Abstract
Phenylalanine ammonia-lyase (PAL), polyphenol oxidase (PPO), and peroxidase (POD) are considered important biochemical markers in host plant resistance against pest insects. Constitutive activity of these enzymes was analyzed in resistant and susceptible wheat cultivars against cereal aphid Sitobion avenae (F.) at various developmental stages, i.e., tillering, stem elongation, flag leaf, and ear. Following aphid infestation, the activity of these enzymes was determined at the flag leaf and ear stages. Resistant cultivars exhibited greater constitutive PAL activity than susceptible ones at the tillering, stem elongation, and flag leaf stages. Aphid infestation enhanced levels of PAL activity in the flag leaf and ear stages in both resistant and susceptible cultivars. Constitutive PPO activity was higher in the resistant cultivars at all developmental stages. Aphid infestation induced increases in PPO activity in the flag leaf and ear stages of one susceptible cultivar, whereas induction in resistant cultivars was weaker. Resistant cultivars showed greater constitutive POD activity in the tillering, stem elongation, and flag leaf stages, while aphid infestation induced POD activity in all cultivars, especially in susceptible ones. The potential role of PAL, PPO, and POD in wheat defense against aphid infestation is discussed.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Host plant resistance to herbivores can suppress herbivore population densities and offers a promising approach for managing insect pests in a sustainable, economical, and environmentally safe manner (Heng-Moss et al. 2004). Plant secondary metabolites play an important role in plant defense against insects and pathogens (Cai et al. 2004), the levels of which are often mediated by defense-related enzymes, such as phenylalanine ammonia-lyase (PAL), polyphenol oxidase (PPO), and peroxidase (POD; Carver et al. 1992; Rosenthal and Berenbaum 1992; Bi and Felton 1995; Bi et al. 1997a, b, c).
PAL is a key enzyme in the phenylpropanoid pathway that converts phenylalanine into trans-cinnamic acid. Trans-cinnimaic acid can be further hydroxylated and methylated to produce compounds (i.e., coumaric, caffeic, and ferulic acids) that are toxic to herbivores and pathogens (Cole 1984; Leszczynski et al. 1989; Verpoorte and Alfermann 2000; Morelló et al. 2005; Wang et al. 2006). PPO and POD are oxidases that catalyze the formation of quinones from various phenolic precursors (Hildebrand et al. 1986; Mayer 1987; Felton et al. 1989; Duffey and Stout 1996).
PAL, PPO, and POD are associated with plant defense against insects (Felton et al. 1992; Stout et al. 1999; Chaman et al. 2001, 2003; Ni et al. 2001). However, little is known about the dynamic changes of these enzymes during plant development and the influence of aphid infestation on their activity in resistant vs. susceptible cultivars, especially in wheat. The present research was initiated to investigate the constitutive and induced activity of these enzymes in Sitobion avenae-resistant and S. avenae-susceptible wheat cultivars at various developmental stages.
Methods and Materials
Plants and Insects
Wheat cultivars KOK1679, L1, Beijing 411, and Beijing 837 were grown in 2003 and 2004 at the Science Park of China Agricultural University, Beijing, China. Wheat seeds were sown 20 cm apart in a 2-m single-row plot on October 14, 2003. Each plot was used as one replicate and covered with white nylon mesh to protect plants from insects and mechanical damage during the growth.
KOK1679 and L1 cultivars are resistant and repellant to the cereal aphid species S. avenae, while Beijing 411 and Beijing 837 are susceptible and attractive to this species (Liu et al. 2001). In addition, both KOK1679 and L1 have antibiotic properties against S. avenae compared to Beijing 411 and Beijing 837 (Cai et al. 2004 and unpublished data).
S. avenae nymphs and adults were collected from field-grown wheat and maintained on the aphid-susceptible wheat cultivar, Beijing 411, under field conditions. Insects over one generation were used for the experiment. When the unfolded flag leaf and ear emerged in 49–56 and 58–60 of Zadoks code (Zadoks et al. 1974), 20 flag leaves or ears from each experimental plot were selected and evenly divided for experimental and control treatments. Ten nymphs (second to third instar) were released onto each flag leaf and ear in each treatment. The flag leaves and ears were covered with nylon mesh cages (15 × 4 cm) to prevent the aphids from escaping (Rafi et al. 1996; Argandona et al. 2001). The experiments were arranged in a randomized complete block design, and each treatment was replicated three times.
