fig. 3).
Metabolic Syndrome Prevalence in Relation to Body Fat and Lifestyle Behavioral Practices
In this study, analyses of anthropometric parameters in relation to MetS risk factors showed that students with at least one MetS risk factor had significantly higher BMI, body fat percentage, and visceral fat scores than students with no MetS risk factors, as shown in Table 4.
In addition, results of the association between lifestyle-related practices and MetS risk factors revealed that a higher percentage of students with no MetS risk factors reported daily consumption of breakfast intake and three meals a day compared to students with at least one metabolic risk factor. Likewise, students with no MetS risk factors were more likely to be more active and less likely to smoke than students with at least one MetS risk factor (Table 5). However, sleep patterns did not differ between students with at least one MetS risk and those without (Table 5).
Discussion
Despite the high rate of obesity in Michigan [23], there have been no published studies on the prevalence of MetS among Michigan university students or young adults. In the present study, we aimed to assess the prevalence of MetS among a sample of Central Michigan University students and which criteria of MetS were most prevalent in our participants. Also, we tried to evaluate whether there were any differences in the number of criteria of MetS found among students based on their weight status, eating habits, sleep pattern, or physical activity levels.
Using the NCEP-ATP III criteria, the overall prevalence of MetS in our sample was 0.3%. This figure is lower than that reported in previous studies among college students in the U.S., where MetS prevalence was in the range of 0.6–13% [4, 5, 40–44]. For instance, a study by Huang et al. conducted among a sample of 163 undergraduate students (primarily white), aged 18–24 years, at the University of Kansas, reported a prevalence of MetS of 0.6% [5]. Another study by Fernandes and Lofgren, conducted among 189 first year college students, aged 18–24 years old, at the University of Rhode Island, reported a prevalence of MetS of 3.7% [43]. Likewise, Morrell et al. found the prevalence of MetS to be 7.7% in a sample of 299 undergraduate students aged 18–24 years from three geographically diverse universities [40]. Similarly, Keown et al. reported a prevalence of MetS of 10% in a sample of 4192 college students, aged 18–30 years old, at a southeastern university [44]. The variation in the prevalence of MetS in these studies could be due to racial/ethnic differences or to the prevalence of overweight/obesity in the study sample. Findings from a large national study indicated that the prevalence of MetS varies with race and ethnicity, and increases with age and BMI, with non-Hispanic black males being one-half as likely as non-Hispanic white males to have metabolic syndrome, and non-Hispanic black and Mexican–American females being 1.5 times as likely as non-Hispanic white females to have this condition [45]. In our study, the majority of students were white (87.4%) with a low rate of obesity (6.6%) [45].
Alarmingly, the prevalence of MetS among college students was not only reported in the U.S. but also in other countries [9, 15, 46, 47]. For example, Yen et al. reported that 4.6% of university freshmen students (mean age 19.2 years) met the definition of MetS in North Taiwan [15]. Cha et al. reported a MetS prevalence of 12% among a sample of 73 college students (aged 18–29 years) in Korea [46]. Barbosa et al. found a MetS prevalence of 20.5% in a sample of 590 university students of São Luís in Brazil [9]. Also, in Brazil, de Freitas et al. reported that 1.7% of Brazilian college students (mean age 21.5 years) met the criteria of MetS [47]. Literature indicates that individuals with MetS appear to have a two-fold increase in risk for CVD and a four-fold increase in risk for type 2 diabetes [19, 20, 27]. Thus, despite that college students appear to be healthy and at lower risk for chronic disease [24, 27, 40], the findings of these studies reveal the need for early screening to identify those students who may be at risk for MetS so that appropriate intervention strategies can be implemented at an early stage. Otherwise, students who may be at risk for MetS but unaware of it may go undiagnosed until type 2 diabetes and, eventually, CVD occur later in their lives.
