Abstract
Tannins are believed to function as antiherbivore defenses, in part, by acting as prooxidants. However, at the high pH found in the midguts of caterpillars, the oxidative activities of different types of tannins vary tremendously: ellagitannins >> galloyl glucoses > condensed tannins. Ingested ascorbate is utilized by caterpillars to minimize phenolic oxidation in the midgut. Thus, leaves that contain higher levels of reactive tannins and lower levels of ascorbate were hypothesized to produce higher levels of phenolic oxidation in caterpillars. We tested this hypothesis with eight species of deciduous trees by measuring their foliar phenolic and ascorbate compositions and measuring the semiquinone radical (oxidized phenolic) levels formed in caterpillars that ingested each species. When the generalist caterpillars of Orgyia leucostigma (Lymantriidae) fed on the leaves of tree species in which condensed tannins were predominant (i.e., Populus tremuloides, P. deltoides, and Ostrya virginiana), semiquinone radical levels were low or entirely absent from the midgut contents. In contrast, species that contained higher levels of ellagitannins (or galloyl rhamnoses; i.e., Quercus alba, Acer rubrum, and A. saccharum) produced the highest levels of semiquinone radicals in O. leucostigma. Low molecular weight phenolics contributed relatively little to the overall oxidative activities of tree leaves compared with reactive tannins. Ascorbate levels were lowest in the species that also contained the highest levels of oxidatively active tannins, potentially exacerbating phenolic oxidation in the gut lumen. We conclude that the tannin compositions of tree leaves largely determine the effectiveness of foliar phenolics as oxidative defenses against caterpillars such as O. leucostigma.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
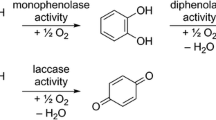
Phenolics are ubiquitous chemical defenses in leaves, playing protective roles against a wide variety of biotic agents and abiotic stresses. Tannins are the predominant phenolics produced in tree leaves, often reaching levels of 5–20% dry weight (DW) in temperate deciduous species. Whereas tannins were long believed to act as protein-binding agents that reduced the digestive efficiencies of insect herbivores, protein binding in vivo is now known to be precluded by high pH and/or surfactants generated from lipid digestion (Martin et al. 1987). Instead, as defenses against caterpillars, tannins can act as anti-feedants or have toxic or anti-nutritive effects. Because tannins are not absorbed, their post-ingestive effects are thought to result from their oxidation in the midgut lumen (Barbehenn 2001; Barbehenn et al. 2005). The oxidation of phenolics produces semiquinone radicals and other reactive oxygen species. If levels of these products overwhelm the antioxidant defenses in the midgut lumen or tissues, oxidative stress ensues. The oxidative damage to a wide variety of biological molecules (including nutrients, cell membrane components, and DNA) that occurs from oxidative stress is believed to contribute to reduced fitness in insect herbivores (Summers and Felton 1994; Bi and Felton 1995; Bi et al. 1997).
Ascorbate is the main low molecular weight (MW) antioxidant found in the gut fluids and tissues of leaf-feeding insects (Felton and Duffey 1992; Barbehenn et al. 2001, 2003a). However, antioxidant defense systems that utilize ascorbate can be overwhelmed when insects feed on leaves that contain either large amounts or highly reactive types of tannins (Barbehenn et al. 2005). The most reactive types of tannins in the high pH conditions found in caterpillar midguts are ellagitannins, followed by galloyl glycosides (or “gallotannins”) and, distantly, by condensed tannins (Barbehenn et al. 2006b; Moilanen and Salminen 2008). Whether this pattern of tannin structure and reactivity also occurs in the complex chemical mixtures found in the midgut fluids of leaf-feeding herbivores has been examined with only one pair of tree species. Acer saccharum (containing relatively high levels of ellagitannins) produced high levels of semiquinone radicals in the midgut contents of caterpillars, while Quercus rubra (primarily containing condensed tannins) produced low levels of semiquinone radicals in caterpillars (Barbehenn et al. 2005, 2006a). In this study, the association between foliar chemical composition and phenolic oxidation was examined in a diverse group of tree species when eaten by the generalist caterpillar Orgyia leucostigma (Lymantriidae). The caterpillars of O. leucostigma are highly polyphagous and include the wide variety of tree species in this study in their diet (Baker 1972; Wagner 2005). Phenolics and ascorbate were quantified in the leaves of eight North American deciduous trees: Populus tremuloides (quaking aspen), P. deltoides (cottonwood), Ostrya virginiana (ironwood), Carya glabra (bitternut hickory), Q. rubra (red oak), Q. alba (white oak), A. rubrum (red maple), and A. saccharum (sugar maple). Phenolic oxidation in the midgut fluids of O. leucostigma was compared between tree species by measuring levels of semiquinone radicals with electron paramagnetic resonance (EPR) spectrometry. EPR spectrometry permits the sensitive and specific detection of free radicals in the complex chemical environment of biological samples (Rosen et al. 1999; Barbehenn et al. 2005). This study tested the hypothesis that the proportion of oxidatively active tannins to ascorbate in tree leaves is positively associated with levels of phenolic oxidation in caterpillars.
