Abstract
Plant volatiles play a key role in host plant location of phytophagous insects. Cydia molesta is an important pest of pear fruit late in the growing season. We identified and quantified volatiles from immature and mature fruits of six pear varieties by using gas chromatography–mass spectrometry (GC-MS). Attractiveness of synthetic blends to adults based on gas chromatography-electroantennogram detection (GC-EAD) activity was investigated in both field and laboratory. Consistent electroantennographic activity was obtained for 12 compounds from headspace collections of the mature fruits of the six pear varieties. Qualitative and quantitative differences were found among six odor profiles. Among the six mixtures, the mixture of 1-hexanol, nonanal, ethyl butanoate, butyl acetate, ethyl hexanoate, hexyl acetate, hexyl butanoate, and farnesene (different isomers) with a 1:1:100:70:7:5:1:4 ratio from the variety Jimi and the mixture of nonanal, ethyl butanoate, 3-methylbutyl acetate, ethyl hexanoate, hexyl acetate, and farnesene with a 1:100:1:32:1:2 ratio from the variety Huangjin were highly attractive to both sexes in the field. However, male captures were much higher than those of females. Further wind tunnel tests proved that both sexes exhibited upwind flight to the lures, but only males landed on the source. Our finding indicates that mixtures mimicking Jimi and Huangjin volatiles attract both females and males of C. molesta, and these host volatiles may be involved in mate finding behavior.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Plant-derived volatiles play a significant role in guiding female herbivores insects to host plants for oviposition (Angioy et al., 2003; Mitchell et al., 2008). Evidence has been provided that plant volatiles elicit specific responses in females of several species of insects both in the laboratory (Tasin et al., 2005, 2007, 2009; Casado et al., 2008) and in the field (Cha et al., 2008; Anfora et al., 2009; Bruce et al., 2011; Zhang et al., 2011).
The oriental fruit moth (OFM), Cydia (= Grapholitha) molesta (Busck) (Lepidoptera: Tortricidae), presumably originating from north-west China, has become widely distributed throughout the stone fruit-growing area in the world (Roehrich, 1961). All of its host plants belong to the family Rosaceae, including mainly peach, pear, apple, but also nectarine, plum, cherry, and others. In North China, the moth first infests the growing peach and apple shoots in the early growing season and then shifts to pear and apple fruits. In the late season, pear trees are the primary OFM host plants. Females mainly lay eggs on mature pear fruits (Rothschild and Vickers, 1991; Dorn et al., 2001; Myers et al., 2006; Yang and Liu, 2010). Because of the economic importance of the OFM, its sex pheromone has been studied since the 1960s (Roelofs et al., 1969), and pheromone-based techniques have been established to monitor and control its populations (Trimble et al., 2004; Stelinski et al., 2005; Kovanci et al., 2009). However, an important drawback in practice is that use of sex pheromone affects only males, while females are directly responsible for infestations by laying eggs on fruits and making inter-orchard flights (Yetter and Steiner, 1932; Steiner and Yetter, 1933). The flight performance of females significantly exceeds that of males (Hughes and Dorn, 2002), and thereby females represent a serious threat to adjacent orchards.
For the OFM, olfactory cues from either shoots or fruits of peach and apple have been studied extensively. Excised peach shoots emitting (Z)-3-hexenyl acetate, (Z)-3-hexenol, and benzaldehyde at 4:1:1 mixture proportions were attractive to mated OFM females (Natale et al., 2003). Behavioral experiments were performed to identify the key components among all the volatiles emitted from peach shoots (Piñero and Dorn, 2007). Volatiles emitted from peach and apple fruits were also attractive to mated females in a dual choice arena (Natale et al., 2004), and the level of attraction of OFM females to peach shoots and fruits varied at three phenological stages (Piñero and Dorn, 2009). However, all of these attraction studies were conducted in the laboratory. The only field work was reported by Il’ichev et al. (2009), who carried out headspace analysis of volatiles from intact young shoot tips of peach. They found that a mixture of (Z)-3-hexenyl acetate, (E)-β-ocimene and (E)-β-farnesene at mixture proportions 1:2:2 was the best attractant, but only OFM males and no females were captured in the field.
Up to now, there are no reports on olfactory orientation of OFM to pear fruit. Pears are substantial post-peach resources during the late fruit-growing season. In China and some other Asian countries, pears are as important as peaches, and inter-planting and mixed cultivation of pears and peaches is common (Makaji, 1987; Zhao et al., 1989; Yang et al., 2002). Such a situation poses a serious threat to pear cultivation in the vicinity of peach trees (Zhao et al., 1989; Yang and Liu, 2010). In the peach-pear system, the role of volatile organic compounds (VOCs) of pear fruits still is little understood, but such knowledge should be helpful to elucidate mechanisms underlying the host shift of OFMs from peach to pear.
The objectives of the present study were: (1) to identify potentially attractive VOCs from six pear varieties; and (2) to test behavioral responses of OFM adults to synthetic blends of these VOCs in a laboratory wind tunnel and in the field.
Methods and Materials
Insects
The OFMs used were obtained from an experimental orchard at the Institute of Forestry and Pomology (IFP), Beijing Academy of Agriculture and Forestry (BAAF), Beijing, China (39°58′N, 116°13′E). Larvae were collected from field-infested shoots of peach, Prunus persica L. Batsch cv. Dajiubao, in late June, and were maintained in a climatic chamber at 24 ± 1 °C and 65–70 % RH under a photo:scoto regime of 16 L: 8 D, with the photophase starting at 05:00 hr am. Larvae were mass-reared on apple, Malus domestica L. Borkh. cv. Hongfushi, in a glass container (27 cm diameter, 13 cm high) for 3 instars and then transferred individually to smaller glass containers (2.5 cm diam., 8 cm high) until eclosion. Adults were maintained in a bell-shaped glass container (diam. of the two openings 6 and 15 cm, 41 cm high). Both sides of the container were covered with fine nylon mesh. From a hole pierced through the mesh in the small side, 15 % honey solution was provided on water-soaked cotton. The moths were reared in the laboratory for 3 generations before testing. For electrophysiological experiments (GC-EAD), 2- to 3-d-old females were used. For the wind-tunnel bioassay, 2- to 4-d-old mated female and male moths were chosen without a conscious bias. Mated females were obtained by placing groups of ca. 20 newly emerged females together with 30 males in the same cage for two scotophases to ensure mating. Adult moths were used only once and were not exposed to synthetic odor sources before the bioassay.