In the present study, although the prevalence of MetS was low, about one-third (29.1%) of our students had at least one MetS risk factor. The most prevalent criteria in our sample were low HDL-cholesterol levels (22.0%) and elevated waist circumference (12.6%). Low HDL-C was more common among women than men (22.8% vs. 19.0%, respectively). This finding was not expected since young women, in general, are believed to have higher HDL levels than men [48, 49]. Research indicates that women with low high-density lipoprotein cholesterol levels have higher risk than men for CVD later in life [50]. Nevertheless, the prevalence of low HDL-cholesterol levels in our study was comparable to the 24% reported by Huang et al. [5], possibly due to similarities in students’ age range and ethnicity, but lower than that of Fernandes and Lofgren (20%) [43] and Tope et al. (37.3%) [41], while higher than that of Morrell et al. (20.1%) [40].
As for the second prevalent criterion of MetS (elevated waist circumference) in our study, central obesity was significantly more common in females (15.4%) than in male students (2.5%) (P = 0.001). This observation was unexpected. Generally, during college years, women are more likely to be concerned about their body weight and shape than men [51–53]. Indeed, among women, central obesity is a leading risk factor for MetS and future development of cardiovascular disease (2, 47). Similar to this study, previous research also reported excess adiposity and central obesity among students with MetS (43, 44). In fact, in our study, students who had at least one metabolic risk factor had a significantly higher percentage of body fat and visceral fat score than students with no metabolic risk factors (Table 4).
Concerning gender, a greater percentage of female students (37.5%), although not significant, presented with at least one MetS abnormality than male students (27.8%). Likewise, previous studies have also indicated more MetS risk factors in females than males (42, 48). However, some studies noted that men had more MetS risk factors than women (39, 40, 44). Irrespective of gender, the presence of MetS risk factors among students is of concern given the future health problems associated with MetS and underscores the importance of early screening to identify students at risk for MetS, especially since most college students are unaware of their risks and perceive themselves as healthy [54]. Thus, early identification and management of metabolic risk factors are essential to reduce the onset or progression of MetS and lifetime CVD risk. Simple prevention strategies such as adherence to lifestyle changes involving healthful diet and physical activity can lead to an 82% reduction in chronic diseases [55]. Accordingly, developing a primary prevention program with a gender-specific approach to lower MetS risk factors among students within the university setting would be beneficial.
In this study, the association between lifestyle-related practices and MetS risk factors revealed that daily breakfast consumption and eating three meals a day, in addition to physical activity and not smoking, were associated with lower metabolic risk factors. This finding was in line with previous research [56–58]. Smith et al. found a higher prevalence of MetS risk factors among students who reported a high consumption of fast food and poor eating habits, and low levels of physical activity [59]. Accordingly, the International Diabetes Federation (IDF) recommended healthy lifestyle practices, such as a healthy diet plan and a moderate increase in physical activity, as primary management strategies to combat MetS [60]. Although previous research suggested that smoking and exposure to smoke were associated with increased risk of MetS [9, 61], in our study, smoking did not appear to impact the incidence of metabolic risk factors in part due to the majority of our students reported being non-smokers.
Collectively, findings from this study highlight the importance of routine screening and early detection of metabolic risk factors among college students so that appropriate intervention strategies can be implemented early in life to reduce the lifetime risk of developing CVD and type 2 diabetes.
During college, students establish independence in decision making and experience more freedom in their lifestyle choices [34]. Thus, dietary practices and lifestyle choices developed during this transitional stage in life may persist into adulthood and may have long-term influences on future health [62]. College life can thus be considered an important opportunity for students to adopt healthy eating patterns, choose healthier food, get enough sleep, exercise regularly, and reduce sedentary time, which all are critical factors in preventing the risk of metabolic diseases [63, 64]. Adopting a Mediterranean-style diet rich in polyunsaturated fatty acids (25% fat), soluble fiber, flavonoids, and antioxidants has been shown to lower metabolic syndrome risk factors among obese children and adolescents with obesity [63].
