Abstract
The Complior device (Alam Medical, France) has contributed to the rise of arterial stiffness as a measure of cardiovascular risk. In its latest version (Complior Analyse) the sensor records pressure instead of distension waveforms thus allowing the measurement of central pressure and pulse wave analysis. The aim of our study was to verify that the new sensor measures pressure waveforms accurately in both time and frequency domain. Invasive and non-invasive signals were recorded simultaneously at the radial artery and compared in the frequency and time domain in haemodynamically stable intensive care unit patients. Twelve patients entered the study (8 men, 4 women, mean age 69 ± 17 years). Heart rate was 90 ± 15 bpm, systolic blood pressure 133 ± 19 mmHg and diastolic blood pressure 68 ± 15 mmHg. There was no statistical difference in the amplitude of harmonics between the invasive signal and Complior signal. When superimposing waveforms in the time domain, there was a small difference in the form factor (4.2 ± 2.8 %) and in the absolute area between the 2 waveforms (3.3 ± 1.7 mmHg·s−1). These differences were of the same magnitude as the beat-to-beat variation of the form factor (3.3 %) and of the absolute area (3.1 mmHg·s−1), respectively. The second systolic peak was detectable in 4 subjects, with no statistical difference between invasive and non-invasive values. The new pressure sensor of the Complior Analyse device recorded pressure waveforms accurately and could be used to perform pressure wave analysis.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Non-invasive central pressure (cBP) assessment is receiving more and more attention as several studies have shown that cBP is better related to cardiovascular surrogate end-points than conventional peripheral pressure, and is a better predictor of cardiovascular events and mortality [1–5]. Drug intervention studies have shown it is possible to observe differential effects on cBP values despite similar peripheral pressure reduction [6–8]. There are several methods to assess cBP non-invasively. One can use a peripheral waveform (radial or brachial) and apply a mathematical transformation called a transfer function. This technique is used by commercial devices such as SphygmoCor (AtCor, Australia) [9], Mobil-o-graph (IEM, Germany) [10], B-Pro and CASP devices (Healthstats, Singapore) [11], Vicorder (Skidmore medical, UK) [12] and cBP301 (Centron Diagnostics, UK) [13]. A second method is based on the observation that the second systolic peak (SBP2) of the peripheral waveform is nearly identical to the systolic cBP value [14–17]. This method is used by the commercial device HEM-9000AI (Omron, Japan), Vasotens (BPlab, Russia) and Arteriograph (Tensiomed, Hunary).
Despite this large number of devices, the superiority of cBP compared to brachial cuff to predict cardiovascular risk has been established mainly from studies using carotidograms with no use of a transfer function nor detection of SBP2 [2–4]. The carotid artery is geographically and functionally close to the ascending aorta and studies have shown that pressure waveforms measured at the carotid level were very similar to ascending aortic pressure [18, 19].
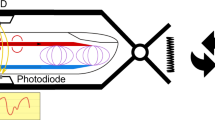
In parallel to the rise of new non-invasive techniques of cBP measurement aortic stiffness, and more specifically its gold standard measure carotid-femoral pulse wave velocity (cf-PWV) [20, 21], has been extensively studied. Following numerous epidemiological studies showing the predictive value of cf-PWV [20, 22], cf-PWV has been introduced in the ESH-ESC guidelines for the management of arterial hypertension [23]. It is a measure of organ damage and is recommended for the diagnostic and follow-up of hypertensive patients [22, 23]. All epidemiological studies of cf-PWV have been performed with simultaneous measurements of the carotid and femoral signals to calculate cf-PWV. While some of these studies have used non-commercial equipment, a great number have used the Complior device (Alam Medical, France) either in its Complior II version or in its Complior SP version. With these versions, carotid and femoral distension waveforms were recorded simultaneously and then a validated “foot-to-foot” algorithm based on the second derivative was used to determine transit time and hence pulse wave velocity [24–26]. In its latest version, the Complior device, now called Complior Analyse, offers the possibility to use the internationally recommended “intersecting tangent” algorithm to detect the foot of the waveforms. Complior Analyse also records non-invasive pressure waveforms directly instead of distension waveforms. Hence it allows measuring and analysing carotid pressure waveforms during standard measure of cf-PWV (Fig. 1).