Methods and Materials
Tree Leaf Chemistry
Six trees of each species were tagged at ten sites, primarily in parks and woodlands in Ann Arbor, MI, USA. Trees of each species were sampled from at least three different sites. A mixture of wild and planted trees of Q. rubra and the Acer species were used. All other trees were growing wild. Trunk diameters at breast height were measured to estimate the following tree sizes (mean [centimeters] ± SE): P. tremuloides, 14.5 ± 2.0; P. deltoides, 39.6 ± 4.5; O. virginiana, 13.6 ± 2.6; C. glabra, 59.5 ± 24.0; Q. rubra, 32.6 ± 4.6; Q. alba, 39.6 ± 11.0; A. rubrum, 19.4 ± 4.5; and A. saccharum, 27.7 ± 7.7. Branch tips were cut from the sunny sides of trees at a height of 2–4 m and immediately placed in flasks of water. Leaves from the same collection used for feeding O. leucostigma were kept intact on twigs in containers of water overnight in an incubator to control for potential changes in foliar chemistry during the feeding period. Leaf midribs were removed with a razor blade, and the remaining leaf lamina were weighed. To measure ascorbic acid, leaves were ground in liquid nitrogen, extracted in 5% metaphosphoric acid (containing 1 mM ethylenediaminetetraacetic acid), and stored at −80°C until analyzed with high-performance liquid chromatography (HPLC; Barbehenn 2003). To measure phenolics, leaves were frozen (−80°C), lyophilized, and ground. Phenolic analyses were performed in Turku, Finland with HPLC coupled with a diode array detector and mass spectrometer, as described previously (Salminen et al. 1999; Barbehenn et al. 2006a). Hydrolyzable tannins were quantified in pentagalloyl glucose equivalents (280 nm), flavonoids in quercetin equivalents (349 nm), chlorogenic acids in chlorogenic acid equivalents (315 nm), coumaroylquinic acids in coumaric acid equivalents (315 nm), and the remaining phenolics (other than condensed tannins) as gallic acid equivalents (280 nm). In addition, total condensed tannins were estimated with the acid–butanol assay (Ossipova et al. 2001), with purified birch leaf condensed tannins as a standard. The chemical compositions of the leaves fed to larvae during a second experiment with multiple tree species were also analyzed as described above.
Free Radicals in Caterpillars
Eggs of O. leucostigma were obtained from the Canadian Forest Pest Management Institute (Sault Ste. Marie, Ontario). Larvae were reared until the final instar in an incubator (23°C, 16:8 h, light/dark) on an artificial diet, as described previously (Barbehenn et al. 2005). Larvae were randomly assigned to feed on the leaves of one of two tree species examined at one of four dates: Acer species on July 4, 2006, Quercus species on July 7, Populus species on July 11, and O. virginiana and C. glabra on July 14. In the first experiments, a single tree from each species was used, providing seven to nine insect replicates on each foliar chemical profile. To demonstrate that results for each species tested were not limited by the use of a single tree per species, tree leaves from five of the tree species were fed to separate groups of O. leucostigma on the same day (July 15), using trees from each species different from those used in the first set of experiments. Confirmatory EPR measurements were made on two to three larvae per tree species. Q. rubra and A. saccharum showed similar results in the first set of experiments as were found previously (Barbehenn et al. 2005; unpublished data), and these species were not reexamined. Leaves were surface sterilized in a dilute bleach solution (2.5 ml/l; 10 min) and rinsed in water (10 min). Twigs containing clusters of leaves were kept in tubes of water to maintain leaf turgor. Groups of final-instars were placed on the leaves of each species in separate ventilated plastic boxes (30 × 20 × 10 cm) and placed in an incubator (23°C, 16:8 h, light/dark). Leaves of each species were readily eaten, although a lower amount of feeding damage was observed on Q. alba. Fresh leaves were provided on two consecutive days, using the same tree from each species. A collection of leaves from each tree was made on its second day for chemical analysis, as described above. On the third day of feeding, larvae were individually chilled (−20°C, 6 min), and their midguts were dissected. Midgut fluids were extracted in 300 µl of low-oxygen pH 10 carbonate buffer (70 mM, containing 10% dimethyl sulfoxide; Barbehenn et al. 2005).