Screening students for metabolic risk factors should be a priority for college educators and college administrators. As seen in the current study, although the prevalence of MetS was low in our sample of students, 30.2% of female participants and 25.3% of the male participants had at least one MetS abnormality. These findings are alarming given that the majority of college students consider themselves in good health [1] and are unaware of their health risk [65]. Thus, there is a need for new approaches to diagnosing early stages of metabolic diseases without the evidence of visible clinical manifestations. Previous studies have indicated that some obese individuals do exhibit a normal metabolic profile despite having high BMI values [referred to as metabolically healthy obese (MHO)] [66]. However, research shows that metabolically healthy obese subjects were at higher risk for developing long-term cardiovascular complications and all-cause mortality compared to those individuals whose BMI was within a healthy weight range [67]. It has been suggested that “healthy” obesity is a transient state that is associated with younger age, lower waist circumferences, and a more peripheral fat distribution [67, 68]. Accordingly, for younger adults who are obese without metabolic abnormalities targeted intervention opportunities including modifications in diet, physical activity, and modifiable lifestyle factors are recommended to reduce the incidence of type 2 diabetes and CVD later in life.
Limitations
As the first study in Michigan to explore the prevalence of MetS in a sample of college students using standardized methodologies, the results make a significant contribution to the literature. However, the study had limitations. One limitation is the cross-sectional design, which does not permit causal inferences. Other limitations are the small sample size and the fact that the majority of the students were Caucasian, with ethnic minorities underrepresented, making it difficult to generalize about all university students. Also, more female than male students participated in this study despite our attempts to recruit a balanced sample. The unequal ratio of female to male participants in this study might be due to the nature of the student recruitment and student motivation. In this study, student participation was voluntary and not mandatory. Students were not provided with any incentive to participate in this study. Further, it is possible that female students may have been more interested in research related to health issues than male students (given that students voluntarily entered into the study) [69]. Another possible factor is the blood testing requirement. This study involved a venous blood sampling which is not comfortable for many students. Undoubtedly, having more men may have allowed for detection of gender differences in MetS prevalence. Despite trying to recruit students (both male and female) from all majors to reflect the student population, few men chose to participate in this study. Nevertheless, the dominance of female participants reflects the university’s student body data and is consistent with the gender composition of previous studies [1, 40, 43]. In the American College Health Association-National College Health Assessment (ACHA-NCHA) survey conducted among 19,861 university students at 2- and 4-year institutions to assess health-related variables, 64.7% of the study participants were females and 32.4% were male students [1]. The study by Morrell et al. conducted among 360 undergraduate students from three large universities in the U.S. also reported that more than two-thirds (68%) of their participants were women [40]. Another study by Fernandez and Lofgren indicated that 68% of the study participants were female students [43]. Therefore, an important recommendation for future studies is to increase the number of males so that gender differences can be detected.
Lastly, a possible additional limitation is the voluntary nature of the recruitment (self-selected), which may introduce a sample bias. Students who were interested in health or highly motivated to measure their body fat composition may have participated in the study more readily, while students who were having weight problems or were sensitive about their weight may not have chosen to participate in this study, leading to underestimation of MetS prevalence in our sample. Despite these limitations, baseline data about MetS prevalence and the most common MetS criteria among students were obtained. Also, this study did not use self-reported weight and height; instead, all anthropometric variables were measured by the same faculty professor and well-trained senior students using a standardized protocol. Unlike in other studies, biochemical measures were collected by a venous blood sampling rather than via fingerstick to obtain accurate measurements of FBS and blood lipids. Therefore, the study’s results may help researchers develop more appropriate prevention strategies tailored to students’ risk profile to reduce possible metabolic risk factors among students and improve health in the campus community.
Conclusion
Findings from this study indicate that despite the low prevalence of MetS in our sample, about one-third of the students had at least one MetS criterion. Accordingly, these findings underscore the need for early screening to identify those students who are at risk so appropriate prevention strategies can be implemented to reduce or prevent the development of MetS and future related co-morbidities. Without early screening, metabolic risk factors among students may go unrecognized and get worse over time leading to disease progression. Overall, findings from this study may help to inform future longitudinal studies and highlight the need for early screening for metabolic risk factors among college students to reduce the risk of MetS as well as related comorbidities later in life.