Chemicals
Decane (99 %), tetradecane (99 %), pentadecane (99 %), hexadecane (99 %), nonadecane (99 %), 2-methylbutyl acetate (99 %), 3-methylbutyl acetate (97 %), ethyl pentanoate (98 %), butyl butanoate (98 %), 6-methyl-5-hepten-2-one (98 %), butyl hexanoate (98 %), (E)-β-ocimene (60 %), and (E,E)-α-farnesene (49 %) were obtained from Sigma-Aldrich Co. (St. Louis, MO, USA). The farnesene obtained from Sigma-Aldrich also contained in addition to (E,E)-alpha-farnesene (49 %) (E)-beta-farnesene (26 %), (Z)- beta-farnesene (18 %), and (Z,E)-alpha-farnesene (7 %). We used this mixture of farnesene isomers for our EAG and field studies and refer to it as “farnesene”. Heptadecane (99 %), hexanol (99 %), 2-ethyl-1-hexanol (99 %), octanal (98 %), nonanal (97 %), decanal (97 %), pentyl acetate (99 %), hexyl acetate (99 %), and racemic linalool (97 %) were obtained from Fluka Production GmbH (Buchs, Switzerland). Ethyl butanoate (99 %) and butyl acetate (99 %) were bought from Acros Organics (New Jersey, USA). Ethyl hexanoate (98 %) and hexyl butanoate (98 %) were purchased from Tokyo Chemical Industry Co. (Tokyo, Japan). Compounds that did not elicit antennal responses, and for which no standards were available, were tentatively identified using the NIST-database.
Plant Materials
Pears were cultivated in an experimental orchard at the IFP. Six varieties of 3 pear species were selected: Xinyali (XY), Xuehua (XH), and Jimi (JM) of Pyrus bretschneideri, Huangjin (HJ) and Fengshui (FS) from Pyrus pyrifolia, and Conference (KF) from Pyrus communis. All these varieties are commonly grown in the Beijing area. No insecticides or any specific treatment against OFM were used in the orchard during tests. Unripe and ripe fruits were healthy and were picked from trees according to the phenological development of corresponding varieties in the Beijing area in 2009 and 2010 (Cao et al., 2000, 2006; Jiang et al., 2002). Fruits were picked within 20 min before sampling of VOCs or wind-tunnel assays.
Collection of VOCs
We used a push-pull system to collect headspace VOCs. Fruits (ca. 1,500 g) were placed immediately after harvesting into a 2,000 ml glass jar for extraction. Air aspirated with a vacuum pump (Qianxi Air Company, Beijing, China) was filtered through an activated-charcoal filter, then passed through the jar at 300 ml/min, and finally through a sorbent cartridge (Porapak Q, 50 mg, 80/100 mesh, Supelco, Bellefonte, PA, USA). The sorbents were held between plugs of glass wool in a glass tube (10 cm long, 0.5 cm inner diam). Samples were collected for 8 hr at 24 ± 1 °C and 65–70 % RH. Volatiles were desorbed by eluting the sorbent cartridge with 500 μl hexane (HPLC grade, Sigma-Aldrich) at room temperature. Five samples were collected for each unripe and ripe pear variety, respectively. Samples were analyzed by GC-EAD and GC-MS. For quantitative analyses, 0.5 μg benzaldehyde (99 %, Fluka, Switzerland) was added as an internal standard to each sample. Preliminary analyses showed that this compound was not detectable in the headspace of the pear varieties studied here. Sample volumes were reduced to 50 μl by using a slow stream of nitrogen and then analyzed. If not used immediately, extracts were sealed in glass vials and stored at −18 °C until used.
GC-MS
Headspace VOCs of the unripe and ripe fruits of the 6 varieties and mixtures of synthetic compounds were analyzed with an Agilent Technologies 5973 MS (Agilent) coupled to an Agilent Technologies 6890 N GC (Agilent) equipped with polar DB-WAX fused-silica column (30 m × 0.25 mm ID, 0.25 μm film, J&W Scientific Inc., Folsom, CA, USA) or nonpolar DB-5 fused-silica column (30 m × 0.25 mm ID, 0.25 μm film, J&W Scientific Inc., Folsom, CA, USA). The temperature program was as follows: 50 °C for 1 min, then 3 °C/min to 120 °C, then 10 °C/min to 240 °C and finally held at 240 °C for 10 min. Windows NT/MASS Spectral Search Program (Version 1.7) software was used for data analysis. Injections were made in the splitless mode. Helium was used as carrier gas (1.0 ml/min). For electron impact (EI) mass spectra, the ionization voltage was 70 eV, and the temperatures of the ion source and of the interface were 230 °C and 280 °C, respectively. The emission current was 34.6 μA. Identification of VOCs was verified by comparison with authentic samples.
