Abstract
We examined the role of macrophage migration inhibitory factor (MIF) in the generation of the Th2 response using MIF-deficient mice in a model of epicutaneous sensitization to ovalbumin. Lymph node cells from sensitized MIF-deficient mice produce lower levels of Th2 cytokines after antigen challenge when compared to their wild-type counterparts. Sensitized mice lacking MIF show less pulmonary inflammation after intranasal antigen exposure. Mice deficient in CD74, the MIF receptor, also are unable to generate an inflammatory response to epicutaneous sensitization. Examination of the elicitation phase of the atopic response using DO11.10 OVA TCR transgenic animals shows that T cell proliferation and IL-2 production are strongly impaired in MIF-deficient T cells. This defect is most profound when both T cells and antigen-presenting cells are lacking MIF. These data suggest that MIF is crucial both for the sensitization and the elicitation phases of a Th2-type immune response in allergic disease.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Atopic diseases (asthma, allergic rhinitis, and atopic dermatitis) are being increasingly recognized among populations in the developing and developed world and lead to a high degree of morbidity and even mortality. The pathogenesis of these diseases, which is incompletely understood, involves the activation of the Th2 subset of CD4 T cells. A specific profile of cytokines (e.g., interleukin (IL)-4, IL-5, and IL-13) also characterizes the acute, Th2-type inflammatory response [1–6]. IL-4 and IL-13 promote the production of IgE by class switching, upregulate major histocompatibility complex (MHC) class II molecules on macrophages, induce VCAM expression by endothelial cells, and suppress Th1 inflammatory cytokine production [7–12]. The receptors for IL-4 and IL-13 share the IL-4 receptor α-chain, which likely accounts for their similar effects on a number of cellular processes [13–15]. However, notable differences in the functions of IL-4 and IL-13 exist. For example, IL-4, but not IL-13, directs naive T cells to differentiate into Th2-type effector cells [16, 17]. Moreover, IL-13-deficient mice are impaired in the generation of an epicutaneously induced Th2 immune response, while this response is intact in IL-4-deficient mice [18, 19]. IL-5 induces eosinophil differentiation, proliferation, activation, and survival [20–24]. Cells producing these Th2 cytokines are found in abundance at sites of acute allergic inflammation and are crucial for the development of the inflammatory response.
IL-4 is considered to promote Th2 differentiation by enhancing the expression of the transcription factor GATA3, which promotes cellular proliferation, cytokine production, and inhibition of the Th1 response [25, 26]. Additionally, Th2 differentiation is influenced by the presence of regulatory T cells (Tregs), the deficiency of which leads to a syndrome characterized by heightened allergic responses and autoimmunity [27]. The transcription factor Foxp3 is expressed by Treg cells and has been implicated in driving the cells into the regulatory lineage, while suppressing other factors which promote cytokine production in effector T cells [28]. The presence of Tregs has been negatively correlated with the development of Th2 responses in both mouse models and human allergic disease [27, 29].
Macrophage migration inhibitory factor (MIF) initially was described 40 years ago as an activity elaborated by mitogen-activated lymphocytes that inhibited the random migration of macrophages [30, 31]. Since that time, MIF has been cloned and an array of pro-inflammatory, anti-apoptotic, and proliferative functions have been ascribed to this protein. It induces a sustained extracellular signal-regulated kinase (ERK 1/2) phosphorylation [32], upregulates toll-like receptor (TLR)-4 expression [33], and suppresses p53-mediated apoptosis [34]. MIF’s proliferative and anti-apoptotic functions are believed to sustain inflammation in environments where activation-induced apoptosis may result in the attrition of immune cells [35]. MIF also is produced by many non-immune cells in response to diverse stimuli [36, 37].
MIF is essential for T cell activation and the development of adaptive immune responses. Anti-MIF antibodies inhibit T cell proliferation and IL-2 production in vitro and suppress antigen driven T cell activation and antibody production in vivo [38]. In a mixed lymphocyte reaction (MLR), the T cell proliferative response to an alloantigen is decreased in lymphocytes derived from mif −/− (MIF-KO) mice when compared with lymphocytes from wild-type (WT) mice [39]. In two classic models of Th1-biased immune responses, contact hypersensitivity to the topically applied hapten, trinitrochlorobenzene (TNCB), and the delayed-type hypersensitivity reaction to tuberculin antigen, MIF-KO show attenuated inflammation [39, 40]. Importantly, MIF-KO mice and mice treated with an anti-MIF antibody display impaired immune responses in both the afferent and efferent arms of the contact hypersensitivity response [39].
In addition to its role in promoting Th1-type inflammation, a variety of experimental evidence suggests an important role for MIF in the development of allergic, Th2-type immune responses. Th2 T cell clones stimulated with the mitogen concanavalin A release MIF, while Th1 clones do not [38]. In murine models, MIF is required for the maximal development of allergic rhinitis and an asthma-like condition induced by ovalbumin (OVA) priming and inhalation challenge [41, 42]. Furthermore, MIF deficiency attenuates the generation of IgE and IgG1 antibodies in the murine asthma model [42, 43]. Recently, the role of MIF also has been implicated in a model of atopic dermatitis (AD) induced by epicutaneous (EC) sensitization with OVA [44]. High expression polymorphisms in the promoter of the human MIF gene are significantly associated with asthma severity and the development of atopic dermatitis [42, 45, 46]. MIF expression is elevated in the skin and serum of patients with atopic dermatitis [47–49] and in the sputum, serum, and bronchoalveolar lavage (BAL) fluid from patients with asthma [50, 51]. Human eosinophils also produce MIF in a time- and concentration-dependent fashion in response to an activating stimulus [50]. Finally, inhibition of MIF by an antibody or a small molecule inhibitor leads to reduced airway inflammation and remodeling in an asthma-hyperresponsiveness model [52, 53].
MIF is known to be important for OVA-induced Th2 lung inflammation when the sensitization phase of the immune response involves intraperitoneal (IP) administration of OVA with an adjuvant [42]. In this report, we use EC sensitization and adoptive transfer of in vitro sensitized T cells to examine more precisely the role of MIF in the generation of a Th2 immune response. These techniques offer several advantages to systemic sensitization with OVA. They allow the role of MIF in the Th2 immune responses of both the skin and lung to be studied; they offer an opportunity for the relative contribution of MIF to the sensitization and elicitation to be studied more precisely; and the use of EC sensitization without co-administration of an adjuvant represents a considerably more physiologic model system than systemic immunization with adjuvant [54].
Materials and Methods
Animals
BALB/c mice were purchased from Charles River Laboratories. MIF-KO were backcrossed onto the BALB/c background at the Yale Animal Resources Center and were used at generation N10 [55]. Cd74 −/− (CD74-KO) mice in the BALB/c background (N10) were obtained from the Weizmann Institute (Rehovot, Israel) and subsequently maintained at the Yale Animal Resources Center [56]. BALB/c mice transgenic for the T cell receptor (TCR) recognizing OVA residues 323–339 (DO11.10H2dTg) were provided by Lauren Cohn (Yale University, New Haven, CT, USA). DO11.10H2d mice on the MIF-KO (BALB/c) background were prepared by crossing the mice from each of the two strains. All mice were bred and maintained on OVA-free diets in a pathogen-free environment at the Yale Animal Resources Center. Age-matched female mice, 8–12 weeks old, were used in all experiments.