Abbreviations
- MetS:
-
Metabolic syndrome
- CVD:
-
Cardiovascular disease
- ACHA-NCHA:
-
American College Health Association-National College Health Assessment
- CDC:
-
Centers for Disease Control and Prevention
- NCEP-ATP:
-
National Cholesterol Education Program’s Adult Treatment Panel
- BRFSS:
-
Behavioral Risk Factor Surveillance System
References
American College Health Association. (2016). American College Health Association-National College Health Assessment II: Reference group executive summary fall 2015. Hanover, MD: American College Health Association. http://www.acha-ncha.org/docs/NCHA-II%20FALL%202015%20REFERENCE%20GROUP%20EXECUTIVE%20SUMMARY.pdf. Accessed 7 April 2016.
Grundy, S. M., Brewer, H. B. Jr., Cleeman, J. I., Smith, S. C. Jr., Lenfant, C., & American Heart Association, & National Heart, Lung, and Blood Institute. (2004). Definition of metabolic syndrome: Report of the National Heart, Lung, and Blood Institute/American Heart Association conference on scientific issues related to definition. Circulation, 109(3), 433–438.
Carnethon, M. R., Loria, C. M., Hill, J. O., Sidney, S., Savage, P. J., Liu, K., & Coronary Artery Risk Development in Young Adults Study. (2004). Risk factors for the metabolic syndrome: The Coronary Artery Risk Development in Young Adults (CARDIA) study, 1985–2001. Diabetes Care, 27(11), 2707–2715.
Huang T. T. K, Shimel, A., Lee, R. E., Delancey, W., & Strother, M. L. (2007). Metabolic risks among college students: Prevalence and gender differences. Metabolic Syndrome and Related Disorders, 5(4), 365–372.
Huang, T. T., Kempf, A. M., Strother, M. L., Li, C., Lee, R. E., Harris, K. J., & Kaur, H. (2004). Overweight and components of the metabolic syndrome in college students. Diabetes Care, 27(12), 3000–3001.
Galassi, A., Reynolds, K., & He, J. (2006). Metabolic syndrome and risk of cardiovascular disease: A meta-analysis. The American Journal of Medicine, 119(10), 812–819. doi:10.1016/j.amjmed.2006.02.031.
Malik, S., Wong, N. D., Franklin, S. S., Kamath, T. V., L’Italien, G. J., Pio, J. R., & Williams, G. R. (2004). Impact of the metabolic syndrome on mortality from coronary heart disease, cardiovascular disease, and all causes in United States adults. Circulation, 110(10), 1245–1250.
Patel, T. P., Rawal, K., Bagchi, A. K., Akolkar, G., Bernardes, N., da Silva Dias, D., et al. (2016). Insulin resistance: An additional risk factor in the pathogenesis of cardiovascular disease in type 2 diabetes. Heart Failure Reviews, 21(1), 11–23.
Barbosa, J. B., Santos A. M. D., Barbosa, M. M., Barbosa, M. M., Carvalho C. A. D., Fonseca, P. C. D. A., et al. (2016). Metabolic syndrome, insulin resistance and other cardiovascular risk factors in university students. Ciencia & Saude Coletiva, 21(4), 1123–1136.
Eckel, R. H., Grundy, S. M., & Zimmet, P. Z. (2005). The metabolic syndrome. The Lancet, 365(9468), 1415–1428. doi:10.1016/S0140-6736(05)66378-7.
Alberti, K. G., Zimmet, P., Shaw, J., & Group IDFETFC. (2005). The metabolic syndrome—a new worldwide definition. The Lancet, 366(9491), 1059–1062. doi:10.1016/S0140-6736(05)67402-8.
Burke, J. D., Reilly, R. A., Morrell, J. S., & Lofgren, I. E. (2009). The University of New Hampshire’s young adult health risk screening initiative. Journal of the American Dietetic Association, 109(10), 1751–1758.
Huang, T. T. K., Kempf, A. M., Strother, M. L., Li, C., Lee, R. E., Harris, K. J., & Kaur, H. (2004). Overweight and components of the metabolic syndrome in college students. Diabetes Care, 27(12), 3000–3001.