GC-EAD
VOCs were analyzed with an Agilent Technologies 6890 N GC with a flame ionization detector, using a polar DB-WAX (30 m × 0.25 mm ID, 0.25 μm film, J&W Scientific, Folsom, CA, USA) or nonpolar DB-5 (30 m × 0.25 mm ID, 0.25 μm film, J&W Scientific Inc., Folsom, CA, USA) capillary column coupled with an electroantennographic detector (EAD; Syntech, Hilversum, The Netherlands). The oven temperature program was the same as in the GC-MS analysis. Nitrogen was used as carrier gas (1.0 ml/min). A micromanipulator assembly (MP-15, Syntech) was connected to a stimulus controller (CS-55, Syntech). All signal sources were connected to a serial data acquisition interface (IDAC-4, Syntech). Antennae from the heads of OFM females were excised using micro-scissors. A few segments from the tips of antennae were clipped off and mounted on the antenna holder with two metal electrodes using conductive gel (Spectra 360, Parker Lab, NJ, USA), and then the electrode holder was inserted into the EAD probe. Testing began after a relative stable baseline had been achieved. The outlet of the GC column was split in a 2:1 ratio between the electroantennographic detector, a cut antenna of an OFM female, and the flame ionization detector (FID). The mounted antenna was positioned in the charcoal-filtered and humidified air stream that carried the VOCs eluting from the GC column. The antennal and FID signals were amplified and recorded simultaneously using Syntech software (GC-EAD 32, version 4.4, Syntech, Hilversum, The Netherlands). Each sample was tested 6 times. Identities of EAD-active compounds were verified by comparison of mass spectra and retention times with those of synthetic standards. Each tested antenna was derived from a different female. In addition, blends of 12 EAD-active synthetic compounds (1-hexanol, nonanal, ethyl butanoate, butyl acetate, 2-methylbutyl acetate, 3-methylbutyl acetate, pentyl acetate, ethyl hexanoate, hexyl acetate, butyl hexanoate, hexyl butanoate, and farnesene as a mixture of different isomers, see above) were analyzed by GC-EAD (10 ng of each).
Field Experiment 1
On the basis of the results of the GC-EAD analyses, VOCs from the mature fruits of 6 varieties eliciting antennal responses in female OFM were formulated in blends for the field tests. Six blends of synthetic compounds were prepared in the ratios of GC-EAD-active VOCs as emitted by the corresponding 6 varieties (Table 1). The formulations contained 100 mg of the most abundant compound and the others in the same proportion as in the natural volatile mixture. Preliminary field experiments showed that such concentrations can provide an adequate emission under field conditions. The rubber septa are goblet shaped (10 mm depth, 6 mm inter diam.) and have a maximum volume of 400 μl. They were made to order by Shunyi Rubber Company, Beijing, China. This volume is enough to load all the treatment solutions in the study. The mixtures of compounds were prepared 1–2 hr prior to bioassays in the field trial. They were added into the rubber septa and then fixed upward on the bottom of sticky delta trap in the dusk, during which OFM-flight intensity peaks, for all the treatments. We deployed the rubber septa with solutions directly in the trap, before the mixture solution was fully impregnated into the rubber septa. All odor blends were deployed in this way. Unbaited traps (HPLC-grade hexane, Sigma-Aldrich) and OFM sex-pheromone lures (Geruibiyuan Technology Company, Beijing, China) were used as controls.
The field test was conducted in a 7-hectare peach orchard and a 6.5-hectare pear orchard at the IFP with a history of OFM infestation. The peach orchard was chosen with the aim of avoiding full overlap between the background odor in the pear orchard and synthetic blends derived from pear fruits. A sticky delta trap (35 cm long × 20 cm high × 20 cm wide) was used to trap the insects. Trials were carried out in a randomized complete-block design. In each block, baited traps were installed approximately 1.5 m above the ground, and were set up at a distance of at least 30 m to minimize interference between traps. Each treatment was replicated four times in four blocks.
Traps were monitored twice weekly. Captured OFMs were transferred to the laboratory, and their number and sex were recorded. Trials lasted 2 week. The data of captures over a 14-d-period were pooled. The field test was conducted from late June to early August 2010 in peach and pear orchards, during the 2nd and 3rd OFM flights in the Beijing area.
Field Experiment 2
In field experiment 1, an eight-component mixture of 1-hexanol, nonanal, ethyl butanoate, butyl acetate, ethyl hexanoate, hexyl acetate, hexyl butanoate, and farnesene (different isomers, see above) at 1:1:100:70:7:5:1:4 proportions in JM and a six-component mixture of nonanal, ethyl butanoate, 3-methylbutyl acetate, ethyl hexanoate, hexyl acetate, and farnesene at 1:100:2:32:1:2 proportions in HJ were more attractive to the moths than the other blends. For field experiment 2, the common VOCs and the specific VOCs in two mimics were grouped separately based on the ratio in HJ and JM, and then six blends—JM, JMS, JMC, HJ, HJS, and HJC—were compared for their attractiveness to the moths in the peach orchard (Table 2).
Field Experiment 3
Six mixtures of synthetic VOCs in the proportions of GC-EAD-active VOCs emitted by the corresponding six varieties were further evaluated in a peach orchard in 2011 (Table 1). The field test was conducted from late June to early July, during the second OFM flight period, in a 15-hectare peach orchard located in the fruit-production area of Tianbaoyuan, Huairou, Beijing. All other details of the experiment were the same as for field experiment 1.
Wind Tunnel Assay
The laboratory wind tunnel measured 1.6 m long, 0.5 m wide, 0.5 m high. A fan at the upwind end generated a steady airflow into the tunnel, set at 0.3 m/s at the point of release of moths. The light intensity in the tunnel was about 250 lux. The room was kept at 23 ± 2 °C and 50–70 % RH.
Moths were transferred into the test room 2 hr before experiments. Batches of 10 moths were placed in a small screen metal release cage (7 cm diam., 9 cm height) with a side door. The air could flow through the release cages. Tests began 2 hr before the beginning of the scotophase and lasted 3 hr. The release cage containing a moth was placed on a holder at the downwind end of the tunnel, 30 cm above its floor and ca. 140 cm from the VOC source. The side door faced the upwind end of the tunnel, and the door was opened to allow the moths to leave the cage. Each batch of 10 mated moths was tested for 20 min, and 6 batches of moths were used per day. Each VOC mixture was tested with 9 batches of moths on different days. Moths were tested only once. Synthetic compounds were loaded in rubber septa. The septum loaded with one of the mixtures was placed on a holder at the upwind end in the center of the tunnel, 10 cm from the upwind end. Individual moths were scored for the following behaviors: (1) departure from the release cage and flight upwind; (2) arrival within 10 cm of the VOC source; and (3) landing on the source. For data analysis, we categorized each moth based on the most complete behavior that it displayed within 20 min.
For these experiments, mature pear fruits (ca. 220 g, 8–10 cm diam.) were freshly cut and immediately transferred into the laboratory for bioassays. VOCs collected from the fruit were diluted with hexane (HPLC grade, Sigma-Aldrich). Mixtures were prepared in proportions of GC-EAD-active VOCs found in the natural blends emitted by the corresponding varieties (Table 3). The formulation of the lure was similar to that in the field test. The predominant VOC in the mixture was dosed at 0.5 mg. Preliminary tests in the wind tunnel showed that these concentrations were adequate to elicit moth responses.