Epicutaneous Sensitization and Lymph Node Cytokine Production
The EC sensitization protocol, described previously [18], entailed application of OVA (100 μg, Grade V; Sigma Chemical Co.) to the shaved back of mice in 75 μl of PBS on day 0. Skin-draining axillary lymph nodes (LN) from EC-sensitized mice were isolated on day 4 after initial exposure. Cells were cultured at 4 × 106 cells/ml with an equal number of syngeneic, mitomycin-treated, T cell-depleted splenocytes (prepared as previously described, [18]) and OVA (100 μg/ml) in Bruff’s/10% FCS at 37°C for 48 h, in the presence or absence of IL-2 (20 ng/ml, R&D Systems) [57]. This 1:1 ratio of antigen-presenting cells (APCs) to T cells has been described in prior studies of MLRs [18, 43]. In experiments with bone marrow-derived macrophages, LN cells (4 × 106 cells/ml) were cultured with an equal number of syngeneic T-depleted macrophages. CD4+ T cells were prepared by negative selection, as described previously [58], using monoclonal antibodies (Abs) to CD8, class II MHC I-Ad (clone 212.A1), FcR, B cell precursors (clone TIB164), CD49b (pan NK cell, BD Biosciences), and anti-Ig-coated magnetic beads (Advanced Magnetics, Inc.). Cytokines in the culture supernatants: IL-4, IL-5, IL-13, and interferon-gamma (IFN-γ) were detected with commercially available ELISA kits (IL-4, IL-5, and IFN-γ, ΒD Biosciences, and IL-13, R&D Systems).
Lymph node cultures that were used for flow cytometric analysis were treated with Golgi Plug (BD Biosciences) 6 h prior to analysis. Cells were harvested and washed in PBS containing 1% BSA. After treating with Fc Block (BD Biosciences), staining for surface markers was performed using PerCP-conjugated anti-CD4 and APC-conjugated anti-CD25 (eBioscience). Cells then were fixed and permeabilized using the BD cytofix/cytoperm kit according to the manufacturer’s instructions (BD Biosciences), and staining for intracellular cytokines and transcription factors was performed using PE-conjugated anti-IL-4, APC-conjugated IFN-γ, and PE-conjugated Foxp3 (eBioscience). Flow cytometry was performed using a BD FACSCalibur instrument, and data were analyzed using the FlowJo software.
qRT-PCR for Transcription Factors
Non-adherent cells from the lymph node cultures described above were collected after 48 h. Cells were treated with RLT buffer, and RNA isolated using RNeasy mini kit (QIAGEN) according to the manufacturer’s protocol. Complementary DNA was reverse-transcribed using 500 ng of RNA with the QuantiTect kit (QIAGEN). The template complementary DNA was used with 0.5 μM of forward and reverse primers for murine β-actin, Tbet (TBX-21), Gata3, and Foxp3 (QIAGEN) and SYBR Green PCR Mix (BIO-RAD) according to the manufacturer’s instructions. Reactions were carried out using the iCycler (BIO-RAD) system, and quantitative comparisons were obtained using the ΔΔCT method.
Inhaled Ag Challenge and Assessment of Lung Inflammation and Serum Antibody Level
On day 14 after EC sensitization, mice were challenged with intranasal (IN) OVA over a 6-day period, as previously described [18]. Briefly, 50 μl of OVA in PBS (0.5 mg/ml) was administered by IN droplet on days 14, 15, 18, and 19. On day 21, mice were sacrificed to allow for assessment of lung inflammation and determination of serum antibody level. BAL was performed from the lungs and the total cell number recovered from each lung was enumerated. Cell counts based on the morphology and staining characteristics of 200 cells per sample were determined. H&E-stained lung sections were examined in a blinded fashion to determine the degree of inflammation present using a subjective scale. In this scale, we counted the number of perivascular and peribronchial inflammatory foci in a whole lung section, giving weight to areas of inflammation that appeared significantly denser than average by counting them twice [42]. The levels of OVA-specific IgG1, IgG2a, and total IgE from serum collected on day 21 were determined as described previously [18].
Bone Marrow Macrophage Isolation
To obtain bone marrow-derived macrophages, bone marrow from the femurs and tibias of 6–12-week-old mice was cultured for 4 days in RPMI medium with 20% fetal calf serum and 30% L929 conditioned medium. As described previously [59], the L929 conditioned medium consisted of RPMI and 10% fetal calf serum in which near-confluent L929 cells were allowed to grow for 3 days.
Generation of Th2-Skewed OVA-Sensitized DO11.10 T Cells
To generate OVA-sensitized Th2-skewed cells from DO11.10 mice, CD4 T cells were isolated by negative selection, as described above. Sygeneic T cell-depleted splenocytes, prepared as above, were used as APCs. Cultures were established in flasks with a twofold excess of APCs (2.5 × 106/ml) over CD4 T cells (1.25 × 106/ml). IL-4 at 10 ng/ml (Collaborative Research, Inc.), IL-2 at 5 μg/ml, and anti-IFN-γ (XMG1.2) at 3.8 mg/ml, an inhibitory concentration, were added with 5 μg/ml of OVA (323–339) peptide. These cultures were maintained for 4 days prior to preparing the cells for transfer into mice.
Transfer of OVA-Sensitized DO11.10 Cells and IN Administration of OVA
Cultured DO11.10 cells were harvested after day 4, purified by lymphocyte separation medium (MP Biomedical, LLC), and washed with PBS, and 5 × 06 cells were injected IP into mice on day 1. Four to 7 days later, mice were challenged with 50 μl of OVA in PBS (0.5 mg/ml) administered by IN droplet. The mice were challenged IN again on days 1, 4, and 5 following the first challenge. The mice were sacrificed 2 days after the last challenge to allow for assessment of lung inflammation as detailed above.
T Cell Proliferation Assays and IL-2 Measurements
T lymphocytes were prepared from splenocytes and LN cells of 6–10-week-old WT or MIF-KO mice by negative selection with MACS (Miltenyi Biotech, Auburn, CA, USA) columns, as per the manufacturer’s instructions. Anti-B220, CD8, CD16/32, MHC class II (I-Ab KH74), CD11b, and γδ antibodies (eBioscience) were used in the depletion; 2 × 106 cells/ml were suspended in 2 ml of Click’s media supplemented with 10% FBS [60]. Two milliliters of either complete media or media supplemented with 2 μg/ml anti-CD28 Ab (eBioscience) was added. Two hundred microliters of the resulting cell suspension was plated in the wells of a 96-well plate that was coated at 37°C for 2 h with anti-CD3 Ab (eBioscience) at various concentrations prior to use [61]. Cells were cultured for 2 days at 37°C in a 5% CO2 incubator. Subsequently, 1 μCi of [3H] thymidine was added to each well and this was allowed to incubate for 18 h. The cells were harvested and counts per minute were determined.
For T cell transfer studies, WT or MIF-KO DO11.10 T cells were prepared by negative selection from pooled unsensitized lymph node and spleen cells of two mice, using anti-Ig antibody-coated magnetic beads (Advanced Magnetics, Inc.) by the same protocol described above. Mitomycin-treated, T cell-depleted splenocytes from WT or MIF-KO mice for use as APCs were prepared as described above; 3 × 105 T cells were mixed with a twofold excess of APCs (6 × 105) in 96-well plates with varying doses of OVA peptide. The MTT assay for cellular proliferation was performed as per manufacturer’s instruction (Promega), and IL-2 concentration in the supernatant was determined at 40 h by ELISA (eBioscience).