Morrell, J. S., Lofgren, I. E., Burke, J. D., & Reilly, R. A. (2012). Metabolic syndrome, obesity, and related risk factors among college men and women. Journal of American College Health, 60(1), 82–89.
Yen, S. L., Chiu, T. Y., Lin, Y. C., Lee, Y. C., Lee, L. T., Huang, K. C. (2008). Obesity and hepatitis B infection are associated with increased risk of metabolic syndrome in university freshmen. International Journal of Obesity (London), 32(3), 474–480.
Mattsson, N., Ronnemaa, T., Juonala, M., Viikari, J. S., & Raitakari, O. T. (2007). The prevalence of the metabolic syndrome in young adults. The Cardiovascular Risk in Young Finns Study. Journal of Internal Medicine, 261(2), 159–169. doi:10.1111/j.1365-2796.2006.01752.x.
Yahia, N. H., Hayek, G., & Shahin, S. (2011). Obesity, metabolic syndrome, and the Mediterranean diet in a sample of Lebanese university students. Nutrition Today, 46(3), 138–145. doi:10.1097/NT.0b013e31821a82dd.
Schilter, J., & Dalleck, L. (2010). Fitness and fatness: Indicators of metabolic syndrome and cardiovascular disease risk factors in college students? Journal of Exercise Physiology Online, 13(4), 29–39.
Sparling, P. B., Beavers, B. D., & Snow, T. K. (1999). Prevalence of coronary heart disease (CHD) risk factors in a college population. Medicine & Science in Sports & Exercise, 31(5), S254.
Spencer, L. (2002). Results of a heart disease risk-factor screening among traditional college students. Journal of American College Health, 50(6), 291–296.
Hlaing, W., Nath, S. D., & Huffman, F. G. (2007). Assessing overweight and cardiovascular risks among college students. American Journal of Health Education. doi:10.1080/19325037.2007.10598948.
Robert Wood Johnson Foundation. (2016). About state of obesity: The state of obesity. Princeton: Robert Wood Johnson Foundation. http://stateofobesity.org/about/.
DPH, & CDC. (2016). Behavioral risk factor surveillance system (BRFSS) prevalence & trends data: Explore by location. http://nccd.cdc.gov/BRFSSPrevalence/rdPage.aspx?rdReport=DPH_BRFSS.ExploreByLocation&rdProcessAction=&SaveFileGenerated=1&islLocation=26&islClass=CLASS01&islTopic=Topic03&islYear=2014&hidLocation=26&hidClass=CLASS01&hidTopic=Topic03&hidTopicName=Alcohol+Consumption&hidYear=2014&irbShowFootnotes=Show&iclIndicators_rdExpandedCollapsedHistory=&iclIndicators=DRNKANY5&hidPreviouslySelectedIndicators=&DashboardColumnCount=2&rdShowElementHistory=&go=GO&rdScrollX=0&rdScrollY=0&rdRnd=28900.
Turchiano, M., Sweat, V., Fierman, A., & Convit, A. (2012). Obesity, metabolic syndrome, and insulin resistance in urban high school students of minority race/ethnicity. Archives of Pediatrics and Adolescent Medicine, 166(11), 1030–1036. doi:10.1001/archpediatrics.2012.1263.
Becker, M. H., Maiman, L. A., Kirscht, J. P., Haefner, D. P., & Drachman, R. H. (1977). The health belief model and prediction of dietary compliance: A field experiment. Journal of Health and Social Behavior, 18(4), 348–366.
Rosenstock, I. M., Strecher, V. J., & Becker, M. H. (1988). Social learning theory and the health belief model. Health Education Quarterly, 15(2), 175–183.
Yahia, N., Brown, C., Rapley, M., & Chung, M. (2014). Assessment of college students’ awareness and knowledge about conditions relevant to metabolic syndrome. Diabetology & Metabolic Syndrome, 6(1), 111. doi:10.1186/1758-5996-6-111.
