Abstract
Spatial patterns and morphology of hydrothermal vents and the occurrence of vent-associated megafauna were investigated in the back-arc basin system of the Okinawa Trough, western Pacific. Amongst hydrothermal vent fields located on the rising slopes to the NE and S of the basin, the Iheya North area has been subjected to a series of intensive diving surveys. Hydrothermal vents demonstrated concentrated patterns of distribution on different spatial scales. In Iheya North, the majority of vents occurred in lines parallel to the NE to W/SW alignment of the spreading axis, within a radius of ca. 200 m around the most active sulphide structure with the highest recorded fluid temperature of over 300°C. The morphology of hydrothermal vents varied greatly from an incipient flat rock with crevices to a 20- to 30-m-tall, multi-flanged structure with concomitant variation in the distribution and abundance of vent-associated megafauna, particularly a galatheid Shinkaia crosnieri and Bathymodiolus mussels. Comparison of active and inactive vents revealed that the spatial extent of Shinkaia ‘aggregation’ (a group of individuals with short nearest-neighbour distances) effectively defined a habitat unit of this species, and active hydrothermal chimneys contained more of these units, leading to greater spatial occupancy by Shinkaia. Given the temporally unstable nature of vent structures as recognised by repeated surveys, vent assemblages are thought to be closely dictated by the spatio-temporal dynamics of vents in the Okinawa Trough back-arc system.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Since the first discovery in the 1970s of organisms associated with deep-sea geothermal activities on the Galapagos Spreading Centre, much research effort has been directed towards gathering information on hydrothermal vents and vent-associated assemblages around the world. Whilst the occurrence of non-vent deep-sea assemblages largely depends upon resources transported from photosynthesis-dominated shallower waters (Gage and Tyler 1991; Gooday 2002; Tokeshi 2002, 2003; Johnson et al. 2007; Galéron et al. 2009; Wei et al. 2010), chemoautotrophic bacterial production dictates the diversity of hydrothermal vent assemblages in different oceanic systems (Parson et al. 1995; Kojima 2002; Van Dover 2000; Nakagawa and Takai 2008). A steadily increasing volume of work on these unique systems has been undertaken, though there is still a paucity of detailed information on the spatio-temporal dynamics and variability of hydrothermal vents and vent-associated assemblages (Lutz and Kennish 1993; Jollivet 1996; Copley et al. 2007; Kato et al. 2010).
While hydrothermal vent assemblages associated with mid-ocean ridge (MOR) systems have been widely known and received much attention (Grassle 1986; Childress and Fisher 1992; Lutz and Kennish 1993; Copley et al. 1997; Gebruk et al. 1997; Tunnicliffe et al. 1998; Desbruyères et al. 2000, 2001; Van Dover 2000; Cuvelier et al. 2009), those associated with non-MOR systems have a shorter research history and, consequently, are scientifically less known. Upwelling of magmas which generates hydrothermal vents in back-arc systems is distinct in that it is associated with episodic or periodic extension of spreading centres, rather than continuous extension as seen in MOR systems (Van Dover 2000; Glasby and Notsu 2003). Thus, hydrothermal vents in a back-arc system constitute a distinct entity that is unrelated in space and time to vents in other systems. Consequently, vent structures in back-arc systems are considered to provide unique examples of varied geomorphological settings for organisms’ existence.
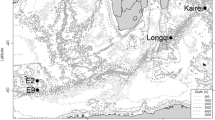
Of the two back-arc systems present in the north-western Pacific, the Ryukyu Arc running through the Okinawa Trough (Fig. 1a) is known to harbour very active vent fields. The Okinawa Trough is located on the western side of the Nansei-shoto Archipelago in a region of complex tectonic movements (Glasby and Notsu 2003). Along the south-eastern edge of the Nansei-shoto Archipelago, the Philippine Sea Plate sinks beneath the Eurasian Plate, forming a deep trench (the Nansei-shoto Trench—maximum depth 7,460 m) and pulling the eastern edge of the Eurasian Plate into the subduction zone. As the Eurasian Plate itself is retreating W/NW, the seafloor of the Eastern China Sea to the west of the Nansei-shoto Archipelago is gradually stretched and spreading, now forming a basin that reaches ca. 2,200 m depth.
Since their discovery in 1988 (Kimura et al. 1989), the hydrothermal systems in the Okinawa Trough have been the subject of an intensive series of geophysical and biological surveys (e.g. Aoki and Nakamura 1989; Hashimoto et al. 1995; Yamamoto et al. 1999; Fujikura et al. 2001; Ohta and Kim 2001; Watanabe 2001; Glasby and Notsu 2003; Nakagawa et al. 2005) and six active vent fields have been reported to date. These are, from NE to SW, Minami-Ensei Knoll (#1; Fig. 1b), Iheya North (#2), Iheya Ridge (#3), Izena Hole (#4), Hatoma Knoll (#5) and Daiyon-Yonaguni Knoll (#6). All these areas are located along the edges of the rising slope between 700 and 1,500 m depth, while the deepest part of the Trough is formed by a relatively flat, central plain of 1,500–2,000 m depth. The present study deals mainly with the Iheya North area (designated as ‘North Knoll of Iheya Ridge’ in Fujikura et al. 2008) (#2, Fig. 1b) at 950–1,050 m depth in the north-eastern part of the Okinawa Trough where the depth gradient runs from NE (shallower) to SW (deeper). More data are available from this area, as it has been subjected to a series of intensive surveys.
The objective of the present study is to examine the morphological characteristics and spatial patterns of hydrothermally active sulphide structures and vent-associated megafauna, using an extensive collection of photographic and video recordings obtained through manned submersible surveys. In particular, the present study complements the existing physicochemical and microbiological information on the hydrothermal system of Iheya North (Glasby and Notsu 2003; Nakagawa et al. 2005) by presenting photographic records of variable edifice morphologies from the Iheya North area, alongside the analysis of spatial distribution patterns of megafauna on medium to small spatial scales.