Results
Skin-Draining LN Cells from MIF-Deficient Mice Are Impaired in Their Ability to Produce Th2 Cytokines After EC Sensitization
MIF is important for the sensitization phase of a Th1 immune response in the skin after TNCB application [39]. We initially sought to demonstrate whether MIF is similarly important for the sensitization phase of a Th2 immune response in the skin. Four days after EC OVA exposure, skin-draining LN cells from WT and MIF-KO mice were collected and restimulated in vitro by culturing them with mitomycin-treated, T cell-depleted splenocytes and OVA (Fig. 1). A robust production of Th2 cytokines was observed in the supernatants from cultures with WT LN cells, while the cultures with MIF-KO LN cells displayed markedly attenuated levels of these cytokines. Although absolute levels of the Th1 cytokine IFN-γ were lower after EC OVA sensitization than after cutaneous sensitization to a stimulus known to have more of a Th1 bias (such as TNCB), MIF-KO LN cells also produced a lower amount of this cytokine after restimulation with OVA in vitro. This latter finding is consistent with published results that MIF is important for the production of Th1 cytokines after antigenic challenge [40, 42]. Notably, prior EC sensitization with OVA had no effect on IFN-γ production after restimulation with the protein. Thus, together with previously published data, our findings suggest that MIF-KO mice are impaired in their ability to generate both Th1-type and Th2-type effector cytokines in response to a topically applied antigen.
MIF-KO mice display attenuated cytokine production in skin-draining LN cells after EC OVA exposure. Production of Th2 cytokines (IL-4, IL-5, and IL-13) are compromised in the MIF-KO system despite supplementation with IL-2. Mice were EC-exposed to OVA on day 0 and axillary LNs were harvested on day 4. LN cells were restimulated in vitro with OVA. After 48 h, cytokine levels in culture supernatants were measured. WT (n = 5), MIF-KO (n = 5). Each panel is representative of three separate experiments. Statistical significance was determined using an unpaired Student’s t test or ANOVA. Error bars represent one standard deviation from the mean. ***p < 0.001, **p < 0.01, *p < 0.05
Mechanism of the MIF-Dependent Defect in Th2 Cytokine Production Appears to Involve Decreased Number of Th2 Cells and Increased Numbers of Tregs
MIF is known to be important in T cell proliferation in response to antigen stimulation [38]. We investigated the number of Th1 and Th2 cells in lymph node cultures from EC-sensitized WT and MIF-KO lymph node cultures using flow cytometry (Fig. 2). Using intracellular staining for IFN-γ and IL-4 to designate Th1 and Th2 subsets of T cells, respectively, we observed that the population of Th2-skewed T cells is reduced in the MIF-KO lymph node cultures, compared to WT (5.37% vs. 2.67%, p < 0.05, Fig. 2a). We also noted a trend towards an increased population of Th1-skewed cells in the WT compared to MIF-KO lymph node cultures (1.88% vs. 1.41%, p > 0.05, data not shown), but it did not reach statistical significance. Further investigation of the expression of Th1 and Th2 transcription factors by qRT-PCR showed a significant decrease in the induction of both Tbet and Gata3 after OVA stimulation in the MIF-KO cultures compared to WT (Fig. 2b, c).
Th2 differentiated CD4 T cells are reduced in the lymph nodes of EC OVA-sensitized MIF-KO mice. a OVA stimulation increases the number of IL-4 producing CD4 T cells, measured by flow cytometry, to a greater degree in the WT, compared to MIF-KO system. b, c Both Th1 and Th2 differentiation, measured by upregulation of transcription factors Tbet and Gata3, are decreased in MIF-KO lymph nodes. Unsensitized (n = 3), WT (n = 3), MIF-KO (n = 3). Summary data are pooled from three independent experiments. ***p < 0.001
Prior work by our group has shown lower IL-2 production in the MIF-dependent reduction in antigen-specific T cell response [38]. We supplemented the WT and MIF-KO lymph node cultures with exogenous IL-2 to determine whether restoring IL-2 might lead to a recovery in the MIF-dependent defect in cytokine production. Interestingly, in the case of the Th2 cytokines, IL-4, IL-5, and IL-13, IL-2 addition augmented cytokine production; however, the level in the MIF-KO system remained below that in the unsupplemented WT (IL-4, p < 0.05; IL-13, p < 0.05; and IL-5, p = 0.053; Fig. 1). By contrast, the addition of IL-2 increased the level of IFN-γ produced by the MIF-KO culture beyond that of the WT.
We next quantified the Treg population in the WT and MIF-KO lymph node cultures. We demonstrated a larger population of Treg cells (Foxp3+/CD25+) in the MIF-KO compared to WT cultures (n = 4 animals, p < 0.05). Although we noticed a similar trend in the level of the expression of the Foxp3 transcription factor in the MIF-KO and WT lymph node cultures, the difference did not reach statistical significance (p = 0.097; Fig. 3).
Tregs are increased in the MIF-KO lymph node cultures. a, b The number of CD4 T cells which display Treg markers (CD25+, Foxp3+) are increased in the MIF-KO compared to WT systems. c Foxp3 upregulation, measured by qRT-PCR, is also increased in the MIF-KO compared to WT, although this increase does not reach statistical significance. WT (n = 4), MIF-KO (n = 3). Summary data are pooled from three independent experiments. *p < 0.05
MIF-KO Mice Display Attenuated Lung Inflammation and Antibody Responses After EC Sensitization
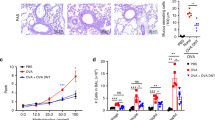
We next aimed to determine whether the impaired sensitization to an EC-applied antigen in MIF-KO mice was functionally significant in vivo. Accordingly, we assessed lung inflammation and antibody responses in MIF-KO and WT mice after EC OVA sensitization and subsequent IN challenge. Both MIF-KO and WT EC OVA-immunized mice displayed significantly increased numbers of cells in their BAL when compared with sham-immunized mice (Fig. 4). The numbers of total cells and eosinophils recovered by BAL were significantly greater in WT than in MIF-KO mice. Histologic evaluation (Figs. 4 and 5), which included a blinded measurement of inflammatory index in sections from the lungs of these mice, confirmed that MIF-KO mice exhibited an attenuated inflammatory response after IN challenge with OVA when compared with their WT counterparts. The inflammatory response after EC OVA sensitization and IN challenge in both groups of mice was generally a mixed perivascular and peribronchial inflammatory response with a moderate to abundant number of eosinophils; this finding also is in agreement with prior reports (Fig. 5) [18, 19].
MIF-KO mice display attenuated airway inflammation after EC sensitization and IN challenge with OVA antigen. WT and MIF-KO mice were exposed to EC OVA while control mice were treated with PBS alone. All mice were subsequently challenged IN with OVA. After sacrifice, BAL was performed and inflammatory cells recovered from individual mice were counted. Histologic sections from the lungs of individual mice were subsequently prepared and an inflammatory index scored for the number of pockets of perivascular and peribronchial pockets of inflammation as described previously [42]. WT: n = 5, MIF-KO: n = 5, WT: control n = 3, MIF-KO: control n = 2. **p < 0.01, *p < 0.05
MIF-KO mice were impaired in their ability to mount a total IgE and OVA-specific IgG1 antibody response after OVA sensitization and challenge (Fig. 6), which is in accord with prior studies of Th2 sensitization using alum as adjuvant [42, 43]. The present results both confirm and extend these prior findings, but now in the physiologically more relevant model of Th2 immune responses, EC sensitization without co-administration of adjuvant. OVA-specific IgE could not be detected in the serum in the EC sensitization model, as has been described previously [18]. Negligible levels of anti-OVA IgG2a, which is an isotype more associated with IFN-γ production by Th1 cells, were detected in the serum of both WT and MIF-KO mice in the present study (data not shown). Notably, the lack of IgG2a production in MIF-KO mice is similar to that observed in IL-13-deficient mice and stands in contrast to the antibody profile observed in IL-4- and STAT-6-deficient mice [18, 19]. In the latter two strains, the attenuation in Th2-associated antibodies in serum is accompanied by a rise in this Th1-associated antibody, presumably because of the absence of IL-4-driven class switching. The findings of decreased Th2-related antibody production in MIF-KO mice, without an attendant increase in Th1 antibody, may result from a direct effect of MIF on antibody production (i.e., an effect of MIF on T and/or B cells), an effect of MIF on the production of cytokines necessary for an appropriate antibody response (with IL-13, rather than IL-4, as a leading candidate), or a combination of these two factors.