Sarti, C., & Gallagher, J. (2006). The metabolic syndrome: Prevalence, CHD risk, and treatment. Journal of Diabetes and Its Complications, 20(2), 121–132. doi:10.1016/j.jdiacomp.2005.06.014.
Isomaa, B., Almgren, P., Tuomi, T., Forsen, B., Lahti, K., Nissen, M. et al. (2001). Cardiovascular morbidity and mortality associated with the metabolic syndrome. Diabetes Care, 24(4), 683–689.
Centers for Disease Control and Prevention. (2004). National health and nutrition examination survey (NHANES): Anthropometry procedures manual. Atlanta: Centers for Disease Control and Prevention. http://www.cdc.gov/nchs/data/nhanes/nhanes_03_04/bm.pdf.
About Adult BMI | Healthy Weight | CDC. (2016). https://www.cdc.gov/healthyweight/assessing/bmi/adult_bmi/index.html.
Expert Panel on the Identification, Evaluation, and Treatment of Overweight in Adults. (1998). Clinical guidelines on the identification, evaluation, and treatment of overweight and obesity in adults: Executive summary. The American Journal of Clinical Nutrition, 68(4), 899–917.
National Cholesterol Education Program Expert Panel on Detection E, Treatment of High Blood Cholesterol in A. (2002). Third report of the National Cholesterol Education Program (NCEP) expert panel on detection, evaluation, and treatment of high blood cholesterol in adults (Adult Treatment Panel III) final report. Circulation, 106(25), 3143–3421.
Yahia, N., Wang, D., Rapley, M., & Dey, R. (2016). Assessment of weight status, dietary habits and beliefs, physical activity, and nutritional knowledge among university students. Perspectives in Public Health, 136(4), 231–244. doi:10.1177/1757913915609945.
Turconi, G., Guarcello, M., Maccarini, L., Cignoli, F., Setti, S., Bazzano, R., & Roggi, C. (2008). Eating habits and behaviors, physical activity, nutritional and food safety knowledge and beliefs in an adolescent Italian population. Journal of the American College of Nutrition, 27(1), 31–43.
Craig, C. L., Marshall, A. L., Sjostrom, M., Bauman, A. E., Booth, M. L., Ainsworth, B. E., et al. (2003). International physical activity questionnaire: 12-country reliability and validity. Medicine and Science in Sports and Exercise, 35(8), 1381–1395.
Lee, P. H., Macfarlane, D. J., Lam, T. H., & Stewart, S. M. (2011). Validity of the international physical activity questionnaire short form (IPAQ-SF): A systematic review. International Journal of Behavioral Nutrition and Physical Activity, 8, 115.
Yahia, N., Abdallah, A., Achkar, A., & Rizk, S. (2010). Physical activity and smoking habits in relation to weight status among Lebanese University students. International Journal of Health Research, 3(1), 21–27. http://www.ajol.info/index.php/ijhr/article/view/70265.
Buysse, D. J., Reynolds, C. F. 3rd, Monk, T. H., Berman, S. R., & Kupfer, D. J. (1989). The Pittsburgh Sleep Quality Index: A new instrument for psychiatric practice and research. Psychiatry Research, 28(2), 193–213.
Morrell, J. S., Byrd-Bredbenner, C., Quick, V., Olfert, M., Dent, A., & Carey, G. B. (2014). Metabolic syndrome: comparison of prevalence in young adults at 3 land-grant universities. Journal of American College Health, 62(1), 1–9. doi:10.1080/07448481.2013.841703.
Tope, A. M., & Rogers, P. F. (2013). Metabolic syndrome among students attending a historically black college: prevalence and gender differences. Diabetology & Metabolic Syndrome, 5(1), 2. doi:10.1186/1758-5996-5-2.
Dalleck, L. C., & Kjelland, E. M. (2012). The prevalence of metabolic syndrome and metabolic syndrome risk factors in college-aged students. American Journal of Health Promotion, 27(1), 37–42.
Fernandes, J., & Lofgren, I. E. (2011). Prevalence of metabolic syndrome and individual criteria in college students. Journal of American College Health, 59(4), 313–321. doi:10.1080/07448481.2010.508084.