Sample Collection
A total of 60 tillers from each plot were sampled randomly at various developmental stages to determine the constitutive enzyme activity in wheat tissue. Whole plants, including stems and leaves at Zodaks stage 21–23 and the upper 3–5 young leaves and stem at Zadoks stage 31–34, were sampled (Zadoks et al. 1974). Flag leaves at stage 49–56 and ears at stage 58–60 also were sampled. Flag leaves and ears of all the treatments and controls were individually excised and collected 9 days after aphid release (Ni et al. 2001). Upon collection, samples were wrapped in aluminum foil and immediately dropped into liquid nitrogen for transport to a −80°C freezer before enzymatic analyses.
Plant Protein and Enzyme Assays
Plant tissue (100 mg) from each sample was ground in liquid nitrogen and homogenized in 20 mM HEPES buffer (500 μl; pH 7.2). The resulting mixture was centrifuged (Himac CR22E, Hitachi Koki Co. Ltd. Japan) at 10,000×g for 20 min at 4°C. The supernatant was used for analyses of total protein content and enzymatic activity.
The Bradford method (Bradford 1976) was used to determine total protein content in samples. Three aliquots (20 μl) of each sample were mixed with Bio-Rad reagent (180 μl, Bio-Rad, Richmond, CA, USA). Absorbance of the reaction mixture was determined at 595 nm, and protein content was determined from a standard curve established by using known quantities of bovine serum albumin (from Sigma Chemical Co.) and the above reagent.
PAL activity was determined following the method of Wang and Xue (1980) with enzyme extract (0.25 ml) in sodium borate buffer (1.5 ml, 30 mM; pH 8.8). The decrease of l-phenylalanine was monitored at 290 nm. PAL activity was estimated from dA 290. The assay was replicated three times.
POD activity was measured following a method previously described (Hildebrand et al. 1986; Hori et al. 1997). Enzyme extract (10 μl) was mixed with 1 ml of substrate containing hydrogen peroxide (10 μl; 30%, w/w), guaiacol (1 μl; 20 mM), and HEPES (100 μl; 200 mM, pH 7.0) in deionized water. POD activity was estimated from the increase in A 470. The measurement was repeated three times.
PPO activity was determined following the method of Hori et al. (1997). Enzyme extract (100 μl) was mixed with a solution containing catechol (500 μl; 1.6% w/w), HEPES buffer (100 μl; 200 mM, pH 6.0), and deionized water (800 μl). PPO activity was estimated from the increase in A 470. The analysis was repeated three times.
Statistical Analysis
Analysis of variance (ANOVA) in SPSS (SPSS 11.0) was used to analyze PAL, POD, and PPO activity in wheat cultivars at various developmental stages. Pairs of treatment means were compared and separated (α = 0.05) by the least significant difference (LSD) procedure. Before the ANOVA and LSD procedure, PAL, POD, and PPO activity was transformed using the logarithm transformation (ln) to normalize the data.
Results
Protein Content
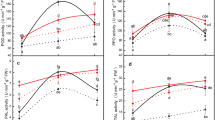
Total protein content among wheat cultivars was significantly different at the different developmental stages (i.e., tillering, stem elongation, flag leaf, and ear stages), as well as at the flag leaf and ear stages in plants infested by aphids (df = 3, 11, F TS = 7294.6, F SES = 4887.1, F FL = 11040.8, F H = 9939.9, F AFL = 4883.9, F AH = 8267.3, P < 0.001). The constitutive protein content of KOK1679 and L1 was lower than that of Beijing 411 and Beijing 837 at all stages, with the exception that the protein content of KOK1679 was higher than that of Beijing 837 at the flag leaf stage (Table 1, Fig. 1). However, after flag leaf and ear stages were infested by aphids, total protein content increased in flag leaves of all cultivars and in ears of KOK1679 and L1 (P < 0.05; Fig. 1).