Total IgE and OVA-specific IgG1 antibody responses are decreased in EC-sensitized and IN challenged MIF-KO mice as compared with control mice. WT and MIF-KO mice were exposed to EC OVA and were subsequently challenged IN with OVA. Total IgE and anti-OVA IgG1 antibody levels from serum collected after sacrifice on day 21 were measured by ELISA. WT: n = 5, MIF-KO: n = 5. ***p < 0.001, *p < 0.05
MIF Is Not Required for Antigen Presentation During EC OVA Sensitization
We next examined whether MIF produced by APCs is required for proper antigen presentation. Four days after EC OVA exposure, skin-draining LN cells from WT mice were collected and CD4+ T cells were purified by immunodepletion. These T cells were restimulated in vitro by co-culture with OVA and mitomycin-treated, T cell-depleted splenocytes (Fig. 7a) or bone marrow-derived macrophages (Fig. 7b) from non-immunized WT or MIF-KO donors. No significant differences were observed in the levels of IL-4, IL-5, IL-13, and IFN-γ in the culture supernatants when splenic or bone marrow APCs were derived from WT or MIF-KO mice.
Splenocytes and bone marrow-derived macrophages from MIF-KO mice do not show defects in vitro in antigen presentation. WT mice were EC-exposed to OVA on day 0 and axillary LNs were harvested on day 4. LN cells were restimulated in vitro with OVA and mitomycin-treated T-depleted splenocytes (a) or bone marrow-derived macrophages (b) as APCs. After 48 h, cytokine levels in culture supernatants were measured. WT: n = 5, MIF-KO: n = 5. Each panel is representative of three separate experiments
The MIF Cell Surface Receptor, CD74, Is Required for EC Sensitization to OVA
MIF binds to cell surface CD74 with nanomolar affinity, leading to the phosphorylation of the CD74 intracytoplasmic domain and the membrane recruitment of CD44, which then activates a Src family member non-receptor tyrosine kinase to initiate signal transduction [62, 63]. CD74 deficiency in mice also results in reduced, but not absent MHC class II cell surface expression [56]. Interestingly, T cells from CD74-deficient mice (CD74-KO) proliferate normally in vitro and in vivo after immunization with a protein Ag and adjuvant but their cytokine secretion profiles are strongly biased to a Th1 profile [64]. CD74-KO mice also have been shown to be impaired in their ability to generate Th2 immune responses in the murine OVA asthma model [64]. We thus sought to determine whether CD74-KO mice were impaired in the production of Th2-type cytokines after exposure to an EC stimulus and, moreover, to compare the degree of impairment to that seen in MIF-KO mice.
Indeed, draining LN cells from EC-sensitized CD74-KO mice, when compared with LN cells isolated from WT mice, displayed significantly reduced levels of the Th2 cytokines, IL-4, IL-5, and IL-13 after in vitro restimulation with OVA antigen (Fig. 8). Furthermore, the levels of these key Th2 cytokines, produced by LN cells from CD74-KO mice, were significantly lower than observed in MIF-KO LN cells. While the LN cells from the CD74-KO mice reproducibly displayed marked defects in the production of Th2 cytokines, they were able to produce some Th2 cytokines as evidenced by the fact that the levels of IL-4, IL-5, and IL-13 in the supernatants from these stimulated cells were significantly higher than in control supernatants (i.e., from cultures to which OVA was not added). Interestingly, despite the pronounced Th2 defects seen after EC sensitization in CD74-KO mice, the LN cells from these mice displayed a somewhat lesser decrease in production of the Th1 cytokine IFN-γ in response to OVA when compared to WT LN cells. Although our experiments were not designed to assess Th1 responses, these data support the prior work of Topilski et al. who observed a severe attenuation of the Th2 response in CD74-KO mice but a relatively preserved Th1 response [64]. The attenuated Th2 cytokine secretion profile from CD74-KO LN cells after EC OVA sensitization is consistent with the findings observed in MIF-KO mice. Whether the more severely impaired response in the CD74-KO than in the MIF-KO mouse results from additional defects in antigen presentation or to the absence of activating signals provided by MIF or other potential CD74 ligands [65] remains to be established.
MIF receptor (CD74) deficiency attenuates Th2 but not Th1 cytokine production in skin-draining LN cells after EC OVA exposure to a greater extent than does MIF deficiency. Mice were EC-exposed to OVA on day 0 and axillary LNs were harvested on day 4. LN cells were restimulated in vitro with OVA, and after 48 h, cytokine levels in culture supernatants were measured. WT: n = 5, MIF-KO: n = 5, CD74-KO n = 5. Each panel is representative of three separate experiments. ***p < 0.001, **p < 0.01, *p < 0.05
MIF Is Required for the Elicitation Phase of a Th2 Immune Response
MIF is known to be important for the sensitization and elicitation phases of a Th1 response [39]. In this study, we have shown that MIF is important for the sensitization phase of a Th2 immune response (Fig. 1). The differences we observed between WT and MIF-KO mice in the elicitation phase of a Th2 immune response (i.e., in lung inflammation and antibody production) as well as differences observed in previous studies [42] could be entirely explained by a lack of sensitization to the antigenic stimulus, OVA, in MIF-KO animals.
To specifically address whether MIF is important for the elicitation phase of a Th2 immune response independently of the sensitization phase, we utilized the DO11.10 transgenic mouse, in which all of the T cells express a single TCR directed against a specific peptide present in OVA (residues 323–339). T cells from this mouse strain were isolated and sensitized in vitro to OVA peptide in the presence of cytokines and antibodies which skewed the resulting T cells to a Th2 phenotype [66]. These T cells were injected IP into WT or MIF-KO mice and the mice then were challenged IN with OVA antigen.
As shown in Fig. 9, the numbers of total cells as well as eosinophils recovered from BAL fluid in MIF-KO mice were significantly lower than the numbers found in WT mice. Moreover, histopathologic examination of the lungs from these mice (Figs. 9 and 10) was remarkable for an attenuated inflammatory response in the setting of MIF deficiency. Thus, MIF is a crucial mediator not only of the sensitization, but also of the elicitation phase of a Th2 immune response.
MIF is required for the elicitation phase of a Th2 inflammatory response. WT and MIF-KO mice were injected IP with OVA-sensitized Th2 cells derived from DO11.10 mice and were then IN challenged with OVA antigen or PBS. Inflammatory cells recovered from the BAL of five individual mice were counted. Histologic sections from the lungs were subsequently prepared and an inflammatory index scored for the number of pockets of perivascular and peribronchial pockets of inflammation. WT: n = 5, MIF-KO: n = 5, WT control: n = 3, MIF control: n = 3. **p < 0.01
T Cells from MIF-KO Mice Display Attenuated Proliferation in Response to Anti-CD3 Stimulation
WT T cells treated with anti-MIF antibody mice display impaired activation, proliferation, and IL-2 production [38], which may be an important mechanism for explaining the attenuation in immune responses observed in MIF-KO mice. We wanted to confirm that impaired T cell proliferation is a feature not only of anti-MIF-treated WT cells but also cells derived from MIF-KO mice. Thymidine incorporation in response to stimulation with anti-CD3 alone or anti-CD3 plus anti-CD28 was compared in naive WT and MIF-KO T cells. After CD3 ligation, a significant decrease in proliferation was observed in the MIF-KO lymphocytes in the presence and absence of CD28 ligation (Fig. 11). The concentration of anti-CD3 eliciting a proliferative response was lower with the addition of anti-CD28.