Data Analysis
Mean numbers of OFM males and females captured in traps baited with different lures in the field and attractiveness of mated OFM females and males to the VOC source in the wind tunnel were analyzed by one-way ANOVA. The means were separated by Tukey’s multiple range tests (P < 0.05). Significant differences in three flight behaviors between both sexes were analyzed by Mann–Whitney U-tests. All data were analyzed with the statistical program SPSS (version 13.0).
Results
Chemical Identification of VOCs from Different Pear Varieties
In order to understand which volatiles attract OFMs to pears, we analyzed the VOCs of unripe and ripe fruits of six different varieties. Several compounds were identified, belonging to different chemical classes: hydrocarbons, alcohols, aldehydes, esters, benzene derivatives, ketones, and terpenoids (Table 3). Profiles of the six varieties consisted of the same chemical classes, but differed greatly between ripe and unripe fruits. In particular, decanal, octanal, methyl salicylate, (E)-2-hexenyl benzoate, ocimene, and linalool were characteristic of unripe pears and absent from ripe fruits, whereas the concentrations of other VOCs, such as several hydrocarbons, 2-ethyl-1-hexanol, nonanal, and (E, E)-α-farnesene, decreased with maturation. In mature fruits, esters were the dominant VOCs, mainly ethyl butanoate, butyl acetate, ethyl hexanoate, and hexyl acetate. In particular, butyl acetate was the most abundant VOC identified in KF and XH varieties, and ethyl butanoate in the others (Table 3).
Antennal Responses to VOCs from Ripe Pears
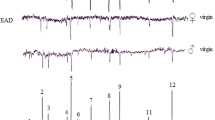
By using GC-EAD, we identified the compounds detected by the antenna of the moth (Fig. 1). Their chemical identities were then verified by MS, and comparison of mass spectra and retention times were made with those of authentic compounds.
Simultaneously recorded GC-EAD responses to VOCs collected from mature fruits of the varieties Jimi (JM) of Pyrus bretschneideri (a), Huangjin (HJ) of Pyrus pyrifolia (b) and Conference (KF) of Pyrus communis (c) using a polar DB-WAX capillary column. The upper trace is the flame ionization detector response (FID), and the lower displays the Cydia molesta female antennal response (EAD). Consistently EAD-active compounds in six different tests were showed as follows: For JM (a): (1) ethyl butanoate; (2) butyl acetate; (3) ethyl hexanoate; (4) hexyl acetate; (5) 1-hexanol; (6) nonanal; (7) hexyl butanoate; (8) (E, E)-α-farnesene. For HJ (b): (1) ethyl butanoate; (2) 3-methylbutyl acetate; (3) ethyl hexanoate; (4) hexyl acetate; (5) nonanal; (6) (E, E)-α-farnesene. For KF (c): (1) butyl acetate; (2) 2-methylbutyl acetate; (3) pentyl acetate; (4) unidentified; (5) ethyl hexanoate; (6) hexyl acetate; (7) unidentified; (8) 1-hexanol; (9) unidentified; (10) (E, E)-α-farnesene
In total, 12 compounds from the headspace of ripe fruits belonging to six varieties elicited antennal responses from OFM females: 1-hexanol, nonanal, ethyl butanoate, butyl acetate, 2-methylbutyl acetate, 3-methylbutyl acetate, pentyl acetate, ethyl hexanoate, hexyl acetate, butyl hexanoate, hexyl butanoate, and (E, E)-α-farnesene (Fig. 1). The EAD-active VOCs were different in quality and quantity among the six varieties. However, ethyl hexanoate, hexyl acetate, and (E, E)-α-farnesene were common components of all 6 varieties, while 1-hexanol, nonanal, ethyl butanoate, and butyl acetate were present in five of them (Table 3). Different esters frequently were present among GC-EAD-active volatiles in the mature fruits of all varieties.
In order to quantify antennal sensitivity, a second series of GC-EAD analyses was performed using 10 ng of each VOC. The strongest antennal response in OFM mated females was evoked by farnesene (Table 4).
Field Experiment 1
Based on the results of the GC-EAD analysis, six mixtures, each containing the most GC-EAD-active VOCs from the mature fruits of each variety, were evaluated both in a peach and a pear orchard in the early season (Fig. 2). A larger OFM population apparently was present in the peach orchard than in the pear orchard, based on the number of male moths trapped with the sex pheromone. Traps baited with the blends mimicking the odor of variety JM (1-hexanol, nonanal, ethyl butanoate, butyl acetate, ethyl hexanoate, hexyl acetate, hexyl butanoate, and farnesene in a 1:1:100:70:7:5:1:4 ratio) and variety HJ (nonanal, ethyl butanoate, 3-methylbutyl acetate, ethyl hexanoate, hexyl acetate, farnesene with a 1:100:1:32:1:2 ratio) caught significantly more OFM males than the control or the traps baited with the other four blends. Only a few females were trapped with no significant difference between traps, including the control (Fig. 2a). In the pear orchard, where in the early season a much smaller OFM population was present based on the monitoring by sex pheromone-baited traps (9.25 ± 2.22 males/trap), no moths were caught in the traps baited with pear volatiles.