Semiquinone and ascorbyl radicals were quantified with EPR spectrometry, as described previously (Barbehenn et al. 2006b). Standard solutions of the stable free radical, 2,2,6,6-tetramethyl-1-piperidinyloxy (TEMPO; Aldrich Chemical Co.) were run to confirm that the method was consistent through time and to provide a free radical standard for converting double integrals of spectra to a concentration basis (nanomolar). Radical concentrations are expressed as TEMPO radical equivalents. Radical concentrations in midgut fluid volumes were calculated by using a value of 90% water in the midgut contents (Barbehenn et al. 2003a). In some cases, EPR spectra were either too weak to integrate accurately (e.g., negative double integral values) or produced double integral values that were overestimated because the baseline regions surrounding the signal contributed (incorrectly) to the overall double integral values. In these cases, WinEPR software (Bruker Instruments, Billerica, MA, USA) was used to correct each spectrum as follows. The first derivative spectrum was baseline corrected by using the baseline area on either side of the signal region. The corrected first derivative spectrum was then integrated. The resulting absorption spectrum was again baseline corrected by using the baseline areas on either side of the signal region. This produced a signal region flanked by flat baseline regions abutting a Y-axis value of zero. The corrected double integral value was then determined. For spectra that contained signals from both semiquinone and ascorbyl radicals, an additional calculation was made to separate the two component signals. The ascorbyl radical double integral value was determined from a regression of the peak heights of ascorbyl radical absorption spectra (downfield peaks) vs. the double integral values of these spectra. The semiquinone portion of the mixed spectrum was then calculated as the difference between the total radical and the ascorbyl radical double integral values.
To test the possibility that the unusual EPR spectra from caterpillars on C. glabra might have been caused by the reduction of quinones by ascorbate, EPR was used to examine juglone (Sigma Chemical Co.), a common quinone from species of Juglandaceae. Juglone (0.9 mM final concentration) was run alone and in mixtures with ascorbic acid (0.4–0.8 mM final concentration). The same parameters and experimental conditions for EPR were used as described above.
The relative contributions of low MW phenolics to the semiquinone levels measured in the midgut fluid O. leucostigma was estimated by using representative compounds from each of the groups of phenolic compounds measured: gallic acid, p-coumaric acid, chlorogenic acid, catechin, rutin, and its aglycone quercetin (Sigma Chemical Co.). In addition, salicortin, a “phenolic glycoside” purified from P. tremuloides, was examined (Lindroth et al. 1987). All compounds were solubilized in 70% ethanol, prepared from nitrogen-purged solvents, with the exception of rutin, which was solubilized in 90% methanol. Semiquinone concentrations formed from each phenolic were measured at three phenolic concentrations (ranging between 22–648 μg/ml; N = 3 independent preparations/phenolic). The slopes of the regressions of semiquinone concentration on phenolic concentration normalize semiquinone radical concentrations for the effect of phenolic concentration (Barbehenn et al. 2006b). Two scans of each sample were run by using parameters and other experimental conditions identical to those described for examining gut fluid extracts.
Statistical Analysis
Foliar tannins, low MW phenolics and ascorbic acid were compared among tree species with one-way analysis of variance (Wilkinson 2000). Post hoc comparisons among means were made with a Bonferroni adjustment of α for unplanned comparisons. EPR spectra from O. leucostigma were not significantly different among trees (experiments) within species in terms of semiquinone concentration (P > 0.414; see Fig. 3a) or in line shape. Therefore, EPR data were pooled within tree species, providing results from two trees for most species and sample sizes of 9–12 replicate larvae per tree species. Larvae were fed leaves from only one tree from C. glabra, thus limiting generalization from these results because of pseudoreplication. No replication of A. saccharum and Q. rubrum trees was done after obtaining results that confirmed previous results from these species (Barbehenn et al. 2005; unpublished data). Free radical levels in O. leucostigma (replicates) were compared among tree species with Mann–Whitney U tests, with a Bonferroni adjustment for unplanned comparisons (Wilkinson 2000). This experimental design was intended to examine the main hypothesis that caterpillar gut biochemistry varies among trees with specific foliar phenolic profiles. Associations between foliar chemistry in individual trees fed to O. leucostigma and semiquinone radical levels in the midgut fluids of each larva were examined with Pearson correlations (SAS 2003).