2 Materials and methods
The data in the present study consist of video recordings and photographs taken on board the manned submersible “Shinkai 2000” between 1997 and 2002. The submersible was equipped with CCD video and stereo camera systems, both specially adapted for the deep-cruising vehicle and adjusted to photograph the seafloor from different angles. Video recording was carried out continuously throughout a bottom cruise and stereo photographs were taken at either regular (45–60 s; surveys in May 2002) or irregular intervals (all other surveys). Still photographs were also taken from an observation port of the vehicle using a hand-held digital camera, especially when the submersible was temporarily resting on the seabed. A total of over 100 h of video recordings from 20 manned dives, 1,200 high-quality photographs (obtained mainly in the May 2002 survey) and data obtained through unmanned towing surveys were used in the present analysis. All video recordings were reviewed on a high-resolution monitor and parts of the recordings were digitised to obtain still images for cross-comparisons and image analyses on a computer. Similarly, all analogue photographs were also digitised for a computer-based analysis. For the assessment of sulphide structures, still images derived from both analogue/digital photography and video recordings were compared and reconstructed to obtain enlarged and extended images. Occurrence of megafaunal species and hydrothermal vents were carefully noted with reference to the map data obtained through diving operations and deep-tow surveys. In order to facilitate size estimation of objects, video and photographic images often included objects of known sizes such as vehicle parts. In May 2002 surveys, a measuring quadrat (25 × 25 cm) was also deployed. Qualitative samples of benthic organisms were collected using the manipulator and brought back on-board and then to the laboratory for taxonomic and other works.
Two edifice structures of contrasting activity levels (high and low) were chosen for detailed analyses of small-scale spatial patterns of vent assemblages. In the video-reconstructed photographs the edifice surface from the top downwards was covered by 50 × 50 cm grids and the occurrence of organisms (particularly galatheid crabs) in each grid was noted and their two-dimensional cover was quantified as digitised data; these were then used to estimate density and occupancy values [proportion (%) of area occupied]. Density here refers to the minimum estimated value, as those individuals (especially small-sized ones) completely hidden beneath other individuals in a dense aggregation and/or in (parts of) images, where illumination was not sufficient to allow individual recognition, were inevitably overlooked. A total of ca. 300 grids were thus examined for each edifice structure. Further, in order to quantify aggregations of galatheid crabs, an ‘aggregation’ was defined as a group of individuals in which each individual has the first and the second nearest-neighbour distance (NND) of less than half its total body width. The size of an aggregation was thus calculated as area encompassing all the individuals belonging to an aggregation. This working definition is based on the observation that Shinkaia crabs generally formed such a strong concentration in the centre of their local distribution (see Sect. 3). The consideration behind this definition was that (1) one or two isolated individuals cannot be considered as forming aggregation, and (2) only those existing in apparent proximity should be counted as aggregation members, for the definition to be of practical use. Alongside aggregation size, aggregation perimeter length was also measured to assess variation in aggregation pattern, through the examination of area–perimeter relationships using ANCOVA.
3 Results
3.1 Occurrence of hydrothermal vents
Figure 2 shows the occurrence of hydrothermal vents in the Iheya North area on the spatial scales of 104 to 102 m. Hydrothermal vents were found in the middle of a ridge-plateau system of approximately 15 km (E–W) × 6 km (N–S) that rose from a seafloor of ca. 1,400 m depth to the west and 1,300 m to the east, within a restricted area of ca. 700 × 700 m at a depth of around 1,000 m (Fig. 2, upper). Outside this area, no hydrothermal assemblage has been found. Within this area, the majority of vent assemblages occurred within a radius of approximately 200 m centred around Edifice 3 (E3; Fig. 2, lower) (designated as ‘North Big Chimney’; Nakagawa et al. 2005) at 950–1,050 m depth. Four types of vents have been recognised: (1) vents with edifice structures with aggregations of the galatheid Shinkaia crosnieri and the mussels Bathymodiolus platifrons and B. japonicus (the latter in smaller proportions, see below); (2) vents without (well-developed) edifice structure, i.e. essentially flat substrata, with recognisable shimmering hydrothermal fluids, where aggregations of Shinkaia crosnieri and Bathymodiolus mussels occur; (3) Bathymodiolus aggregations without Shinkaia crosnieri; and (4) aggregations of Calyptogena in soft substrate. Of these, the first and the second categories represent active hydrothermal vents, while the third and the fourth represent weak or dying vents and peripheral zones. The analysis of the orientation of animal aggregations in categories (1) and (2) vents using videos and photographs showed that these active vents were positioned along NE–W/SW curving lines (ridges) which were separated by a distance of about 70 m from one another (Fig. 2, lower). Clearly, the central ridge encompassing Edifices 3 and 4 (E3 ‘North Big Chimney’ and E4 ‘Central Big Chimney’) was the centre of hydrothermal activity in Iheya North, with the most extensive occurrence of Shinkaia crosnieri and Bathymodiolus aggregations and hydrothermal fluid reaching the maximum temperature of 311°C (Nakagawa et al. 2005).
Bathymetry of Iheya North area (upper, lines indicate deep-tow survey lines) and the distribution of hydrothermal vents and megafaunal assemblages at around 1,000 m depth (lower). Sites where either Bathymodiolus or Calyptogena patches were observed without physically apparent vent structures are separately indicated. Curved broken lines indicate the observed aligning of assemblages
3.2 Mound structures and vent assemblages
Examination of hydrothermal sites in Iheya North revealed morphological variation of sulphide mound structures in this back-arc system, with associated variation in the abundance and distribution of vent-associated megafauna. Figures 3, 4 and 5 show representative edifice morphologies and pattern of occurrence of vent assemblages. Edifice 1 (E1 in Figs. 2, and 3a–d) had a massive columnar structure (ca. 20 m in height) showing an apparent decline in hydrothermal activity, particularly in the top part of the structure without conspicuous aggregations of animal assemblages normally observed in more active edifices. Edifice tops were either devoid of vent-associated animals (Fig. 3d) or covered by bacterial mat of unknown identity (Fig. 3c), but hydrothermal activity still continued in the lower part of the edifice with the existence of shimmering fluid and patches of animals. An example of hydrothermally more active structures is shown by Edifice 2 (ca. 15 m in height) with numerous cone-shaped chimney tops (ca. 50 cm tall) (Fig. 3e, f) on a raised mound, inhabited by the galatheid Shinkaia crosnieri and the mussel Bathymodiolus platifrons. Close examination of this edifice showed an irregular, lava-field like surface with numerous cracks and fissures which allowed the establishment of Bathymodiolus aggregations with accompanying mobile fauna (Fig. 3g, h).