The aim of this study was to confirm that the new sensor of the Complior Analyse device measures pressure waveforms accurately. It was judged unethical to measure simultaneous invasive and non-invasive pressure waveforms at the carotid artery level. As Complior sensors can be used at any arterial points where the pressure pulse can be manually felt, radial signals were studied assuming that results on the comparison of radial waveforms could be extrapolated to any pressure waveforms. As blood pressure (BP) and hence pressure waveforms may differ significantly between left and right arm, we compared signals measured simultaneously at the same radial location.
2 Methods
This is a prospective monocentric study (Antoine Béclère Hospital, Clamart, France). The study began in July 2011 and ended in December 2011. All haemodynamically stable intensive care unit (ICU) patients were screened. They were eligible in the present study if an arterial radial line had been inserted for clinical reasons (Seldicath 3F radial fluid filled catheter; Plastimed, Le Plessis Bouchard, France). The study was approved by ethics comity of the French Society of Intensive Care Medicine (“Société de Réanimation de Langue Française”, agreement n° 12-376) and all patients gave informed consent to participate. Patients with frequent premature ventricular beats or movement artefacts were excluded from the study. The Complior sensor was positioned with its specific hand-free holder above the invasive point of measurement, i.e., circa 8 cm proximally from the radial puncture point (Fig. 1d). It must be noted that this is not the standard positioning of the Complior sensor which is normally positioned above the radius head (Fig. 1b). However, this is the puncture point where the catheter sheath is present so it was not possible to position the non-invasive sensor at this location. When positioned above the invasive point of measurement, there are more underlying tissues between the Complior sensor and the artery and hence this may reduce the quality of the recorded signal. However, in our opinion, this may be viewed as a reasonable compromise in patients equipped with an arterial indwelling catheter while putting more constraints on the quality of the sensor. Complior sensors are piezo-electric sensors which record pressure waveforms. The natural frequency of the overall system (sensor + electronic) is 24 Hz and the damping coefficient is 0.27.
Just prior to recordings, the invasive line was flushed to avoid any possible damping of the signal. The analog signal of the invasive catheter and the signal from the Complior sensor were recorded simultaneously via a PC-acquisition unit, sampled at 1,000 Hz and stored for later off-line analysis with Matlab (Mathworks, USA).
Invasive and Complior sensor signals were analyzed independently by an operator unaware of recording procedure and patient status. As least five consecutive heart beats of simultaneous invasive and Complior signals per patient were selected for further analysis. The selected pulses had to be free of ectopic beats or movement artefact. No additional off-line filtering was performed. The foot of each pulse was automatically detected by the “intersecting tangent algorithm” as described by Chui et al. and recommended by the arterial stiffness collaboration group [27, 28]. Average of invasive and Complior pulses were calculated for each subject by calibrating pulses duration to patient’s heart rate and pulse amplitude to identical value.
The Complior sensor records relative pressure. Like any tonometric signal, the off-set and amplitude of the signal depend on signal strength as well as on thickness of underlying tissues and hold-on pressure. Complior waveforms were calibrated with invasive systolic and diastolic BP.
Amongst the 16 subjects entering the study, four subjects were excluded due to poor quality signal (50 Hz noise or analog to digital amplifier saturation) such that 12 subjects completed the final analysis. In order to verify wave shape agreement, Complior and invasive signals were superimposed in the time domain and compared in the Fourier domain. In the time domain, the mean error between the 2 curves was calculated as the absolute area between the 2 averaged waveforms and their form factor was calculated [(mean BP − diastolic BP)/pulse pressure × 100] [29, 30]. When possible, the 2nd systolic shoulder (SBP2) was also detected and the radial augmentation index (AIx) was derived as Aix = (SBP2 − DBP)/(SBP − DBP). As information on pressure waveform is only on the first harmonics, we performed frequency analysis on the first 8 harmonics.