Results
Tannins comprised the bulk of the phenolic compounds in most of the tree species (Table 1, Fig. 1). P. tremuloides, P. deltoides, O. virginiana, and Q. rubra contained little or no ellagitannins or galloyl glycosides. By contrast, Q. alba, A. saccharum, and C. glabra contained substantial amounts of ellagitannins. A. rubrum and A. saccharum were the only species that contained galloyl glycosides; A. rubrum contained large amounts of digalloyl rhamnose and lower levels of tri-, tetra- and pentagalloyl rhamnose (unpublished data), whereas A. saccharum primarily contained small amounts of pentagalloyl glucose. Condensed tannins represented a major fraction (30–70%) of the phenolics in five of the species (P. tremuloides, P. deltoides, O. virginiana, C. glabra, and Q. rubra). By comparison, in three of the most oxidatively active species (Q. alba, A. rubrum, and A. saccharum), condensed tannin was a minor fraction (4–16%) of the phenolics (Fig. 1).
Low MW phenolics were present in similar, small amounts (0.5–2.6% DW) in most species (Table 2), comprising between 6% and 41% of the total phenolics (Fig. 1; excluding ascorbic acid). However, in P. deltoides, low MW phenolics were both at higher absolute levels (3.6% DW) and comprised a major fraction of the total phenolics (69%). Chlorogenic acid (and its derivatives) and flavonoid glycosides were the main types of low MW phenolics in each species (Table 2). While chlorogenic acid had relatively high oxidative activity, the oxidative activity of rutin (a representative flavonoid glycoside) was low (Table 3). The aglycone of rutin, quercetin, had substantially higher activity than the glycoside, but aglycones were below detectable levels in the tree leaves. The oxidative activities of catechin, rutin, and p-coumaric acid appeared to be negligible. Although gallic acid was highly active, it was present only in small amounts in A. rubrum. Salicortin produced no EPR spectrum. Based on these representative examples and the low foliar concentrations of low MW phenolics, their contributions to the semiquinone radical levels observed in the midgut contents of caterpillars appeared to be small.
Ascorbate levels varied widely between tree species: C. glabra and P. tremuloides ≫ P. deltoides and O. virginiana > Q. rubra and Q. alba > A. rubrum and A. saccharum (Table 1). For comparison with results provided on a fresh weight basis, ascorbate levels ranged from 6.0 μmol/g in A. rubrum to 25.6 μmol/g in C. glabra.
In the midgut contents of O. leucostigma that fed on tree species containing low levels of oxidatively active phenolics (i.e., P. tremuloides, P. deltoides, and O. virginiana), semiquinone radicals were absent or at low levels (Fig. 2; Table 4). In larvae on P. tremuloides and P. deltoides, only ascorbyl radicals (benign) were present. In larvae on Q. rubra and O. virginiana, EPR spectra were consistent with the line shapes of condensed tannin radicals. By contrast, species with substantial levels of ellagitannins, or galloyl glycosides, and low levels of ascorbic acid (i.e., Q. alba, A. saccharum, and A. rubrum) produced high levels of semiquinone radicals (Figs. 1 and 2). These EPR spectra were similar to those of hydrolyzable tannins (Barbehenn et al. 2006b). EPR spectra from larvae on C. glabra were unique, showing substantial levels of both ascorbyl and semiquinone radicals (Fig. 2, Table 4). Similar “hybrid” EPR spectra were produced in pH 10 reaction mixtures that contained juglone and ascorbic acid (data not shown).
To quantify the association between foliar chemistry and levels of semiquinone radicals in O. leucostigma, correlations between these measures were plotted. The resulting correlations provided some support for the hypothesis that the proportion of oxidatively active tannins to ascorbate is positively associated with semiquinone radical levels. The correlation between total phenolics/ascorbate and semiquinone radical levels was relatively weak, in part, because species that contained high proportions of condensed tannins produced little or no measurable semiquinone radicals (r = 0.61, P < 0.001; Fig. 3a). The correlation between oxidatively active tannins/ascorbate and semiquinone radical levels appears to improve the correlation because weakly active species are positioned near the origin (r = 0.68, P < 0.001; Fig. 3b). However, Q. alba remained an exception to the pattern presented by the other species. The correlation between the percentage of oxidatively active tannins and semiquinone radical levels fits Q. alba and most other species closely (r = 0.78, P < 0.001; Fig. 3c). It is noteworthy that negative correlations were observed between ascorbate and semiquinone radical levels (r = −0.62; P < 0.001) and between condensed tannins and semiquinone radical levels (r = −0.53; P < 0.001).