a–d Edifice 1; e–h Edifice 2. a Upper part of Edifice 1 with scattered patches of Shinkaia. b Viewed from the opposite side. c Chimney top covered by filamentous bacteria. d Bare chimney top. e, g Details of the top part of Edifice 2. Scale bars refer to the centre of each frame. Photographs: M. Tokeshi
a Perspective view of Edifice 3 (west side), showing approximately the upper two-thirds of its ca. 30 m high, multi-flange structure; inset shows the top part. Note the occurrence of 5–6 flange layers in this image. b Edifice top with an active vent. Note dark, friable anhydrite deposits around the vent orifice and the aggregation of Shinkaia crosnieri 25–30 cm away from the vent. c Assemblage of vent-associated organisms on Edifice 3, including Shinkaia, Bathymodiolus, Alvinocaris and zoarcid fishes. Photographs: M. Tokeshi
a–e Edifice 4; f, g Edifice 5. a Upper part of Edifice 4 with an extensive aggregation of Shinkaia. b A flange structure of the chimney top. c, d Vestimentiferan tubeworms on the side wall of Edifice 4. e Shinkaia aggregation on steep sides and ridges close to the chimney top where vent fluids tend to gather. f An aggregation of vent organisms on Edifice 5. g Edifice top covered by Bathymodiolus. Scale bars refer to the centre of each frame. Photographs: M. Tokeshi (except c and d which are from the archives of JAMSTEC)
Edifice 3 (‘North Big Chimney’) was the most active mound reaching ca. 32 m in height and pyramid-shaped with well-developed, multi-layered flange structures (type 3) (Fig. 4a), with massive aggregations of vent animals, especially towards the peak of the structure. The mound top had an active orifice where hydrothermal fluid gushed out (Fig. 4b), surrounded, at some distance away, by aggregations of Shinkaia crosnieri. Vent organisms included not only invertebrates but also a Zoarcid fish species, which tended to seek shelter in bare patches of sulphide substrate (Fig. 4c). Edifice 4 (‘Central Big Chimney’) was also an active mound with multiple tops and flange structures, covered by massive aggregations of Shinkaia crosnieri with some patches of vestimentiferan tubeworms (Fig. 5a–e). Edifice 5 (ca. 18 m in height) was similar in morphology to Edifice 2 with multiple mound tops but, unlike Edifice 2, its top parts were covered predominantly by Bathymodiolus mussels with no or few Shinkaia crosnieri, while scattered aggregations of Shinkaia crabs occurred on mound surfaces (Fig. 5f, g). Of the three Bathymodiolus species known to occur in the Okinawa Trough (Fujikura et al. 2008), the materials collected from vent fields of Iheya North during dive No. 1,350 in May 2002 included no specimen of B. aduloides, while B. platifrons accounted for the majority of 128 specimens (ca. 89%) with the rest being identified as B. japonicus. In the samples collected, the less abundant B. japonicus always occurred in mixed patches with B. platifrons without forming a single-species aggregation. In contrast, B. platifrons often achieved near 100% dominance in the areas under study.
3.3 Spatial pattern of vent assemblages
While sulphide mounds varied in structure, vent assemblages demonstrated a recognisable spatial pattern with a vent orifice functioning as a pivot of organisation. In areas immediately surrounding an active orifice (Fig. 6, zone 1, <0.2 m from orifice), no organism was observed except white bacteria mat and occasional intrusion by Caridean shrimps (see also Fig. 4b). Outside this zone, Shinkaia crosnieri individuals aggregated with virtually no space in between them and often on top of each other, consequently forming two or three layers of individual (zone 2, 0.2–0.8 m from orifice). They often took up positions that allow them to have some direct contact with hydrothermal fluid, e.g. on side walls and ridges along which vent fluid travelled upwards (Fig. 5e). In these aggregated conditions, Shinkaia crosnieri showed no or very little movement except when disturbed by the submersible’s manoeuvring; the only visible sign was in their antennae and mouthparts. Other organisms in this zone included Caridean shrimps of Alvinocaris and Lebbeus. The surrounding area (zone 3, 0.8–2.5 m from orifice) was characterised by the coexistence of Shinkaia crosnieri and bathymodiolid mussels, the latter forming a bed on which the former perched, with small crab individuals finding refugia in between mussel shells (Fig. 6, upper photo). Limpets of genera Bathyacmaea and Lepetodrilus occurred commonly on the mussel shell surfaces (Fig. 6, upper photo) and Caridean shrimps were also common. The outermost zone (zone 4) was characterised by an extension of Bathymodiolus beds marked by the absence of Shinkaia crosnieri. In some cases, mussel beds and patches occurred away from an active vent, particularly in sites of Edifices 2 and 5. Individuals of Paralomis crabs were occasionally observed (probably not an obligate vent-associate but a frequent visitor from outside). The spatial extent of this outermost zone was often difficult to assess with accuracy due to the dark colour of Bathymodiolus mussels (without accompanying Shinkaia crosnieri which would stand out as clear white objects).
On two mounds with low (Edifice 1) and high (Edifice 3) hydrothermal activity, Shinkaia aggregations showed similar size ranges (210–17,600 cm2) and no significant difference in the area–perimeter relationship (ANCOVA, p > 0.05). However, there was a clear difference in terms of spatial occupancy pattern (Fig. 7, left). On Edifice 1, the majority of grid areas (47% of total) were unoccupied by Shinkaia crosnieri and its occupancy rarely exceeded 50% with no grid reaching 70% or more occupancy, leading to a single-peaked pattern of the frequency distribution of occupancy values. In contrast, Edifice 3 was characterised by the occurrence of multiple peaks in occupancy pattern, corresponding to (1) 100% occupancy by Shinkaia crosnieri, (2) intermediate values of occupancy centred around 30–40% of occupancy, (3) area without Shinkaia crosnieri (=zero occupancy). Estimated minimum density of Shinkaia crosnieri also showed similar contrasting patterns on Edifice 1 and 3 (Fig. 7, right). The vast majority (92%) of grids on Edifice 1 had densities of <240 individuals m−2 of Shinkaia crosnieri, and only 1.9% of grids were estimated to have densities of >320 individuals m−2. In contrast, on Edifice 3, 30% of grids had >320 individuals m−2 of Shinkaia crosnieri, of which 14% exceeded densities of 560 individuals m−2. Thus, Shinkaia crosnieri was more strongly aggregated on Edifice 3 than on Edifice 1.