MIF-KO T Cells Display Impaired Proliferation and IL-2 Production in Response to Antigen
Taken together, our data indicate that MIF produced by T cells is crucial for the generation of an adequate proliferative response to an antigenic challenge (Fig. 11), while its production by APCs appears to be less important (Fig. 7). These data are consistent with published findings [39], which showed that in parallel MLRs, lymphocyte proliferation to alloantigen was not impaired when MIF-KO cells were used as APCs with WT lymphocytes, but was impaired when APCs were WT and lymphocytes were MIF-KO. To support and extend these findings, we generated DO11.10 transgenic mice on a MIF-KO background and compared proliferation and IL-2 production by T cells derived from these and WT DO11.10 mice after exposure to OVA peptide in the context of WT or MIF-KO APCs.
DO11.10 TCR transgenic T cells derived from MIF-KO mice were significantly impaired in their proliferative and IL-2 production responses when compared with cells derived from WT mice regardless of whether the APCs employed were WT or MIF deficient (Fig. 12). Conversely, in cultures where the T cells were derived from WT mice, nearly identical T cell proliferative responses were observed after antigenic challenge irrespective of whether antigen presentation was performed by MIF-KO or WT splenocytes. T cell proliferation was similar despite the fact that there was a lower level of IL-2 in the supernatants when the APCs were MIF deficient. The reason for this result likely reflects the adequacy of the lower level of IL-2 in triggering the available IL-2 receptors on the T cells. When T cells were derived from MIF-KO mice, however, the presence or absence of APC-encoded MIF had more substantial functional consequences. MIF-KO T cells presented antigen by MIF-KO splenocytes displayed significant defects in both proliferation and IL-2 production when compared with T cells where antigen presentation was performed by WT APCs. Thus, when there is no source at all of MIF in the experimental system (i.e., from either T cells or APCs), T cell proliferation and IL-2 production are most severely impaired (Fig. 12).
MIF-KO T cells display impaired proliferation and IL-2 production after exposure to an antigen. a Proliferation of T cells isolated from either WT or MIF-KO DO11.10 transgenic mice after exposure to the antigen was determined by MTT assay. Splenocytes from either WT or MIF-KO mice were used as APCs. b IL-2 production from WT or MIF-KO DO11.10 T cells was assessed after exposure to the antigen. Splenocytes from either WT or MIF-KO mice were used as APCs. Similar results were found in two separate experiments. Comparison of all data points singly against each other on both graphs (a, b) at 5 and 25 μg/ml OVA peptide reveals highly statistically significant differences (p < 0.001) except for two labeled points on a
Discussion
Atopic diseases are present in a large and growing segment of our population. The lifetime prevalence of AD has been estimated at levels as high as 20% of the US population and the lifetime prevalence of asthma at approximately 10% [67]. Despite the significant morbidity and mortality associated with atopic diseases, our understanding of the molecular mechanisms that underlie an individual’s predisposition to develop an exaggerated immune reaction to an allergic stimulus is still incomplete. For example, we still do not understand the most relevant route of antigen exposure during the sensitization phases of these diseases. Interestingly, it has been suggested that in human atopic diseases, EC exposure to antigens may be an important route of sensitization [54]. Thus, the EC OVA sensitization model system employed in the present studies may well be the most physiologically relevant one in which to study atopic diseases.
MIF is a cytokine that has pro-inflammatory effects in both in vivo and in vitro models of Th1- and Th2-type inflammation. In several model systems, MIF was found to “override” or counter-regulate the broad, immunosuppressive action of glucocorticoids on cells of the immune system [68], which is notable because glucocorticoids remain the mainstay of therapy for many Th1- and Th2-type inflammatory diseases. MIF also is encoded in functionally distinct alleles, and polymorphisms in MIF have been associated with Th2 diseases such as atopic dermatitis, asthma as well as several Th1-biased inflammatory conditions [42, 45, 46].
We investigated herein the role of MIF in EC sensitization to a Th2 antigenic stimulus, OVA, and also examined its role in the elicitation phase of the response. After EC sensitization, LN cells from MIF-KO mice, when compared to WT mice, produced lower levels of key Th2 cytokines when restimulated with the OVA antigen. Notably, EC sensitization does not require administration of an adjuvant, although there is evidence that mice genetically deficient in the TLR-2 or the TLR adaptor protein, MyD88, are impaired in the production of Th1 and Th2 cytokines after EC sensitization (C. Herrick, unpublished data) [69, 70]. Thus, a natural adjuvant, perhaps one derived from a microbe colonizing the surface of the skin, may be stimulating a TLR signaling cascade during EC OVA sensitization. Since APCs from MIF-KO mice are known to display reduced cell surface TLR-4 [33], impaired signaling through this or perhaps another TLR may explain certain of the defects in sensitization observed in MIF-KO mice after EC OVA application. Our experiments nevertheless indicate that once T cells are sensitized to OVA, comparable antigen presentation appears to be achieved by WT and MIF-KO APCs (splenocytes and bone marrow-derived macrophages). That MIF-KO APCs function comparably to WT APCs is supported by the observation that the proliferative response of WT T cells to an antigenic stimulus is very similar regardless of MIF expression by APCs.
The key defect in the sensitization and elicitation phases of a Th2 response in MIF-KO mice is likely an intrinsic property of MIF deficiency by T cells. This is an attractive hypothesis because MIF is known to have direct effects on T cell activation, proliferation, and IL-2 production [38]. Furthermore, in studies of the MLR, lymphocyte proliferation to alloantigen was not impaired when APCs were MIF-KO and lymphocytes were WT, but was impaired when APCs were WT and lymphocytes were MIF-KO [39]. Finally, our data also indicate that the presence of MIF in T cells is vital for the ability to respond maximally to an antigen. This T cell defect in the MIF-KO lymph nodes, which is manifested by a reduced number of Th2 cells and increased number of Treg cells, is not compensated by adding IL-2 to the cultures.
These different experiments imply that there is a defect in the responsiveness of MIF-KO T cells that cannot be compensated by the presence of WT APCs. Nevertheless, our results also suggest that MIF from sources other than sensitized T cells does play a role in the expression of Th2 inflammation. We observed diminished lung inflammation after the transfer of OVA-sensitized, WT DO11.10 T cells into WT and MIF-KO mice and challenge with IN OVA. This experiment therefore showed that MIF not only is required for the elicitation phase of a Th2 immune response but also that MIF produced by sensitized T cells alone is insufficient to compensate for a lack of MIF in the recipient mouse.