Keown, T. L., Smith, C. B., & Harris, M. S. (2009). Metabolic syndrome among college students. The Journal for Nurse Practitioners, 5(10), 754–759. doi:10.1016/j.nurpra.2009.02.013.
Ervin, R. B. (2009) Prevalence of metabolic syndrome among adults 20 years of age and over, by sex, age, race and ethnicity, and body mass index: United States, 2003–2006. National Health Statistics Reports, 13, 1–7.
Cha, E., Burke, L. E., Kim, K. H., Shin, Y. A., & Kim, H. Y. (2010). Prevalence of the metabolic syndrome among overweight and obese college students in Korea. The Journal of Cardiovascular Nursing, 25(1), 61–68. doi:10.1097/JCN.0b013e3181b848be.
de Freitas, R. W. Jr., de Araujo, M. F., Marinho, N. B., de Vasconcelos, H. C., Lima, A. C., Pereira, D. C., et al. (2013). Prevalence of the metabolic syndrome and its individual components in Brazilian college students. Journal of Clinical Nursing, 22(9–10), 1291–1298. doi:10.1111/jocn.12015.
Eapen, D. J., Kalra, G. L., Rifai, L., Eapen, C. A., Merchant, N., & Khan, B. V. (2010). Raising HDL cholesterol in women. International Journal of Womens Health, 1, 181–191.
The National Heart L, and Blood Institute (NHLBI), The Healthy Heart Handbook for Women, HHS, NIH, NHLBI. U.S. Department of Health & Human Services. http://www.nhlbi.nih.gov/health/educational/hearttruth/downloads/html/hhh/high-blood-cholesterol.htm.
Tan, Y. Y., Gast, G. C., & van der Schouw, Y. T. (2010). Gender differences in risk factors for coronary heart disease. Maturitas, 65(2), 149–160. doi:10.1016/j.maturitas.2009.09.023.
Yahia, N., El-Ghazale, H., Achkar, A., & Rizk, S. (2011). Dieting practices and body image perception among Lebanese university students. Asia Pacific Journal of Clinical Nutrition, 20(1), 21–28.
Lewinsohn, P. M., Seeley, J. R., Moerk, K. C., & Striegel-Moore, R. H. (2002). Gender differences in eating disorder symptoms in young adults. The International Journal of Eating Disorders, 32(4), 426–440. doi:10.1002/eat.10103.
Anderson, C. B., & Bulik, C. M. (2004). Gender differences in compensatory behaviors, weight and shape salience, and drive for thinness. Eating Behaviors, 5(1), 1–11. doi:10.1016/j.eatbeh.2003.07.001.
Munoz, L. R., Etnyre, A., Adams, M., Herbers, S., Witte, A., Horlen, C., et al. (2010). Awareness of heart disease among female college students. Journal of Women’s Health, 19(12), 2253–2259.
Stampfer, M. J., Hu, F. B., Manson, J. E., Rimm, E. B., & Willett, W. C. (2000). Primary prevention of coronary heart disease in women through diet and lifestyle. The New England Journal of Medicine, 343(1), 16–22.
Pan, Y., & Pratt, C. A. (2008). Metabolic syndrome and its association with diet and physical activity in US adolescents. Journal of the American Dietetic Association, 108(2), 276–286 (discussion 286).
Huang TTK, Harris, K. J., Lee, R. E., Nazir, N., Born, W., & Kaur, H. (2003). Assessing overweight, obesity, diet, and physical activity in college students. Journal of American College Health, 52(2), 83–86.
Shafiee, G., Kelishadi, R., Qorbani, M., Motlagh, M. E., Taheri, M., Ardalan, G., et al. (2013). Association of breakfast intake with cardiometabolic risk factors. Jornal de Pediatria, 89(6), 575–582.
Smith, C., & Essop, M. F. (2009). Gender differences in metabolic risk factor prevalence in a South African student population. Cardiovascular Journal of Africa, 20(3), 178–182.