Correlations between the foliar chemistry of ingested tree leaves and semiquinone radical levels in the midgut contents of final-instar O. leucostigma. Pearson correlation coefficients were calculated with individual data (P < 0.001 for each correlation), but means, from each experiment, are plotted for clarity. Total phenolics is the sum of all individually measured phenolics. Filled square Q. rubra, open square Q. alba, filled diamond A. saccharum, open diamond A. rubrum, open circle C. glabra, filled circle O. virginiana, ex mark P. deltoides, asterisk P. tremuloides. Where not visible, data points are overlapping
Discussion
A major goal of this study was to examine the association between foliar chemistry (phenolics and ascorbic acid) and phenolic oxidation in the midgut contents of caterpillars. The percentage of the total phenolics composed of oxidatively active tannins (ellagitannins and galloyl glycosides) was strongly correlated with oxidative stress in O. leucostigma. A broad range of oxidative activities in tree species could be distinguished based on the levels of semiquinone radicals they produced in O. leucostigma: (1) species that produced a large percentage of oxidatively active tannins (Q. alba, A. rubrum, and A. saccharum), (2) intermediate species (C. glabra and Q. rubra), and (3) species that produced a small percentage of oxidatively active tannins (P. tremuloides, P. deltoides, and O. virginiana). Although only one insect species was examined, O. leucostigma is relatively phenolic tolerant (Barbehenn et al. 2005), and higher levels of oxidative stress would be expected in many less tannin-tolerant species.
As expected, ascorbate levels were negatively associated with phenolic oxidation in O. leucostigma: Low ascorbate levels were generally present in oxidatively active species, and high ascorbate levels were commonly present in species with low oxidative activities. Together with the analyses of foliar phenolics, these results generally support the hypothesis that the proportion of oxidatively active phenolics to ascorbate is associated with phenolic oxidation in caterpillars. However, Q. alba was an exception to this pattern. Q. alba produced higher levels of semiquinone radicals than expected, given its ellagitannin and ascorbate levels. One potential reason for this exception is the presence of highly reactive types of ellagitannins in Q. alba, including vescalagin, castalagin, cocciferin D2, and castavaloninic acid (Moilanen and Salminen 2008). These ellagitannins are based on an acyclic glucose, whereas the main ellagitannins of the other species in this study (i.e., Q. rubra, A. saccharum, and C. glabra) have glucopyranose backbones (Salminen, unpublished data). Secondly, Q. alba contained low levels of condensed tannins, both in absolute terms and relative to the levels of the oxidatively active phenolics present (Table 1; Fig. 1). Because condensed tannins can decrease the rate of oxidation of more reactive tannins (Barbehenn et al. 2006a), a low percentage of condensed tannins means that the more reactive tannins can oxidize near their maximum rates. This may explain the improved correlation between the percentage of oxidatively active phenolics and semiquinone radicals in O. leucostigma (Fig. 3c). The correlation in Fig. 3c is also a useful improvement over previous work, in which oxidative stress was associated with the proportion of total phenolics to ascorbic acid (Barbehenn et al. 2003b, 2005).
Tannins, especially ellagitannins and galloyl glycosides, were the primary sources of oxidative stress in the midgut contents of caterpillars in this study. This was due both to their intrinsic reactivity and to the high levels of tannins produced in oxidatively active leaves. Low MW phenolics were present in much lower levels than tannins in oxidatively active tree species, and many of these compounds have relatively low oxidative activities in the low-oxygen, high pH conditions of caterpillar midguts. Even in P. deltoides, which contained the highest levels of chlorogenic acid (1.5% DW), no measurable levels of semiquinone radicals were produced in the midguts of O. leucostigma. Although only one representative flavonoid and flavonoid glycoside was examined in this study, the low rates of browning of a wide range of these phenolics compared with ellagitannins support the generality of the EPR results on quercetin and rutin (Salminen, unpublished data). At sufficiently high levels, low MW phenolics could play other roles as chemical defenses in tree leaves. Unlike tannins, these phenolics can be absorbed from the midgut contents (Summers and Felton 1994; Barbehenn 2001). Therefore, low MW phenolics, and the quinones that they form, might produce oxidative stress and/or toxicity in insect tissues (Gant et al. 1988; Summers and Felton 1994; but see Johnson and Felton 2001). Other potential toxins in the trees in this study include alkaloids in species of Aceraceae, quinones in the Juglandaceae (e.g., C. glabra), and salicylates in the Salicaceae (Barbosa and Krischik 1987; Lindroth and Peterson 1988; Hemming and Lindroth 1995; Thiboldeaux et al. 1998).