Patterns of area occupancy (panels on the left) and density (number of individuals 50 × 50 cm−2) (panels on the right) of Shinkaia crosnieri on Edifice 1 (n = 299) and Edifice 3 (n = 305). Proportional frequencies of quadrats falling into increasing occupancy/density intervals are shown; note that the case of occupancy = 0 is separately represented
4 Discussion
4.1 Spatial distribution and morphology of hydrothermal vents
The occurrence of hydrothermal vents in the Okinawa Trough is apparently non-random and has demonstrated aggregated patterns on different spatial scales. On the scale of 102 to 103 km, i.e. the entire basin, hydrothermal vents were concentrated in the northern and southern parts with no vent being found in the central plain of the Trough (Fig. 1). On the scale of 1–10 km, hydrothermal vents in Iheya North were concentrated within an area of 1 km2 (Fig. 2, upper). Further, on the scale of 102 to 103 m, the majority of vents occurred within a radius of 200 m (Fig. 2, lower), a situation somewhat similar to the occurrence of clusters of sulphur mounds at the Main Endeavour Field of the Juan de Fuca Ridge (Delaney et al. 1992; Robigou et al. 1993) and the PACMANUS site of the Manus Basin (Hashimoto et al. 1999). It is notable here that the occurrence of vents and vent-associated assemblages demonstrated directional, parallel patterns with Edifices 3 and 4 forming the central axis (Fig. 2, lower) and other vents lying on approximately parallel lines. Given the fact that, on the scale of the Okinawa Trough as a whole, the axis of basin expansion runs in the NE to W/SW direction (Glasby and Notsu 2003), it is interesting to note that a similar directionality in hydrothermal activity occurs on a much smaller spatial scale. These patterns of spatial distribution of hydrothermal vents are apparently different from those found in the East Pacific Rise where vent sites occurred in an extensive linear manner (Haymon et al. 1991).
Observations of vent structures in the study area suggest a general sequence of mound development in this system. In the 1st stage, hydrothermal fluid seeps out of fissures in hard, flat substrate and small aggregations of sulphate bacteria, galatheid crabs and bathymodiolid mussels occur around seepages. In the 2nd stage, the accumulation of sulphide material begins with an appearance of incipient flange structures, i.e. the lateral outgrowth of a sulphide mound. The 3rd stage refers to a fully developed edifice with multi-layered flange structures, demonstrated most notably by Edifice 3. Organisms occur all over the edifice walls except underneath a flange shelf where hot liquid tends to accumulate. More animals occur towards the top part of an edifice where vents are more numerous and active, as hot liquid and gas tend to go upwards. The 4th stage shows a degenerative state of mound development where hydrothermal activity is going down (e.g. Edifice 1; Fig. 3) with the erosion and eventual loss of flange structures and declining abundances of vent-associated organisms.
Heterogeneity in vent structures as revealed in the present study is considered to reflect variation in the elapsed time and cycle of hydrothermal activity at particular sites. This also means that, though a general developmental pathway may exist as discussed above, vents do not always follow the same destiny. It has been suggested that massive sulphide structures made of sulphide–anhydrite–silica breccias result from episodes of hydrothermal activity–inactivity, rather than a constant accumulation of sulphides due to continuous hydrothermal activity (Humphris et al. 1995). It is possible that many 1st-stage vents would remain in the same state without further development and disappear with declining hydrothermal activity. In fact, numerous 1st-stage vents occurred as ‘subordinate vents’ on the lower part and walls of an edifice. Therefore, a very small proportion of 1st-stage vents are sufficiently strong and long-lasting to develop into a full-scale sulphide edifice. On the other hand, all vent structures would eventually cease their hydrothermal activity, as the centre of activity gradually but inevitably shifts in position with the expansion of a back-arc basin. Consequently, active vents ‘migrate’ with passage of time, a phenomenon which is thought to underlie the current disjointed distribution of hydrothermal vent sites in the Okinawa Trough. In other words, active vents in the back-arc systems are probably less stable temporally and short-lived on average compared with their counterparts in mid-ocean spreading ridges where the centre of hydrothermal activity is relatively stable in space (see Embley and Chadwick 1994). Thus, hydrothermal vents of a back-arc system are considered to present a highly dynamic environment to their inhabitants, on comparable or smaller scales of time and space as seen in hydrothermal systems of MORs (Fustec et al. 1987; Gebruk et al. 2000a, b; Copley et al. 1997, 2007).
The massive pyramid-shaped structure with fully developed flanges (Edifice 3; Fig. 4) was revealed in its entirety for the first time from the Okinawa Trough in the present analysis. This is of a much larger scale in comparison to a pyramidal flange structure (ca. 2 m high) previously reported from a nearby area (Glasby and Notsu 2003). While this has a clear structural similarity to sulphide mounds found on the Juan de Fuca Ridge (Robigou et al. 1993; Tivey et al. 1999), the broad pyramidal shape rather than a tall chimney shape of the whole construct suggests that the deposition of sulphide material in this system has been slower than in most mid-ocean hydrothermal systems where simple columnar chimneys commonly occur along fast-spreading ridges (Wilcock and Delaney 1996). The fact that shimmering fluid was often visible on the upper side of a flange and that macrofauna tended to be sparse on the upper side of a horizontally expanding flange structure suggests that the elevated temperature occurring there (see Woods and Delaney 1992) may be inhospitable to invertebrates. An experiment under high pressure conditions (Lee 2003) indicated that the temperature tolerance of deep-sea metazoan life would be under 60°C.
Even if the position of a vent and its level of hydrothermal activity do not appreciably change over a relatively short period of time (e.g. several weeks to several years), the physical structure of a sulphide mound is constantly changing. This is due to the fact that sulphide structures, especially surface projections such as chimney tops and flanges, are mechanically weak and their increasing weight and volume cannot permanently be supported. Similarly, in the case of vents with decreasing hydrothermal activity, the accumulation of new sulphide deposits is less than what is being lost by gradual erosion of surface structures. In the present study, a lateral projection (part of a flange, ca. 1 m length × 1 m height) on the top part of Edifice 3 was apparently lost sometime between 1999 and 2002, with a concomitant loss of a colony of vestimentiferan tubeworms (the largest aggregation on Edifice 3) that was observed in earlier surveys. This clearly indicates that sulphide mounds undergo constant morphological changes that would affect the development of vent-associated assemblages.