Defense-Related Enzymes
KOK1679 and L1 exhibited greater constitutive PAL activity than Beijing 411 and Beijing 837 in the tillering and stem elongation stages (df = 3, 11; F TS = 28.13, P < 0.01; F SES = 6.53, P = 0.02; Table 2). The constitutive PAL activity was also higher in KOK1679 and L1 than in Beijing 411 and Beijing 837 in the flag leaf (df = 3, 11; F FL = 26.58, P < 0.001), but not in the ear stage (Fig. 2). Aphid infestation strongly enhanced the levels of PAL activity in the flag leaf and ear stages of all four cultivars (P < 0.05; Fig. 2).
Constitutive PPO activity was higher in KOK1679 and L1 than in Beijing 837 and Beijing 411 at all developmental stages (df = 3, 11; F TS = 9.81, P = 0.005; F SES = 7.34, P = 0.011; F FL = 18.72, P = 0.001; F E = 21.33, P < 0.001; Table 3, Fig. 3). Aphid infestation caused significant increases in PPO activity in the flag leaf and the ear stages of Beijing 837 and Beijing 411 and in the ear stage of KOK 1679 (Fig. 3).
KOK1679 and L1 exhibited greater constitutive POD activity than Beijing 837 and Beijing 411 in the tillering and stem elongation stages (df = 3, 11; F TS = 21.78, P < 0.001; F SES = 15.28, P = 0.001; Table 4). The constitutive POD activity was also higher in the flag leaf stage of KOK1679 and L1 than in Beijing 837 and Beijing 411 (df = 3, 11; F FL = 15.79, P = 0.001; Fig. 4). Aphid infestation boosted POD activity in all cultivars, especially in Beijing 837 and Beijing 411 (P < 0.05; Fig. 4).
Discussion
Active defense in plants can be induced by both biotic and abiotic factors (Karban and Baldwin 1997; Kant et al. 2004). PAL, PPO, and POD are among the most important enzymes involved in the defensive responses of plants to insects and pathogens. PAL plays a key role in corn resistance to the maize sheath blight Rhizoctonia solani (Jin et al. 2003) as well as to Helminthosporium maydis and Helminthosporium turcicum (Wang and Xue 1980). Cole (1984) demonstrated that PAL activity is related to lettuce resistance to the lettuce root aphid Pemphigus bursarius (L). S. avenae infestation resulted in increased PAL activity in the flag leaf and ear stages of resistant and susceptible winter wheat cultivars (Ciepiela 1989; Havlickova et al. 1996). Increased PAL activity also was found in the leaves of Brassica oleracea seedlings infested by green peach aphid Myzus persicae (Sulzer; Zhang et al. 2005).
S. avenae-resistant wheat cultivars (i.e., KOK1679 and L1) had significantly higher levels of PAL activity than susceptible cultivars (Beijing 411 and Beijing 837) at the tillering and stem elongation developmental stages. PAL activity in the flag leaf and ear stages of aphid-resistant cultivars increased more dramatically than that in the aphid-susceptible ones after S. avenae infestation, suggesting that the resistant cultivars may have a stronger capability to synthesize secondary metabolites related to insect susceptible ones.
Increased PPO activity in susceptible wheat can be induced by both aphids and methyl jasmonate (Leszczynski 1985; Boughton et al. 2006). Our study showed that constitutive PPO activity was higher in nutritional organs of resistant wheat cultivars compared to susceptible ones (Table 3). PPO activity in the ear and flag leaf stages of both resistant and susceptible cultivars increased when plants were infested with aphids; the increase was greater in susceptible cultivars (Fig. 3). However, Chrzanowski et al. (2003) reported that PPO activity in the ear stages of both resistant and susceptible spring wheat cultivars was decreased by S. avenae infestation. This discrepancy may be due, at least in part, to genetic differences in the wheat cultivars. Ni et al. (2001) reported that Diuraphis noxia feeding did not elicit changes in PPO activity in either resistant or susceptible cultivars of wheat or barley. This difference may be associated with the specific feeding behaviors exhibited by different aphid species, e.g., D. noxia feeding elicits leaf-chlorosis while S. avenae feeding does not.