Salicylates have often been called phenolic glycosides, and it has been suggested that products of their metabolism act as prooxidants in the midgut lumen (Clausen et al. 1989; Ruuhola et al. 2001). We were unable to detect the formation of measurable levels of semiquinone radicals in O. leucostigma that consumed the leaves of two salicylate-producing species, P. tremuloides and P. deltoides. In addition, salicortin produced no measurable semiquinone radicals and had a negligible rate of browning at pH 10 (Barbehenn, unpublished data). Previous work has noted the difficulty of detecting the oxidation of phenolics from P. tremuloides leaf tissues (Haruta et al. 2001), and work on the chemical fate of ingested salicylates from Salix species found little, if any, catechol (an oxidizable salicylate metabolite) in the frass of Operophtera brumata (Ruuhola et al. 2001). Together, these observations suggest that the chemical defenses of trees in the Salicaceae function most effectively as antifeedants and toxins in caterpillars.
We suspected that quinones in C. glabra, in combination with high levels of ascorbate, produced the unique EPR spectra observed in O. leucostigma. These spectra indicated the presence of both high levels of semiquinone and ascorbyl radicals (Table 4; Fig. 2). Mixtures of juglone and ascorbic acid produced EPR line shapes that were similar to those in O. leucostigma on C. glabra, suggesting that the chemical reduction of quinones by ascorbate could produce large concentrations of semiquinone and ascorbyl radicals simultaneously. Only a single tree of C. glabra was examined, greatly limiting the extent to which results on this species can be interpreted. It is noteworthy, therefore, that, similar “hybrid” EPR spectra were also observed when O. leucostigma larvae fed on another species of hickory (C. ovata) (Barbehenn, unpublished data). Foliar quinone levels were not measured in this study, and further work is needed to determine their potential impact on oxidative stress in caterpillars via redox cycling in the gut contents or tissues.
The phenolic composition of A. rubrum was unusual among the trees examined, containing large amounts of rhamnose-based galloyl glycosides (primarily digalloyl rhamnose). Previous work on A. rubrum foliar chemistry identified the presence of 1-O-galloyl-α-l-rhamnose (Abou-Zaid and Nozzolillo 1999), but found that the major phenolic component was ethyl m-digallate (Abou-Zaid et al. 2001). The complete absence of the latter compound in our analyses, and the presence of much higher levels of galloyl rhamnosides, lead us to question whether ethyl m-digallate could have been produced during the 2-day extraction period in ethanol that was used in previous studies. For example, methanol is known to cleave the m-depside bonds of digalloyl groups, producing methyl gallate (Hofmann and Gross 1990).
The results of this study suggest that trees such as O. virginiana, Populus species, and possibly Q. rubra might be superior host plants for generalist caterpillars. These tree species contain low levels of oxidatively active phenolics and produced little phenolic oxidation in the midgut lumen when ingested by O. leucostigma. Some field observations are consistent with this expectation: Q. rubra and O. virginiana are among the top host plant species for Lymantria dispar (Lymantriidae) (Liebhold et al. 1995), and Populus species are excellent host plants for Malacosoma disstria (Lasiocampidae) in its northern range (Parry and Goyer 2004). By comparison, A. saccharum and A. rubrum are relatively poor host plants for L. dispar and M. disstria (Liebhold et al. 1995; Parry and Goyer 2004). Given the high levels of oxidative stress in O. leucostigma on Q. alba in this study, it is surprising that Q. alba is considered a favored host for the closely related larvae of L. dispar. There is a large amount of geographical variation in the tannin compositions of oak species, including Q. alba (Salminen, unpublished data), suggesting that the suitability of species such as Q. alba as host plants could vary geographically.
We conclude that the tannin compositions of tree leaves largely determine the effectiveness of foliar phenolics as oxidative defenses against O. leucostigma. The basis for this conclusion includes the following main findings: (1) Oxidatively active tree leaves contained high proportions of ellagitannins or galloyl rhamnoses among their foliar phenolics; (2) low MW phenolics contributed relatively little to the overall oxidative activities of tree leaves compared with oxidatively active tannins; (3) ascorbic acid levels were lowest in the species that also contained the highest levels of oxidatively active tannins, potentially exacerbating phenolic oxidation in the gut lumen; and (4) even a “tannin-tolerant” caterpillar such as O. leucostigma was unable to control the oxidation of ingested phenolics when trees such as A. rubrum, A. saccharum, or Q. alba were eaten.