4.2 Vent structures and their assemblages
The hydrothermal vent assemblages of the Okinawa Trough are characterised uniquely by the dominance of a galatheid Shinkaia crosnieri (Baba and Williams 1998) around virtually all active vents (this study; Kimura et al. 1989; Fujikura et al. 2001). While galatheid crabs are common in both deep-sea hydrothermal and non-hydrothermal habitats (e.g. Gage and Tyler 1991; Desbruyères and Segonzac 1997; Van Dover 2000; Tokeshi 2003), dense aggregations of galatheids seem to characterise in particular high-temperature vents of the Okinawa Trough (e.g. observations in Hatoma and Dai-yon Yonaguni Knolls; Tsuchida et al. 2003). In hydrothermal systems of the East Pacific Rise and the Northeast Pacific Ridges, vestimentiferan tubeworms formed dense aggregations around vent orifices (Fustec et al. 1987; Sarrazin and Juniper 1999; Tsurumi and Tunnicliffe 2003), while in Mid-Atlantic and Indian Ocean hydrothermal systems aggregations of free-swimming rimicarid shrimps characterised active diffusers (Gebruk et al. 1997; Segonzac 1992; Segonzac et al. 1992; Polz et al. 1998; Van Dover et al. 1996, 2001; Hashimoto et al. 2001; Copley et al. 2007).
Amongst decapod crustaceans, bythograeid crabs have been reported as being closely associated with hydrothermal vents on the East Pacific Rise (Bythograea thermydron, Williams 1980) the back-arc basins of western Pacific (Mariana: Austinograea williamsi, Hessler and Lonsdale 1991; Manus and Lau: A. alayseae, Desbruyères and Segonzac 1997; Izu-Ogasawara: Gandalfus yunohana, Hashimoto and Fujikura 1992) and also the Mid-Atlantic Ridge (Segonzacia mesatlantica, Desbruyères and Segonzac 1997; Cuvelier et al. 2009). The spatial aggregations of these bythograeid crabs, however, do not seem to reach the magnitude observed for Shinkaia crosnieri in the Okinawa Trough.
Apart from tube worms and crustaceans, bathymodiolid mussels are common and often achieve a high abundance in most hydrothermal vent systems around the world (Gebruk et al. 2000a; Van Dover 2002; Cuvelier et al. 2009). On the other hand, bathymodiolids do not normally occur very close to an active vent orifice but some distance from it (note, however, the report by Cuvelier et al. 2009 documenting the occurrence of Bathymodiolus azoricus aggregations close to fluid exits at Lucky Strike, MAR), as observed in the present study for Bathymodiolus platifrons with its distribution extending to the periphery of a vent. This may reflect their lower tolerance levels of temperature and/or hydrogen sulphide than aggregation-forming tubeworms (Lee 2003).
The observed spatial arrangements of the two large-sized macroinvertebrates that characterise hydrothermal vent assemblages of the Okinawa Trough (this study; Fujikura et al. 2001) reflect the underlying spatial pattern of hydrothermal activity. In particular, Shinkaia aggregations were invariably observed in places where a visible shimmering of hydrothermal fluids occurred. As free-ranging animals, Shinkaia crabs appear to have a clear advantage over sessile Bathymodiolus in colonising vent habitats, similar to a free-ranging rimicarid Rimicaris exoculata in Mid-Atlantic systems (Segonzac 1992; Segonzac et al. 1992). In hydrothermal vents, physicochemical aspects including the flow regimes of hydrothermal fluids, temperature and hydrogen sulphide concentrations are highly variable in space and time, leading to high variability in faunal abundance and distribution (Hessler et al. 1985; Fustec et al. 1987; Segonzac 1992; Chevaldonné and Jollivet 1993; Van Dover et al. 1996; Copley et al. 1997, 2007; Sarrazin et al. 1997, 1999; Sarrazin and Juniper 1999; Gebruk et al. 2000a, b). In such variable environments, mobile Shinkaia crosnieri is well-adapted for forming aggregations in optimum microhabitats, as demonstrated by their pattern of spatial occupancy on edifices of contrasting activity levels (Fig. 7). Thus, Shinkaia crosnieri formed strong aggregations in hydrothermally active sites with their abundances closely reflecting the strength of hydrothermal activity. This also agrees with the observation that Shinkaia crosnieri did not aggregate around a relatively low-temperature vent under an artificial rearing condition (Miyake et al. 2007).
The predominance of Shinkaia crosnieri with Bathymodiolus mussels in the Okinawa Trough makes a contrast to hydrothermal vent systems of the Eastern Pacific where vestimentiferan tubeworms are predominant around active orifices, especially at early stages of vent development (Desbruyères 1995; Fustec et al. 1987; Tsurumi and Tunnicliffe 2001, 2003). This gives credence to the view that the faunal assemblages of the Okinawa Trough constitute a biogeographically distinct entity, largely due to the fact that the Okinawa Trough is tectonically separate from MOR systems. This in turn points to the importance of plate tectonics in the formation of hydrothermal vent faunas (Tunnicliffe and Fowler 1996; Tunnicliffe et al. 1998). The rapid colonisation of a vent site by vestimentiferans as observed in the East Pacific Rise (Shank et al. 1998) seems to be replaced in the Okinawa Trough by Shinkaia crabs capable of making quick responses to vent appearance. It may be hypothesised that their tendency to make dense aggregations (‘patches’) around a vent orifice hinders colonisation by vestimentiferans in this system. Further, aggregations of Bathymodiolus could also hinder vestimentiferan colonisation. It remains to be seen whether such spatial interactions as observed for sessile intertidal assemblages (Tokeshi 1999) could account for the relatively low abundance (i.e. lack of large aggregations, though they are commonly observed as scattered patches/individuals in all types of vent environments) of vestimentiferans in the hydrothermal vent systems of the Okinawa Trough.