Previous studies have demonstrated that POD activity can be enhanced by insects and elicitors such as ethephon and methyl jasmonate (Bi and Felton 1995; Bi et al. 1997a, b; Takahama and Oniki 1997; Kielkiewicz 1998). Buffalograss that is resistant to Blissus occiduus had higher POD activity compared to susceptible plants (Heng-Moss et al. 2004). Andres et al. (2001) reported that wheat root resistance to the cereal cyst nematode was associated with increased POD activity. POD activity in B. oleracea leaves infested with M. persicae increased significantly compared with control leaves (Zhang et al. 2005). Macrosiphum euphorbiae (Thomas) feeding induced greater POD activity in tomato leaves (Stout et al. 1998). Our results revealed that at the tilling and stem elongation stages, resistant wheat cultivars had a greater constitutive POD activity compared with susceptible cultivars, whereas at the flag leaf stage, the constitutive POD activities were similar between the resistant and susceptible cultivars (Table 4, Fig. 4). Also, aphid infestation of susceptible wheat cultivars at the flag leaf and ear stages induced a higher POD activity (Fig. 4). The differences in POD activities among the growing stages may be attributed to normal wheat plant growth (Ni et al. 2001).
In summary, S. avenae-resistant wheat cultivars had greater constitutive PAL, PPO, and POD activity at the tillering, stem elongation, and flag leaf stages of development. When the flag leaf and ear stages were attacked by aphids, PAL activity increased more dramatically in the resistant cultivars than in the susceptible ones. Aphid infestation increased activities of PPO and POD more dramatically in the susceptible cultivars than in the resistant ones.
References
Andres, M. F., Melillo, M. T., Delibes, A., Romero, M. D., and Bleve-Zacheo, T. 2001. Changes in wheat root enzymes correlated with resistance to cereal cyst nematodes. New Phytol. 152:343–354.
Argandona, V. H., Chaman, M., Cardemil, L., Munoz, O., Zuniga, G. E., and Corcuera, L. J. 2001. Ethylene production and peroxidase activity in aphid-infested barley. J. Chem. Ecol. 27:53–68.
Bi, J. L., and Felton, G. W. 1995. Foliar oxidative stress and insect herbivory: primary compounds, secondary metabolites and reactive oxygen species as components of induced resistance. J. Chem. Ecol. 21:1511–1530.
Bi, J. L., Murphy, J. B., and Felton, G. W. 1997a. Antinutritive and oxidative components as mechanisms of induced resistance in cotton to Helicoverpa zea. J. Chem. Ecol. 23:97–117.
Bi, J. L., Murphy, J. B., and Felton, G. W. 1997b. Does salicylic acid act as a signal in cotton for induced resistance to Helicoverpa zea? J. Chem. Ecol. 23:1805–1818.
Bi, J. L., Felton, G. W., Murphy, J. B., Howles, P. A., Dixon, R. A., and Lamb, C. J. 1997c. Do plant phenolics confer resistance to specialist and generalist insect herbivores? J. Agri. Food Chem. 45:4500–4504.
Boughton, A. J., Hoover, K., and Felton, G. W. 2006. Impact of chemical elicitor applications on greenhouse tomato plants and population growth of the green peach aphid, Myzus persicae. Entomol. Exp. Appl. 120:175–188.
Bradford, M. M. 1976. A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72:248–254.
Cai, Q. N., Zhang, Q. W., and Cheo, M. 2004. Contribution of indole alkaloids to Sitobion avenae (F.) resistance in wheat. J. Appl. Entomol. 128:517–521.
Carver, T. L. W., Robbins, M. P., Zeyen, R. J., and Dearne, G. A. 1992. Effects of PAL-specific inhibition on suppression of activated defence and quantitative susceptibility of oats to Erysiphe graminis. Physiol. Mol. Plant Pathol. 41:149–163.
