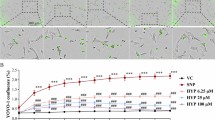
Abstract
Although genetic and environmental factors contribute to neurodegenerative disease, the underlying etiology common to many diseases might be based on metabolic demand. Mitochondria are the main producer of ATP, but are also the major source of reactive oxygen species. Under normal conditions, these oxidants are neutralized; however, under environmental insult or genetic susceptibility conditions, oxidative stress may exceed cellular antioxidant capacities, leading to degeneration. We tested the hypothesis that loss in mitochondrial reserve capacity plays a causative role in neuronal degeneration and chose a cone photoreceptor cell line as our model. 661W cells were exposed to agents that mimic oxidant stress or calcium overload. Real-time changes in cellular metabolism were assessed using the multi-well Seahorse Biosciences XF24 analyzer that measures oxygen consumption (OCR) and extracellular acidification rates (ECAR). Cellular stress resulted in an early loss of mitochondrial reserve capacity, without affecting basal respiration; and ECAR was increased, representing a compensatory shift of ATP productions toward glycolysis. The degree of change in energy metabolism was correlated with the amount of subsequent cell death 24-hours post-treatment, the concentration-dependent loss in mitochondrial reserve capacity correlated with the number of live cells. Our data suggested first, that loss in mitochondrial reserve capacity is a major contributor in disease pathogenesis; and second, that the XF24 assay might represent a useful surrogate assay amenable to the screening of agents that protect against loss of mitochondrial reserve capacity. In future experiments, we will explore these concepts for the development of neuroprotective agents.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Acosta ML, Fletcher EL, Azizoglu S, Foster LE, Farber DB, Kalloniatis M (2005) Mol Vis 11:717–728
Ames A 3rd, Li YY, Heher EC, Kimble CR (1992) J Neurosci 12:840–853
Balaban RS, Nemoto S, Finkel T (2005) Cell 120:483–495
Bandy B, Davison AJ (1990) Free Radic Biol Med 8:523–539
Beckman KB, Ames BN (1998) Physiol Rev 78:547–581
Beeson CC, Beeson GC, Schnellmann RG (2010) Anal Biochem 404:75–81
Brand MD (2000) Exp Gerontol 35:811–820
Brand MD, Nicholls DG (2011) Biochem J 435:297–312
Brand MD, Affourtit C, Esteves TC, Green K, Lambert AJ, Miwa S, Pakay JL, Parker N (2004) Free Radic Biol Med 37:755–767
Cocheme HM, Murphy MP (2008) J Biol Chem 283:1786–1798
Cruickshanks KJ, Klein R, Klein BE (1993) Arch Ophthalmol 111:514–518
de Grey AD (2006) Free Radic Res 40:1244–1249
Dranka BP, Hill BG, Darley-Usmar VM (2010) Free Radical Biol Med 48:905–914
Dranka BP, Benavides GA, Diers AR, Giordano S, Zelickson BR, Reily C, Zou L, Chatham JC, Hill BG, Zhang J, Landar A, Darley-Usmar VM (2011) Free Radical Biol Med 51:1621–1635
Finnegan S, Mackey AM, Cotter TG (2010) Eur J Neurosci 32:322–334
Fliesler SJ, Richards MJ, Miller CY, McKay S, Winkler BS (1997) Exp Eye Res 64:683–692
Fox DA, Poblenz AT, He L (1999) Ann N Y Acad Sci 893:282–285
Fukushima T, Tanaka K, Lim H, Moriyama M (2002) Environ Health Prev Med 7:89–94
Gandhi V, Estey E, Du M, Nowak B, Keating MJ, Plunkett W (1995) Clin Cancer Res 1:169–178
Grimm C, Wenzel A, Hafezi F, Yu S, Redmond TM, Reme CE (2000) Nat Genet 25:63–66