Flow cytometric analysis of splenocytes from MIF-KO and WT mice showed no significant abnormalities in the total number of cells when comparing several important lymphocyte subpopulations [42, 71]. Specifically, WT and MIF-KO mice displayed equivalent numbers of splenocytes with surface expression of CD3, CD4, CD8, and B220. Furthermore, on the CD4+ cells, cell surface expression of CD25, CD69, and CD44/62L was similar. At baseline, the numbers of Th1, Th2, and Treg cells were not different. Thus, a diminished number of a specific subset of circulating T cells is unlikely to explain the defects that we and others have described in the development of Th2-type inflammation in MIF-KO mice [39, 47, 48, 51]. Additionally, it is likely a functional defect in the Th2 response, rather than an intrinsic defect in B cells, which explains the diminished IgG1 and total IgE responses that develop without a compensatory increase in IgG2a in the EC OVA-sensitized MIF-KO animals.
The molecular mechanism of MIF’s action in immunologic responses is mediated in large part by signaling through the ERK 1/2 MAPK pathways, which are activated by MIF/receptor interaction. In many cell types, this receptor is CD74, with ligand–receptor interaction occurring either on the cell surface or within an endosome [72, 73]. Our results, which demonstrate similar Th2 cytokine defects in both MIF and CD74-KO animals, may be explained by reduced activation of MIF/CD74 signaling leading to reduced inflammatory cytokine production and/or impaired cell survival. The increased impairment in Th2 cytokine production in the CD74-KO lymph node cultures, compared to MIF-KO, may be due to further defects in these cells, such as in antigen presentation, or by additional ligands for CD74 [64].
In contrast to most cytokines, which can be characterized as “predominantly” Th1 or Th2, MIF upregulates both Th1- and Th2-type inflammatory responses. The important effect of MIF on both types of inflammation is consistent with the current hypotheses indicating that MIF is (1) an upstream mediator of innate and acquired immunity and (2) a key regulator of the immunosuppressive action of endogenous glucocorticoids [74]. Glucocorticoids are used clinically to prevent and control both Th1- and Th2-type inflammation. Therefore, if MIF is to be viewed as an “anti-glucocorticoid”, the results from the current study may be expected.
The suggestion that MIF is a central, upstream immune activator is based on the observations that MIF is stored preformed in cells, is released rapidly after exposure to an inflammatory stimulus, and activates downstream molecules in different inflammatory cascades. In response to infections and other inflammatory stimuli, for example, MIF-KO mice display attenuated levels of TNF, IL-1, IL-12, MCP-1, and PGE2 [55, 71, 75]. Since MIF affects both Th1- and Th2-type inflammation, it can be inferred that this cytokine is important globally for the initiation and the propagation of inflammation and is not as important in directing the specific cytokine or cellular milieu of an inflammatory environment. There is evidence that the main role of MIF in inflammation is to prevent the loss of immune cells and to promote their growth via sustained ERK activation [35]. To address the mechanism of MIF action on T cells after exposure to an antigenic stimulus, we showed that the numbers of Th2-skewed T cells are reduced in the lymph nodes of OVA-sensitized MIF-KO mice. Whether this decrease is due to reduced proliferation (related to IL-2), increased apoptosis, direct effects on regulating transcription factors (such as Tbet or Gata3), or more than one of these mechanisms remains to be elucidated. An additional element that requires investigation is the MIF-related decrease in Treg cells in the WT sensitized lymph node cultures, which may be considered permissive for Th2 differentiation. A recent work by Park and colleagues also has suggested a role for MIF in Treg populations by demonstrating that MIF from the helminth Anisakis simplex suppresses Th2 inflammatory responses by antagonizing the host MIF and increasing the subset of Treg cells [76].
In summary, we found that MIF is crucial for both the afferent and efferent arms of a Th2-type inflammatory response using EC sensitization to OVA antigen and adoptive transfer of in vitro sensitized T cells. These findings may prove important clinically especially since individuals with low vs. high expression alleles of MIF display different phenotypes in atopic dermatitis and in asthma [42, 45, 46]. We postulate that MIF inhibitors, which are currently in clinical development, may prove efficacious in palliating atopic inflammation [52, 77].
Abbreviations
- AD:
-
Atopic dermatitis
- APCs:
-
Antigen-presenting cells
- BAL:
-
Bronchoalveolar lavage
- EC:
-
Epicutaneous
- ERK 1/2:
-
Extracellular signal-regulated kinase
- IFN-γ:
-
Interferon-gamma
- IL:
-
Interleukin
- IN:
-
Intranasal
- IP:
-
Intraperitoneal
- LN:
-
Lymph node
- MHC:
-
Major histocompatibility complex
- MIF:
-
Macrophage migration inhibitory factor
- TCR:
-
T cell receptor
- TNCB:
-
Trinitrochlorobenzene
- TLR:
-
Toll-like receptor
References
Hamid Q, Naseer T, Minshall EM, Song YL, Boguniewicz M, Leung DY. In vivo expression of IL-12 and IL-13 in atopic dermatitis. J Allergy Clin Immunol. 1996;98:225–31.
Huang SK, Xiao HQ, Kleine-Tebbe J, Paciotti G, Marsh DG, Lichtenstein LM, et al. IL-13 expression at the sites of allergen challenge in patients with asthma. J Immunol. 1995;155:2688–94.
Robinson DS, Hamid Q, Ying S, Tsicopoulos A, Barkans J, Bentley AM, et al. Predominant TH2-like bronchoalveolar T-lymphocyte population in atopic asthma. N Engl J Med. 1992;326:298–304.
van Reijsen FC, Bruijnzeel-Koomen CA, Kalthoff FS, Maggi E, Romagnani S, Westland JK, et al. Skin-derived aeroallergen-specific T-cell clones of Th2 phenotype in patients with atopic dermatitis. J Allergy Clin Immunol. 1992;90:184–93.
Robinson D, Hamid Q, Bentley A, Ying S, Kay AB, Durham SR. Activation of CD4+ T cells, increased TH2-type cytokine mRNA expression, and eosinophil recruitment in bronchoalveolar lavage after allergen inhalation challenge in patients with atopic asthma. J Allergy Clin Immunol. 1993;92:313–24.
Hamid Q, Boguniewicz M, Leung DY. Differential in situ cytokine gene expression in acute versus chronic atopic dermatitis. J Clin Invest. 1994;94:870–6.
Brombacher F. The role of interleukin-13 in infectious diseases and allergy. Bioessays. 2000;22:646–56.
Doherty TM, Kastelein R, Menon S, Andrade S, Coffman RL. Modulation of murine macrophage function by IL-13. J Immunol. 1993;151:7151–60.
Finkelman FD, Katona IM, Urban Jr JF, Holmes J, Ohara J, Tung AS, et al. IL-4 is required to generate and sustain in vivo IgE responses. J Immunol. 1988;141:2335–41.
Grunig G, Warnock M, Wakil AE, Venkayya R, Brombacher F, Rennick DM, et al. Requirement for IL-13 independently of IL-4 in experimental asthma. Science. 1998;282:2261–3.
McKenzie AN, Culpepper JA, de Waal Malefyt R, Briere F, Punnonen J, Aversa G, et al. Interleukin 13, a T-cell-derived cytokine that regulates human monocyte and B-cell function. Proc Natl Acad Sci U S A. 1993;90:3735–9.
Wills-Karp M, Luyimbazi J, Xu X, Schofield B, Neben TY, Karp CL, et al. Interleukin-13: central mediator of allergic asthma. Science. 1998;282:2258–61.
Zurawski SM, Chomarat P, Djossou O, Bidaud C, McKenzie AN, Miossec P, et al. The primary binding subunit of the human interleukin-4 receptor is also a component of the interleukin-13 receptor. J Biol Chem. 1995;270:13869–78.
Lin JX, Migone TS, Tsang M, Friedmann M, Weatherbee JA, Zhou L, et al. The role of shared receptor motifs and common Stat proteins in the generation of cytokine pleiotropy and redundancy by IL-2, IL-4, IL-7, IL-13, and IL-15. Immunity. 1995;2:331–9.