Chaman, M. E., Corcuera, L. J., Zuniga, G. E., Cardemil, L., and Argandona, V. H. 2001. Induction of soluble and cell wall peroxidases by aphid infestation in barley. J. Agr. Food Chem. 49:2249–2253.
Chaman, M. E., Copaja, S. V., and Argandona, V. H. 2003. Relationships between salicylic acid content, phenylalanine ammonia-lyase (PAL) activity, and resistance of barley to aphid infestation. J. Agr. Food Chem. 51:2227–2231.
Chrzanowski, G., Ciepiela, A. P., Sprawka, I., Sempruch, C., Sytykiewicz, H., and Czerniewicz, P. 2003. Activity of polyphenoloxidase in the ears of spring wheat and triticale infested by grain aphid (Sitobion avenae (F.)). Electronic J. Polish Agricult. Universities, Biol., 6:2, art-04.
Ciepiela, A. 1989. Changes in phenylalanine and tyrosine content and metabolism in ears of susceptible and aphid resistant winter wheat cultivars upon infestation by Sitobion avenae. Entomol. Exp. Appl. 51:277–281.
Cole, R. A. 1984. Phenolic acids associated with the resistance of lettuce cultivars to the lettuce root aphid. Ann. Appl. Biol. 105:129–145.
Duffey, S. S., and Stout, M. J. 1996. A nutritive and toxic compounds of plant defense against insects. Arch. Insect Biochem. Physiol. 32:3–37.
Felton, G. W., Donato, R. J., Vecchio, D., and Duffey, S. S. 1989. Activation of plant foliar oxidases by insect feeding reduces nutritive quality of foliage for noctuid herbivores. J. Chem. Ecol. 15:2667–2694.
Felton, G. W., Donato, K. K., Broadway, R. M., and Duffey, S. S. 1992. Impact of oxidized plant phenolics on the nutritional quality of dietary protein to a noctuid herbivore, Spodoptera exigua. J. Insect Physiol. 38:277–285.
Havlickova, H., Cvikrova, M., and Eder, J. 1996. Changes in the pattern of phenolic acids induced by aphid infestation in two winter wheat cultivars. Bulletin OILB/SROP 19:106–110.
Heng-Moss, T., Sarath, G., and Baxendale, F. 2004. Characterization of oxidative enzyme changes in buffalograsses challenged by Blissus occiduus. J. Econ. Entomol. 97:1086–1095.
Hildebrand, D. F., Rodriguez, J. G., Brown, G. C., Lui, K. T., and Volden, C. S. 1986. Peroxidative responses of leaves in two soybean genotypes injured by two spotted spider mites (Acari: Tetranychidae). J. Econ. Entomol. 79:1459–1465.
Hori, K., Wada, A., and Shibuta, T. 1997. Changes in phenoloxidase activities of the gals on leaves of Ulmus davidana formed by Tetraneura fusiformis (Homoptera: Eriosomatidae). Appl. Entomol. Zool. 32:365–371.
Jin, Q., Ye, H., and Zhang, M. 2003. Relationship between the activity of PAL and resistance of corn to maize sheath blight. J. Sichuan Agricult. University 21:116–118. (In Chinese with English abstract).
Kant, M. R., Ament, K., Sabelis, M. W., Haring, M. A., and Schuurink, R. C. 2004. Differential timing of spider mite-induced direct and indirect defenses in tomato plants. Plant Physiol. 135:483–495.
Karban, R., and Baldwin, I. T. 1997. Induced Responses to Herbivory. The University of Chicago Press, Chicago.
Kielkiewicz, M. 1998. Concentration of some phenylpropanoid compounds and the activity of oxidative enzymes in the intra-tomato plant (Lycopersicon esculentum Mill.) locally infested by the carmine spider mite (Tetranychus cinnabarinus Boisd.). Zeszyty Naukowe Akademii Techniczno Rolniczej w Bydgoszczy (Poland) 214:41–47.
Leszczynski, B. 1985. Changes in phenols content and metabolism in leaves of susceptible and resistant winter wheat cultivars infested by Rhopalosiphum padi (L.) (Homoptera: Aphididae). Zeitschrift fur Angewandte Entomologie 100:343–348.