Mean total number ± SD of Cydia molesta males and females captured in each trap (N = 4) in a peach orchard on June 26-July 9, 2010 (a) and males captured in each trap (N = 4) in peach and pear orchard on July 21-August 3, 2010 (b). Each lure baited with rubber septa with synthetic VOC mixtures from different varieties, a hexane control (CK) and sex pheromone (SP). The six varieties of 3 pear species were Xinyali (XY), Xuehua (XH), and Jimi (JM) of Pyrus bretschneideri, Huangjin (HJ) and Fengshui (FS) from Pyrus pyrifolia, and Conference (KF) from Pyrus communis. Formulation of each synthetic lure is indicated below the graph. Each lure contained 100 mg of the most abundant component. Farnesene = mixture of (E,E)-alpha-farnesene (49 %), (E)-beta-farnesene (26 %), (Z)- beta-farnesene (18 %), and (Z,E)-alpha-farnesene (7 %). The experiment was conducted at an experimental orchard at the Institute of Forestry and Pomology (IFP), Beijing Academy of Agricultural and Forestry (BAAF), Beijing, China. Different letters on bars indicate significant differences (one-way ANOVA followed by Tukey’s multiple comparison test, P < 0.05)
Another field test also was performed in late July in both peach and pear orchards. Similar numbers of OFMs were found in both orchards, as monitored by sex pheromone (172.50 ± 4.04 and 174.25 ± 4.92 males/trap in peach and pear orchards, respectively). As in the early season test, traps baited with blends of the JM and HJ varieties caught significantly more OFM males than the other traps (Fig. 2b).
Field Experiment 2
In order to screen the VOCs essential for moth attraction in the synthetic odor blends that mimicked JM and HJ, the components of the JM and HJ blends were further evaluated in the field for their importance in attraction. We set up the following treatments: (1) JM mimic blend; (2) HJ mimic blend; (3) JMC: common components released by both HJ and JM, using the component ratio typical for JM; (4) HJC: common components released by both HJ and JM, using the component ratio typical for HJ; (5) JMS: VOCs selectively released from JM; and (6) HJS: VOCs selectively released from HJ (Fig. 3). All the other details of the experiment were same as for field experiment 1. The six blends were compared for attractiveness to moths in the peach orchard. The complete JM mixture was the most effective, followed by the complete HJ mixture, and the complete mixtures were significantly more effective than the reduced mixtures (Fig. 3).
Mean total number ± SD of oriental fruit moth (OFM) males and females captured in each trap (N = 4), each baited with rubber septa with the synthetic VOC mixtures based on headspace VOC composition from mature fruits of the varieties Jimi (JM) of Pyrus bretschneideri and Huangjin (HJ) of Pyrus pyrifolia, their subsets and a hexane control (CK) in a peach orchard. Formulation of synthetic lures is indicated below the graph. Each lure contained 100 mg of the most abundant component. Farnesene = mixture of (E,E)-alpha-farnesene (49 %), (E)-beta-farnesene (26 %), (Z)- beta-farnesene (18 %), and (Z,E)-alpha-farnesene (7 %). The experiment was conducted in Beijing, on July 10-July 23, 2010. Different letters (capital letters for males captured and small letters for females) on bars indicate significant differences (one-way ANOVA followed by Tukey’s multiple comparison test, P < 0.05)
Field Experiment 3
The field trials to evaluate further the response of OFMs to six blends that mimicked the six varieties were replicated in 2011. A larger OFM population was present in the peach orchard during the early season in 2011 than in 2010, based on the high number of male moths trapped with the sex pheromone (Figs. 2 and 4). As in the first field test in 2010, traps baited with mixtures of the JM and HJ varieties caught significantly more OFMs than the control, and the traps baited with the other four mixtures, but the total captures were significantly greater in 2011 than 2010 (Fig. 2 and 4). In particular, female captures significantly increased in 2011. The JM blend was the most effective attractant for females.
Mean total number ± SD of Cydia molesta males and females captured in each trap (N = 4) in a peach orchard on June 28–July 11, 2011. Each trap was baited with a rubber septum with synthetic VOC mixtures corresponding to those emitted by different varieties, a hexane control (CK), and sex pheromone (SP). The six varieties of 3 pear species were Xinyali (XY), Xuehua (XH), and Jimi (JM) of Pyrus bretschneideri, Huangjin (HJ) and Fengshui (FS) from Pyrus pyrifolia, and Conference (KF) from Pyrus communis. Formulation of synthetic lure is indicated below the graph. Each lure contained 100 mg of the most abundant compounds. Farnesene = mixture of (E,E)-alpha-farnesene (49 %), (E)-beta-farnesene (26 %), (Z)- beta-farnesene (18 %), and (Z,E)-alpha-farnesene (7 %). The experiment was conducted in a peach orchard located in the fruit production area of Tianbaoyuan, Huirou, Beijing. Different letters on bars indicate significant differences (one-way ANOVA followed by Tukey’s multiple comparison test, P < 0.05)
Wind Tunnel Bioassays
In field experiments, we found that VOC mixtures attracted significantly more male than female OFMs. In order to evaluate the sexual differences in the flight performance toward synthetic mixtures in the laboratory, we used the eight-component JM mixture and the six-component HJ mixture as lures in wind-tunnel tests. Mature JM fruits also were applied as lures. We then investigated in more detail the differences between males and females in responding to JM and HJ mixtures, as well as to mature fruits of the JM variety. All three samples stimulated upwind oriented flight in both females and males (Fig. 5a, b), but only males reached and contacted the source (Fig. 5b). Males were more strongly attracted than females to synthetic mixtures.
Attraction of mated Cydia molesta females (a) and males (b) in a wind tunnel to mature pear synthetic VOC mixtures mimicking headspace composition of mature fruits of the varieties Jimi (JM) of Pyrus bretschneideri and Huangjin (HJ) of Pyrus pyrifolia. JM mimic = 1-hexanol, nonanal, ethyl butanoate, butyl acetate, ethyl hexanoate, hexyl acetate, hexyl butanoate, farnesene; HJ mimic = nonanal, ethyl butanoate, 3-methylbutyl acetate, ethyl hexanoate, hexyl acetate, farnesene with ratios shown in Table 3. Farnesene = mixture of (E,E)-alpha-farnesene (49 %), (E)-beta-farnesene (26 %), (Z)- beta-farnesene (18 %), and (Z,E)-alpha-farnesene (7 %). Within columns with same color, means followed by different letters are significantly different (one-way ANOVA followed by Tukey’s multiple comparison test, P < 0.05). Moths were scored for upwind orientation (black bar), arrival at the source within 10 cm (white bar), and contact with the lures (grey bar). Significant differences in three flight behaviors between both sexes were analyzed by the Mann–Whitney U-test (P < 0.05; ns, no significant difference)
Mature pear fruits of variety JM proved to be the most attractive to both sexes in the wind tunnel. In such experiments, 67 % of the females flew upwind and 50 % arrived within 10 cm of the source, but none landed on the fruit (Fig. 5a). By contrast, 96 % of males showed upwind orientation, 91 % arrived within 10 cm of the source, and 17 % landed on the source (Fig. 5b). In summary, the behaviors of female and male OFMs to JM pears were very similar to those observed with the corresponding artificial VOC blends (Fig. 5a, b).