5 Conclusion
Hydrothermal vents in the Okinawa Trough back-arc system had varied spatial structures on regional to local scales, including the morphological variability of sulphide edifices. These structural variations coupled with the changing pattern of hydrothermal activity were closely accompanied by variation in vent-associated assemblages, particularly an endemic galatheid Shinkaia crosnieri and bathymodiolid mussels. Comparative geomorphological and biogeographical studies of different vent structures may give further insight into the workings of these unique hydrothermal systems.
References
Aoki M, Nakamura K (1989) The occurrence of chimneys in Izena Hole No. 2 ore body and texture and mineral composition of the sulfide chimneys. Proc JAMSTEC Symp Deep Sea Res 5:197–210 (in Japanese with English summary)
Baba K, Williams AB (1998) New Galatheoidea (Crustacea, Decapoda, Anomura) from hydrothermal systems in the West Pacific Ocean: Bismarck Archipelago and Okinawa Trough. Zoosystema 20:143–156
Chevaldonné P, Jollivet D (1993) Videoscopic study of deep-sea hydrothermal vent alvinellid polychaete populations: biomass estimation and behaviour. Mar Ecol Prog Ser 95:251–262
Childress JJ, Fisher CR (1992) The biology of hydrothermal vent animals: physiology, biochemistry and autotrophic symbioses. Oceanogr Mar Biol Annu Rev 30:337–441
Copley JTP, Tyler PA, Murton BJ, Van Dover CL (1997) Spatial and interannual variation in the faunal distribution at Broken Spur vent field (29°N, Mid-Atlantic Ridge). Mar Biol 129:723–733
Copley JTP, Jorgensen PBK, Sohn RA (2007) Assessment of decadal-scale ecological change at a deep Mid-Atlantic hydrothermal vent and reproductive time-series in the shrimp Rimicaris exoculata. J Mar Biol Assoc UK 84:859–867
Cuvelier D, Sarrazin J, Colaço A, Copley J, Desbruyères D, Glover AG, Tyler P, Serrão Santos R (2009) Distribution and spatial variation of hydrothermal faunal assemblages at Lucky Strike (Mid-Atlantic Ridge) revealed by high-resolution video image analysis. Deep Sea Res Part I 56:2026–2040
Delaney JR, Robigou V, McDuff RE, Tivey MK (1992) Geology of a vigorous hydrothermal system on the Endeavour Segment, Juan de Fuca Ridge. J Geophys Res 97:19663–19682
Desbruyères D (1995) Temporal variations of deep-sea hydrothermal communities at 13°N/EPR. InterRidge News 4:6–10
Desbruyères D, Segonzac M (1997) Handbook of deep-sea hydrothermal vent fauna. Èditions Ifremer, Brest
Desbruyères D, Almeida A, Biscoito M, Comtet T, Khripounoff A, Le Bris N, Sarradin PM, Segonzac M (2000) A review of the distribution of hydrothermal vent communities along the northern Mid-Atlantic Ridge: dispersal vs. environmental controls. Hydrobiologia 440:201–216
Desbruyères D, Biscoito M, Caprais JC, Colaco A, Comtet T, Crassous P, Fouquet Y, Khripounoff A, Le Bris N, Olu K, Riso R, Sarradin PM, Segonzac M, Vangriesheim A (2001) Variations in deep-sea hydrothermal vent communities on the Mid-Atlantic Ridge near the Azores plateau. Deep Sea Res Part I 48:1325–1346
Embley RW, Chadwick WW (1994) Volcanic and hydrothermal processes associated with a recent phase of sea-floor spreading at the Northern Cleft segment—Juan-de-Fuca Ridge. J Geophys Res 99:4741–4760
Fujikura K, Fujiwara Y, Ishibashi J, Katayama S, Komatsu T, Maezawa Y, Maki Y, Miyazaki J, Tsuchida S, Yamaguchi T, Yamanaka T, Watabe H, Watanabe H, Zielinski S, Kato K (2001) Report on investigation of hydrothermal vent ecosystems by the crewed submersible ‘Shinkai 2000’ on the Dai-yon (No. 4) Yonaguni Knoll and the Hatoma Knoll, the Okinawa Trough. JAMSTEC J Deep Sea Res 19:142–154 (in Japanese with English summary)
Fujikura K, Okutani T, Maruyama T (2008) Deep-sea life—biological observations using research submersibles. Tokai University Press, Tokyo (in Japanese with English captions)
Fustec A, Desbruyères D, Juniper SK (1987) Deep-sea hydrothermal vent communities at 13N on the East Pacific Rise: microdistribution and temporal variations. Biol Oceanogr 4:121–164
Gage JD, Tyler PA (1991) Deep-sea biology. Cambridge University Press, Cambridge
Galéron J, Menot L, Renaud N, Crassous P, Khripounoff A, Treignier C, Sibuet M (2009) Spatial and temporal patterns of benthic macrofaunal communities on the deep continental margin in the Gulf of Guinea. Deep Sea Res Part II 56:2299–2312
Gebruk AV, Galkin SV, Vereshchaka AL, Moskalev LI, Southward AJ (1997) Ecology and biogeography of the hydrothermal vent fauna of the Mid-Atlantic Ridge. Adv Mar Biol 32:93–144
Gebruk AV, Chevaldonné P, Shank T, Lutz LA, Vrijenhoek RC (2000a) Deep-sea hydrothermal vent communities of the Logatchev area 14°45′N, Mid-Atlantic Ridge): diverse biotopes and high biomass. J Mar Biol Assoc UK 80:383–393
Gebruk AV, Southward EC, Kennedy H, Southward AJ (2000b) Food sources, behaviour, and distribution of hydrothermal vent shrimps at the Mid-Atlantic Ridge. J Mar Biol Assoc UK 80:485–499
Glasby GP, Notsu K (2003) Submarine hydrothermal mineralization in the Okinawa Trough, SW of Japan: an overview. Ore Geol Rev 23:299–339
Gooday AJ (2002) Biological responses to seasonally varying fluxes of organic matter to the ocean floor: a review. J Oceanogr 58:305–332
Grassle JF (1986) The ecology of deep-sea hydrothermal vent communities. Adv Mar Biol 23:301–362