Leszczynski, B., Wright, L. C., and Bakowski, T. 1989. Effect of secondary plant substances on winter wheat resistance to grain aphid. Entomol. Exp. Appl. 52:135–139.
Liu, Y., Ni, H., Sun, J., and Hu, C. 2001. Different olfactory responses of wheat aphids to the volatiles of wheat plant with different resistant levels. Scientia Agricultura Sinica 34:391–395. (In Chinese with English abstract).
Mayer, A. M. 1987. Polyphenol oxidase in plants—recent progress. Phytochemistry 26:11–20.
Morelló, J. R., Romero, M. P., Ramo, T. M., and Motilva, J. 2005. Evaluation of L-phenylalanine ammonia-lyase activity and phenolic profile in olive drupe (Olea europaea L.) from fruit setting period to harvesting time. Plant Sci. 168:65–72.
Ni, X., Quisenberry, S. S., Heng-Moss, T., Markwell, J., Sarath, G., Klucas, R., and Baxendale, F. 2001. Oxidative responses of resistant and susceptible cereal leaves to symptomatic and non-symptomatic cereal aphid (Hemiptera: Aphididae) feeding. J. Econ. Entomol. 94:743–751.
Rafi, M. M., Zemetra, R. S., and Quisenberry, S. S. 1996. Interaction between Russian wheat aphid (Homoptera: Aphididae) and resistant and susceptible genotypes of wheat. J. Econ. Entomol. 89:239–246.
Rosenthal, G. A., and Berenbaum, M. R. 1992. Herbivores: Their Interactions with Secondary Plant Metabolites. Academic, New York.
Stout, M. J., Workman, K. V., Bostock, R. M., and Duffey, S. S. 1998. Specificity of induced resistance in the tomato, Lycopersicon esculentum. Oecologia 113:74–81.
Stout, M. J., Fidantsef, A. L., Duffey, S. S., and Bostock, R. M. 1999. Signal interactions in pathogen and insect attack: systemic plant-mediated interactions between pathogens and herbivores of the tomato, Lycopersicon esculentum. Physiol. Mol. Plant Pathol. 54:115–130.
Takahama, U., and Oniki, T. 1997. A peroxidase/phenolics/ascorbate system can scavenge hydrogen peroxide in plant cells. Physiol. Plant. 101:845–852.
Verpoorte, R., and Alfermann, A. W. 2000. Metabolic engineering of plant secondary metabolism. Kluwer, Dordrecht.
Wang, J. W., and Xue, Y. L. 1980. Studies on plant phenylalanine ammonia-lyase. Acta Phytophysiologia Sinica 7:374–380. (In Chinese with English abstract).
Wang, Y., Cai, Q. N., Zhang, Q. W., and Han, Y. 2006. Effect of the secondary substances from wheat on the growth and digestive physiology of cotton bollworm Helicoverpa armigera (Lepidoptera: Noctuidae). Eur. J. Entomol. 103:255–258.
Zadoks, J. C., Chang, T. T., and Konzak, C. F. 1974. A decimal code for the growth stages of cereals. Weed Res. 14:415–421.
Zhang, C. N., Wu, J. X., Dai, W., and Chen, L. 2005. Activities of some isoenzymes in the leaves of Brassica oleracea seedlings infested by peach aphid (Myzus persicae). Acta Botanica Sinica 25:1566–1569.
Acknowledgments
This research was funded by the National Basic Research Program of China (“973” Program, 2006CB100206), National Support Program (2006BAD08A05), Ministry of Science and Technology of China, and the Initiation Research Project (2004050) of China Agricultural University. We thank Dr. Jinping Du (Beijing Ecoman Biotech Co. LTD) for the grammatical assistance and two anonymous reviewers for the helpful comments.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Han, Y., Wang, Y., Bi, JL. et al. Constitutive and Induced Activities of Defense-Related Enzymes in Aphid-Resistant and Aphid-Susceptible Cultivars of Wheat. J Chem Ecol 35, 176–182 (2009). https://doi.org/10.1007/s10886-009-9589-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-009-9589-5