Discussion
We identified 12 EAD-active VOCs from 6 pear varieties. The VOC mixtures from mature JM fruit and mature HJ fruit were significantly more attractive in the field to OFM females and males than the blends from other varieties. The results indicate that olfactory cues from pears play a role in orientation to host plants by both sexes of OFM. Most studies on plant VOC mixtures that attract OFM have been performed in the laboratory. Our studies verified the attractiveness of synthetic host-plant VOCs to female and male moths under field conditions.
Different VOC profiles influence OFM behavior at different phenological stages. At the beginning of the season, the pest mainly infests the growing shoots of peach. Natale et al. (2003) reported that a mixture of (Z)-3-hexenyl acetate, (Z)-3-hexenol, and benzaldehyde from peach shoots (4:1:1) elicited significant bioactivity in dual-choice tests with a bioassay-assisted fractionation that used different sorbent polymers. Piñero and Dorn (2007) found that a mixture of benzaldehyde, benzonitrile, (Z)-3-hexenyl acetate, (Z)-3-hexenol, and (E)-2-hexenal from peach shoots was as attractive as natural VOCs in a Y-tube olfactometer by using a subtraction approach. In field tests, Il’ichev et al. (2009) reported that a mixture of (Z)-3-hexenyl acetate, (E)-β-farnesene, and (E)-β-ocimene at 1:2:2 proportions was the most effective lure.
We found that headspace collections from pear fruits were greatly different from peach-shoot VOCs. Esters were the major components from mature pear fruits, and several of them elicited antennal responses. The increase in ester production in mature fruits corresponded with pest damage in mature fruit. The seasonal flight period for OFM starts before the onset of pear-fruit ripening, and females oviposit on the mature fruit. Our findings suggest that esters are essential for attraction of OFMs to pear fruits during the late fruit-growth stage. Some multi-voltine insects such as the OFM exploit several plant species as their hosts in different phenological stages. This is an important strategy for optimization of resources in order to complete the life cycle, and it is important for understanding the evolution of VOC-based recognition of host plants by herbivorous insects and for development of a possible semiochemical-based synthetic lure for use in the field.
By comparing the attractiveness of different VOC mixtures from pear fruits, we found both quality and quantity of components in the mixtures to be important. First, minor components are essential for optimal attraction. Ethyl butanoate and butyl acetate were the most abundant active VOCs from six varieties, but the minor components were significantly different, suggesting that they are responsible for the different attractiveness of the different varieties. The minute amounts of benzonitrile from peach shoots have been shown to be important for olfactory responses in OFMs (Piñero et al., 2008). Second, the mixture proportions of ubiquitous plant VOCs are crucial for attraction of OFMs. Many insects use specific compounds to recognize host plants (Fraenkel, 1959; Szafranek et al., 2006; Hilker and McNeil, 2008). In our studies, however, EAD-active compounds were relatively ubiquitous plant VOCs. In such a case, species-specific mixture proportions of common VOCs would be crucial for insects to locate their hosts reliably. The mixture proportions of common plant VOCs are well-known to be important for host-plant recognition (Visser and Avé, 1978; Buttery et al., 1984; Reddy and Guerrero, 2000; Bruce et al., 2005). Similarly, in our study system, JM emitted the same EAD-active VOCs as XH, and the mixture proportions of these compounds were responsible for the significantly higher attractiveness of JM in the field.
Male captures, however, were higher although the lures were based on the EAD-active compounds for females. Similar results have been reported by Il’ichev et al. (2009). They identified potentially effective attractants from young peach shoot tips, but traps with all tested mixtures in all concentrations caught only males in the field. In our wind-tunnel study, we showed that females also were attracted by true pear fruits and synthetic lures although the performance of males exceeded that of females especially at close range (Fig. 5). Similar results have been found for the codling moth, Cydia pomonella, an important pest of apple (Wildbolz, 1958; Coracini et al., 2004). Female codling moths have frequently been observed to fly upwind over several meters toward branches with green apples, but contact the apple less frequently, which suggests that the females employ a different search strategy to find a suitable oviposition site, especially at close range (Witzgall et al., 1999).
Why are more male OFMs than females captured by traps with lures that emit plant-derived VOCs? Some authors have hypothesized that males use plant VOCs to distinguish environments where they can find females more easily (Ansebo et al., 2004). A partial explanation might be related to the large number of olfactory receptor neurons that are found in male antennae (Bäckman et al., 2000). Moreover, the flight mode of males may be different from that of females. Males fly toward the plant in search of females, and their orientation behavior is adapted to locate a point source of sex pheromone. By contrast, females searching for suitable oviposition sites may not be attracted to point sources of plant VOCs, and therefore trap design could be quite important. Furthermore, females may use optical cues in addition to VOCs to orient to an oviposition site. The greatest flight activity of mated and unmated male and female OFMs occurred during the first hour of dusk, during which time light intensity decreased from 3,750 to 57 lux, suggesting that the females use optical cues during orientation to oviposition sites (Hughes and Dorn, 2002). Females may land at the oviposition site based on both chemical and visual cues. The grape berry moth, Paralobesia viteana, a crepuscular species, oviposits less in the absence of light (Clark and Dennehy, 2002). In addition, physical structure and low-volatility VOCs present on the fruit surfaces could also affect the landing of females. In our study, OFMs always laid eggs on the smooth surface of waxed paper in the laboratory. The sticky base of the traps might be a repellent for female landing. The high male capture rates in our study could be applied in further research to establish a novel approach for monitoring males, especially in orchards treated with sex pheromone for mating disruption (Il’ichev et al., 2009). A logical follow-up, therefore, would be to investigate putative synergism between host-plant VOCs and the sex pheromone.