Hashimoto J, Fujikura K (1992) Vent communities at the Suiyo Seamount, Mokuyo Seamount and Nikko Seamount. Proc JAMSTEC Symp Deep Sea Res 8:48–51 (in Japanese with English summary)
Hashimoto J, Ohta S, Fujikura K, Miura T (1995) Microdistribution pattern and biogeography of the hydrothermal vent communities of the Minami-Ensei Knoll in the Mid-Okinawa Trough, western Pacific. Deep Sea Res Part I 42:577–598
Hashimoto J, Ohta S, Fiala-Médioni A, Auzende J-M, Kojima S, Segonzac M, Fujiwara Y, Hunt JC, Gena K, Miura T, Kikuchi T, Yamaguchi T, Toda T, Chiba H, Tsuchida S, Ishibashi J, Henry K, Zbinden M, Pruski A, Inoue A, Kobayashi H, Birrien J-L, Naka J, Yamanaka T, Laporte C, Nishimura K, Yeats C, Malagun S, Kia P, Oyaizu M, Katayama T (1999) Hydrothermal vent communities in the Manus Basin, Papua New Guinea: results of the BIOACCESS cruise’96 and’98. InterRidge News 8:12–18
Hashimoto J, Ohta S, Gamo T, Chiba H, Yamaguchi T, Tsuchida S, Okudaira T, Watabe H, Yamanaka T, Kitazawa M (2001) First hydrothermal vent communities from the Indian Ocean discovered. Zool Sci 18:717–721
Haymon RM, Fornari D, Edwards M, Carbotte S, Wright D, Macdonald KC (1991) Hydrothermal vent distribution along the East Pacific Rise crest (9°09′-54′N) and its relationship to magmatic and tectonic processes on fast-spreading mid-ocean ridges. Earth Planet Sci Lett 104:513–534
Hessler RR, Lonsdale PF (1991) Biogeography of Mariana Trough hydrothermal vent communities. Deep-Sea Res 38:185–199
Hessler RR, Smithey WM, Keller CH (1985) Spatial and temporal variation of giant clams, tubeworms and mussels at deep-sea hydrothermal vents. In: Jones ML (ed) Hydrothermal vents of the eastern Pacific: an overview. Bull Biol Soc Wash 6:411–428
Humphris SE, Herzig PM, Miller DJ, Alt JC, Becker K, Brown D, Brugmann G, Chiba H, Fouquet Y, Gemmell JB, Guerin G, Hannington MD, Holm NG, Honnorez JJ, Iturrino GJ, Knott R, Ludwig R, Nakmura K, Petersen S, Reysenbach AL, Rona PA, Smith S, Sturz AA, Tivey MK, Zhao X (1995) The internal structure of an active sea-floor massive sulfide deposit. Nature 377:713–716
Johnson NA, Campbell JW, Moorre TS, Rex MA, Etter RJ, McClain CR, Dowell MD (2007) The relationship between the standing stock of deep-sea macrobenthos and surface production in the western North Atlantic. Deep Sea Res Part I 54:1350–1360
Jollivet D (1996) Specific and genetic diversity at deep-sea hydrothermal vents: an overview. Biodivers Conserv 5:1619–1653
Kato S, Takano Y, Kakegawa T, Oba H, Inoue K, Kobayashi C, Utsumi M, Marumo K, Kobayashi K, Ito Y, Ishibashi J, Yamagishi A (2010) Biogeography and biodiversity in sulfide structures of active and inactive vents at deep-sea hydrothermal fields of the southern Mariana Trough. Appl Environ Microbiol 76:2968–2979
Kimura M, Tanaka T, Kyo M, Ando M, Oomori T, Izawa E, Yoshikawa I (1989) Study of topography, hydrothermal deposits and animal colonies in the Middle Okinawa Trough hydrothermal areas using the submersible “SHINKAI 2000” system. Proc JAMSTEC Symp Deep Sea Res 5:223–244 (in Japanese with English summary)
Kojima S (2002) Deep-sea chemoautosynthesis-based communities in the northwestern Pacific. J Oceanogr 58:343–363
Lee RW (2003) Thermal tolerances of deep-sea hydrothermal vent animals from the Northeast Pacific. Biol Bull 205:98–101
Lutz RA, Kennish MJ (1993) Ecology of deep-sea hydrothermal vent communities: a review. Rev Geophys 31:211–242
Miyake H, Kitada M, Tsuchida S, Okuyama Y, Nakamura K (2007) Ecological aspects of hydrothermal vent animals in captivity at atmospheric pressure. Mar Ecol 28:86–92
Nakagawa S, Takai K (2008) Deep-sea vent chemoautotrophs: diversity, biochemistry, and ecological significance. FEMS Microbiol Ecol 65:1–14
Nakagawa S, Takai K, Inagaki F, Chiba H, Ishibashi J, Kataoka S, Hirayama H, Nunoura T, Horikoshi K, Sako Y (2005) Variability in microbial community and venting chemistry in a sediment-hosted backarc hydrothermal system: impacts of subseafloor phase-separation. FEMS Microbiol Ecol 54:141–155
Ohta S, Kim D (2001) Submersible observations of the hydrothermal vent communities on the Iheya Ridge, Mid-Okinawa Trough. Jpn J Oceanogr 57:663–677
Parson LM, Walker CL, Dixon DR (1995) Hydrothermal vents and processes. Royal Geological Society, London
Polz MF, Robinson JJ, Cavanaugh CM, Van Dover CL (1998) Trophic ecology of massive shrimp aggregations at a Mid-Atlantic Ridge hydrothermal vent site. Limnol Oceanogr 43:1631–1638
Robigou V, Delaney JR, Stakes DS (1993) Large massive sulfide deposits in a newly discovered active hydrothermal system, the Highrise Field, Endeavor Segment, Juan-de-Fuca Ridge. Geophys Res Lett 20:1887–1890
Sarrazin J, Juniper SK (1999) Biological characteristics of a hydrothermal edifice mosaic community. Mar Ecol Prog Ser 185:1–19
Sarrazin J, Robigou V, Juniper SK, Delaney JR (1997) Biological and geological dynamics over four years on a high-temperature sulfide structure at the Juan de Fuca Ridge hydrothermal observatory. Mar Ecol Prog Ser 153:5–24
Sarrazin J, Juniper SK, Massoth G, Legendre P (1999) Physical and chemical factors influencing species distributions on hydrothermal sulfide edifices of the Juan de Fuca Ridge, northeast Pacific. Mar Ecol Prog Ser 190:89–112
Segonzac M (1992) Les peuplements associés a l’hydrothermalisme océaniue du Snake Pit (dorsale médio-atlantique; 23°N, 3480 m): composition et microdistribution de la mégafaune. C R Acad Sci Paris Sér III 314:593–600