2.1 Statistical Analyses
Data are given as mean ± SD. Non parametric Wilcoxon test was used to compare harmonics’ moduli and form factors even when normality test passed. Differences between invasive and Complior variables were expressed as mean difference ± SD as recommended in Bland and Altman analysis. Statistical significance was declared when p < 0.001.
3 Results
Characteristics of the 12 subjects entering the analysis are presented in Table 1. BP values are from the invasive recording as no brachial cuff BP was taken. Reasons for ICU admittance were acute respiratory distress syndrome (n = 5), septic shock (n = 3), post operative shock (n = 1), left ventricular insufficiency (n = 1), coma (n = 1) and intentional self overdose (n = 1). Four patients were given noradrenaline or isoprenaline at the time of measurement and six were intubated.
Figure 2 shows the average invasive and Complior waveforms recorded in one of the subjects. Potential differences in the wave shape between invasive and non-invasive pressure were studied in the frequency-domain and in the time-domain and presented in Table 2. Figure 3 presents the frequency analysis of the invasive and Complior signals. There was no statistical difference between the moduli of both signals.
In the time-domain, the absolute area between the averaged invasive and Complior signals was 3.3 ± 1.7 mmHg·s−1. In order to evaluate the significance of this value, we calculated the absolute mean area among pulses from invasive and Complior signals. The within-subject absolute area difference between invasive pulses from the same subject was 1.8 ± 1.9 mmHg·s−1 and 3.1 ± 2.0 mmHg·s−1 for Complior Analyse signals. The wave shape difference between invasive and Complior traces was hence of the same order of magnitude as intra-signal wave shape variability. The Bland–Altman difference between the invasive and Complior Form factor was 4.2 ± 2.8 % (invasive Form factor = 35.6 ± 5.2 % and Complior form factor = 39.9 ± 6.0 %, p < 0.001) (Table 2; Fig. 4). The beat-to-beat form factor standard deviation was 3.3 % for Complior signal and 1.6 % for invasive signal. Hence, after the systolic peak, Complior waveforms were slightly above invasive waveforms as shown in Fig. 2. This trend was of the same order of magnitude as the beat to beat variation of the form factor.
SBP2 and hence AIx were not detectable in 8 out of the 12 subjects on both invasive and non-invasive tracings. In the remaining 4 subjects there was no statistical difference between invasive and non-invasive SBP2 and Aix values (Table 2). The sample was too small to perform a Bland–Altman analysis on SBP2 and AIx.
4 Discussion
Non-invasive tonometric pressure-recording commercial devices must be validated against invasive signals. The Millar tonometer (Millar instruments, USA) used by SphygmoCor (AtCor, Australia) was tested in 1989 by Kelly et al., against invasive femoral signals in dogs and against invasive radial signals in 62 patients undergoing radial cannulation for clinical monitoring purposes with short (<30 cm) fluid-filled lines [31]. Signals were compared in the frequency domain only. Small differences were observed as the Millar tonometer tends to overestimate the 1st harmonic (p < 0.02) and underestimate moduli above the 4th harmonics [31].
Likewise, Pulse Pen’s tonometer (Diatecne, Italy) was compared in the frequency domain in patient undergoing coronary angiography [32]. Invasive signals were recorded at the origin of the ascending aorta and at the initial tract of the carotid artery using fluid-filled catheters, while non invasive waveforms were recorded at the common carotid artery and at the base of the neck (bottom carotid) and calibrated to brachial non-invasive BP. The authors observed no statistical differences between the frequency analysis of the invasive aortic/carotid bifurcation pressure and the non-invasive bottom carotid tonometry (comparison with the least geographical difference). When comparing invasive ascending aorta with non-invasive common carotid signals, a slightly bigger, but still not significant, difference in their frequency analysis was found [32]. These results confirm previous ones showing that differences between carotid and aortic signals are negligible [18, 19] and that carotid waveforms can be used as direct measure of cBP waveforms.