References
Anfora, G., Tasin, M., Cristofaro, A. D., Ioriatti, C., and Lucchi, A. 2009. Synthetic grape volatiles attract mated Lobesia botrana females in laboratory and field bioassays. J. Chem. Ecol. 35:1054–1062.
Angioy, A. M., Desogus, A., Barbarossa, I. T., anderson, P., and Hansson, B. S. 2003. Extreme sensitivity in an olfactory system. Chem. Senses 28:279–284.
Ansebo, L., Coracini, M. D. A., Bengtsson, M., Liblikas, I., Ramirez, M., Borg Karlsson, A.-K., Tasin, M., and Witzgall, P. 2004. Antennal and behavioural response of codling moth Cydia pomonella to plant volatiles. J. Appl. Entomol. 128:488–493.
Bäckman, A. C., anderson, P., Bengtsson, M., Löfqvist, J., Unelius, C. R., and Witzgall, P. 2000. Antennal response of codling moth males, Cydia pomonella L. (Lepidoptera: Tortricidae), to the geometric isomers of codlemone and codlemone acetate. J. Comp. Physiol. A 186:513–519.
Bruce, T. J. A., Wadhams, L. J., and Woodcock, C. M. 2005. Insect host location: a volatile situation. Trends Plant Sci. 10:269–274.
Bruce, T. J. A., Martin, J. L., Smart, L. E., and Pickett, J. A. 2011. Development of semiochemical attractants for monitoring bean seed beetle, Bruchus rufimanus. Pest Manag. Sci. 67:1303–1308.
Buttery, R. G., Kamm, J. A., and Ling, L. C. 1984. Volatile components of red clover leaves, flowers, and seed pods: possible insect attractants. J. Agric. Food Chem. 32:254–256.
Cao, Y. F., Li, S. L., Huang, L. S., Sun, B. J., and Tan, X. W. 2000. Comprehensive evaluation of pear variety resource in China. China Fruit 4:42–44.
Cao, Y. F., Liu, F. Z., Hu, H. J., and Zhang, B. B. 2006. Descriptors and Data Standard for Pear (Pyrus spp.). China Agric Press, Beijing.
Casado, D., Gemeno, C., Avilla, J., and Riba, M. 2008. Diurnal variation of walnut tree volatiles and electrophysiological responses in Cydia pomonella (Lepidoptera: Tortricidae). Pest Manag. Sci. 64:736–747.
Cha, D. H., Nojima, S., Hesler, S. P., Zhang, A. J., Linn Jr., C. E., Roelofs, W. L., and LOEB, G. M. 2008. Identification and field evaluation of grape shoot volatiles attractive to female grape berry moth (Paralobesia viteana). J. Chem. Ecol. 34:1180–1189.
Clark, L. G. and Dennehy, T. J. 2002. Oviposition behaviour of grape berry moth. Entomol. Exp. Appl. 47:223–230.
Coracini, M., Bengtsson, M., Liblikas, L., and Witzgall, P. 2004. Attraction of coding moth males to apple volatiles. Entomol. Exp. Appl. 110:1–10.
Dorn, S., Hughes, J., Molinari, F., and Cravedi, P. 2001. Cydia molesta and Cydia pomonella: comparison of adult behaviour. IOBC wprs Bull. 24:133–137.
Fraenkel, G. S. 1959. The raison d’être of secondary plant substances. Science 129:1466–1470.
Hilker, M. and McNEIL, J. 2008. Chemical and behavioral ecology in insect parasitoids: how to behave optimally in a complex odourous environment, pp. 92–112, in E. Wajnberg, C. Bernstein, and J. van Alphen (eds.), Behavioral Ecology of Insect Parasitoids. Blackwell, Malden.
Hughes, J. and Dorn, S. 2002. Sexual differences in the flight performance of the oriental fruit moth, Cydia molesta. Entomol. Exp. Appl. 103:171–182.
Il’ichev, A. L., Kugimiya, S., Williams, D. G., and Takabayashi, J. 2009. Volatile compounds from young peach shoots attract males of oriental fruit moth in the field. J. Plant Interact. 4:289–294.
Jiang, W. B., Gao, G. L., Yu, K. J., and Ma, K. 2002. Creation of pear variety resource in China over the past decade and its prospects. J. Fruit Sci. 19:314–320.
Kovanci, O. B., Gencer, N. S., and Larsen, T. E. 2009. The deposition and retention of a microencapsulated oriental fruit moth pheromone applied as an ultra-low volume spray in the canopy of three peach cultivars. Bull. Insectol. 62:69–74.
Makaji, T. 1987. Diagnosis of Fruit Tree Diseases and Pests with Color Plates. Agriculture Publication Incorporation, Tokyo.
Mitchell, V. J., Manning, L. A., Cole, L., Suckling, D. M., and El-Sayed, A. M. 2008. Efficacy of the pear ester as a monitoring tool for codling moth Cydia pomonella (Lepidoptera: Tortricidae) in New Zealand apple orchards. Pest Manag. Sci. 64:209–214.
Myers, C. T., Hull, L. A., and Krawczy, K. G. 2006. Effects of orchard host plants on preference of the oriental fruit moth (Lepidoptera: Tortricidae). J. Econ. Entomol. 99:1176–1183.
Natale, D., Mattiacci, L., Hern, A., Pasqualini, E., and Dorn, S. 2003. Response of female Cydia molesta (Lepidoptera: Tortricidae) to plant derived volatiles. Bull. Entomol. Res. 93:335–342.
Natale, D., Mattiacci, L., Pasqualini, E., and Dorn, S. 2004. Apple and peach fruit volatiles and the apple constituent butyl hexanoate attract female oriental fruit moth, Cydia molesta, in the laboratory. J. Appl. Entomol. 128:335–342.
Piñero, J. C. and Dorn, S. 2007. Synergism between aromatic compounds and green leaf volatiles derived from the host plant underlies female attraction in the oriental fruit moth. Entomol. Exp. Appl. 125:185–194.
Piñero, J. C. and Dorn, S. 2009. Response of female oriental fruit moth to volatiles from apple and peach trees at three phenological stages. Entomol. Exp. Appl. 131:67–74.