Smerz-Bertling C, Duschl A. Both interleukin 4 and interleukin 13 induce tyrosine phosphorylation of the 140-kDa subunit of the interleukin 4 receptor. J Biol Chem. 1995;270:966–70.
Swain SL, Weinberg AD, English M, Huston G. IL-4 directs the development of Th2-like helper effectors. J Immunol. 1990;145:3796–806.
Zurawski G, de Vries JE. Interleukin 13, an interleukin 4-like cytokine that acts on monocytes and B cells, but not on T cells. Immunol Today. 1994;15:19–26.
Herrick CA, MacLeod H, Glusac E, Tigelaar RE, Bottomly K. Th2 responses induced by epicutaneous or inhalational protein exposure are differentially dependent on IL-4. J Clin Invest. 2000;105:765–75.
Herrick CA, Xu L, McKenzie AN, Tigelaar RE, Bottomly K. IL-13 is necessary, not simply sufficient, for epicutaneously induced Th2 responses to soluble protein antigen. J Immunol. 2003;170:2488–95.
Foster PS, Hogan SP, Ramsay AJ, Matthaei KI, Young IG. Interleukin 5 deficiency abolishes eosinophilia, airways hyperreactivity, and lung damage in a mouse asthma model. J Exp Med. 1996;183:195–201.
Lee JJ, McGarry MP, Farmer SC, Denzler KL, Larson KA, Carrigan PE, et al. Interleukin-5 expression in the lung epithelium of transgenic mice leads to pulmonary changes pathognomonic of asthma. J Exp Med. 1997;185:2143–56.
Resnick MB, Weller PF. Mechanisms of eosinophil recruitment. Am J Respir Cell Mol Biol. 1993;8:349–55.
Yamaguchi Y, Hayashi Y, Sugama Y, Miura Y, Kasahara T, Kitamura S, et al. Highly purified murine interleukin 5 (IL-5) stimulates eosinophil function and prolongs in vitro survival. IL-5 as an eosinophil chemotactic factor. J Exp Med. 1988;167:1737–42.
Yamaguchi Y, Suda T, Suda J, Eguchi M, Miura Y, Harada N, et al. Purified interleukin 5 supports the terminal differentiation and proliferation of murine eosinophilic precursors. J Exp Med. 1988;167:43–56.
Zheng W, Flavell RA. The transcription factor GATA-3 is necessary and sufficient for Th2 cytokine gene expression in CD4 T cells. Cell. 1997;89:587–96.
Zhu J, Yamane H, Cote-Sierra J, Guo L, Paul WE. GATA-3 promotes Th2 responses through three different mechanisms: induction of Th2 cytokine production, selective growth of Th2 cells and inhibition of Th1 cell-specific factors. Cell Res. 2006;16:3–10.
Chapoval S, Dasgupta P, Dorsey NJ, Keegan AD. Regulation of the T helper cell type 2 (Th2)/T regulatory cell (Treg) balance by IL-4 and STAT6. J Leukoc Biol. 2010;87:1011–8.
Bettelli E, Dastrange M, Oukka M. Foxp3 interacts with nuclear factor of activated T cells and NF-kappa B to repress cytokine gene expression and effector functions of T helper cells. Proc Natl Acad Sci USA. 2005;102:5138–43.
Choi JM, Shin JH, Sohn MH, Harding MJ, Park JH, Tobiasova Z, et al. Cell-permeable Foxp3 protein alleviates autoimmune disease associated with inflammatory bowel disease and allergic airway inflammation. Proc Natl Acad Sci USA. 2010;107:18575–80.
Bloom BR, Bennett B. Mechanism of a reaction in vitro associated with delayed-type hypersensitivity. Science. 1966;153:80–2.
David JR. Delayed hypersensitivity in vitro: its mediation by cell-free substances formed by lymphoid cell-antigen interaction. Proc Natl Acad Sci USA. 1966;56:72–7.
Mitchell RA, Metz CN, Peng T, Bucala R. Sustained mitogen-activated protein kinase (MAPK) and cytoplasmic phospholipase A2 activation by macrophage migration inhibitory factor (MIF). Regulatory role in cell proliferation and glucocorticoid action. J Biol Chem. 1999;274:18100–6.
Roger T, David J, Glauser MP, Calandra T. MIF regulates innate immune responses through modulation of Toll-like receptor 4. Nature. 2001;414:920–4.
Hudson JD, Shoaibi MA, Maestro R, Carnero A, Hannon GJ, Beach DH. A proinflammatory cytokine inhibits p53 tumor suppressor activity. J Exp Med. 1999;190:1375–82.
Mitchell RA, Liao H, Chesney J, Fingerle-Rowson G, Baugh J, David J, et al. Macrophage migration inhibitory factor (MIF) sustains macrophage proinflammatory function by inhibiting p53: regulatory role in the innate immune response. Proc Natl Acad Sci USA. 2002;99:345–50.
Bucala R. MIF re-discovered: pituitary hormone and glucocorticoid-induced regulator of cytokine production. Cytokine Growth Factor Rev. 1996;7:19–24.
Nishihira J. Macrophage migration inhibitory factor (MIF): its essential role in the immune system and cell growth. J Interferon Cytokine Res. 2000;20:751–62.
Bacher M, Metz CN, Calandra T, Mayer K, Chesney J, Lohoff M, et al. An essential regulatory role for macrophage migration inhibitory factor in T-cell activation. Proc Natl Acad Sci USA. 1996;93:7849–54.
Shimizu T, Abe R, Nishihira J, Shibaki A, Watanabe H, Nakayama T, et al. Impaired contact hypersensitivity in macrophage migration inhibitory factor-deficient mice. Eur J Immunol. 2003;33:1478–87.
Bernhagen J, Bacher M, Calandra T, Metz CN, Doty SB, Donnelly T, et al. An essential role for macrophage migration inhibitory factor in the tuberculin delayed-type hypersensitivity reaction. J Exp Med. 1996;183:277–82.
Nakamaru Y, Oridate N, Nishihira J, Takagi D, Furuta Y, Fukuda S. Macrophage migration inhibitory factor (MIF) contributes to the development of allergic rhinitis. Cytokine. 2005;31:103–8.
Mizue Y, Ghani S, Leng L, McDonald C, Kong P, Baugh J, et al. Role for macrophage migration inhibitory factor in asthma. Proc Natl Acad Sci USA. 2005;102:14410–5.
Wang B, Huang X, Wolters PJ, Sun J, Kitamoto S, Yang M, et al. Cutting edge: deficiency of macrophage migration inhibitory factor impairs murine airway allergic responses. J Immunol. 2006;177:5779–84.
Yoshihisa Y, Makino T, Matsunaga K, Honda A, Norisugi O, Abe R, Shimizu H, and Shimizu T. Macrophage migration inhibitory factor is essential for eosinophil recruitment in allergen-induced skin inflammation. J Invest Dermatol. 2011;131:925–931
Hizawa N, Yamaguchi E, Takahashi D, Nishihira J, Nishimura M. Functional polymorphisms in the promoter region of macrophage migration inhibitory factor and atopy. Am J Respir Crit Care Med. 2004;169:1014–8.
Wu J, Fu S, Ren X, Jin Y, Huang X, Zhang X, et al. Association of MIF promoter polymorphisms with childhood asthma in a northeastern Chinese population. Tissue Antigens. 2009;73:302–6.