Segonzac M, de Saint Laurent M, Casanova B (1992) L’énigme du comportement trophique des crevettes Alvinocarididae des sites hydrothermaux de la dorsale médio-atlantique. Cah Biol Mar 34:535–571
Shank TM, Fornari DJ, Von Damm KL, Lilley MD, Haymon RM, Lutz RA (1998) Temporal and spatial patterns of biological community development at nascent deep-sea hydrothermal vents (9°N, East Pacific Rise). Deep Sea Res Part I 45:465–516
Tivey MK, Stakes DS, Cook TL, Hannington MD, Petersen S (1999) A model for growth of steep-sided vent structures on the Endeavour Segment of the Juan de Fuca Ridge: results of a petrologic and geochemical study. J Geophys Res 104:22859–22883
Tokeshi M (1999) Species coexistence: ecological and evolutionary perspectives. Blackwell, Oxford
Tokeshi M (2002) Spatial distribution of a deep-sea crinoid Pentametrocrinus tuberculatus in the Izu-Ogasawara Arc, western Pacific. J Zool (Lond) 258:291–298
Tokeshi M (2003) Habitat utilization by macroinvertebrates in deep-sea sandy fields: examples from the An’ei Seamount, western Pacific. JAMSTEC J Deep Sea Res 23:87–97
Tsuchida S, Fujiwara Y, Fujikura K (2003) Distribution and population structure of the galatheid crab Shinkaia crosnieri (Decapoda: Anomura: Galatheidae) in the southern Okinawa Trough. Jpn J Benthol 58:84–88 (in Japanese with English summary)
Tsurumi M, Tunnicliffe V (2001) Characteristics of a hydrothermal vent assemblage on a volcanically active segment of Juan de Fuca Ridge, northeast Pacific. Can J Fish Aquatic Sci 58:530–542
Tsurumi M, Tunnicliffe V (2003) Tubeworm-associated communities at hydrothermal vents on the Juan de Fuca Ridge, northeast Pacific. Deep Sea Res Part I 50:611–629
Tunnicliffe V, Fowler CMR (1996) Influence of sea-floor spreading on the global hydrothermal vent fauna. Nature 379:531–533
Tunnicliffe V, McArthur AG, McHugh D (1998) A biogeographical perspective of the deep-sea hydrothermal vent fauna. Adv Mar Biol 34:353–442
Van Dover C (2000) The ecology of deep-sea hydrothermal vents. Princeton University Press, Princeton
Van Dover CL (2002) Community structure of mussel beds at deep-sea hydrothermal vents. Mar Ecol Prog Ser 230:137–158
Van Dover CL, Desbruyères D, Segonzac M, Comtet T, Saldanha L, FialaMedioni A, Langmuir C (1996) Biology of the Lucky Strike hydrothermal field. Deep Sea Res Part I 43:1509–1526
Van Dover CL, Humphris SE, Fornari D, Cavanaugh CM, Collier R, Goffredi SK, Hashimoto J, Lilley MD, Reysenbach AL, Shank TM, Von Damm KL, Banta A, Gallant RM, Gotz D, Green D, Hall J, Harmer TL, Hurtado LA, Johnson P, McKiness ZP, Meredith C, Olson E, Pan IL, Turnipseed M, Won Y, Young CR, Vrijenhoek RC (2001) Biogeography and ecological setting of Indian Ocean hydrothermal vents. Science 294:818–823
Watanabe K (2001) Mapping of the hydrothermal activity area on the Hatoma Knoll in the southern Okinawa Trough. JAMSTEC J Deep Sea Res 19:87–94 (in Japanese with English summary)
Wei CL, Rowe GT, Hubbard GF, Scheltema AH, Wilson GDF, Petrescu I, Foster JM, Wicksten MK, Chen M, Davenport R, Soliman Y, Wang Y (2010) Bathymetric zonation of deep-sea macrofauna in relation to export of surface phytoplankton production. Mar Ecol Prog Ser 399:1–14
Wilcock WSD, Delaney JR (1996) Mid-ocean ridge sulfide deposits: evidence for heat extraction from magma chambers or cracking fronts? Earth Planet Sci Lett 145:49–64
Williams AB (1980) A new crab family from the vicinity of submarine thermal vents on the Galapagos Rift (Crustacea: Decapoda: Brachyura). Proc Biol Soc Wash 93:443–472
Woods AW, Delaney JR (1992) The heat and fluid transfer associated with the flanges on hydrothermal venting structures. Earth Planet Sci Lett 112:117–129
Yamamoto T, Kobayashi T, Nakasone K, Nakao S (1999) Chemosynthetic community at North Knoll, Iheya Ridge, Okinawa Trough. JAMSTEC J Deep Sea Res 15:19–24
Acknowledgments
Thanks are due to the Captain of the support vessel “Natsushima”, Mr. F. Saito, the Commander, Mr. Y. Yoda, and the piloting team of the “Shinkai 2000” submersible (in particular the Pilots, Messrs K. Matsumoto, H. Ueki and Y. Oono) and other crew members for unstinting logistical support during the research cruise in May 2002 during which a major portion of the data used in the present article was gathered. Thanks are also due to Dr K. Fujikura of the Japan Agency for Marine-Earth Science and Technology for providing video records and related data of past diving surveys and all those involved in “Shinkai 2000” surveys until the submersible ceased its active operation in November 2002. I am also indebted to members of the Amakusa Marine Biological Laboratory for various forms of help with data processing and manuscript preparation. This work was supported in part by the P&P programs and the GCOE program of the Kyushu University and by “the Grants-In-Aid for Scientific Research” (Nos. 14255013, 14340246) from the Japan Society for the Promotion of Science.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tokeshi, M. Spatial structures of hydrothermal vents and vent-associated megafauna in the back-arc basin system of the Okinawa Trough, western Pacific. J Oceanogr 67, 651–665 (2011). https://doi.org/10.1007/s10872-011-0065-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10872-011-0065-9