References
Abou-Zaid, M. M., and Nozzolillo, C. 1999. 1-O-galloyl-α-l-rhamnose from Acer rubrum. Phytochemistry 52:1629–1631.
Abou-Zaid, M. M., Helson, B. V., Nozzolillo, C., and Arnason, J. T. 2001. Ethyl m-digallate from red maple, Acer rubrum L., as the major resistance factor to forest tent caterpillar, Malacosoma disstria Hbn. J. Chem. Ecol. 27:2517–2527.
Baker, W. L. 1972. Eastern forest insects. USDA miscellaneous publication no. 1175. Washington, DC.
Barbehenn, R. V. 2001. Roles of peritrophic membranes in protecting herbivorous insects from ingested plant allelochemicals. Arch. Insect Biochem. Physiol. 47:86–99.
Barbehenn, R. V. 2003. Antioxidants in grasshoppers: higher levels defend the midgut tissues of a polyphagous species than a graminivorous species. J. Chem. Ecol. 29:683–702.
Barbehenn, R. V., Bumgarner, S. L., Roosen, E., and Martin, M. M. 2001. Antioxidant defenses in caterpillars: role of the ascorbate recycling system in the midgut lumen. J. Insect Physiol. 47:349–357.
Barbehenn, R. V., Walker, A. C., and Uddin, F. 2003a. Antioxidants in the midgut fluids of a tannin-tolerant and a tannin-sensitive caterpillar: effects of seasonal changes in tree leaves. J. Chem. Ecol. 29:1099–1116.
Barbehenn, R. V., Poopat, U., and Spencer, B. 2003b. Semiquinone and ascorbyl radicals in the gut fluids of caterpillars measured with EPR spectrometry. Insect Biochem. Mol. Biol. 33:125–130.
Barbehenn, R. V., Cheek, S., Gasperut, A., Lister, E., and Maben, R. 2005. Phenolic compounds in red oak and sugar maple leaves have prooxidant activities in the midguts of Malacosoma disstria and Orgyia leucostigma caterpillars. J. Chem. Ecol. 31:969–988.
Barbehenn, R. V., Jones, C. P., Karonen, M., and Salminen, J.-P. 2006a. Tannin composition affects the oxidative activities of tree leaves. J. Chem. Ecol. 32:2235–2251.
Barbehenn, R. V., Jones, C. P., Hagerman, A. E., Karonen, M., and Salminen, J.-P. 2006b. Ellagitannins have greater oxidative activities than gallotannins and condensed tannins at high pH: potential impact on caterpillars. J. Chem. Ecol. 32:2253–2267.
Barbosa, P., and Krischik, V. A. 1987. Influence of alkaloids on feeding preference of eastern deciduous forest trees by the gypsy moth Lymantria dispar. Amer. Nat. 130:53–69.
Bi, J. L., and Felton, G. W. 1995. Foliar oxidative stress and insect herbivory: primary compounds, secondary metabolites, and reactive oxygen species as components of induced resistance. J. Chem. Ecol. 21:1511–1530.
Bi, J. L., Murphy, J. B., and Felton, G. W. 1997. Antinutritive and oxidative components as mechanisms of induced resistance in cotton. J. Chem. Ecol. 23:97–117.
Clausen, T. P., Reichardt, P. B., Bryant, J. P., Werner, R. A., Post, K., and Frisby, K. 1989. Chemical model for short-term induction in quaking aspen (Populus tremuloides) foliage against herbivores. J. Chem. Ecol. 15:2335–2346.
Felton, G. W., and Duffey, S. S. 1992. Ascorbate oxidation reduction in Helicoverpa zea as a scavenging system against dietary oxidants. Arch. Insect Biochem. Physiol. 19:27–37.
Gant, T. W., Ramakrishna, R., Mason, R. P., and Cohen, G. M. 1988. Redox cycling and sulphydryl arylation; their relative importance in the mechanism of quinone cytotoxicity to isolated hepatocytes. Chem.-Biol. Interactions 65:157–173.
Haruta, M., Pedersen, J. A., and Constabel, P. 2001. Polyphenol oxidase and herbivore defense in trembling aspen (Populus tremuloides): cDNA cloning, expression, and potential substrates. Physiol. Plant 112:552–558.