Hagen TM, Yowe DL, Bartholomew JC, Wehr CM, Do KL, Park JY, Ames BN (1997) Proc Natl Acad Sci USA 94:3064–3069
Hassan HM (1984) Methods Enzymol 105:523–532
Hoch FL (1992) Biochim Biophys Acta 1113:71–133
Jarrett SG, Lin H, Godley BF, Boulton ME (2008) Prog Retin Eye Res 27:596–607
Koutalos Y, Yau KW (1996) Trends Neurosci 19:73–81
Krishnamoorthy RR, Crawford MJ, Chaturvedi MM, Jain SK, Aggarwal BB, Al-Ubaidi MR, Agarwal N (1999) J Biol Chem 274:3734–3743
Linsenmeier RA, Braun RD (1992) J Gen Physiol 99:177–197
Lipton SA, Rasmussen H, Dowling JE (1977) J Gen Physiol 70:771–791
Lohr HR, Kuntchithapautham K, Sharma AK, Rohrer B (2006) Exp Eye Res 83:380–389
Lynch RM, Balaban RS (1987) Am J Physiol 252:C225–C231
Mecocci P, MacGarvey U, Kaufman AE, Koontz D, Shoffner JM, Wallace DC, Beal MF (1993) Ann Neurol 34:609–616
Medrano CJ, Fox DA (1995) Exp Eye Res 61:273–284
Miquel J (1992) Mutat Res 275:209–216
Nichols WW, Epstein BJ (2009) Curr Pharm Des 15:304–320
Okawa H, Sampath AP, Laughlin SB, Fain GL (2008) Curr Biol 18:1917–1921
Redinbaugh MG, Turley RB (1986) Anal Biochem 153:267–271
Richter C (1988) FEBS Lett 241:1–5
Sharma AK, Rohrer B (2004) J Biol Chem 279:35564–35572
Sharma AK, Rohrer B (2007) Curr Eye Res 32:259–269
Shigenaga MK, Hagen TM, Ames BN (1994) Proc Natl Acad Sci USA 91:10771–10778
Smith PK, Krohn RI, Hermanson GT, Mallia AK, Gartner FH, Provenzano MD, Fujimoto EK, Goeke NM, Olson BJ, Klenk DC (1985) Anal Biochem 150:76–85
Stackley KD, Beeson CC, Rahn JJ, Chan SS (2011) PLoS One 6:e25652
Tan E, Ding XQ, Saadi A, Agarwal N, Naash MI, Al-Ubaidi MR (2004) Invest Ophthalmol Vis Sci 45:764–768
Tuszynski GP, Murphy A (1990) Anal Biochem 184:189–191
Usachev Y, Verkhratsky A (1995) Cell Calcium 17:197–206
Winkler BS, Arnold MJ, Brassell MA, Sliter DR (1997) Invest Ophthalmol Vis Sci 38:62–71
Winkler BS, Arnold MJ, Brassell MA, Puro DG (2000) Invest Ophthalmol Vis Sci 41:3183–3190
Winkler BS, Sauer MW, Starnes CA (2003) Exp Eye Res 76:715–723
Winkler BS, Sauer MW, Starnes CA (2004a) J Neurochem 89:514–525
Winkler BS, Starnes CA, Sauer MW, Firouzgan Z, Chen SC (2004b) Neurochem Int 45:311–320
Wu M, Neilson A, Swift AL, Moran R, Tamagnine J, Parslow D, Armistead S, Lemire K, Orrell J, Teich J, Chomicz S, Ferrick DA (2007) Am J Physiol Cell Physiol 292:C125–C136
Wu S-B, Wei Y-H (2012) Biochim Biophys Acta 1822:233–247
Yarfitz S, Hurley JB (1994) J Biol Chem 269:14329–14332
Yoshida Y, Singh I, Darby CP (1992) Acta Neurol Scand 85:191–196
Zglinicki Tv (2003) Aging at the molecular level. Kluwer, Dordrecht
Zhang X, Feng Q, Cote RH (2005) Invest Ophthalmol Vis Sci 46:3060–3066
Author information
Authors and Affiliations
Corresponding authors
Additional information
This work was supported in part by National Institutes of Health grant EY013520, EY019320 (B.R.), a Department for Veterans Affairs merit award RX000444 (B.R.), the WG-TRAP award by Foundation Fighting Blindness (B.R. and C.B.), and an unrestricted grant to MUSC from Research to Prevent Blindness (RPB), New York, NY. B.R. is a RPB Olga Keith Wiess Scholar.
Rights and permissions
About this article
Cite this article
Perron, N.R., Beeson, C. & Rohrer, B. Early alterations in mitochondrial reserve capacity; a means to predict subsequent photoreceptor cell death. J Bioenerg Biomembr 45, 101–109 (2013). https://doi.org/10.1007/s10863-012-9477-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10863-012-9477-5