International Diabetes Federation. (2006). The IDF consensus worldwide definition of the Metabolic Syndrome. IDF Communications.
Kelishadi, R., Noori, A., Qorbani, M., Rahimzadeh, S., Djalalinia, S., Shafiee, G., et al. (2016). Are active and passive smoking associated with cardiometabolic risk factors in adolescents? The CASPIAN-III Study. Paediatrics and International Child Health. doi:10.1080/20469047.2015.1109271.
Wengreen, H. J., & Moncur, C. (2009). Change in diet, physical activity, and body weight among young-adults during the transition from high school to college. Nutrition Journal, 8, 32.
Young Intellectual Development. (1997). Guidelines for school health programs to promote lifelong healthy eating. The Journal of School Health, 67(1), 9–26.
Centers for Disease Control and Prevention. (1996). Guidelines for school health programs to promote lifelong healthy eating. MMWR Recommendations and Reports, 45(RR-9), 1–41.
Becker, B. M., Bromme, R., & Jucks, R. (2008). College students’ knowledge of concepts related to the metabolic syndrome. Psychology, Health & Medicine, 13(3), 367–379. doi:10.1080/13548500701405525.
Stefan, N., Haring, H. -U., Hu, F. B., & Schulze, M. B. (2013). Metabolically healthy obesity: epidemiology, mechanisms, and clinical implications. The Lancet Diabetes & Endocrinology, 1(2), 152–162.
Roberson, L. L., Aneni, E. C., Maziak, W., Agatston, A., Feldman, T., Rouseff, M., Tran, T., Blaha, M. J., Santos, R. D., Sposito, A., Al-Mallah, M. H., Blankstein, R., Budoff, M. J., & Nasir, K. (2014). Beyond BMI: the “Metabolically healthy obese” phenotype & its association with clinical/subclinical cardiovascular disease and all-cause mortality—a systematic review. BMC Public Health, 14, 14.
Munoz-Garach, A., Cornejo-Pareja, I., & Tinahones, F. J. (2016). Does metabolically healthy obesity exist? Nutrients, 8(6), 320. doi:10.3390/nu8060320.
Yahia, N., Brown, C. A., Rapley, M., & Chung, M. (2016). Level of nutrition knowledge and its association with fat consumption among college students. BMC Public Health, 16(1), 1047. doi:10.1186/s12889-016-3728-z.
Acknowledgements
A special note of appreciation and a heartfelt thank you goes to Dr. Bill Lawrence, CEO and Director of McLaren Central Michigan Hospital and Ms. Marybeth Mey, Laboratory Manager, for their indispensable help in the laboratory analysis. Also, I would like to extend my sincere appreciation to Dr. Allan Geliebter and Professor Steven Couture for reviewing the draft. Last, but not least, I would like to thank the FRCE for providing the grant to conduct this study, Manuela Uribe for her help in data entry, and to all CMU students who participated in this study.
Author Contributions
NY carried out questionnaire design, manuscript preparation, data collection and study coordination. CB performed all the statistical analysis. ES, SC, AL, CT, and CG contributed in data collection and data entry. All authors read and approved the final manuscript.
Funding
This study was partially funded by the Faculty Research and Creative Endeavors (FRCE) grant (Type B grant), and the FRCE Premier Display grant at CMU.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Informed Consent
Students agreeing to participate in the study were asked to sign a consent form, in harmony with the Helsinki declaration, and were informed that they could withdraw from the study at any time without penalty. The CMU Institutional Review Board (IRB) approved the study protocol (IRB Project ID: 678896-11) and IRB can be reached at: cmuirb@cmich.edu. Informed consent was obtained from all individual participants included in the study.
Availability of Data and Materials
Data are available at cbrown03@bu.edu.
Rights and permissions
About this article
Cite this article
Yahia, N., Brown, C.A., Snyder, E. et al. Prevalence of Metabolic Syndrome and Its Individual Components Among Midwestern University Students. J Community Health 42, 674–687 (2017). https://doi.org/10.1007/s10900-016-0304-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10900-016-0304-5