The results from the new Complior sensors showed no differences in the frequency domain with simultaneous invasive signal recorded at the same location, confirming at least the same accuracy as the 2 above mentioned commercial devices.
Despite the validation of SphygmoCor and PulsePen signals, a recent publication has shown that the form factor of pressure signals recorded with SphygmoCor and Pulse Pen were significantly different with a significant impact on the value of SBP2 [33]. For this reason, we studied waveforms not only in the frequency domain but also in the time domain where their superimposition allows a more sensitive comparison.
There was a small but significant difference in the form factor between the invasive and non-invasive signal (fig. 4). We did not perform a reproducibility study but this difference is of the same order as the beat-to-beat form factor variability. The error calculated from the area between the two curves was considered negligible (3.3 ± 1.7 mmHg·s−1) as the error between successive pulses was of the same magnitude (3.1 ± 2.0 mmHg·s−1 for the area between successive pulses). As many of our ICU patients had significant peripheral vasodilation, SBP2 was detectable in only 4 out of the 12 studied subjects. Among the remaining subjects, there was no difference in SBP2 from the invasive and Complior signal.
5 Limitation of the study
With the Complior Analyse device, central pulse wave analysis is performed at the carotid artery level. For ethical reasons it is not possible to compare simultaneously invasive and non-invasive common carotid pressure waveforms. An alternative would be to use invasive ascending aortic signals versus non-invasive carotid signals. However if a discrepancy occurs it would not be possible to conclude as to its origin between physiological wave shape differences and sensor sensitivity. The Complior sensor was applied above the catheter tip, at the middle of the forearm where there are more underlying tissues and where applanation against a bony surface is more difficult. Nevertheless, we demonstrated the accuracy of the sensor using radial signals in patients with a large spectrum of wave shape, heart rate and BP values (Table 1). Hence we feel that our setting was a good compromise and that results could be transposed to any pressure location.
The second limitation of our study is the relatively small number of subjects. However as mentioned previously, we had a heterogeneous group of patients from the ICU exhibiting a wide range of wave shape morphology. Having a small group of patients only increases the possibility of finding a difference by chance, as a discrepancy for one type of subject will have a higher weighting in a smaller cohort. Hence we feel confident that increasing the number of subjects would not change the conclusion of our study.
While we showed data regarding the beat to beat reproducibility, we have not documented the reproducibility of pressure recordings. Further studies should be performed in standard clinical settings to assess the reproducibility of pressure recordings and pressure analysis results.
In conclusion, perfect agreement between invasive and Complior Analyse pressure waves was observed in the frequency domain. In the time domain, there were small differences in the form factor or area between the two curves. However these differences were of the same order of magnitude as beat to beat variations. Hence, the new pressure sensor of the Complior Analyse device records pressure waveforms accurately and could be used to perform pressure wave analysis.
References
Vlachopoulos C, Aznaouridis K, O’Rourke MF, Safar ME, Baou K, Stefanadis C. Prediction of cardiovascular events and all-cause mortality with central haemodynamics: a systematic review and meta-analysis. Eur Heart J. 2010;31:1865–71.
Safar ME, Blacher J, Pannier B, Guerin AP, Marchais SJ, Guyonvarc’h PM, et al. Central pulse pressure and mortality in end-stage renal disease. Hypertension. 2002;39:735–8.
Pini R, Cavallini MC, Palmieri V, Marchionni N, Di BM, Devereux RB, et al. Central but not brachial blood pressure predicts cardiovascular events in an unselected geriatric population: the ICARe Dicomano Study. J Am Coll Cardiol. 2008;51:2432–9.