Hemming, J. D. C., and Lindroth, R. L. 1995. Intraspecific variation in aspen phytochemistry: Effects on performance of gypsy moths and forest tent caterpillars. Oecologia 103:79–88.
Hofmann, A. S., and Gross, G. G. 1990. Biosynthesis of gallotannins: Formation of polygalloylglucoses by enzymatic acylation of 1,2,3,4,6-penta-O-galloylglucose. Arch. Biochem. Biophys. 283:530–532.
Johnson, K. S., and Felton, G. W. 2001. Plant phenolics as dietary antioxidants for herbivorous insects: A test with genetically modified tobacco. J. Chem. Ecol. 27:2579–2597.
Liebhold, A. M., Gottschalk, K. W., Muzika, R.-M., Montgomery, M. E., Young, R., O’Day, K., and Kelley, B. 1995. Suitability of North American tree species to the gypsy moth: a summary of field and laboratory tests. USDA Forest Service, General Technical Report NE-211.
Lindroth, R. L., and Peterson, S. S. 1988. Effects of plant phenols on performance of southern armyworm larvae. Oecologia 75:185–189.
Lindroth, R. L., Hsia, M. T. S., and Scriber, J. M. 1987. Seasonal patterns in the phytochemistry of three Populus species. Biochem. Syst. Ecol. 15:681–686.
Martin, J. S., Martin, M. M., and Bernays, E. A. 1987. Failure of tannic acid to inhibit digestion or reduce digestibility of plant protein in gut fluids of insect herbivores: implications for theories of plant defense. J. Chem. Ecol. 13:605–621.
Moilanen, J., and Salminen, J.-P. 2008. Ecologically neglected tannins and their biologically relevant activity: chemical structures of plant ellagitannins reveal their in vitro oxidative activity at high pH. Chemoecology, (in press).
Ossipova, S., Ossipov, V., Haukioja, E., Loponen, J., and Pihlaja, K. 2001. Proanthocyanidins of mountain birch leaves: quantification and properties. Phytochem. Anal. 12:128–133.
Parry, D., and Goyer, R. A. 2004. Variation in the suitability of host tree species for geographically discrete populations of forest tent caterpillar. Environ. Entomol. 33:1477–1487.
Rosen, G. M., Britigan, B. E., Halpern, H. J., and Pou, S. 1999. Free Radicals: Biology and Detection by Spin Trapping. Oxford University Press, NY.
Ruuhola, T., Tikkanen, O.-P., and Tahvanainen, J. 2001. Differences in host use efficiency of larvae of a generalist moth, Operophtera brumata on three chemically divergent Salix species. J. Chem. Ecol. 27:1595–1615.
Salminen, J.-P., Ossipov, V., Loponen, J., Haukioja, E., and Pihlaja, K. 1999. Characterisation of hydrolyzable tannins from leaves of Betula pubescens by high-performance liquid chromatography-mass spectrometry. J. Chrom. A. 864:283–291.
Sas institute 2003. The SAS system for Windows. Version 9.1. SAS Institute, Cary, NC, USA.
Summers, C. B., and Felton, G. W. 1994. Prooxidant effects of phenolic acids on the generalist herbivore Helicoverpa zea (Lepidoptera: Noctuidae): Potential mode of action for phenolic compounds in plant anti-herbivore chemistry. Insect Biochem. Mol. Biol. 24:943–953.
Thiboldeaux, R. L., Lindroth, R. L., and Tracy, J. W. 1998. Effects of juglone (5-hydroxy-1,4-naphthoquinone) on midgut morphology and glutathione status in Saturniid moth larvae. Comp. Biochem. Physiol. 120:481–487.
Wagner, D. M. 2005. Caterpillars of Eastern North America. Princeton University Press.
Wilkinson, L. 2000. SYSTAT: The system for statistics. SYSTAT, Inc., Evanston, IL.
Acknowledgments
This work was supported by NRI-CSREES-USDA grants 2004-35302-14940 (to RVB) and 2007-35302-17803 (to RVB and J-PS). Additional support for J-P Salminen was provided by grant 119659 from the Academy of Finland. We thank Rick Lindroth for providing salicortin and KimHang Dinh and Rasika Ranganathan for measuring ascorbic acid.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Barbehenn, R., Weir, Q. & Salminen, JP. Oxidation of Ingested Phenolics in the Tree-Feeding Caterpillar Orgyia leucostigma Depends on Foliar Chemical Composition. J Chem Ecol 34, 748–756 (2008). https://doi.org/10.1007/s10886-008-9478-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-008-9478-3