Piñero, J. C., Galizia, C. G., and Dorn, S. 2008. Synergistic behavioral responses of female oriental fruit moths (Lepidoptera: Tortricidae) to synthetic host plant-derived mixtures are mirrored by odor-evoked calcium activity in their antennal lobes. J. Insect Physiol. 54:333–343.
Reddy, G. V. and Guerrero, A. 2000. Behavioral responses of the diamondback moth, Plutella xylostella, to green leaf volatiles of Brassica oleracea subsp. capitata. J. Agric. Food Chem. 48:6025–6029.
Roehrich, R. 1961. Contribution a l’étude écologique des populations de la tordeuse de pêcher (Grapholita molesta Busck) dans la région Aquitaine. Annales de l’Institut National de la Recherche Agronomique, Paris.
Roelofs, W. L., Comeau, A., and Selle, R. 1969. Sex pheromone of the oriental fruit moth. Nature 224:723.
Rothschild, G. H. L. and Vickers, R. A. 1991. Biology, ecology and control of the oriental fruit moth, pp. 389–412, in L. P. S. Van der Geest and H. H. Evenhuis (eds.), Tortricid Pests: Their Biology. Natural Enemies and Control. Elsevier, Amsterdam.
Steiner, L. F. and Yetter, W. P. 1933. Second report on the efficiency of bait traps for the oriental fruit moth as indicated by the release and capture of marked moths. J. Econ. Entomol. 26:774–788.
Stelinski, L. L., Gut, L. J., Mallinger, R. E., Epstein, D., Reed, T. P., and Miller, J. R. 2005. Small plot trials documenting effective mating disruption of oriental fruit moth by using high densities of wax-drop pheromone dispensers. J. Econ. Entomol. 98:1267–1274.
Szafranek, B., Chrapkowska, K., Waligora, D., Palavinskas, R., Banach, A., and Szafranek, J. 2006. Leaf surface sesquiterpene alcohols of the potato (Solanum tuberosum) and their influence on Colorado beetle (Leptinotarsa decemlineata Say) feeding. J. Agric. Food Chem. 54:7729–7734.
Tasin, M., Anfora, G., Ioriatti, C., Carlin, S., and de Cristofaro, A. 2005. Antennal and behavioral responses of grapevine moth Lobesia botrana females to volatiles from grapevine. J. Chem. Ecol. 31:77–87.
Tasin, M., Bäckman, A. C., Coracini, M., Casado, D., Ioriatti, C., and Witzgall, P. 2007. Synergism and redundancy in a plant volatile blend attracting grapevine moth females. Phytochemistry 68:203–209.
Tasin, M., Bäckman, A. C., Anfora, G., Carlin, S., Ioriatti, C., and Witzgall, P. 2009. Attraction of female grapevine moth to common and specific olfactory cues from 2 host plants. Chem. Senses 35:57–64.
Trimble, R. M., Pree, D. J., Barszcz, E. S., and Carter, N. J. 2004. Comparison of a sprayable pheromone formulation and two hand-applied pheromone dispensers for use in the integrated control of oriental fruit moth (Lepidoptera: Tortricidae). J. Econ. Entomol. 97:482–489.
Visser, J. H. and Avé, D. A. 1978. General green leaf volatiles in the olfactory orientation of the Colorado beetle, Leptinotarsa decemlineata. Entomol. Exp. Appl. 24:738–749.
Wildbolz, T. 1958. Uber die Orientierung des Apfelwicklers bei der Eiablage. Mitt. Schweiz. Entomol. Ges. 31:25–34.
Witzgall, P., Bäckman, A. C., Svensson, M., Koch, U. T., Rama, F., El-Sayed, A., Brauchli, J., Arn, H., Bengtsson, M., and Löfqvist, J. 1999. Behavioral observations of codling moth, Cydia pomonella, in orchards permeated with synthetic pheromone. Biocontrol 44:211–237.
Yang, X. Y. and Liu, J. L. 2010. The reason of serious damage of Grapholitha molesta in 2008. Shanxi Fruit 1:38.
Yang, C. Y., Jung, J. K., Han, K. S., Boo, K. S., and Yiem, M. S. 2002. Sex pheromone composition and monitoring of the oriental fruit moth, Grapholitha molesta (Lepidoptera: Tortricidae) in Naju pear orchards. J. Asia Pac. Entomol. 5:201–207.
Yetter, W. P. and Steiner, L. F. 1932. Efficiency of bait traps for the oriental fruit moth as indicated by the release and capture of marked adults. J. Econ. Entomol. 25:106–116.
Zhang, Q. H., Ma, J. H., Yang, Q. Q., Byers, J. A., Klein, M. G., Zhao, F. Y., and Luo, Y. Q. 2011. Olfactory and visual responses of the long-legged chafer Hoplia spectabilis Medvedev (Coleoptera: Scarabaeidae) in Qinghai Province, China. Pest Manag. Sci. 67:162–169.
Zhao, Z. R., Wang, Y. G., and Yan, G. Y. 1989. The report of primary study on Grapholitha molesta in north of Jiangshu Province. Entomol. Knowl. 26:17–19.
Acknowledgments
We thank Rui Wang and Xiao-Wei Qin in our laboratory for technical assistance in GC-EAD and GC-MS, and Jun Liu from the Institute of Forestry and Pomology, Beijing Academy of Agriculture and Forestry Sciences for supplying facilities for field trials. We also thank Paolo Pelosi, University of Pisa, and Joop van Loon, Wageningen University, for help in improving the paper, and three anonymous reviewers for critical comments and suggestions. This work was supported by Public Welfare Project from Ministry of Agriculture of China (grant no. 200803006) and projects of Postdoctoral Science Foundation (grant no. 201104140) and the National Natural Science Foundation of China (grant no. 30925026, 30621003).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lu, PF., Huang, LQ. & Wang, CZ. Identification and Field Evaluation of Pear Fruit Volatiles Attractive to the Oriental Fruit Moth, Cydia molesta . J Chem Ecol 38, 1003–1016 (2012). https://doi.org/10.1007/s10886-012-0152-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-012-0152-4