Wang KL, Cheng HM, Chuang SY, Spurgeon HA, Ting CT, Lakatta EG, et al. Central or peripheral systolic or pulse pressure: which best relates to target organs and future mortality? J Hypertens. 2009;27:461–7.
Roman MJ, Devereux RB, Kizer JR, Lee ET, Galloway JM, Ali T, et al. Central pressure more strongly relates to vascular disease and outcome than does brachial pressure: the Strong Heart Study. Hypertension. 2007;50:197–203.
Williams B, Lacy PS, Thom SM, Cruickshank K, Stanton A, Collier D, et al. Differential impact of blood pressure-lowering drugs on central aortic pressure and clinical outcomes: principal results of the Conduit Artery Function Evaluation (CAFE) study. Circulation. 2006;113:1213–25.
Boutouyrie P, Achouba A, Trunet P, Laurent S. Amlodipine-valsartan combination decreases central systolic blood pressure more effectively than the amlodipine-atenolol combination. the EXPLOR Study. Hypertension. 2010;55:1314–22.
Matsui Y, Eguchi K, O’Rourke MF, Ishikawa J, Shimada K, Kario K. Association between aldosterone induced by antihypertensive medication and arterial stiffness reduction: the J-CORE study. Atherosclerosis. 2011;215:184–8.
Pauca AL, O’Rourke MF, Kon ND. Prospective evaluation of a method for estimating ascending aortic pressure from the radial artery pressure waveform. Hypertension. 2001;38:932–7.
Weber T, Wassertheurer S, Rammer M, Maurer E, Hametner B, Mayer CC, et al. Validation of a brachial cuff-based method for estimating central systolic blood pressure. Hypertension. 2011;58:825–32.
Williams B, Lacy PS, Yan P, Hwee CN, Liang C, Ting CM. Development and validation of a novel method to derive central aortic systolic pressure from the radial pressure waveform using an N-point moving average method. J Am Coll Cardiol. 2011;57:951–61.
Pucci G, Cheriyan J, Hubsch A, Hickson SS, Gajendragadkar PR, Watson T, et al. Evaluation of the Vicorder, a novel cuff-based device for the noninvasive estimation of central blood pressure. J Hypertens. 2013;31:77–85.
Brett SE, Guilcher A, Clapp B, Chowienczyk P. Estimating central systolic blood pressure during oscillometric determination of blood pressure: proof of concept and validation by comparison with intra-aortic pressure recording and arterial tonometry. Blood Press Monit. 2012;17:132–6.
Pauca AL, Kon ND, O’Rourke MF. The second peak of the radial artery pressure wave represents aortic systolic pressure in hypertensive and elderly patients. Br J Anaesth. 2004;92:651–7.
Guilcher A, Brett S, Munir S, Clapp B, Chowienczyk PJ. Estimating central SBP from the peripheral pulse: influence of waveform analysis and calibration error. J Hypertens. 2011;29:1357–66.
Takazawa K, Kobayashi H, Kojima I, Aizawa A, Kinoh M, Sugo Y, et al. Estimation of central aortic systolic pressure using late systolic inflection of radial artery pulse and its application to vasodilator therapy. J Hypertens. 2012;30:908–16.
Lin MM, Cheng HM, Sung SH, Liao CF, Chen YH, Huang PH, et al. Estimation of central aortic systolic pressure from the second systolic peak of the peripheral upper limb pulse depends on central aortic pressure waveform morphology. J Hypertens. 2012;30:581–6.
Kelly RP, Karamanoglu M, Gibbs H, Avolio AP, O’Rourke MF. Noninvasive carotid pressure wave registration as an indicator of ascending aortic pressure. J Vasc Med Biol. 1989;1:241–7.
Chen CH, Ting CT, Nussbacher A, Nevo E, Kass DA, Pak P, et al. Validation of carotid artery tonometry as a means of estimating augmentation index of ascending aortic pressure. Hypertension. 1996;27:168–75.