Shimizu T, Abe R, Nakamura H, Ohkawara A, Suzuki M, Nishihira J. High expression of macrophage migration inhibitory factor in human melanoma cells and its role in tumor cell growth and angiogenesis. Biochem Biophys Res Commun. 1999;264:751–8.
Shimizu T, Abe R, Ohkawara A, Mizue Y, Nishihira J. Macrophage migration inhibitory factor is an essential immunoregulatory cytokine in atopic dermatitis. Biochem Biophys Res Commun. 1997;240:173–8.
Shimizu T, Abe R, Ohkawara A, Nishihira J. Increased production of macrophage migration inhibitory factor by PBMCs of atopic dermatitis. J Allergy Clin Immunol. 1999;104:659–64.
Rossi AG, Haslett C, Hirani N, Greening AP, Rahman I, Metz CN, et al. Human circulating eosinophils secrete macrophage migration inhibitory factor (MIF). Potential role in asthma. J Clin Invest. 1998;101:2869–74.
Yamaguchi E, Nishihira J, Shimizu T, Takahashi T, Kitashiro N, Hizawa N, et al. Macrophage migration inhibitory factor (MIF) in bronchial asthma. Clin Exp Allergy. 2000;30:1244–9.
Chen PF, Luo YL, Wang W, Wang JX, Lai WY, Hu SM, et al. ISO-1, a macrophage migration inhibitory factor antagonist, inhibits airway remodeling in a murine model of chronic asthma. Mol Med. 2010;16:400–8.
Amano T, Nishihira J, Miki I. Blockade of macrophage migration inhibitory factor (MIF) prevents the antigen-induced response in a murine model of allergic airway inflammation. Inflamm Res. 2007;56:24–31.
Spergel JM, Paller AS. Atopic dermatitis and the atopic march. J Allergy Clin Immunol. 2003;112:S118–27.
Bozza M, Satoskar AR, Lin G, Lu B, Humbles AA, Gerard C, et al. Targeted disruption of migration inhibitory factor gene reveals its critical role in sepsis. J Exp Med. 1999;189:341–6.
Shachar I, Flavell RA. Requirement for invariant chain in B cell maturation and function. Science. 1996;274:106–8.
Koni PA, Flavell RA. Lymph node germinal centers form in the absence of follicular dendritic cell networks. J Exp Med. 1999;189:855–64.
Levin D, Constant S, Pasqualini T, Flavell R, Bottomly K. Role of dendritic cells in the priming of CD4+ T lymphocytes to peptide antigen in vivo. J Immunol. 1993;151:6742–50.
Alexopoulou L, Holt AC, Medzhitov R, Flavell RA. Recognition of double-stranded RNA and activation of NF-kappaB by Toll-like receptor 3. Nature. 2001;413:732–8.
Click RE, Benck L, Alter BJ. Immune responses in vitro. I. Culture conditions for antibody synthesis. Cell Immunol. 1972;3:264–76.
Mukherjee S, Ahmed A, Malu S, Nandi D. Modulation of cell cycle progression by CTLA4-CD80/CD86 interactions on CD4+ T cells depends on strength of the CD3 signal: critical role for IL-2. J Leukoc Biol. 2006;80:66–74.
Leng L, Metz CN, Fang Y, Xu J, Donnelly S, Baugh J, et al. MIF signal transduction initiated by binding to CD74. J Exp Med. 2003;197:1467–76.
Shi X, Leng L, Wang T, Wang W, Du X, Li J, et al. CD44 is the signaling component of the macrophage migration inhibitory factor–CD74 receptor complex. Immunity. 2006;25:595–606.
Topilski I, Harmelin A, Flavell RA, Levo Y, Shachar I. Preferential Th1 immune response in invariant chain-deficient mice. J Immunol. 2002;168:1610–7.
Coleman AM, Rendon BE, Zhao M, Qian MW, Bucala R, Xin D, et al. Cooperative regulation of non-small cell lung carcinoma angiogenic potential by macrophage migration inhibitory factor and its homolog, D-dopachrome tautomerase. J Immunol. 2008;181:2330–7.
Cohn L, Homer RJ, Marinov A, Rankin J, Bottomly K. Induction of airway mucus production By T helper 2 (Th2) cells: a critical role for interleukin 4 in cell recruitment but not mucus production. J Exp Med. 1997;186:1737–47.
Spergel JM. From atopic dermatitis to asthma: the atopic march. Ann Allergy Asthma Immunol. 2010;105:99–106; quiz 107–109, 117
Calandra T, Bernhagen J, Metz CN, Spiegel LA, Bacher M, Donnelly T, et al. MIF as a glucocorticoid-induced modulator of cytokine production. Nature. 1995;377:68–71.
Akei HS, Brandt EB, Mishra A, Strait RT, Finkelman FD, Warrier MR, et al. Epicutaneous aeroallergen exposure induces systemic TH2 immunity that predisposes to allergic nasal responses. J Allergy Clin Immunol. 2006;118:62–9.
Jin H, Kumar L, Mathias C, Zurakowski D, Oettgen H, Gorelik L, et al. Toll-like receptor 2 is important for the T(H)1 response to cutaneous sensitization. J Allergy Clin Immunol. 2009;123:875–82. e871.
Fingerle-Rowson G, Petrenko O, Metz CN, Forsthuber TG, Mitchell R, Huss R, et al. The p53-dependent effects of macrophage migration inhibitory factor revealed by gene targeting. Proc Natl Acad Sci USA. 2003;100:9354–9.
Xie L, Qiao X, Wu Y, Tang J. β-Arrestin1 mediates the endocytosis and functions of macrophage migration inhibitory factor. PLoS One. 2011;6(1):e16428.
Lue H, Dewor M, Leng L, Bucala R, Bernhagen J. Activation of the JNK signalling pathway by macrophage migration inhibitory factor (MIF) and dependence on CXCR4 and CD74. Cell Signal. 2010;23:135–44.
Flaster H, Bernhagen J, Calandra T, Bucala R. The macrophage migration inhibitory factor-glucocorticoid dyad: regulation of inflammation and immunity. Mol Endocrinol. 2007;21:1267–80.
Hoi AY, Hickey MJ, Hall P, Yamana J, O’Sullivan KM, Santos LL, et al. Macrophage migration inhibitory factor deficiency attenuates macrophage recruitment, glomerulonephritis, and lethality in MRL/lpr mice. J Immunol. 2006;177:5687–96.
Park SK, Cho MK, Park HK, Lee KH, Lee SJ, Choi SH, et al. Macrophage migration inhibitory factor homologs of Anisakis simplex suppress Th2 response in allergic airway inflammation model via CD4+CD25+Foxp3+ T cell recruitment. J Immunol. 2009;182:6907–14.
Lolis E, Bucala R. Therapeutic approaches to innate immunity: severe sepsis and septic shock. Nat Rev Drug Discov. 2003;2:635–45.
Acknowledgments
We thank Jason Griffith for providing thoughtful discussions and insights, and Idit Shachar of the Weizmann Institute, Rehovot, Israel, for providing the BALB/c CD74-KO mice. We also acknowledge the following grant support: RD NIH grant 2 T32 AR 007107-36 and Yale-New Haven Hospital William Wirt Winchester Grant, RB NIH grants AR050498 and AR049610, CH NIH grant AI069396, and NIH Grant T30-AR07016, Dermatology Foundation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Das, R., Moss, J.E., Robinson, E. et al. Role of Macrophage Migration Inhibitory Factor in the Th2 Immune Response to Epicutaneous Sensitization. J Clin Immunol 31, 666–680 (2011). https://doi.org/10.1007/s10875-011-9541-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10875-011-9541-7