Laurent S, Cockcroft J, Van BL, Boutouyrie P, Giannattasio C, Hayoz D, et al. Expert consensus document on arterial stiffness: methodological issues and clinical applications. Eur Heart J. 2006;27:2588–605.
Van Bortel LM, Laurent S, Boutouyrie P, Chowienczyk P, Cruickshank JK, De BT, et al. Expert consensus document on the measurement of aortic stiffness in daily practice using carotid-femoral pulse wave velocity. J Hypertens. 2012;30:445–8.
Vlachopoulos C, Aznaouridis K, Stefanadis C. Prediction of cardiovascular events and all-cause mortality with arterial stiffness: a systematic review and meta-analysis. J Am Coll Cardiol. 2010;55:1318–27.
Mancia G, De BG, Dominiczak A, Cifkova R, Fagard R, Germano G, et al. 2007 Guidelines for the management of arterial hypertension: the task force for the management of arterial hypertension of the European Society of Hypertension (ESH) and of the European Society of Cardiology (ESC). J Hypertens. 2007;25:1105–87.
Asmar R, Benetos A, Topouchian J, Laurent P, Pannier B, Brisac A-M, et al. Assessment of arterial distensibility by automatic pulse wave velocity measurement: validation and clinical application studies. Hypertension. 1995;26:485–90.
Asmar R, Topouchian J, Pannier B, Benetos A, Safar M. Pulse wave velocity as endpoint in large-scale intervention trial. the Complior study. Scientific, Quality Control, Coordination and Investigation Committees of the Complior Study. J Hypertens. 2001;19:813–8.
Pereira T, Maldonado J. Comparative study of two generations of the Complior device for aortic pulse wave velocity measurements. Blood Press Monit. 2010;15:316–21.
Chiu YC, Arand PW, Shroff SG, Feldman T, Carroll JD. Determination of pulse wave velocities with computerized algorithms. Am Heart J. 1991;121:1460–70.
Arterial Stiffness Collaboration group. Determinants of pulse wave velocity in healthy people and in the presence of cardiovascular risk factors: ‘establishing normal and reference values’. Eur Heart J. 2010.
Kips J, Vanmolkot F, Mahieu D, Vermeersch S, Fabry I, de HJ, et al. The use of diameter distension waveforms as an alternative for tonometric pressure to assess carotid blood pressure. Physiol Meas. 2010;31:543–53.
Chemla D, Hebert JL, Aptecar E, Mazoit JX, Zamani K, Frank R, et al. Empirical estimates of mean aortic pressure: advantages, drawbacks and implications for pressure redundancy. Clin Sci (Lond). 2002;103:7–13.
Kelly RP, Hayward C, Ganis J, Daley J, Avolio A, O’Rourke MF. Noninvasive registration of the arterial pressure pulse waveform; using high-fidelity applanation tonometry. J Vasc Med Biol. 1989;1:142–9.
Salvi P, Lio G, Labat C, Ricci E, Pannier B, Benetos A. Validation of a new non-invasive portable tonometer for determining arterial pressure wave and pulse wave velocity: the PulsePen device. J Hypertens. 2004;22:2285–93.
Zhang Y, Agnoletti D, Protogerou AD, Wang JG, Topouchian J, Salvi P, et al. Radial late-SBP as a surrogate for central SBP. J Hypertens. 2011;29:676–681.
Conflict of interest
Sandrine Millasseau works as a freelance specialist on pulse wave analysis and receives revenues from several medical companies including AtCor, Omron (both cited in this article) and Alam Medical whose sensors have been validated in this study. The other authors have no conflict of interest to declare in the subject matter. Alam Medical freely loaned a Complior Analyse device for the duration of the study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sztrymf, B., Jacobs, F., Chemla, D. et al. Validation of the new Complior sensor to record pressure signals non-invasively. J Clin Monit Comput 27, 613–619 (2013). https://doi.org/10.1007/s10877-013-9477-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10877-013-9477-y